Abstract
Objectives
Although laparoscopic distal gastrectomy has been widely used for distal gastric cancer, the best functional reconstruction type has not yet been established. Based on previous experience, we propose a modified uncut Roux-en-Y anastomosis. This study aimed to compare the outcomes of different intracorporeal anastomoses after laparoscopic distal gastrectomy.
Methods
From April 2015 to August 2020, the data of 215 patients who underwent laparoscopic distal gastrectomy was collected. The patients were divided into 4 groups according to the digestive tract reconstruction method, Billroth-I, Billroth-II, Roux-en-Y, and the modified uncut Roux-en-Y. Clinicopathologic characteristics, surgery details, short-term outcomes, and postoperative nutritional status were analyzed.
Results
The operation time of Billroth-I anastomosis was significantly shorter (216.2 ± 25.8 min, P < .001) than that of other methods. There was no difference in postoperative complications and OS among the 4 reconstruction methods. The incidences of esophagitis, gastritis, and bile reflux were significantly lower in the Roux-en-Y and uncut Roux-en-Y group (P < .001) 1 year after surgery. And the postoperative albumin and PNI levels in uncut Roux-en-Y group were higher than those in other groups(P < .05). On multivariate analysis, age and reconstruction type were independently related to esophagitis, gastritis, and bile reflux. Serum albumin and the prognostic nutritional index were significantly higher in the uncut Roux-en-Y group than other groups (P < .05).
Conclusions
All 4 reconstruction techniques are feasible and safe. The Roux-en-Y and uncut Roux-en-Y are superior to Billroth-Ⅰ and Billroth-Ⅱ+Braun in terms of reflux esophagitis, gastritis, and bile reflux. Uncut Roux-en-Y may result in better PNI than the others.
Introduction
Gastric cancer is the fifth most common malignancy and the third leading cause of cancer-related death in the world. 1 For tumors located in the distal stomach, radical distal gastrectomy is the main curative treatment. 2
The distal gastrectomy was first performed by Billroth in 1881, and open distal gastrectomy (ODG) has a history of over a hundred years. Furthermore, in 1994, Kitano et al. first reported laparoscopic-assisted distal gastrectomy (LADG). 3 Recently, some randomized controlled trials have supported the non-inferiority of LDG compared with ODG for relapse-free survival in early and locally advanced distal gastric cancer, and LDG was superior to ODG in terms of intraoperative blood loss, wound complication, time to first flatus, hospital stay, incisional hernia, and adhesive bowel obstruction.4-6
After distal gastrectomy, Billroth-Ⅰ (BⅠ), Billroth-Ⅱ (BⅡ), Roux-en-Y (RY), and uncut Roux-en-Y (URY) are the 4 most used reconstruction techniques.2,7 BⅠ is widely used in Japan and Korea due to its physiologic nature. 8 However, it may damage nourishing blood vessels while mobilizing the duodenum, increasing the risk of anastomotic fistula, and this approach is also limited by the location of the tumor.9,10 In advanced-stage patients, BⅡ and RY are more commonly adopted. 11 BⅡ is an easier operation than RY gastrojejunostomy, but it is often associated with bile reflux and reflux gastritis, which may be a potential risk factor for canceration of the remnant stomach and lower esophagus. 12 Consequently, Braun anastomosis was added to avoid bile reflux, but its effectiveness is still controversial.13,14 RY can obviously prevent bile reflux through a complex anastomosis.15,16 Nevertheless, some studies reported that Roux stasis syndrome (RSS) was observed in patients who underwent RY gastrojejunostomy after distal gastrectomy. 17 In order to solve this problem, a new reconstruction technique named URY was devised, which blocked the intestine without cutting it off. It could maintain intestinal continuity and facilitate bowel peristaltic movement, and thus reduce gastric residue and decrease reflux. 18 However, patients who receive URY may suffer from afferent recanalization with the passage of time. 19 Therefore, each method has its own advantages and disadvantages and there is no general consensus on the functional benefits of the different reconstructions.
Although there are several studies comparing different reconstructions, BI, BII, RY, and URY have rarely been compared. In this study, we fully compared the short-term surgical outcomes between the 4 groups and evaluated their quality of life using postoperative nutritional status and reflux-related complications.
Materials and Methods
The reporting of this study conforms to STROBE guidelines. 20
Patients
The data of 215 patients were consecutively collected who underwent laparoscopic distal gastrectomy for distal gastric cancer at Tianjin Medical University General Hospital, China, between April 2015 and August 2020. All patients were histologically diagnosed using preoperative endoscopic biopsy, and distant metastasis was excluded using imaging examinations. Parameters such as patient demographics, comorbidities, American Society of Anesthesiologists (ASA) score, and other clinicopathologic data were evaluated. The tumor node metastasis (TNM) staging was based on the 8th edition of the American Joint Committee on Cancer (AJCC). The patients were routinely performed to evaluate the nutritional status of patients. For patients with malnutrition, we often give nutritional support. All patient verbal consents were obtained and details had been de-identified for this study.
Surgical Procedures
All operations were performed by 2 experienced treatment groups. BI was generally performed for small lesion without pyloric invasion. BII + Braun, RY, and URY were more commonly performed for locally advanced or bulky cancer. Surgeons tend to choose one of the three methods depending on what they specialize in. The surgical methods included LADG and totally laparoscopic distal gastrectomy (TLDG); the diatal gastrectomy and D2 lymph node dissection were performed according to the guidelines of the Japanese Gastric Cancer Association. 2
Billroth-Ⅰ Anastomosis
We used a modified delta-shaped gastroduodenostomy in the BI group. After the duodenum was fully mobilized to 3-4 cm distal to the pylorus, the endoscopic linear stapler (ECR60B, Ethicon, USA) was used to transect the duodenum by rotating 90° from back to front and resected the stomach in the predetermined position. The small incisions were made on the posterior side of the duodenum and the greater curvature of the remnant stomach, respectively. The linear stapler was inserted into the small holes, and the duodenal cutting edge was rotated 90° counterclockwise, then we fired the stapler to complete gastroduodenal anastomosis.
Billroth-Ⅱ+Braun Anastomosis
After transecting the duodenum and stomach, the incision was created on the greater curvature of the remnant stomach at 2 cm from the staple line and the anti-mesentery side of the jejunum 20 cm distal to the Treitz ligament. The linear stapler (ECR60B, Ethicon, USA) was used to complete gastrojejunostomy in an isoperistaltic antecolic fashion. A side-to-side jejuno-jejunal anastomosis (Braun anastomosis) was performed 10 cm distal from the gastrojejunostomy using linear stapler (ECR45W, Ethicon, USA).
Roux-en-Y Anastomosis
As for RY, the jejunum was cut off by (ECR45W, Ethicon, USA) at 25 cm away from the Treitz ligament and the anti-mesentery side was open at 2 cm from the jejunal stump after transecting the duodenum and stomach. The incision was created on the greater curvature of the remnant stomach at 2 cm from the staple line. Then the linear stapler (ECR60B, Ethicon, USA) was used to complete gastrojejunostomy in an isoperistaltic antecolic fashion. The entry point was closed and the jejunal stump was resected by another linear stapler. Finally, a side-to-side jejuno-jejunal anastomosis was performed between the afferent and the efferent loop 10 cm and 40 cm from gastrojejunostomy with linear stapler (ECR45W, Ethicon, USA), respectively.
Uncut Roux-en-Y Anastomosis
For the URY gastrojejunostomy, we made some changes based on research by Uyama et al.
18
and Kim et al.
21
After distal gastrectomy, gastrojejunostomy was perforrmed at a site 20 cm distal to the ligament of Treitz using a liner stapler. A 4-cm incision was done to take out the specimen and occlude the jejunal lumen. The jejunal lumen was occluded 3 cm proximal to the gastrojejunostomy. For permanently occluding the jejunal lumen, we made four or five seromuscular stitches with 1-0 silk circularly around the jejunal wall, and tied it snugly. The site of jejunal occlusion was reinforced with five or six interrupted seromuscular sutures to induce artificial permanent serosa-to-serosa adhesion. Approximately 40 cm distal to the gastrojejunostomy, a side-to-side jejunojejunostomy was made for the diverting the duodenal fluid. This anastomosis was at a jejunal site 10 cm distal to the ligament of Treitz (Figure 1A, Supplementary video 1). Modified uncut Roux-en-Y operation. (A) Gastrojejunostomy is established approximately 20 cm distal to the ligament of Treitz. Occlusion is done at 3 cm proximal to the gastrojejunostomy. Jejunojejunostomy is done between the afferent and efferent jejunal limbs approximately 10 cm distal to the ligament of Treitz and 40 cm distal to the gastrojejunostomy site. (B) We performed1-0 silk seromuscular stitches circularly around the jejunal wall (b1), and provided reinforcement using interrupted seromuscular sutures at the occlusion site (b2). (C) Postoperative upper gastrointestinal radiography was done to determine afferent recanalization. This figure shows the results of the 4 patients who accepted uncut Roux-en-Y anastomosis.
Outcomes
The short-term outcomes were fully collected and graded according to the Clavien-Dindo classification. 22 The perioperative charateristics including operation method, operation time, intraoperative blood loss, number of harvested lymph node, time to first flatus, RSS, and postoperative hospital stay were assessed. In this study, we defined RSS as the presence of symptoms such as nausea, vomiting, or abdominal fullness without mechanical obstruction and more than 7 days of continuous fasting after the operation or refasting. 23 All patients were followed up every 3 months for a year after surgery. Nutritional status parameters included total protein, serum albumin, serum hemoglobin, full blood count, and the prognostic nutritional index (PNI). The PNI was calculated using the following equation: PNI = [10×serum albumin (g/dL)] + [.005 × total lymphocyte count]. Postoperative endoscopic findings at 1 year postoperatively were classified according to residue, gastritis, bile (RGB) classification, 24 and the modified Los Angeles classification. 25 When yellowish liquid was observed in the remnant stomach, it was regarded as bile reflux. The Grade 1, M, and above were classified as a symptomatic group, and Grade 0 and N were classified as an asymptomatic group. We estimated the extent of recanalization using upper gastrointestinal radiography.
Statistical Analysis
All variables were analyzed using SPSS 25.0 for Windows (SPSS, Inc., Chicago, IL) with statistical significance set at P < .05. One-way analysis of variance (ANOVA) with a post hoc Holm–Bonferroni method test was used to evaluate the continuous variables among the 4 groups, and chi-square or Fisher’s exact test was used to evaluate categorical variables. For data that did not conform to a normal distribution, a multiple independent sample nonparametric test following the Kruskal–Wallis H test was performed.
Results
Clinicopathologic Characteristics of Patients (n = 215).

Values are presented as number (%), mean ± standard deviation, or median (interquartile range). BI: Billroth I; BII+Braun: Billroth II and Braun; RY: Roux-en-Y; URY: uncut Roux-en-Y.
# The difference was statistically significant between the BII+Braun and BI (P < .001), and URY (P = .026).
Comparison of the Perioperative Surgical Outcomes.

Values are presented as number (%), mean ± standard deviation, or median (interquartile range). BI: Billroth I; BII+Braun: Billroth II and Braun; RY: Roux-en-Y; URY: uncut Roux-en-Y; LADG: laparoscopic-assisted distal gastrectomy; TLDG: totally laparoscopic distal gastrectomy.
* The difference was statistically significant between the BI and BII+Braun (P < .001), and RY (P < .001), and URY (P < .001).
# The difference was statistically significant between the BII+Braun and RY (P = .009) and URY (P = .045)
The Clavien-Dindo Classification of Postoperative Complications.

Values are presented as number (%). BI: Billroth I; BII+Braun: Billroth II and Braun; RY: Roux-en-Y; URY: Uncut Roux-en-Y.
The over survival (OS) curves are plotted in Figure 2. The 3-year OS rate was 83.9%, 63.2%, 68.9%, and 88.3%, respectively. There was no significant difference in the prognosis of the digestive tract methods based on the TNM stage (P > .05). The 3-year OS rate in stage I, stage II, and stage III was 94.4%, 81.6%, and 51.4%, respectively (P < .001). Comparison of the survival curve in different groups. (A) The OS rate of the Billroth-I, Billroth-II+Braun, Roux-en-Y, and uncut Roux-en-Y anastomosis groups (P = .881). (B) The OS rate of TNM stages I, II, and III (P < .001). (C) The overall survival (OS) rate in patients with TNM stage I (P = .888). (D) The OS rate in patients with TNM stage II (P = .811). (E) The OS rate in patients with TNM stage III (P = .155).
The postoperative nutritional status changes are shown in Figure 3(B-F). The changes in full blood count, serum total protein, serum albumin, and PNI showed a similar tendency. Postoperative serum albumin and PNI were significantly higher in the URY group than in the others after 1 year (P < .05). Due to some patients’ refusal to receive endoscopy during the follow-up, only 131 patients were finally evaluated using endoscopy. According to the endoscopic findings, the amount of gastritis (P = .01), reflux esophagitis (P = .013), and bile reflux (P < .001) was significantly less in the RY and URY groups (Figure 3A). Meanwhile, to assess the functional and nutritional outcomes beyond just the different reconstructions, logistic regression models were used to evaluate gastritis, esophagitis, bile reflux regression, and postoperative PNI (Table 4). Reconstruction and age were independent risk factors for gastritis, reflux esophagitis, and bile reflux. The incidence rate of RSS was lower in the URY group than in the RY group (5.9% vs 15.3%), but there was no significant difference. As for patients in URY group, they were advised to proceed with upper gastrointestinal radiography. Twenty-one patients completed the examination and no one suffered afferent recanalization. Some examples are shown in Figure 1C. Endoscopic findings and nutritional changes after laparoscopic distal gastrectomy in the 4 groups. (A) Endoscopic findings one year after surgery; (B) prognostic nutritional index (PNI); (C) albumin (g/L); (D) total lymphocyte count (109/L); (E) hemoglobin (mg/dL); and (F) total protein (g/L). Logistic Regression Analysis of Postoperative Function and Nutrition. The independent factors associated with gastritis, esophagitis, bile reflux, and postoperative PNI were evaluated using univariate and multivariable logistic regression models. † 50 was set as the cut-off value for PNI and classified the patients into a high-PNI (≥50) group and a low-PNI (<50) group.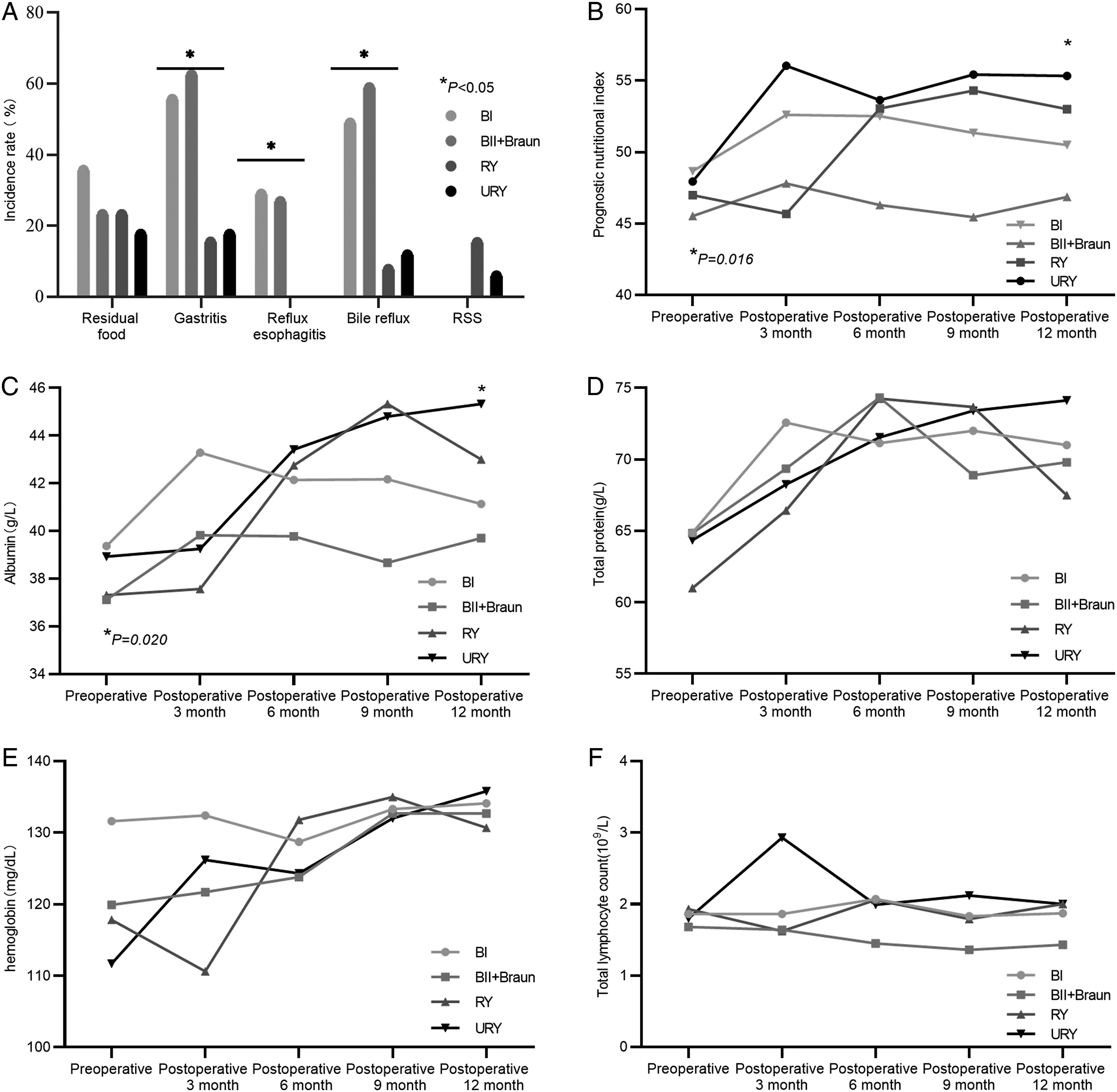

Discussion
In this study, we retrospectively analyzed the differences in short-term and long-term outcomes between the different reconstruction methods after distal gastrectomy. BI was commonly performed when the tumor was located in the antrum, following distal gastrectomy. The incidence of postoperative complications and OS was not statistically different. Changes in the nutritional status were obviously different; the URY group showed a high serum albumin level and PNI. RY and URY could reduce the rate of reflux-related complications. In addition, no recanalization happened in the URY group.
For gastric cancer, radical gastrectomy is the principal therapy for cure,1,2 and reconstruction plays an important role after gastrectomy. The BI, BII, and RY anastomoses have been popularly performed after laparoscopic distal gastrectomy. Although laparoscopic modified delta-shaped gastroduodenostomy is more physiological as it maintains the normal passage of food into the duodenum and allows easy access to the bile duct, it is limited by tumor size and location. 26 Therefore, in those patients who were diagnosed with locally advanced and bulky distal gastric cancer, BII and RY anastomosis were more suitable. BII anastomosis is usually associated with severe bile reflux. 27 And the Braun anastomosis also failed to reduce reflux as expected.14,28 In this study, 58.9% of patients suffered bile reflux in the BII+Braun group. Meanwhile, RY may be related to RSS. 17
URY can reduce the occurrence of RSS by eliminating ectopic pacemakers and preserving the duodenal pacing potential to speed up gastric emptying. 17 As shown in Figure 3, the incidence rate of RSS was lower in the URY group than in the RY group. It is believed that RSS is related to remnant gastric and Y-loop emptying disturbance.29,30 It has been reported that abnormal electrical signals were detected in the Y-shaped loop, which may be due to the loss of pacemaker potential stimulation in the small intestine from the duodenum.30,31 However, with the passage of time, recanalization may occur after URY.19,32 The materials and the types of occlusion lead to different outcomes. Bovine pericardium and Teflon buttress can inhibit recanalization of uncut stapled bowel.33,34 Compared to the 2-row linear stapler, the 6-row stapler significantly reduced the rate of afferent recanalization. 32 In order to reduce the surgical cost, we did some modifications as shown in Figure 1. Patients were examined using upper gastrointestinal radiography half a year after operation, and the longest follow-up time was 38 months. No afferent recanalization has been found till now.
After analyzing our data, we found that URY had similar short-term outcomes compared to other reconstruction types. Generally, the time to first flatus and soft diet are indications of early postoperative gastrointestinal function recovery, and non-physiological reconstruction may lead to slower recovery of gastrointestinal function. 35 Kim et al. reported that the time to first flatus and soft diet in group BⅠ was significantly shortened, which may due to the shorter operation time in group BⅠ causing a relatively small anesthetic inhibitory effect in the gastrointestinal tract. 36 In this study, there were no differences among the 4 groups in terms of the time to first flatus. This may be related to the enhanced recovery after surgery. Some studies have shown that there are differences in postoperative complications according to different reconstruction methods. Anastomotic stricture and bleeding were more common in group BⅠ, duodenal stump leakage was more common in group BⅡ, and the incidence of internal hernia was higher in RY group.37-39 There were no significant differences in postoperative complications, including anastomotic leakage and gastric emptying disorder, according to our data.
Smith et al. 40 pointed out that the long-term complications after distal gastrectomy, such as reflux esophagitis, gastritis, bile reflux, and dumping syndrome, may be related to the type of reconstruction. Some meta-analysis showed that there was no difference in dumping syndrome among the 4 groups. 16 URY and RY were superior to BⅠ and BⅡ anastomosis in preventing bile reflux and residual gastritis, but RSS occurred more frequently in RY and may be related to injury of the celiac branch of the vagus nerve during operation.41,42 Inokuchi et al. 43 reported that RY was associated with less food residue, reflux esophagitis, gastritis, and bile reflux during follow-up than BI. In our study, the incidence of reflux gastritis in the RY and URY groups was significantly lower than that in the BⅡ+Braun group. In terms of bile reflux, the URY and RY groups showed similar results. This is similar to the results shown by Park et al. 44 Bile reflux to the remnant stomach will lead to the occurrence of remnant gastric cancer, as per Werscher’s study 45 URY and RY anastomosis can effectively reduce reflux and obtain better nutritional status than BⅠ and BⅡ+Braun. Yang et al. 37 randomly divided 158 patients who received LADG into URY and BⅡ groups. During the postoperative period, the URY group’s stomach pH values were lower than 7 and the BII group’s pH values were higher than 7. After 1 year of follow-up, patients who received BII presented with a higher incidence of biliary reflux and alkaline gastritis.
Postoperative nutritional status was also related to the quality of life and survival after operation.46,47 Generally, hemoglobin, total lymphocyte count, total protein, and serum albumin are considered to be the factors affecting postoperative nutrition. PNI has been widely used because of its effectiveness, simplicity, and convenience in evaluating preoperative nutritional status and predicting surgical risk in patients with gastrointestinal malignant tumors.47,48 Low PNI is an independent risk factor for short-term postoperative complications and long-term prognosis of many different types of cancer such as gastric, colorectal, and esophageal cancer. 49 In our center, the total lymphocyte count did not change significantly during the follow-up period, but the total protein and albumin fluctuated obviously after operation. The postoperative PNI and serum albumin of the URY group was higher than that of the other 3 groups, which indicated that these patients had better nutritional status than others. Certainly, the postoperative tumor recurrence may be one of the factors influence the nutritional status, too. In this study, we only compared nutritional status among groups at 1 year postoperatively, and survival analysis showed that there was no difference in disease-free survival at 1 year postoperatively, so we did not take it into consideration.
In this study, there was no difference in postoperative complications, and functional and nutritional status between the URY and RY groups. However, the results of a meta-analysis showed that operative time, incidence of reflux gastritis/esophagitis, delayed gastric emptying, and RSS were reduced, and that the level of serum albumin was increased in patients undergoing URY reconstruction compared with those undergoing RY reconstruction. 50 Sah et al. retrospectively analyzed 236 distal gastric cancer patients who underwent curative distal gastrectomy with gastrojejunal RY or URY anastomosis and found that URY could significantly reduce the rate of anastomotic leakage and abdominal infection and shorten the postoperative stay. 51
As discussed above, URY showed excellent long-term functional outcomes compared with BI or BII+Braun, especially in terms of anti-bile reflux and nutritional state. However, this study is only a single-center retrospective study. Most patients were older with advanced-stage tumor, and there was only a small number of patients in the URY group for analysis. In addition, we only analyzed the situation one year after operation. Only total protein, serum albumin, hemoglobin, lymphocyte count, and PNI were included to evaluate the nutritional status. The total cholesterol and nutritional risk index were not investigated. Endoscopy was not performed in a same hospital. In retrospective case-control studies, selection bias is a problem that cannot be ignored. We hope that more samples can be included in the follow-up, and bias can be reduced by random sampling or paired analysis of propensity scores. Therefore, a more comprehensive study will be obtained when we solve the above deficiency.
Conclusion
In conclusion, all 4 reconstruction approaches can be safely and feasibly applied in LDG with similar operative outcomes. URY was superior to BI and BⅡ+Braun in terms of reflux esophagitis, gastritis, and bile reflux. Considering the surgical simplicity, postoperative PNI, and anti-bile reflux ability, URY may be a good choice for LDG.
Supplemental Material
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Investigation and Ethics Committee of the Tianjin Medical University General Hospital, Tianjin, China. All procedures in this study were conducted in accordance with the Investigation and Ethics Committee of the Tianjin Medical University General Hospital (IRB2020-WZ-133) approved protocols.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
