Abstract
Introduction
Neoadjuvant chemoradiation (CRT) is commonly used for esophageal cancer (EC), with pathologic complete response (pCR) linked to improved prognosis. pCR rates range from 25%-30% for adenocarcinoma to 50% for squamous cell carcinoma (SCC). High-volume centers (HVCs) often yield favorable outcomes across various cancers. This study examines whether CRT delivery at an HVC impacts pCR and ypN0 rates in EC patients undergoing esophagectomy.
Methods
In this retrospective cohort analysis, we identified patients with clinical stage T2+/N+ EC who received neoadjuvant CRT and esophagectomy from a prospectively maintained database (1996-2019). CRT was delivered at the HVC or a community center (CC), while all surgeries occurred at the HVC. Primary outcomes were pCR and ypN0.
Results
Among 708 patients, median age was 65 years, with 83.6% male and 95.3% white. Most had adenocarcinoma (87.9%) in the distal esophagus or gastroesophageal junction. CRT was delivered at CCs in 64.8% and at HVC in 35.2%. Radiation dose was higher at the HVC (mean 5195 vs 4944 cGy, P < 0.001), with similar intensity-modulated radiation therapy use (83% vs 79%, P = 0.12). Overall pCR rate was 37.7%, without significant difference between HVC and CC (40.6% vs 36.2%, P = 0.25, multivariable adjusted OR 0.88, P = 0.47). SCC histology was associated with higher pCR (OR 2.39, P = 0.002). Among SCC patients, pCR rate was higher at HVC than CC (75.0% vs 50.0%, P = 0.03) but not significant in multivariable analysis (OR = 2.65, P = 0.11). Conversely, minimal histopathologic response (TRG3) was higher at CCs, not statistically different (17.9% vs 14.5%, P = 0.24). Rate of ypN0 was similar (HVC 70.7% vs CC 68.0%, P = 0.71) with no difference in ratio of positive to resected nodes (P = 0.94).
Conclusion
There were no significant differences in pCR or ypN0 rates by CRT delivery site. pCR was higher for SCC histology at HVC, but did not reach statistical significance. Further research is warranted for SCC specifically.
Plain Language Summary
This study evaluates outcomes for the patients who have esophageal cancer and undergo radiation as well as chemotherapy and surgery for their cancer. The study evaluates whether there are differences in their outcomes whether they had the radiation at the same facility where they had the surgery.
Keywords
Introduction
Locally advanced esophageal cancer (EC), at least clinical T2+ or cN+, has historically been managed with neoadjuvant chemotherapy and radiation based on the CROSS trial data. 1 Pathologic complete response (pCR) and negative pathologic lymph nodes following neoadjuvant therapy (ypN0) are associated with an improved prognosis.2,3 pCR rates commonly range between 25% and 30% for esophageal adenocarcinoma (AC) and up to 50% for squamous cell carcinoma (SCC).2–4
Treatment at high volume centers (HVCs) is associated with more favorable outcomes, including overall survival (OS), for a variety of cancer types.5–7 For example, in patients with non-metastatic rectal cancer, survival following proctectomy improved with increasing facility volume. 5 A retrospective analysis investigating nearly 65 000 patients with advanced prostate cancer from 2004 to 2013 demonstrated a significant association between higher facility volume and improved OS. 6 Within the National Cancer Database, patients treated at HVCs with intensity-modulated radiotherapy for esophageal adenocarcinoma appear to have longer OS compared to those treated at non-HVCs. 7 Another retrospective study assessing the relationship between center volume and outcomes for locally advanced esophageal cancer from 2004 to 2016 within the National Cancer Database have shown similar findings, with patients at HVCs more likely to undergo surgery with lower odds of death during treatment. 8 Another cohort study found significant variability in the diagnosis, evaluation, and treatment of esophageal cancer across regionalized thoracic surgery centers, with outcomes like perioperative mortality significantly worse at low- and medium-volume centers compared to HVCs, despite overall improvements in survival associated with neoadjuvant therapy. 9 Further studies highlight the correlation between high surgical volumes and improved outcomes in esophageal cancer, emphasizing that the centralization of care to high-volume centers is associated with reduced postoperative mortality and morbidity.10,11
It is understood that travel distance is an important consideration for patients undergoing intensive therapy for their cancer care.12,13 As a result, patients will commonly receive a portion of their cancer care, such as chemoradiation, at a different institution that they receive other therapy, such as surgery. 14 In our regional healthcare network, the HVCs serve as a tertiary referral center for esophageal cancer center, providing cancer care to a broad catchment area. However, many patients undergo neoadjuvant chemoradiation at CCs closer to their homes before traveling to the HVC for esophagectomy. This is a direct reflection of some of the most common factors that patients need to consider when planning out their treatment, including travel burden, proximity to care, and insurance constraints. As a result, while the HVC performs a high volume of esophagectomies, it may not necessarily deliver the majority of preoperative chemoradiation for referred patients. Such a dynamic highlights the increased number of patients receiving radiation at a CC in our cohort and underscores the necessity to investigate how the location of neoadjuvant chemoradiation delivery may affect pathologic outcomes. One retrospective study investigating patients with breast cancer and the treatment regimen they underwent (mastectomy vs breast-conserving surgery followed by radiation therapy) compared to the distances they had to travel for their treatments found that patients living further from radiation facilities, such as HVCs, were significantly more likely to undergo mastectomy alone rather than surgery followed by radiation, leading to poorer survival outcomes and thus highlighting the importance of travel distance when patients plan their treatments and a potential urban-rural survival disparity for patients with cancer. 15 Given that pCR and ypN0 rate are thought to be associated with longer OS for esophageal cancer, we sought to examine our institutional experience to assess whether the facility where radiation therapy was administered was associated with pCR and ypN0 rates among our cohort of patients undergoing esophagectomy for locally advanced esophageal cancer. In our clinical practice, patients undergoing surgery at our institution frequently receive neoadjuvant chemoradiation locally, so this naturally presents a varied cohort suitable for addressing this clinical question.
Materials and Methods
Study Cohort
This study was an IRB approved retrospective cohort analysis of a prospectively maintained institutional database (IRB Protocol MCC #16630). Individual consent for this retrospective analysis was waived. The study design was performed in accordance with the STROBE checklist for cohort studies. 16 Patient level data was deidentified prior to analysis. Patients were included for study if they received neoadjuvant CRT for clinical T2+ or clinical N+ esophageal adenocarcinoma or squamous cell carcinoma and a subsequent esophagectomy from 1996 to 2019. Most commonly, Ivor Lewis esophagectomy was performed but any surgical approach to esophagectomy was included in the cohort.
Primary Exposure of Interest
The primary exposure of interest was administration of CRT at a high-volume center (HVC; our institution) vs administration of CRT at a community center (CC). The cohort was split according to this exposure. While patients in the cohort received CRT at different institutions, all patients subsequently received an esophagectomy at the HVC. CRT was prescribed per physician discretion with respect to delivered dose and choice of radiosensitizing chemotherapy regimen. The CCs where CRT was administered varied in size and affiliation but were all facilities staffed by accredited radiation oncologists. It should be noted that the CC cohort includes patients who received CRT at a mix of CC facilities. Details regarding specific therapies are described in the Results.
Primary Outcomes and Covariates
The primary outcomes of interest in our analysis were the differences in pCR and ypN0 rates between patients who received CRT at a HVC vs at a CC. Pathological complete response (pCR) was defined as no residual cancer cells in the primary luminal tumor tissue or any lymph nodes in the surgical specimen. ypN0 was defined as no residual cancer cells detected in any surgically resected lymph node after chemoradiation. Location of the clinically suspicious lymph nodes was considered as a secondary outcome of interest. Other variables that were considered for analysis include patient characteristics (age, sex, race, smoking status), tumor characteristics (histology, anatomic location, differentiation, clinical T stage and clinical N stage), and treatment characteristics (radiotherapy modality, completion of therapy, dosage administered, chemotherapy regimen: carboplatin-taxol regimens [primarily paclitaxel], cisplatin-5-fluorouracil [5-FU], leucovorin/5-FU-oxliplatin [FOLFOX], 5-FU only, other chemotherapy regimens, and no chemotherapy at all). Lastly, treatment era was considered as a variable, designated as treatment before or after publication of CROSS trial in 2012.
Statistical Methods
Descriptive statistics are presented as frequency for categorical variables and median with interquartile range (IQR) for continuous variables. Univariate analyses were performed using Chi-square, Fisher’s exact, and Wilcoxon rank-sum as appropriate. To test for association with the primary outcomes of interest - pCR and ypN0 rates - multivariable logistic regression was used for multivariable analysis. Variables were considered for inclusion in multivariable analysis if associated with the outcome in univariate analysis (P < 0.20) or if considered clinically important. Variables were sequentially removed from the multivariable model if not significantly associated with the outcome (P > 0.05). All statistical analyses were performed using Stata version 14 (StataCorp. 2023. Stata Statistical Software: Release 14. College Station, TX: StataCorp LLC.)
Results
Characteristics for Cohort Undergoing Chemoradiation Followed by Surgery, by Site of Chemoradiation Administration

aNot statistically different after excluding unknown T stage (P = 0.13).
bNot statistically different after excluding unknown N stage (P = 0.10).
The number of patients treated at a HVC was 249 (35.2%), while the number of patients treated at a CC was 459 (64.8%). The mean radiation dosage at a HVC was 5195 cGy, while the mean radiation dosage at a CC was 4944 cGy. The difference in mean radiation dosage was statistically significant (P < 0.001). Patients at an HVC received intensity-modulated radiation therapy (IMRT) at a higher rate than patients at a CC (83% vs 79%, P = 0.12), but this was not statistically significant. Completion of radiation therapy across both the HVC and CCs was very high in both groups (98% vs 98%, P = 0.8). There were no differences in proportion of patients receiving neoadjuvant therapy at HVC vs CC by pre- or post-CROSS era (61% pre-CROSS era for both groups, P = 0.99). There were significant differences in chemotherapy regimens used between the HVC and the CCs (P < 0.001, Table 1). The HVC preferred to use cisplatin-5-FU chemotherapy (n = 167, 67%) while the CCs more commonly administered carboplatin-taxol regimens (typically paclitaxel, n = 161, 35%). In the post-CROSS treatment era, the CCs continued to treat primarily with carboplatin-taxol therapy (n = 119/177, 67%) while the HVC continued to use cisplatin-5FU (n = 57/96, 59%) and in the later years of study preferred FOLFOX therapy (n = 19/96, 20%). There were no differences in number of lymph nodes resected between the HVC or CC. The median nodes removed was 15 vs 14 in the pre-CROSS era (P = 0.43), and 22 vs 21 in the post-CROSS era (P = 0.41)
The overall pCR rate was 37.7% (n = 269). When split by histology, the pCR rate for patients with AC was 35.1%, and the pCR rate for patients with SCC was 57.0% (Figure 1). The pCR rate did not significantly differ between a HVC and CCs (40.6% vs 36.2%, P = 0.25). There was also no significant association between pCR and treatment site (OR 0.88, P = 0.47) after multivariable adjustment for histology, clinical stage, tumor grade, primary tumor site, or treatment era (Table 2). SCC histology was significantly associated with a higher likelihood of pCR (OR 2.63, P < 0.001). Conversely, the rate of minimal histopathologic response (TRG3) was higher at CCs but was not statistically different in univariate or multivariable analysis (17.9% vs 14.5%, P = 0.24; adjusted OR 1.29, P = 0.32). When considering only SCC, pCR rate was significantly higher at the HVC than at the CC in univariate analysis (n = 75.0% vs 50.0%, P = 0.03) but association was lost after multivariable analysis after adjustment for clinical stage, tumor grade, primary tumor site, and treatment era (OR = 3.03, P = 0.063). While significant differences in chemotherapy choice were observed between HVC and CCs, chemotherapy choice was not associated with rate of pCR (P = 0.46). Rates of Pathologic Complete Response (pCR) by Histology whether Chemoradiation Administered at Community Center (CC) Versus High-Volume Center (HVC). Overall, pCR Rate was 37.7%, With pCR Rate for Adenocarcinoma (AC) 35.1%, and pCR Rate for Squamous Cell Carcinoma (SCC) 57.0% Multivariable-Adjusted Association Between Site of Chemoradiation Administration (community center, CC, vs high-volume center, HVC) and pathologic complete response (pCR), reduced multivariable logistic regression model. Squamous cell carcinoma, SCC. aReferent group is high-volume center, HVC.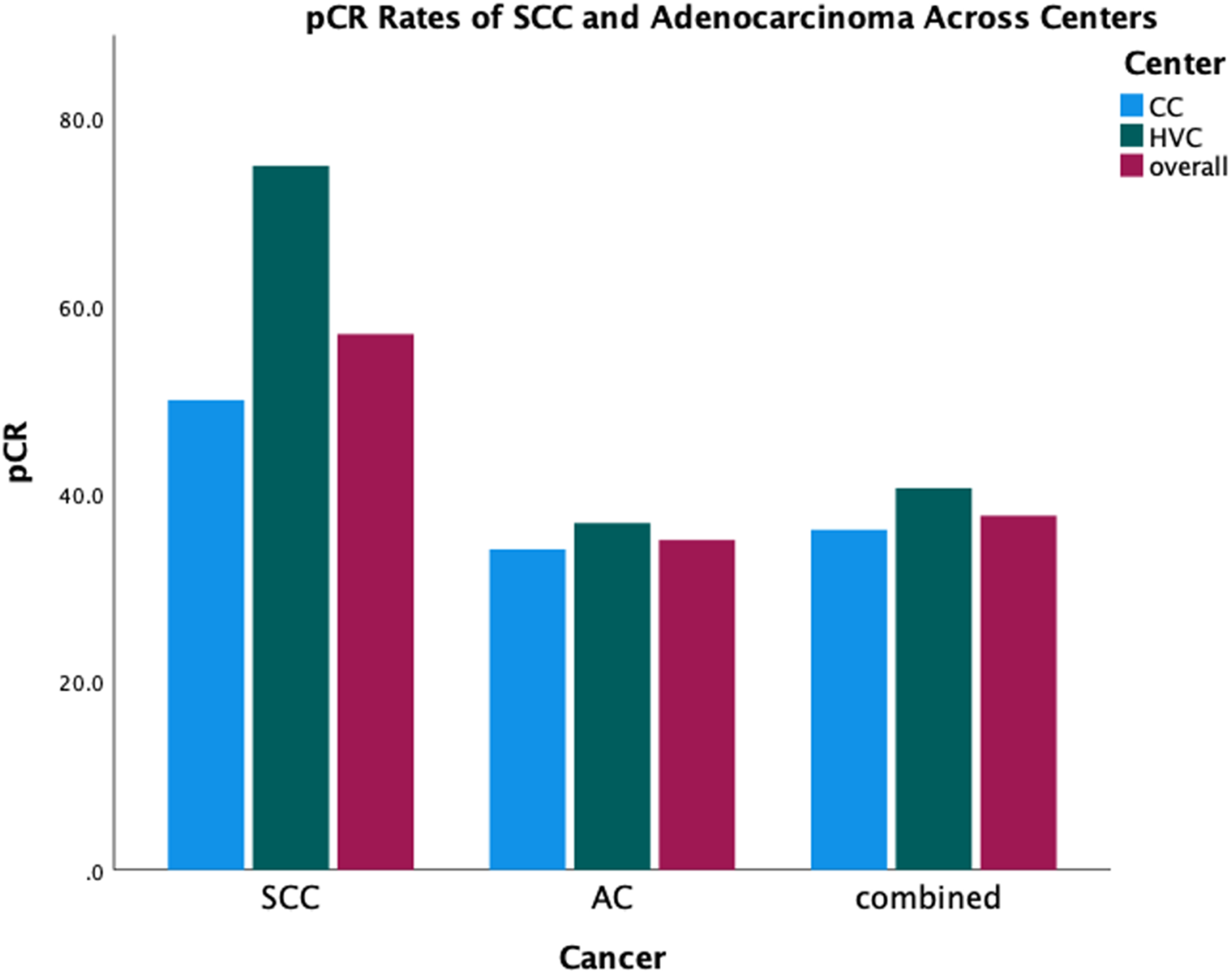

Multivariable-Adjusted Association Between Site of Chemoradiation Administration

(community center, CC, vs high-volume center, HVC) and post-treatment negative nodes (ypN0), reduced multivariable logistic regression model.
aVariable left in model due to clinical importance. There remained no association between site of chemoradiation and ypN0 status with exclusion of this variable.
Location of Clinically Suspicious Lymph Nodes
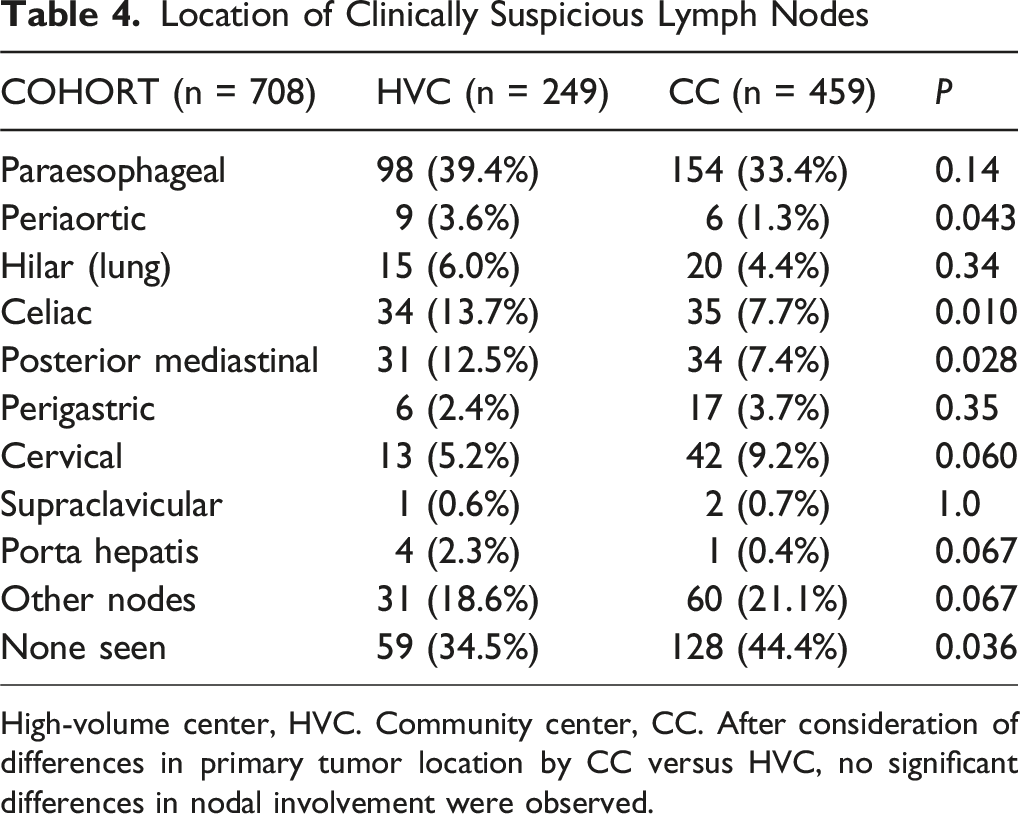
High-volume center, HVC. Community center, CC. After consideration of differences in primary tumor location by CC versus HVC, no significant differences in nodal involvement were observed.
Discussion
In our study, the overall pCR rate was 37.7%, which did not differ significantly between patients receiving therapy at a HVC and CCs, including with adjustment in multivariate analysis. Interestingly, the pCR rate was observed to be higher for patients with SCC receiving CRT at the HVC vs CC on univariate analysis only. This may be secondary to the smaller sample size in the SCC group than AC. Furthermore, the ypN0 rates did not differ significantly between CRT delivery sites, with a rate of ypN0 of 70.7% at the HVC vs 68.0% at the CC. There were some differences observed in nodal basins treated between HVC and community centers. However, due to known phenomenon of skip nodal metastases, it is not possible to say if this was representative of more advanced disease.17,18
We explored whether the HVC may be treating patients with a different distribution of involved lymph nodes to account for a possibility of more advanced disease. However, when accounting for the site of the primary tumor, HVCs and CCs appeared to treat a similar rate of regional nodal disease. Despite the heterogeneity of radiation delivery sites, practitioners, and treatment years, there remained no significant difference in the extent of nodal regions treated. It is important to note that for the early cohort of patients in this study, there were no consensus society guidelines suggesting the clinical nodal target volume for radiation oncologists to include in their elective target volume. In the last decade or so, expert contouring guidelines have been developed to guide individual practice.19–21 Despite this, our study has reinforced that there is consistency of clinical practice based on primary tumor site location.
According to an analysis using the National Cancer Data Base, higher overall survival (OS) rates were associated with receiving trimodal therapy at HVCs, but pCR rates were not different by facility type, which is consistent with the results in this study. 19 This study adds strength to this observation, given the granularity of data available in our institutional experience, compared to that available within the NCDB. While pCR rates were correlated with higher OS, pCR rates were not associated with hospital case volume. Our data indicates that the median RT dosage for patients at HVCs was 51.95 Gy compared to the 49.44 Gy median for patients at CCs. This did not correlate with a higher pCR rate between treatment facilities in the present study. Interestingly, the rate of IMRT utilization for patients treated at an HVC was not statistically different from IMRT use at CCs (83% vs 79%, P = 0.12). This may account for no differences in pathologic differences seen between the groups. We did not find a significant difference in pCR or ypN+ rates between a HVC and CCs. This lack of difference could be due to a multitude of factors, such as effective care coordination and multidisciplinary care between the referring CCs and the HVC. Courrech and colleagues have described comparable outcomes between low-volume esophageal surgery centers and HVCs, suggesting that factors other than hospital volume should be considered when assessing outcomes for surgical approaches to esophageal cancers. 22 While our study investigates the role of CRT in addition to surgery, these comparable findings further underscore the multifaced approach needed due to the complexity of factors influencing treatment outcomes in EC, including radiation dosage, patient demographics, treatment technique, treatment tolerance and concordant co-morbidities.
This is a retrospective analysis which includes several limitations to discuss. First, there is only a single HVC in the analysis. Other HVCs might have different policies and guidelines on which they base their CRT or how they coordinate with CCs. This difference in operation could have affected the rates of pCR and ypN+. Future study from other investigators would be valuable to determine whether the favorable overall outcomes between coordinating HVCs and CCs is seen in other groups which collaborate in patient care. Second, there is a possibility that the patients at the HVC had more anatomically complex disease since they had a significantly higher incidence of celiac, periaortic, and mediastinal clinically suspicious lymph nodes. There is a chance that some difference in management at the HVC (eg, the significantly higher mean radiation dosage used at the HVC) could have improved outcomes for a more challenging patient population, thus masking a true difference in favorable outcomes for EC patients based on facility type. However, as noted above, skip metastases are common in esophageal cancer, and it is difficult to determine whether these differences in nodal distribution represent more advanced disease or not.17,18 Another potential limitation of the study is that patients in the CC cohort received their neoadjuvant therapy at a variety of CCs (as opposed to a single CC). While these centers are staffed by accredited radiation oncologists who typically follow guideline-based treatment protocols for neoadjuvant CRT in esophageal cancer and employed modern treatment planning symptoms, there are likely to be differences between them. Given the retrospective nature of the study and the variability in documentation across these centers, we were unable to ascertain the specific adherence to image guidance protocol, dosimetric quality assurance, or contouring protocols across the CCs. However, the goal of the study was to determine if this practice pattern (receipt of neoadjuvant CRT at a CC, followed by surgery at an HVC) resulted in different outcomes compared to receiving all therapy at the HVC, as opposed to identifying differences between CCs.
Furthermore, results of the current study may be difficult to generalize due to inherent characteristics in the study environment, again warranting future study from others to improve generalizability.
Conclusions
In this analysis of patients with EC undergoing multimodality therapy including surgery at a HVC and CRT at HVCs and CCs, no significant difference in pCR or ypN+ rate was observed nor were there significant differences in the rate of minimal histopathologic response (TRG3). These results support continued coordination of patient care within geographic regions to optimize patient outcomes.
Footnotes
Author Note
Major findings from this study were presented at the 2023 ACRO Summit on March 16, 2023.
Ethical Considerations
This study was reviewed and approved by the Institutional Review Board at Moffitt Cancer Center.
Author Contributions
Conceptualization: AJS, SEH, JMP, Data Collection: PS, SC, JA, MA, Data Analysis: PS, SC, AJS, Resources: SEH, JMP, Writing of initial manuscript: PS, SC, AJS, Critical review and editing of manuscript: ALP, JB, JF, MS, JF, JMP, SEH, Supervision: AJS, SEH, Visualizations: PS, SC, Final review and approval of manuscript: PS, SC, JA, MA, ALP, JB, JF, MS, JF, JMP, SEH, AJS.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Sarah Hoffe is a member of the Editorial Board of Cancer Control and did not participate in the editorial handling or peer review process of the article.
Categories
Oncology, Radiation Oncology.
