Abstract
Epithelial ovarian cancer (EOC), dominated by high-grade serous carcinoma, continues to impose a heavy global burden, particularly in low- and middle-income countries. Although advances in surgery and chemotherapy have extended survival, relapse within 2 years remains common, underscoring the need for novel strategies. Poly adenosine diphosphate ribose polymerase inhibitors (PARPi) have transformed maintenance therapy by prolonging progression-free survival, yet their influence on health-related quality of life (HRQoL) is less well characterized. Patient-reported outcomes (PROs) serve as key tools for evaluating the impact of treatment on physical, emotional, and social well-being. Trials including SOLO1, NOVA, and ARIEL3 demonstrate the increasing recognition of QoL endpoints, though heterogeneity in tools and endpoints limits cross-trial comparisons. Several gaps persist including limited data collection beyond disease progression. This review integrates evidence from published studies, highlights methodological challenges, and proposes directions for future work, emphasizing the centrality of patient-centered outcomes in advancing precision medicine for ovarian cancer. The evidence underscores the imperative of designing trials that incorporate QoL as a core endpoint, ensuring meaningful benefits for patients in both clinical and real-world settings.
Keywords
Epithelial Ovarian Cancer – an Emerging Pandemic
In 2022, global estimates suggested over 320,000 new cases of ovarian cancer and more than 200,000 related deaths. 1 It is eighth most common cancer among women and also the eighth most common cause of death from cancer. According to GLOBOCAN projection, by 2050, the number of cases diagnosed with ovarian cancer (OC) will increase by 55% and the number of women dying from ovarian cancer will increase by almost 70%. 2 Overall survival is well below other cancers affecting women (3). There is a wide variation in the 5-year survival between countries mainly due to the quality of care provided. 5-year survival ranges between 36%–46% in developed nations while it drops down to much lower figures in low-and middle-income countries where almost 70% of diagnosed women live. 2
Epithelial ovarian cancer (EOC) accounts for 90% of all ovarian cancers and high-grade serous carcinoma (HGSC) is the most common histology comprising approximately 75% of EOCs. Majority of women (two-thirds) are diagnosed in advanced stages as the symptoms are often vague. 3 Management of epithelial ovarian cancer typically involves cytoreductive surgery aiming for maximal tumor clearance, followed by platinum-based chemotherapy and, where appropriate, targeted maintenance therapy. In cases who are poor surgical candidates or where upfront surgery is not feasible due to extensive disease, 3 to 4 cycles of neoadjuvant chemotherapy followed by interval cytoreduction and adjuvant chemotherapy and maintenance treatment provides equivalent survival outcomes. 4 Even with intensive treatment, recurrence occurs in the majority of cases, often within 2 years. 1
Poly adenosine ribose polymerase inhibitors (PARPi) have emerged as the most preferred maintenance option both in first line and in recurrent setting. There are options amongst these drugs to be used in BRCA mutated, Homologous Recombination (HR) deficient as well as proficient cases. 5 These oral drugs can be used for long-term disease control and in clinical trial setting they have been used safely for upto 2 years. The disease and its treatment particularly in recurrent setting and for a long term causes a plethora of symptoms affecting the quality of life (QoL). 6
What is Quality of Life?
The World Health Organization (WHO) defines Quality of Life as an individual’s perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns. 7 The US Food and Drug Administration (FDA) defines HRQoL as “a multidomain concept that represents the patient’s general perception of the effect of illness and treatment on physical, psychological, and social aspects of life. Patient-reported outcomes (PROs) are measures of a patient’s health status as reported directly from the patient without added interpretation by a healthcare worker or anyone else. Hence, QoL, health-related quality of life (HRQoL) and patient reported outcomes (PROs) are unique terminologies. 8
History of HRQoL in Ovarian Cancer trials
In 2009 the US FDA issued its guidance on use of PRO measures in clinical trials. However, multiple barriers existed in practice, including the complexity of oncology trials, logistics, cost etc. which let them to review the guidelines and identify practical recommendations in 2014. Key areas for improvement included encouragement of clinical trial sponsors with comprehensive PRO plans during drug development, oncology-specific PRO research agenda and training. 9 The Consolidated Standards of Reporting Trials Statement have also incorporated PROs in clinical trials in 2013. However, despite these guidelines, in practical world there has been deficiencies in reporting PRO outcomes in EOC. Until 2016, among 26 EOC clinical trial protocols, 58% provided a rationale for assessment of PROs, 31% described the PRO objectives, 46% provided instructions on data collection, 15% included plans for monitoring compliance and 62% included an analysis plan. 10
Quality of Life Measurement – a Necessity for PAPRi trials and in Real World
A patient centered treatment approach balancing drug toxicity with quality of life is mandatory in the current era of precision medicine. This is being increasingly recognized by the patients, clinicians, drug regulatory agencies and all other stakeholders. 11 In 2017, The Fifth Ovarian Cancer Consensus Conference of the Gynaecologic Cancer Intergroup (GCIG) have specified trial endpoints incorporating QoL for front line as well as recurrent setting in ovarian cancer drug trials. Scientific societies like European Society of Medical Oncology (ESMO) and American Society of Clinical Oncology (ASCO) are utilizing standardized scores ie, Magnitude of Clinical Benefit Score and Net Health Benefit respectively. Scores based on OS and PFS are upgraded if there is evidence to indicate an improved HRQoL or delayed deterioration in HRQoL or a substantial reduction in adverse events. 12
As of now, clinical trials have incorporated validated measure of QoL, but it is difficult to compare between trials as they have incorporated varied measures and analysis. Incorporating the correct QoL measure is an important step while designing a clinical trial and researchers often struggle while selecting the best which fits their needs and outcomes amongst the many available options. 13 Since QoL and measurement of symptom benefit is an absolute necessity and suggested end point for all trials in front line and recurrent setting, in this review we aim to discuss the QoL measures reported in published trials of PARPi in Epithelial ovarian cancer, the historical perspective, pitfalls, and current recommendations, as well future directions based on published evidence. 14
Tools for Measuring QoL in Epithelial Ovarian Cancer
Specific Modules of the EORTC and FACIT Questionnaires for EOC Trials 13
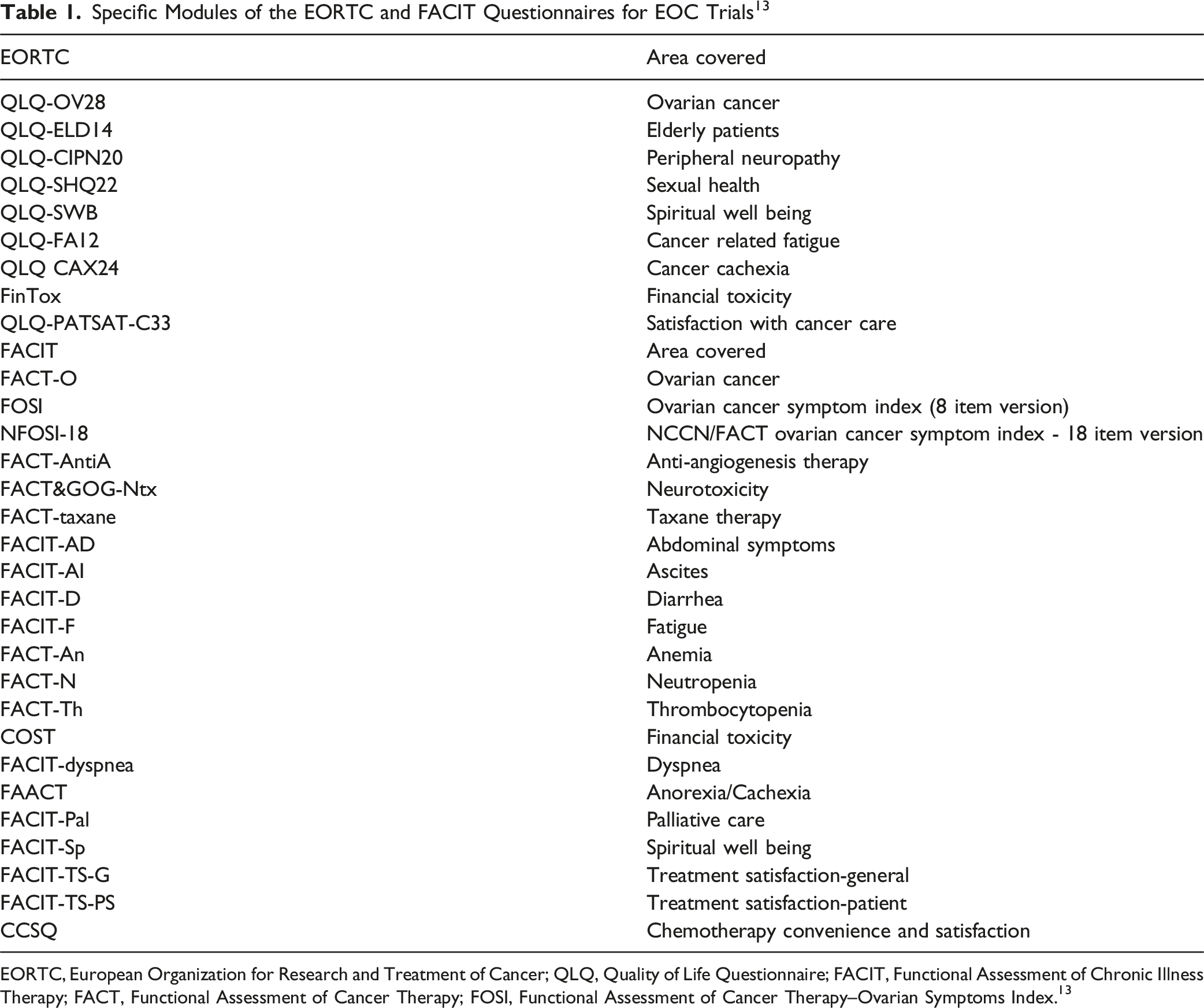
EORTC, European Organization for Research and Treatment of Cancer; QLQ, Quality of Life Questionnaire; FACIT, Functional Assessment of Chronic Illness Therapy; FACT, Functional Assessment of Cancer Therapy; FOSI, Functional Assessment of Cancer Therapy–Ovarian Symptoms Index. 13
In addition, the Gynaecologic Cancer Intergroup has developed a tool called MOST (measure of OC symptoms and treatment concerns) for use in clinical trials of palliative chemotherapy as a measure of OC symptoms and treatment concerns. The MOST will complement existing HRQoL instruments. Additionally, the National Health Institute has developed a Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE) which could lead to a better reporting of adverse effects. 17
QoL Tools Utilized in PARPi trials in EOC
Relapsed Ovarian Cancer
Study 19
Study 19 (NCT00753545) was a phase 2 randomized trial, double-blinded and placebo-controlled, that assessed olaparib as a maintenance therapy in platinum sensitive, relapsed, high grade serous ovarian cancer after 2 or more prior platinum-based regimens with a partial or complete response. Progression-free survival served as the primary study endpoint. This trial also measured HRQoL and disease related symptoms. 18 PRO tools utilized were FACT–O, NFOSI, and the Trial Outcome Index (TOI). Analysis of patient reported outcomes included time to worsening of disease-related symptoms and health-related quality of life. These were analyzed with the use of a Cox proportional-hazards model that included covariates that were used as stratification factors at randomization and rates of improvement in patient-reported outcomes were analyzed with the use of logistic regression. 19
Progression-free survival (PFS) was significantly longer with olaparib than with placebo. There were no significant between-group differences in disease-related symptoms or rates of improvement in HRQoL. The time to worsening of each of these end points was shorter with olaparib than with placebo; however, the difference was not significant. The limitation of this study includes no collection of HRQoL data beyond disease progression and data about time to first subsequent therapy or death (TFST). 20
SOLO2/ENGOT-Ov21
SOLO2/ENGOT-Ov21 (NCT01874353) was a double-blind, randomized, phase 3 trial, evaluating olaparib maintenance treatment in platinum-sensitive, relapsed ovarian cancer patients with a BRCA1/2 mutation who had received at least 2 lines of previous chemotherapy. Primary end point was PFS and this trial demonstrated that olaparib maintenance resulted in significant prolongation of PFS. Frequent but generally mild side effects included fatigue, nausea, and vomiting, which were typically self-limiting. 21
Friedlander et al analyzed the QoL outcomes of this study. The authors hypothesized that olaparib would not negatively affect HRQoL and that there would be additional patient centered benefits. The prespecified primary HRQoL analysis evaluated the change from baseline in the TOI score during the first 12 months of the study. Secondary planned QoL analyses included the duration of good quality of life (defined as time without significant symptoms of toxicity [TWiST] and quality-adjusted progression-free survival [QAPFS]). Olaparib did not have a significant detrimental effect on QOL and there were patient centric benefits in TWiST and QAPFS. In the final analysis, maintenance olaparib provided an improvement of 12.9 months in median overall survival vs placebo. The adjusted average mean change from baseline over the first 12 months in TOI was −2 · 90 (95% CI −4 · 13 to −1 · 67) with olaparib and −2 · 87 (−4·64 to −1 · 10) with placebo (estimated difference −0 · 03; 95% CI −2 · 19 to 2 · 13; P = 0 · 98). Mean QAPFS (13 · 96 [SD 10 · 96] vs 7 · 28 [5 · 22] months; difference 6 · 68, 95% CI 4 · 98-8·54) and mean duration of TWiST (15 · 03 [SD 12 · 79] vs 7 · 70 [6 · 42] months; difference 7 · 33, 95% CI 4 · 70-8·96) were significantly longer with olaparib than with placebo. 22
NOVA
NOVA (NCT018472274) was a double-blind phase 3 trial comparing niraparib with placebo to assess efficacy in platinum sensitive recurrent ovarian cancer with a primary end point being PFS. Patients in the niraparib group had a significantly longer median PFS than in the placebo group. Most common grade 3, 4 adverse events were thrombocytopenia and anaemia and were managed with dose modification. 23
Oza AM et al published the QoL results from this trial. Tools utilised in this study were FOSI and EQ-5D-5 L. PROs were captured during the phase of drug intake as well as at discontinuation and 8 weeks post progression. 24
The mean FOSI score at baseline was similar between the 2 groups. Overall QoL scores remained stable during the treatment and preprogression period in the niraparib group; no significant differences were observed between the niraparib and placebo group, and preprogression EQ-5D-5 L scores were similar between the 2 groups. Disutility analyses showed no significant QoL impairment associated with toxic effects. 25
ARIEL3
ARIEL3 (NCT01968213) was a phase 3 randomized, placebo-controlled study that tested rucaparib after response to second-line or later platinum-based chemotherapy in patients with high-grade, recurrent, platinum-sensitive ovarian carcinoma. The primary outcome was investigator-assessed PFS which significantly improved with rucaparib. Most common grade ≥3 adverse events were anaemia and raised liver enzymes. 26
The PROs were analysed as QA-PFS and Q-TWiST. Tool utilized for measurements was EQ-5D-3 L. Mean QA-PFS was significantly longer with rucaparib vs placebo across all cohorts. Q-TWiST using select grade ≥2 adverse events also consistently favored rucaparib. 27
First Line Maintenance
SOLO1
SOLO1 (NCT01844986) was a phase 3 trial, double-blinded and randomized, designed to evaluate olaparib’s efficacy as maintenance therapy in patients with newly diagnosed advanced high-grade serous or endometrioid ovarian cancer, who had a complete or partial clinical response after platinum-based chemotherapy. 28 The primary end point was PFS and the risk of disease progression or death was 70% lower with olaparib than with placebo. Long term follow up also suggested that benefit derived from 2 years maintenance therapy was sustained beyond the end of treatment, extending median PFS past 4 5 years. 29
Friedlander M analysed the patient centered outcomes and effect of disease progression on health status. Prespecified QoL endpoint was the change from baseline in the FACT-O TOI score for the first 24 months. Prespecified exploratory endpoints were QAPFS and TWiST. The PFS benefit was achieved with no detrimental effect on patients’ HRQoL and was supported by clinically meaningful QAPFS and TWiST benefits. 30
VELIA/GOG-3005
VELIA/GOG-3005 (NCT02470585) was a placebo-controlled phase 3 randomized trial assessing the impact of veliparib when added to first line chemotherapy with carboplatin and paclitaxel and then continued as maintenance monotherapy in advanced high-grade serous OC. The primary end point was PFS which was significantly longer with veliparib induction and maintenance than carboplatin plus paclitaxel therapy alone although it led to a higher incidence of anaemia and thrombocytopenia. 31
The trial utilised the disease related symptom score of the NFOSI-18. The mean change from baseline in the index was compared with the use of a mixed-model, repeated-measures method. The differences in the mean change from baseline in scores between treatment groups were small and not clinically significant. 32
PRIMA/ENGOT-OV26/GOG-3012
PRIMA/ENGOT-OV26/GOG-3012 (NCT02655016) was a double-blinded, phase 3 randomized study that assessed niraparib in patients with newly diagnosed advanced ovarian cancer after first line platinum-based chemotherapy. Progression-free survival served as the primary study endpoint which was significantly longer in niraparib arm than those who received placebo, regardless of the presence or absence of Homologous Recombination Deficiency (HRD). 33 The most common adverse events of grade 3 or higher were anemia, thrombocytopenia and neutropenia. 34
In this trial investigators utilized the FOSI, EQ-5D-5 L, EORTC-QLQ-C30 and EORTC-QLQ-OV28 for QOL assessments which were the secondary outcomes. Pothuri B et al published the PRO data. There was no decline in HRQoL. Scores for abdominal/gastrointestinal symptoms, nausea and vomiting, appetite loss and constipation were worse in niraparib-treated patients, but they resolved over time. 35
PAOLA-1
PAOLA-1 (NCT02477644) was an international, double-blind, phase 3 trial testing olaparib in combination with bevacizumab as maintenance therapy in patients regardless of BRCA mutation status in newly diagnosed, advanced, high-grade ovarian cancer after a response to first-line platinum–taxane chemotherapy plus bevacizumab. 36 The primary end point was investigator assessed PFS which was substantial after adding olaparib in patients with HRD-positive tumors, including those without a BRCA mutation. Adverse events were consistent with the established safety profiles of olaparib and bevacizumab. PROs were a secondary end point and were assessed using EORTC QLQ-C30 and the estimated between-group differences were not clinically significant. 37
ATHENA–MONO/GOG-3020/ENGOT-ov45
ATHENA (NCT03522246) is a currently ongoing phase 3 randomized trial, double-blinded, with 4 different treatment arms (rucaparib, nivolumab, rucaparib + nivolumab, and placebo). The study has 2 separate and fully independently powered comparisons evaluating rucaparib monotherapy (ATHENA–MONO) and rucaparib + nivolumab (ATHENA–COMBO) as first line maintenance treatment in advanced high-grade ovarian cancer. 38 In ATHENA-MONO, the primary end point was investigator-assessed PFS. Rucaparib monotherapy was effective as first-line maintenance, conferring significant benefit vs placebo in patients with advanced ovarian cancer with and without HRD. The most common grade ≥3 treatment-emergent adverse events were anemia and neutropenia. 39
PRO measurement was 1 of the secondary objectives and utilised FACT-O questionnaire. Changes from baseline in scores were similar between rucaparib and placebo. 13
MITO25
MITO25 is an open-label, ongoing phase 1-2 randomized study that aims to evaluate the effect of Carboplatin-Paclitaxel-Bevacizumab (in combination and maintenance) vs Carboplatin-Paclitaxel-Bevacizumab-Rucaparib (Rucaparib only in maintenance) vs Carboplatin-Paclitaxel-Rucaparib (Rucaparib only in maintenance) on PFS in patients with advanced high-grade OC treated according to HRD status. The trial will test the hypothesis that Carboplatin-Paclitaxel-Bevacizumab-Rucaparib and the Carboplatin-Paclitaxel-Rucaparib arms will improve the PFS in comparison to standard Carboplatin-Paclitaxel-Bevacizumab in HRD negative patients and that Carboplatin-Paclitaxel-Bevacizumab-Rucaparib will improve PFS with respect to Carboplatin-Paclitaxel-Rucaparib in HRD positive patients. PROs are secondary objectives which will be calculated using the NFOSI-18 and EQ-5D tool. 40
Summary of QoL Questionnaires and End Points in PARPi Trials of EOC
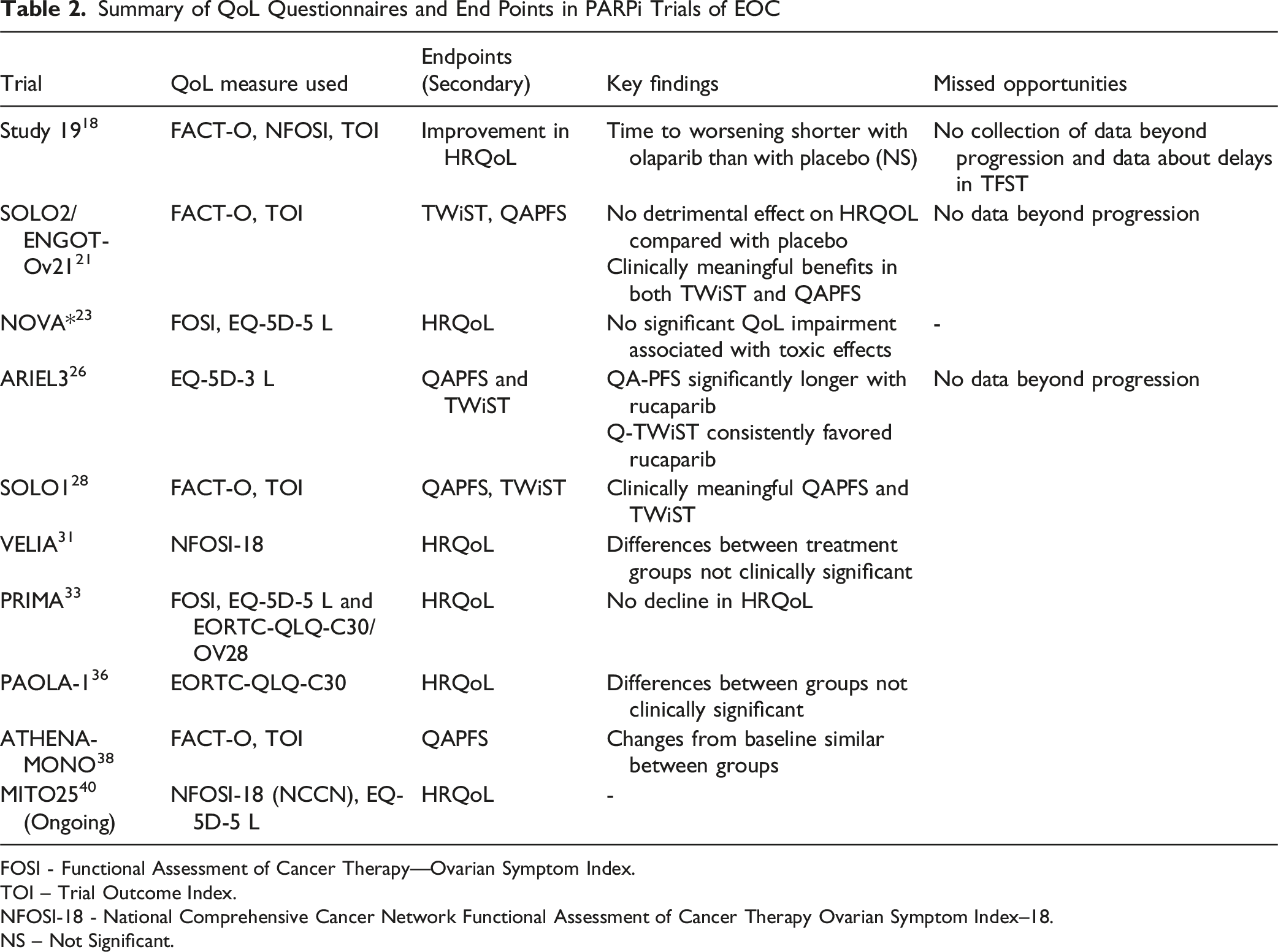
FOSI - Functional Assessment of Cancer Therapy—Ovarian Symptom Index.
TOI – Trial Outcome Index.
NFOSI-18 - National Comprehensive Cancer Network Functional Assessment of Cancer Therapy Ovarian Symptom Index–18.
NS – Not Significant.
Critical Analysis of trials
General Comments
EQ-5D (3 L/5L) is great for cost-utility but blunt for ovarian-specific symptoms (fatigue, neuropathy, abdominal/GI issues). FACT-O/FACT-TOI/FOSI (or EORTC QLQ-C30/OV28) are more sensitive to those domains. Trials that relied mainly on EQ-5D often reported “no difference” while disease-specific scales showed small but real changes. Monthly or every-8-weeks assessments during maintenance generally align with drug kinetics, but in chemo-plus-maintenance designs, cycle-aligned weekly/bi-weekly symptom checks are needed to catch nadirs (eg, day-8/15 fatigue, nausea). Many programs sample too sparsely early on. Multiple scales, domains, timepoints, and subgroups are tested with limited alpha control; “clinically meaningful” change thresholds are inconsistently pre-specified. Some have used ≥10- or ≥12-point FACT-TOI changes or ≥0.08-0.10 EQ-5D utility changes without justification. Mixed modes and language variants are common; few reports document equivalence testing or country-level validation, which can add noise.
Trial Specific Comments
STUDY 19
Maintenance setting with relatively stable symptoms fits monthly HRQoL cadence; disease-specific items capture fatigue and abdominal/GI issues relevant to PARP AEs. EQ-5D lacks sensitivity to low-grade but persistent fatigue/nausea; limited reporting on MCID (Minimal Clinically Important Difference) - based responder rates and on informative drop-out after progression. Hence it was underpowered for small between-arm differences.
SOLO2
Clear maintenance objective; frequent early assessments capture the initial adverse events burden (anemia/fatigue). Multiplicity across domains/timepoints with limited alpha control; reliance on MAR (Missing at Random) in MMRM (Mixed-Effects Model for Repeated Measures); scarce sensitivity analyses for progression-linked missingness. Hence statistical handling likely biased toward “no detriment.”
NOVA
Included separate gBRCA and non-gBRCA cohorts; disease-specific scales were suitable for hematologic-toxicity-driven fatigue. Early high-grade thrombocytopenia/insomnia needed denser sampling during cycles 1-2; MCIDs and responder definitions not consistently pre-specified by cohort. Hence early-cycle under-sampling probably diluted detection of transient symptom decrements.
ARIEL3
Attempt to integrate time and quality—valuable when PFS improves without OS maturity. Utility weights in Q-TWiST/QA-PFS were not patient-elicited within the trial and may not reflect PARP-specific disutility (eg, chronic grade 1-2 fatigue). Threshold analyses help but don’t fix the external-validity gap. State definitions (toxicity vs PFS vs post-progression) can misclassify time if AE data are sparse between visits. Composites were typically exploratory without alpha allocation; missing data from informative drop-out were handled like standard PROs, risking optimistic bias in “time without toxicity.” The composites likely overstated patient-relevant benefit compared with what a PARP-sensitive symptom index would show under rigorous missing-data modelling.
SOLO1
Monthly EQ-5D is acceptable for health-state utilities across long horizons. Ceiling effects—many patients are asymptomatic post-chemo; EQ-5D is insensitive to mild cognitive/fatigue effects; long recall windows risk response shift. Disease-specific scales should have been primary for sensitivity; utilities best reserved for economic modelling.
VELIA
Inclusion of disease-specific symptom constructs appropriate for neuropathy/nausea during chemo. Assessment (often every 3-4 weeks) misses peak chemo toxicities; mode effects if clinic-based paper forms coincide with infusion days; need for cycle-day-specific PROs (eg, day-3, day-8) wasn’t met. Maintenance conclusions more reliable than induction-phase conclusions.
PRIMA
Maintenance-only exposure and dose-individualization strategy make HRQoL detection of fatigue/insomnia meaningful; ovarian-specific items fit the toxicity profile. High early dose reductions complicate interpretation (improvement may reflect titration, not intrinsic tolerability); limited pre-specification of MCID-based hierarchical testing. Early-cycle sampling could have been denser to capture titration period effects.
PAOLA-1
OV28 captures abdominal/GI and treatment side effects relevant to bevacizumab (hypertension, proteinuria has less PRO signal, but GI and fatigue do). Composite exposure (anti-VEGF + PARP) creates overlapping, low-grade chronic symptoms; EQ-5D ceiling effects likely; analyses not powered for small detriments spread across domains. Appropriate disease-specific focus, but generic utilities add little beyond cost-effectiveness inputs.
ATHENA-Mono
Pure maintenance simplifies attribution; ovarian-specific items suit anemia/fatigue profile. Similar to others—MAR assumptions, limited sensitivity analyses, and modest power for small differences across long follow-up. Hence the outcomes would benefit from pre-registered responder analyses and joint modeling of PRO drop-out with PFS.
PROs in Context of Low Middle-Income Countries
It is also imperative to note that these PRO questionnaires are not consistently validated in diverse populations. Most of the trials are conducted in high income countries with a weak representation from women living in lower income nations. Many PROs are developed for specific populations or cultural contexts and may not capture the same concepts or perform reliably in different populations due to cultural or linguistic differences. Addressing these gaps requires cross-cultural adaptation and ongoing validation studies to ensure instruments are fair and appropriate for diverse groups.
GCIG Recommendations and Conclusion
Integration of Patient-Reported Outcome (PRO) End-points in Ovarian Cancer Trials; GCIG Recommendations

PFS, progression-free survival; QoL, quality of life; GCIG, Gynecologic Cancer InterGroup.
New trials should be designed with mandatory implementation of PRO end points with PFS and their incorporation in the statistical analysis plan. The study should be adequately powered to assess these end points. Careful attention and close monitoring are necessary to ensure the integrity of collection of PRO data and avoid missing data. Ideally, HRQoL should be collected even after progression in all trials of maintenance therapy to understand the impact of progression on HRQoL and ascertain whether there are tangible benefits to patients in prolonging PFS. 44
Conclusions
The use of patient-reported outcomes in epithelial ovarian cancer research is no longer optional but essential. While PARP inhibitors provide clear benefits in extending progression-free survival, consistent and rigorous evaluation of QoL ensures that these gains align with what matters most to patients. Future studies must standardize tools, extend follow-up beyond progression, and integrate endpoints such as time to second therapy and definitive QoL deterioration. Such approaches will allow for meaningful comparisons across trials and help establish a patient-centered standard of care. Ultimately, the aim should be to achieve survival benefits without compromising quality of life, thereby delivering treatments that are not only effective but also truly sustainable for patients.
The integration of patient-reported outcomes in epithelial ovarian cancer trials is not just desirable but essential in the era of precision medicine. PARP inhibitors have revolutionized the maintenance treatment landscape by improving progression-free survival; however, their impact on patients’ quality of life requires thorough evaluation. This review underscores the need for robust and consistent use of validated QoL tools, ensuring that patient-centric measures inform treatment decisions.
Clinical trials must adopt a holistic approach, extending QoL assessments beyond disease progression and standardizing instruments to facilitate meaningful comparisons. As recommended by the Gynecologic Cancer InterGroup (GCIG), endpoints such as time to second subsequent therapy (TSST) and time until definitive deterioration of QoL (TUDD) should complement PFS to provide a comprehensive view of therapeutic outcomes. Future studies should prioritize adequately powered trials with clearly defined PRO objectives, ensuring alignment with patient needs and expectations. Incorporating these measures will not only enhance the evaluation of PARPi in clinical settings but also serve as a benchmark for advancing care in ovarian cancer. Ultimately, the goal is to ensure that prolonged survival is achieved without compromising the quality of life, reflecting a truly patient-centered approach to oncology care.
Footnotes
Ethical Considerations
The authors declare that the manuscript is prepared by obeying the Ethical Standards as described in the Committee on Publication Ethics (COPE).
Author Contributions
S.K: Conceptualization; Data curation; Formal analysis; Writing-original draft; Writing-review & editing. K.U: Conceptualization; Writing-original draft; Writing-review & editing. D.S: Investigation; Methodology; Writing-review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
All the authors declare that we don’t have any financial or any other conflict of interests. Given her role as an Editorial Review Board Member, Dr Sarita Kumari had no involvement in the peer-review of this article and had no access to information regarding its peer review.
Data Availability Statement
All data that support the findings of this study are included within the article.
