Abstract
Neoadjuvant chemotherapy was originally designed to convert inoperable cancer. Nowadays, this concept has expanded since it can also offer the possibility to evaluate markers of response such as pathological complete response (pCR) with possible implications in long-term prognostic outcomes. A substantial body of literature tried to evaluate the ability of pCR to fulfill the conditions required to establish a preliminary endpoint, such as pCR, as a surrogate for the final endpoint, the overall survival (OS) but no systematic reviews have been performed yet. In this review we systematically analyzed the prognostic role of pCR in various cancers (breast, gastro-oesophageal, rectal, ovarian, bladder, lung) in which neoadjuvant treatment is a standard of care, evaluating articles published in the English language of phase III or phase II randomized controlled trials and meta-analyses. Since the continue development of immunotherapy in earlier stage, it has also been considered the impact of tumor-infiltrating lymphocyte on pCR.
Keywords
Introduction
Neoadjuvant chemotherapy (NACT) was originally designed to convert inoperable cancer in operable ones. 1 This concept has now expanded since it can also offer the possibility to evaluate markers of response as pathological complete response (pCR) with implications in long-term prognostic outcomes. Moreover, in case of perioperative treatment, the lack of response to neoadjuvant treatment can help to modulate the type of chemotherapy regimen in adjuvant setting.
In 1998, the National Surgical Adjuvant Breast and Bowel Project (NSABP) began enrollment of the B-18 trial, which randomly assigned women with early-stage breast cancer to 4 cycles of doxorubicin and cyclophosphamide given either pre- or post-operatively. 2 Overall survival (OS) was not improved, but the effects of chemotherapy on the primary tumor resulted interesting. pCR was seen in 13% of women treated preoperatively, and these women seemed to have improved disease-free survival (DFS) and OS. Subsequent studies confirmed the correlation between pCR and improved outcomes in many cancer types including rectal, esophageal, and bladder tumors.3,4
Due to the strong association between pCR and survival, in clinical trial drafting many thought that the improvements in the pCR rate could be considered a surrogate for improvement in survival.
In May 2012, the US Food and Drug Administration (FDA) started to support the use of pCR as a surrogate endpoint underlying that the improvements in pCR are “reasonably likely to predict clinical benefit” defined by FDA as clinically and statistically significant improvement in event-free survival (EFS), DFS, or OS.5-8 A substantial body of literature tried to evaluate the ability of pCR to be a surrogate for the final endpoint of OS. However, the prognostic role of pCR in early stage epithelial solid tumors have not been systematically summarized yet. Neoadjuvant treatment represents the standard of care in various solid tumors, and pCR is reached in variable percentage with standard treatments according to tumor histology and site (Figure 1). In the last years, several new treatments have been tested trying to improve the percentage of pCR on surgical specimen and, in particular, the survival. In this review we analyzed the prognostic role of pCR in various cancers, taking in consideration articles published in the English language of phase III or phase II randomized controlled trials and meta-analyses. Table 1 summarized the rate of pCR after neoadjuvant treatments in randomized clinical trials and Table 2 the rate of pCR after neoadjuvant treatments in non-randomized clinical trials and meta-analysis. Since the continue development of new immunological treatments in earlier stage, it has also been considered the role of tumor-infiltrating lymphocyte (TILs) on pCR in different solid tumors. Table 3 summarized the role of the presence of TILs in tumor tissue on pCR after neoadjuvant treatments in the different solid tumors considered. Expected pathological complete response in different solid tumors with standard approved treatments. Rate of Pathologic Complete Response After Neoadjuvant Treatments in Randomized Clinical Trials. Abbreviations: adjuvant, chemotherapy (ACT); cisplatin), neoadiuvant chemotherapy (NCT); delayed, primary surgery (DPS); disease-free, survival (DFS); immediated, primary surgery (IPS); intention, to treat (ITT); major, pathological response (MPR); MVAC, (methotrexate; N, number; overall, survival (OS); pathological, complete response (pCR); radical, cystectomy (RC); vinblastine, doxorubicin. Rate of Pathologic Complete Response After Neoadjuvant Treatments in Non-Randomized Trials and Meta-Analysis. Abbreviations: adjuvant chemotherapy (ACT), chemotherapy (CT); distant recurrence (DR), event-free survival (EFS); 5-year disease free survival (DFS), 5-year overall survival (OS); local recurrence (LR), major pathological response; (MPR), neoadjuvant-chemoradiotherapy (NCRT); neoadjuvant chemotherapy (NACT), pathological complete response (pCR); radical cystectomy (RC), recurrence-free survival (RFS); total neoadjuvant treatment (TNT). Role of the Presence of Tumor Infiltrating Lymphocytes in Tumor Tissue On Pathologic Complete Response After Neoadjuvant Treatments in Different Solid Tumors. Abbreviations: BC, breast cancer; EFS, event-free survival; HER-2, human epidermal growth factor receptor 2; IS, immunoscore; LPBC, lymphocyte-predominant breast cancer; N, number; NACT, neoadjuvant chemotherapy; NCRT, neoadjuvant chemo-radiotherapy; OR, odds ratio; pCR, pathological complete response; SCC, squamous cell carcinoma; TNBC, triple negative breast cancer.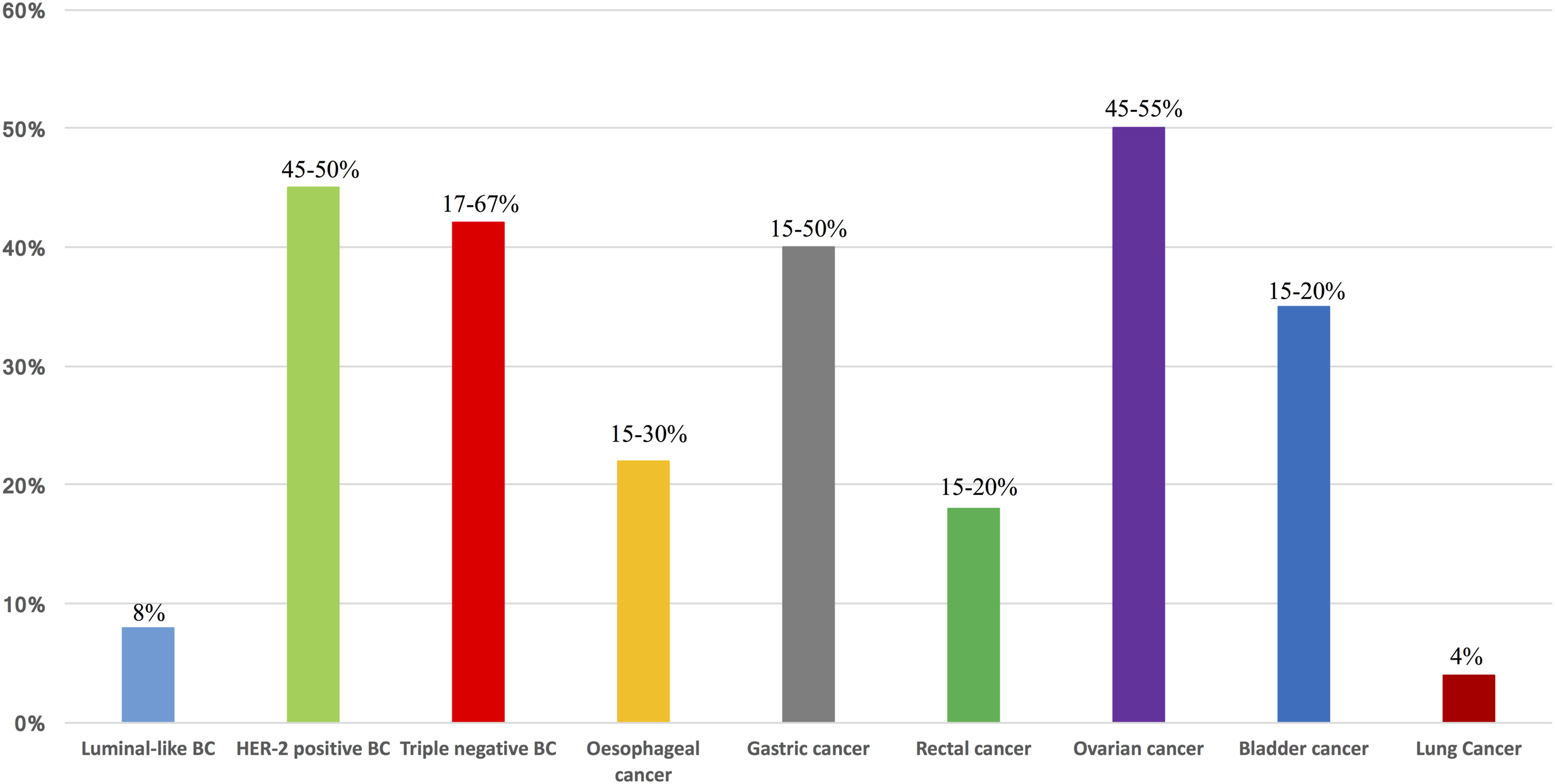



Pathologic Complete Response in Breast Cancer
Female breast cancer is the most common diagnosed cancer with 2.3 million of new cases (11.7% of total cases); it is in fifth position for deaths recorded with 685,000 (6.9%). 9
The pCR percentage is variable, depending on tumor subtype and the regimen of chemotherapy used. Several studies described that patients who achieve pCR have better long terms outcomes, such as DFS and OS.
10
A recent pooled analysis compared many definitions of pCR and investigated their role in predicting risk of recurrence or death.
11
The definitions reported are reported in Figure 2. Currently used definitions of pathological complete response in breast cancer after neoadjuvant treatment and surgical intervention.
The analysis included 7 prospective trials with patients with operable and non-operable primary breast cancer receiving chemotherapy with anthracyclines and taxanes. Patients with Human Epidermal Growth Factor Receptor 2 (HER2)-positive tumors received trastuzumab added to chemotherapy; and, in case of estrogen-receptor (ER)-positive and progesterone-receptor (PgR)-positive disease, an adjuvant endocrine treatment was performed. A total of 6377 patients received NACT; during a median follow-up of 46.3 months, 1466 relapses and 775 deaths were recorded. Stage ypT0 ypN0 was diagnosed in 955 patients (15.0%), ypTis ypN0 in 309 (4.8%), ypT0/is ypN+ in 186 (2.9%), ypT1mic ypN0/+ in 478 (7.5%), and ypT>1mic ypN0/+ in 4449 patients (69.8%). Patients with ypT0 ypN0 tumors experienced better DFS compared with patients with ypTis ypN0 tumors (HR = 1.74; P< .001) and showed a better OS (HR = 1.41; P < .166). Their prognosis was also better than patients with ypT0/is ypN+ in terms of DFS (HR = 3.18; P< .001) and OS (HR = 4.05; P < .001) or ypT1mic ypN0/+ tumors in terms of DFS (HR = 2.33; P < .001) and OS (HR = 2.34; P < .001). Patients with stage ypT0/is ypN + tumors recorded the worst DFS and OS. DFS was highest for ypT0 ypN0 cases and decreased for ypT0/is ypN0, ypT0/is ypN0/+, and ypT0/is/mic ypN0/+ ones. However, pCR was not a predictor for DFS and OS in subgroups associated with lower proliferation, lobular type, grade 1, ER-positive an PgR-positive status. In contrast, pCR was prognostic for DFS and OS in ductal and other histologic subtypes, grade 2 or 3 tumors, ER-negative and PgR-negative status. In luminal-A tumors pCR had no prognostic impact, while in HER2-positive and triple negative breast cancer (TNBC) showed a significant prognostic impact. Moreover, in presence of tumors with poor prognosis, the achievement of pCR gives a prognosis similar to luminal-A tumors. Cortazar and collegues published a meta-analysis of 12 international randomized neoadjuvant trials, in which investigated the potential role of pCR as a surrogate endpoint for long-term outcomes (EFS and OS). 12 Anthracycline-based and taxane-based regimes were adopted, with the adding of trastuzumab in HER2-positive breast cancer. 11955 patients were included in the analysis. Overall frequency of pCR was low: 22% of patients achieved ypT0/is stage after treatment, 18% ypT0/is ypN0 stage, and 13% ypT0 ypN0 stage; and the association between pCR and breast cancer was greater for TNBC and HER2-positive subtypes. In case of the achievement of pCR, the benefit was higher for patients with total eradication of breast and axillary lymph nodes. Within HER2-positive group, the advantage was higher for hormone receptor negative than positive ones, and with the addition of trastuzumab. The pCR was positively associated with EFS and OS for high grade hormone receptor positive/HER2-negative tumors; moreover, the association was stronger in patients with high-grade tumor than in those with low-grade ones. These results were confirmed in HER2-positive tumors independently of hormone receptor status, with a greater benefit for hormone receptor negative patients. Berruti and collegues subsequently performed a meta-regression of 29 trials that included 14641 women treated with neoadjuvant therapy. However, the results showed a minimal association between the effect of the treatment on pCR and the effect on EFS or OS. 13
Pathologic Complete Response in Luminal Breast Cancer
Luminal-A is diagnosed in 50-60% of all breast cancer and, in this context, after NACT, pCR rate is reported in 8% of patients; the survival benefit is less than other breast cancer subtypes. 14 Considering the good prognosis of these tumors, neoadjuvant treatment is indicated only for locally advanced disease. The adoption of neoadjuvant endocrine therapy (NET) for ER-positive hasn’t demonstrated a great benefit in this context. 15 Clinical practice reserves neoadjuvant endocrine therapy for frail patients or unsuitable for surgery or chemotherapy.
ETNA trial was a phase III open label, in which patients with HER2-negative breast cancer, were randomly allocated in 1:1 ratio to paclitaxel (349 patients), or nab-paclitaxel (346 patients). 16 The 2 drugs were given with an anthracycline regimen plus cyclophosphamide, or fluorouracil, epirubicin and cyclophosphamide. The primary endpoint was the pCR rate. The intention-to-treat analysis revealed that the improved pCR rate after nab-paclitaxel was not statistically significant (22.5%) compare to paclitaxel (18.6%; OR .77, p: .19). Patients affected by luminal B-like tumor recorded 13.9% of pCR with nab-paclitaxel treatment vs 10% with paclitaxel regimen.
Pathologic Complete Response in HER2-Positive Breast Cancer
HER2 belongs to the epidermal growth factor receptor family. It is a transmembrane glycoprotein with tyrosine kinase activity. Gene amplification is the primary mechanism for HER2 overexpression and the main predictor to anti-HER2 treatments. Adding trastuzumab to chemotherapy, a humanized monoclonal antibody against extracellular domain of HER2, significantly increased pCR in the context of neoadjuvant treatment in HER2-positive breast cancer. 17 Pertuzumab, a humanized monoclonal antibody against the dimerization domain of HER2, rises significantly pCR rate when added to trastuzumab in neoadjuvant setting.
NeoSphere is a phase II open label trial, in which 417 treatment-naïve patients with early-stage, locally advanced HER2-positive breast cancer were randomized (1:1:1:1) to receive 4 cycles of trastuzumab plus docetaxel, pertuzumab and trastuzumab plus docetaxel, pertuzumab and trastuzumab or pertuzumab plus docetaxel. 18 After surgery, patients received 3 cycles of FEC (fluorouracil, epirubicin and cyclophosphamide), and trastuzumab to complete 1 year of treatment. The primary endpoint was pCR. Patients who received pertuzumab, trastuzumab and docetaxel have a great improvement in the primary end-point (45.8% of patients), compared to patients who received only trastuzumab plus docetaxel (29%). At 5-years analysis progression free-survival (PFS) was greater in trastuzumab plus pertuzumab and docetaxel group (85% of patients) than in the trastuzumab plus docetaxel arm (81%). 19 In the exploratory subgroup analyses, patients who achieved total pCR had improved PFS regardless the regimen of treatment.
The NeoALTTO is a phase III trial in which 455 patients affected by HER2-positive early-stage breast cancer were randomized in a 1:1:1 ratio to receive lapatinib or intravenous trastuzumab or a combination of lapatinib plus trastuzumab in neoadjuvant setting. 20 After 6 weeks of anti-HER2 treatments, patients received weekly paclitaxel for further 12 administrations. As adjuvant treatment, patients received 3 cycles of FEC treatment and subsequently, the same anti-HER2 regimen received before surgery for a total of 34 weeks. The primary endpoint was pCR and it resulted significantly higher in the group who received lapatinib and trastuzumab (51.3%) than in the group who received trastuzumab alone (29.5%). No significant differences in pCR rate between the lapatinib alone group and the trastuzumab alone were recorded. Even if NeoALTTO trial was not powered to detect significant differences in survival outcomes, women with pCR after NACT had significantly better EFS, as well as post-surgery breast cancer relapse, second primary malignancy occurrence or death without recurrence than women without pCR. 21
A recent meta-analysis including a total of 36 studies for a total of 5768 patients evaluated the role of pCR in early stage HER-positive breast cancer. 22 Overall, the improvement in EFS for pCR vs non-pCR was significant (HR = .37, range .32-.43, P< .005). The advantage was greater for patients with hormone receptor-negative disease (HR = .29) than hormone receptor-positive tumors (HR = .52).
Pathologic Complete Response in Triple Negative Breast Cancer
TNBC is defined by the absence of ER, PgR and HER2-receptors. 23 This group shows the highest distant metastases rate and lowest OS of all breast cancer subtypes. Independently from surgery and adjuvant treatment, half of primary TNBC confined to breast and lymph nodes recurs as metastatic disease, in particular in visceral organs. In this context, pCR plays an important role as a surrogate marker for survival outcomes since TNBC is characterized by higher pCR rates than other types of breast cancer. 23 The predictive value of RCB was investigated by Symmans and colleagues in a prospective clinical of NACT. 23 In the triple negative cohort, 10-year relapse-free survival rates were 86%, 81%, 55% and 23%, for residual cancer burden (RCB) 0, RCB I, RCB II, RCB III, respectively, where RCB 0 is equivalent to pCR, while RCB III is associated to no response or tumor progression.
A recent meta-analysis tried to quantify the association between EFS/OS and pCR and to correlate long-term survival outcomes to pCR status. 24 A total of 25 studies were included and different neoadjuvant regimens were considered (anthracycline-based, taxane-based, and platinum plus docetaxel). The pCR rate ranged from 16.7% to 67%. The HR for EFS was reported in 19 studies, and ranged from .06 to .33. OS was reported in 17 studies, and ranged from HR = .06 to HR = .51. Results showed that pCR was related to big improvement in EFS [HR = .24] and OS [HR = .19]: 5-year EFS for patients with pCR was 86% whereas it was 50% respectively for patients who have not obtained pCR. The 5-year OS for patients with 92% and 58% for patients without. Authors conclude that the achievement of pCR was associated with a significant reduction of the risk of progression, recurrence, or death (about 76%), and with lower risk of death.
Moreover, TNBC is associated with an increased deoxyribonucleic acid (DNA) - repair defect cancer cells, caused by germline mutations in genes as BRCA 1/2, PALB2, or by somatic homologous recombination deficiency (HRD). Platinum salt-based chemotherapy induces DNA damage by cross-linking DNA strands leading to the cessation of replication with apoptosis of the tumor cell. Platinum salts have been included among the treatment of TNBC exploiting the fact that TNBC tumors have a limited DNA repair capacity in comparison with other BC histotypes. GeparSixto trial evaluated the efficacy of the addition of carboplatin in neoadjuvant treatment in triple negative and HER2-positive breast cancer patients. 25 Women with stage II-III breast cancer treated with paclitaxel and doxorubicin were randomized in a 1:1 ratio to receive or not carboplatin (296 and 299 patients, respectively). Patients in the HER2-positive group received also lapatinib plus trastuzumab. The primary endpoint was pCR. In the carboplatin group, 129 patients (43.7%) met the primary endpoint compared to 108 patients (36.9%) that not received carboplatin. In the group of patients with TNBC, 84/158 (53.2%) achieved a pCR with the addition of carboplatin compared with 58/157 (36.9%) that did not receive carboplatin. HRD was detected in 136 (70.5%) cases and resulted associated with younger patients age, node negative disease at diagnosis, and higher family risk for developing breast and ovarian cancer. 26 These patients showed higher pCR rates than HR-non deficient cases. The benefit was greater in the carboplatin group patients with a 2 times higher recorded improvement in pCR. At a median follow-up of 46.5 months, patients with TNBC and HRD recorded a significantly higher DFS with the adding of carboplatin (HR = .456, P= .008) and also OS was higher in patients with HRD compared to HR non-deficient tumors (HR = .320, P= .002). Patients who received carboplatin had an improvement in DFS of about 10% independently of the HRD status. The BrighTNess trial was designed to evaluate the addition of a PARP inhibitor, veliparib, plus carboplatin or carboplatin alone to standard NACT in TNBC. 27 This is a phase III randomized, double blind, placebo-controlled trial. 634 patients with a stage II-III were randomized, in 2:1:1 ratio, to receive 4 cycles of paclitaxel plus: carboplatin plus veliparib or carboplatin plus veliparib placebo or carboplatin placebo plus veliparib placebo. All groups were received doxorubicin plus cyclophosphamide for 4 cycles to complete the neoadjuvant regimen. Primary end-point was pCR. The proportion of patients who achieved pCR was higher in the paclitaxel, carboplatin and veliparib group than in patients who received paclitaxel alone (168/316 [53%] vs 49/158 [31%], P < .0001), but no differences were reported compared to patients who received paclitaxel plus carboplatin (92/160 [58%], P = .36). A post-hoc, exploratory analysis comparing the frequency of patients who had a pCR in the paclitaxel plus carboplatin group vs the paclitaxel alone group showed a great improvement by the addition of carboplatin. Moreover, 47/92 patients (51%) with BRCA mutation achieved a pCR vs 262/542 (48%) without BRCA mutation. Keynote 522 trial is a phase III study, in which patients with untreated stage II-III TNBC were randomized in a 2:1 ratio to receive NACT with 4 cycles with paclitaxel and carboplatin plus pembrolizumab every 3 weeks or placebo (390 patients). 28 Subsequently, patients received 4 cycles of pembrolizumab or placebo, and doxorubicin-cyclophosphamide or epirubicin-cyclophosphamide. After definitive surgery, patients were treated with adjuvant pembrolizumab or placebo every 3 weeks for up to 9 cycles. The co-primary endpoints were pCR and EFS. At the first interim analysis, pCR recorded was 64.8% in the pembrolizumab-chemotherapy group (260/401 patients) and 51.2% (103/201 patients) in the placebo-chemotherapy group (P < .001). After a median follow-up of 15.5 months, the HR for disease progression favored the pembrolizumab–chemotherapy group (HR = .63). The benefits in the pembrolizumab-chemotherapy group were consistent across all subgroups, regardless PD-L1 status. In the PD-L1 positive subgroup, the percentage of pCR was 68.9% (230/334 patients) in the pembrolizumab-chemotherapy subgroup and 54.9% (90/164 patients) in the placebo-chemotherapy group. In the PD-L1 negative population pCR was 45.3% (29/64 patients) among pembrolizumab-chemotherapy group and 30.3% (10/33 patients) among who received placebo plus chemotherapy. At a median follow-up of 39 months, PDL-1-positive patients treated with pembrolizumab had a 33% reduced risk of recurrence compared to placebo group (HR = .67; 95% CI, .49-.92). 29 Patients in the PD-L1–negative group who received the pembrolizumab regimen had a reduced risk of 52% reduced risk of recurrence compared to the placebo-chemotherapy group (HR = .48; 95% CI, .28-.85). The 36-month EFS rates were 84.5% vs 76.8%, respectively.
Bevacizumab is a monoclonal antibody against vascular endothelial growth factor 1, that support the growth and maintenance of tumor vasculature necessary for survival and metastasis. 30 The CALGB40603 trial is a 2 × 2 factorial, open label, phase II trial, which examines the addition of carboplatin and/or bevacizumab to conventional NACT in TNBC. Primary endpoint was pCR. A total of 443 patients with stage II-III TNBC received in 1:1:1:1 ratio weekly paclitaxel (for 12 administrations), followed by doxorubicin plus cyclophosphamide every 2 weeks for 4 cycles, and were randomized to concurrent three-weekly carboplatin for 4 cycles and/or bevacizumab every 2 weeks for 9 administrations. Results showed that, thanks to carboplatin administration, the pCR rates improved from 41% to 54% (OR = 1.71; P = .0029), while the bevacizumab treatment improved pCR rate (52% vs 44%) but the results did not reach statistical significance (OR = 1.36; P = .057). Patients who received both regimens had the highest pCR rate (60%).
Among new immunomodulatory therapies, the Impassion 031 trial evaluated the efficacy and safety of atezolizumab or placebo in combination with NACT. 31 A total of 333 patients affected by stage II-III TNBC were randomized in 1:1 ratio to receive chemotherapy plus atezolizumab or placebo every 2 weeks. Chemotherapy regimen included weekly nab-paclitaxel for 12 administrations followed by doxorubicin and cyclophosphamide every 2 weeks for 8 weeks. Co-primary endpoints were pCR in the intention-to-treat population and in PD-L1 positive patients. pCR was recorded in 95 patients (58%) treated with atezolizumab plus chemotherapy vs 69 (41%) with placebo plus chemotherapy (P= .0044). In the PD-L1 positive population, pCR was documented in 53 patients (69%) in the atezolizumab plus chemotherapy group vs 37 (49%) in the placebo plus chemotherapy 1. Poggio F. et al performed a meta-analysis of 9 randomized clinical trial in which they evaluated the platinum-based vs platinum-free NACT in TNBC. 32 The primary objective was the activity of the treatments in terms of pCR. In addition, a further analysis was conducted to assess the benefit of the platinum-based treatment according to germline BRCA mutational status. Overall, 938/2109 (44.5%) patients achieved a pCR after neoadjuvant treatment, 545/1046 (52.1%) in the platinum-based chemotherapy group and 393/1063 (37%) in the platinum-free chemotherapy group (OR = 1.96, P < .001). In 7 RCTs that used anthracycline/taxane-based chemotherapy, 797/1698 (46.9%) patients achieved pCR after neoadjuvant treatment, 461/856 (53.9%) in the platinum-based chemotherapy group and 336/842 (39.9%) in the platinum-free chemotherapy group (OR = 1.85, P < .001). Among BRCA-mutated patients, 54/96 (56.2%) achieved a pCR, 29/50 (58%) in the platinum-based chemotherapy group and 25/46 (54.3%) in the platinum-free chemotherapy group (OR = 1.17, P= .711). No significant differences in EFS (HR = .72, P= .094) nor in OS (HR = .86, P= .651) were observed.
In conclusion, in the context of BC pCR has an important prognostic significance in particular in HER-2 positive and TNBC if patients are treated with standard regimen. The role of pCR on patients’ prognosis with new immunotherapy agents still remains to be better elucidate.
The Role of TILs in Early Breast Cancer
TILs are mononuclear immune cells, which infiltrate tumor tissue and represent an individual immunological response. 33 Breast cancer is not considered an immunogenic tumor, but the association between TILs and outcomes is widely studied. TILs are observed in highly proliferative tumors (TNBC and HER2-positive breast cancer), and in these tumors they are considered to predict better outcomes with higher pCR rate. Gao Zh et al. performed a meta-analysis in which evaluated the prognostic and predictive value of TILs in different molecular subtypes of breast cancer. 34 Thirty-three studies including 18170 patients were analyzed. The meta-analysis showed that high TIL expression was associated with an improvement in pCR rates after NACT in patients with HER2 enriched molecular subtype (OR = 1.137, P < .001), and TNBC (OR = 1.120, P < .001). However, high TILs expression was not significantly associated with high pCR rates after NACT in patients with luminal molecular subtype of breast cancer (OR = 1.154, P= .460). The meta-analysis confirmed that high TILs rate was associated with a great improvement in terms of DFS in patients with HER2 enriched molecular subtype (HR = .940, P = .003) and TNBC (HR = .907, P < .001); while the presence of high TILs rate didn’t improve the DFS rate for luminal subtype (HR = .998; P= .840). The benefits derived by high TILs rate in terms of OS were recorded for patients with HER2 enriched breast cancer (HR = .910, P < .001) and TNBC (HR = .869, P < .001). In contrast, high TILs rate recorded poor OS results in patients with luminal subtype (HR = 1.077, P= .012). In the NeoSphere trial, Bianchini et al evaluated the contribution of the immune system to pCR in BC after neoadjuvant chemotherapy. 35 In the trial, intratumoral (TumTIL) and stromal (StrTIL) tumor-infiltrating lymphocytes were scored. Patients’ samples were than divided into 3 groups: lymphocyte-predominant breast cancer (LPBC) if there was more than 50% of either stromal or intratumoral lymphocytic infiltration; low lymphocyte infiltration (LowTIL) if there was less than 5% of either stromal or intratumoral lymphocytic infiltration. The intermediate group included all other cases. The data were available in 243 patients and 17% were classified as LPBC and 14% as lowTIL. The results of the statistical analysis showed that the association with pCR was not significant. As exploratory analysis, the 3 more similar arms (pertuzumab plus trastuzumab, docetaxel plus trastuzumab and docetaxel plus pertuzumab) were combined and results showed that the group with low-TIL had the lowest rate of pCR (4.3%), compared to intermediate-TIL and LPBC (26.9% and 26.7%, respectively). A secondary analysis of the NeoALTTO trial tested the hypothesis that higher levels of TILs would be associated with better clinical outcomes. 36 The results described that pCR rates increased when levels of stromal TILs were greater than 5%, regardless of treatment group. Patients with higher TILs levels and without pCR had similar EFS to those who achieved pCR; on the other hand, women who did not achieve pCR and had lower levels of TILs were characterized by poor prognosis. Denkert et al evaluated TILs as secondary endpoint of GeparSixto trial. 37 One hundred forty-two patients (24.5%) were lymphocyte-predominant breast cancer (≥ 60% of either intra-tumoral or stromal TILs). LPBC phenotype was recorded in 53 (19.9%) of the 266 HER2 positive tumors and 89 (28.3%) of the TN tumors. LPBC had a pCR rate of 59.9% compared with 33.8% for non-LPBC tumors (P< .001). The percentage of stromal TILs was significantly associated to pCR (OR = 1.22, P< .001).
Pathologic Complete Response in oesophageal Cancer
Criteria Used to Evaluate Response to Neoadjuvant Treatments in Surgical Specimen For Gastroesophageal, Rectal, Ovarian and Bladder Cancer.

In conclusion, a pCR after esophagectomy following NACT for locally advanced carcinoma does not implicates complete healing, but a major pathological response achievement has an impact on LRR and subsequently DFS and OS.
The Role of TILs in Early Oesophageal Cancer
In neoadjuvant setting, TILs predict increased pCR and extended long-term survival rates after NCRT in esophageal SCC (ESCC). 56 Qian and colleagues analyzed 164 patients with locally advanced ESCC (T1-4aN0-3M0) who underwent NCRT or definitive chemoradiotherapy. In the first group (48 cases), patients received radiotherapy (total dose 40 Gy) concomitant to 4 cycles of chemotherapy (docetaxel plus cisplatin). In the second group (116 cases), patients received definitive chemoradiotherapy (60 Gy) concomitant to 4 to 6 cycles of chemotherapy (docetaxel plus cisplatin). In the NCRT group, a pCR rate of 31.25% was reached (15/48 patients), with a significant better survival compared to patients with low pCR. Moreover, in the NCRT group, tumor remission and TILs during NCRT correlated with postoperative pathologic responses. Excellent remission (ER) during chemoradiotherapy and TILs > 60% during chemoradiotherapy predicted pCR. DFS rate of patients with ER and LPE was significantly longer, while it was not significantly different for patients treated with NCRT or definitive CRT.
Pathological Complete Response in Gastric Cancer
In early stage gastroesophageal cancer, surgical resection of the primary tumor and regional lymph nodes is the most effective approach; however tumor often recurs after curative resection. 57 Perioperative chemotherapy or chemoradiotherapy give an improvement in the long-term outcomes. According to the College of American Pathologists’ cancer reporting protocols, the evaluation of the response on surgical specimen after neoadjuvant treatment is performed according to the modified Ryan Scheme for Tumor Regression Score (Table 4). Thanks to the results of the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) trial, that demonstrated a survival benefit for patients with perioperative chemotherapy with epirubicin, cisplatin and infused fluorouracil (ECF) when compared with surgery alone, pre- and postoperative chemotherapy became in Europe the standard of care in resectable gastroesophageal cancer. 58 Patients with stage II or higher tumors and no evidence of metastases were randomized. Chemotherapy was administered for 3 cycles preoperatively and 3 cycle postoperatively. The primary end-point was OS. PCR was evaluated according to the pathologic regression Mandard TRG system (Table 4). 59 The 5-year OS rate for patients with TRG 1-2 who receive chemotherapy was 58.8%, and 28.9% for TRG 3-5. Both pCR and lymph node status significantly correlated with OS in patients who received chemotherapy (TRG 3-5: HR 1.94; P = .021; lymph node metastases: HR 3.63; P < .001). The FLOT trial is a controlled, open-label, phase II-III study in which 716 patients with a clinical stage cT2 or higher, nodal positive stage, or both, resectable tumors, with no evidence of distant metastases, were randomized to receive 3 pre-operative and 3 post-operative 3-week cycles of epirubicin and cisplatin plus fluorouracil or capecitabine (ECF/ECX; control group), or 4 pre-operative and post-operative 2-week cycles of docetaxel, oxaliplatin, leucovorin and fluorouracil (FLOT; experimental group).60,61 The primary outcome of the phase II study was the rate of pathological complete tumor regression according to Becker classification (Table 4), 62 while the primary outcome of the phase III trial was OS. 62 In the phase II trial, FLOT group was associated with significantly higher proportion of patients achieving pathological complete regression than the control arm (16% vs 6%, P= .02). In the phase III trial, while baseline cT and cN stages were similarly distributed among the groups, a higher proportion of stage ypT1 tumors were in the FLOT group than in the ECF/ECX group (88 [25%] of 356 vs 53 [15%] of 360 patients; p: .0008). A higher proportion of tumors were stage ypN0 in the FLOT group than in the control 1 (174 [49%] of 356 vs 146 [41%] 360 patients; p: .025). In the intention to treat population, more patients achieved margin-free (R0) resection in the FLOT group than in the ECF/ECX group (301 [85%] vs 279 [78%]; p: .0162). The median OS was 35 months in the ECF/ECX group and 50 months in the FLOT group (HR .77; p: .012). The estimated OS at 2, 3, and 5 years were 59%, 48%, and 36% in the control group, as compared with 68%, 57% and 45% in the experimental group. A meta-analysis evaluated the prognostic value of pCR after preoperative therapy and 21 studies and 5 types of digestive cancer were included (esophageal cancer (EC), esophagogastric junction AC (EGJAC), gastric AC (GAC), rectal cancer (RC) and pancreatic cancer (PC)). 63 The results showed that pCR was significantly correlated with better OS (HR = .50, P < .001) and DFS (HR = .49, P < .001) for patients who achieved pCR than for those who did not achieve pCR. In the subgroup analysis with EGJAC/GAC patients, the correlation of pCR with OS was significant (HR = .38, p: .02).
In conclusion, although pCR is associated with improved OS and DFS, about 20% to 40% of patients have an early recurrence. Most (73%) recurrences are within the first 2 years and actually, few studies have examined the risk factors for recurrence after a pCR. Most (72%) of the recurrences described are distant metastases, implicating a need for a better systemic tumor control.
The Role of TILs in Gastric Cancer
To date, there are only a few meta-analyses assessing the relationship between TILs and prognosis of gastric cancer.64,65 However, a recent meta-analysis reports that, compared with low TILs density, high TILs density is significantly associated with better OS in patients with gastric cancer (HR = .68, 95% CI 0.52 to .87, P = .003). 66 In addition, TILs resulted associated with lower depth of invasion, less lymph node involvement and earlier TNM stage. No association between TILs and pCR are described in literature.
Pathological Complete Response in Rectal Cancer
The standard treatment for locally advanced rectal cancer includes NCRT, surgery and adjuvant chemotherapy. 67 Downstaging after neoadjuvant treatment is a known prognosticator of survival. NCRT gives a pCR between 10% and 25%. Patients achieving pCR exhibit a low rate of local and distant recurrence and better OS and DFS compared to patients without pCR. According to international guidelines, patients with clinical stage II or III (T3/4 N0, or node positive) are treated by NCRT followed by total mesorectal excision and postoperative adjuvant chemotherapy with fluorouracil and oxaliplatin; this approach confers an excellent local control, with distant recurrence rate more common than local recurrence. 68 The evaluation of the response on surgical specimen after neoadjuvant treatment is performed according to the modified Ryan Scheme for Tumor Regression Score (Table 4). RAPIDO trial is a multicentre, open-label, randomized, controlled phase III trial which included 912 patients with a primary, locally advanced rectal adenocarcinoma, classified as high risk (cT4a or cT4b, extramural vascular invasion, clinical nodal stage cn2, involved mesorectal fascia, or enlarged lateral lymph nodes). 69 Patients were randomized (1:1 ratio) to receive short-course radiotherapy (5 × 5 Gy over a maximum of 8 days) followed by 6 cycles of CAPOX chemotherapy or 9 cycles of FOLFOX4, followed my mesorectal excision (experimental group). Patients in the standard of care group received 28 fractions of 1.8 Gy/day (up to 50.4 Gy) or 25 fractions of 2.0 Gy/day (up to 50 Gy) with concomitant capecitabine followed by total mesorectal excision. Then adjuvant chemotherapy with 8 cycles of CAPOX or 12 cycles of FOLFOX4 was performed. The primary endpoint was 3-year disease-related treatment failure, defined as the first event of locoregional recurrence, distant metastasis, new primary colorectal cancer, or treatment-related death. The results showed that the probability of disease-related treatment failure was 23.7% in the experimental group vs 30.4% in the standard of care group (HR = .75; p: .019). As regarding pCR, 120/423 (28%) patients in the experimental group reached a pCR compared with 57/398 (14%) cases in the standard of care group (OR 2.37; P < .001).
UNICANCER-PRODIGE 23 is a phase III, open-label, randomized, controlled trial which included 461 patients affected by staged cT3 or cT4 rectal adenocarcinoma. 70 Patients were assigned (1:1 ratio) to receive neoadjuvant chemotherapy with 6 cycles of FOLFIRINOX, followed by chemoradiotherapy, surgery and adjuvant chemotherapy or to standard of care as chemoradiotherapy, surgery and adjuvant chemotherapy. Chemoradiotherapy consisted of 50 Gy/total over 5 weeks and concurrent capecitabine whereas adjuvant chemotherapy was performed with modified FOLFOX6 or capecitabine for 3 months in the experimental group and for 6 months in the standard group. Patients in the control group received 12 cycles of modified FOLFOX6 or 8 cycles of capecitabine, and patients in the neoadjuvant chemotherapy group received 6 cycles of modified FOLFOX6 or 4 cycles of capecitabine. The primary endpoint was DFS at 3 years. At a median follow-up of 46.5 months, 3-year DFS rates were 76% in the neoadjuvant chemotherapy group and 69% in the standard-of-care group (HR = .69; p: .034). Rates of ypT0N0 were significantly increased in the neoadjuvant group compared with the standard-of-care group, and tumor regression was improved (P= .003). In the experimental group was recorded a ypN0 rate of 82.5% vs 67.4% in the control group, and ypT0N0 was 27.8% vs 12.1%, respectively.
A systematic review and meta-analysis of 7 studies including 2416 patients evaluated the pCR rate for standard therapy and TNT. 71 The prevalence of pCR was 29.9% in the TNT group and 14.9% in the NCRT plus adjuvant chemotherapy. TNT was associated with improved pCR (OR = 2.44). Only 3 studies presented data on DFS, and pooled analysis showed significantly higher DFS in patients who received TNT (OR = 2.07). Another recent meta-analysis evaluated 16 studies involving 3363 patients underwent NCRT followed by interval proctectomy. 72 The aim was to evaluate the oncological outcomes of patients with a pCR after neoadjuvant therapy. The primary outcomes of interest were the rates of local recurrence, distant recurrence, 5-year OS and 5-year DFS. A total of 1263 patients had a pCR and 2100 had an incomplete or no response. Twelve studies reported a 5-year local recurrence rate of 0% among patients who had pCR, and patients who achieved a pCR had a reduction of developing local failure around 75% (OR = .25; p:0.002). Distant failure or metastasis was recorded in 8.7% of patients who had a pCR, and patients who achieved a pCR had a reduction of 77% of the probability to have distant failure (OR = .23; P < .001). The 5-year OS rate for patients with a pCR was 90.2%; these patients had a 3.3-fold OS advantage compared with incomplete responders (OR = 3.28; P= .001). A 5-year DFS rate was 87% in case of achieving pCR. This group was 4.3 times more likely to be disease-free at 5 years than non-responders (OR = 4.33; P < .001). Bonnetain et al performed a pooled analysis of 2 randomized rectal cancer trials trying to evaluate the effect on pCR and OS of chemoradiation vs radiation alone before surgery.73,74 Results showed that pCR increased from 3.7% to 11.2% (P= .001) but no effects on OS were evident (5-year OS: 65.9% vs 66.3%, respectively).
Lastly, the RTOG 0247 study was a randomized phase II trial which included patients with locally advanced rectal cancer to receive preoperative radiation and capecitabine with irinotecan or oxaliplatin. 75 Irinotecan arm was then closed due to a lower pCR rate of this regimen even if further follow-up showed that the irinotecan arm had a superior survival (4-year OS: 85% v 75%; P= .05). 76
In conclusion, CRT followed by TME is the standard of care since 2004 and there have been no successful trials that have improved metastasis-free or OS since 1997. Local recurrence risk is no longer the major issue in locally advanced rectal cancer patients treated with NCRT and TME since 25-30% of patients develop distant metastases. However, the results of the recent PRODIGE and RAPIDO trials showed an improvement in the 3-year DFS (+7%) with 28% of pCR so that for T4, locally advanced rectal tumor, TNT regimen has now became the new standard of treatment.
The Role of TILs in Early Rectal Cancer
Kong et al performed a meta-analysis including 25 trials with the aim of determining the prognostic and predictive power of TILs in intestinal cancers. 77 Seven trials included 727 patients with locally advanced rectal cancer subjected to NCRT treatment; the levels of CD8+ cells were a significant predictor of tumor regression grade after CRT treatment. There was a consistent correlation between the density of each T-cell subset with TRG, DFS and OS.
Pathologic Complete Response in Ovarian Cancer
In Europe and North America, ovarian cancer remains the most common cause of gynecological cancer-related death. 78 Immediate primary surgery (IPS) followed by adjuvant platinum-based chemotherapy has been considered the standard treatment for women with advanced-stage disease at diagnosis. However, NACT followed by delayed primary surgery (DPS) may be considered an alternative treatment option for women in whom IPS is contraindicated. In this context, the goal of cytoreductive surgery is to complete the removal of macroscopic disease and the histopathologic assessment post-NACT provide a surrogate for response to treatment and information regarding the chemosensitivity of the disease.79,80 The International Collaboration on Cancer Reporting (ICCR) recommended the assessment of tumor regression of ovarian carcinomas treated with NACT using the Chemotherapy Response Score (CRS) proposed by Bohm et al (Table 4). 49 The score is based on the evaluation of the architecture and microenvironment of high grade serous cancer deposits in a section of omentum. Several studies demonstrated that CRS is reproducible and has prognostic significance.81-83 However, despite these growing evidences, the prognostic role of pCR in ovarian cancer remains controversial. A study by Le et al demonstrated that the residual tumor burden after NACT has an independent negative prognostic role, particularly when the residual disease is located in the upper abdomen. 84
Another study by Petrillo et al demonstrates that pCR is an uncommon event in ovarian cancer patients receiving NACT (6% of the cases) and is associated with a longer survival compared with women showing no pCR. 85 The presence of International Federation of Gynecology and Obstetrics (FIGO) stage IV was the only baseline variable showing a significant inverse correlation with pCR and a trend toward a negative predictive role of carcinomatosis for pCR was also observed.3 randomized, phase III trial validated the NACT therapeutic approach in FIGO stage III–IV ovarian cancer reporting non-inferior survival between IPS followed by adjuvant chemotherapy vs NACT followed by DPS. The first study by Vergote et al randomized a total of 632 patients affected by stage IIIC or IV epithelial ovarian carcinoma, fallopian-tube carcinoma, or primary peritoneal carcinoma to receive IPS followed by platinum-based chemotherapy or to NACT platinum-based followed by DPS. 86 The residual tumor was < 1 cm in 41.6% of patients after IPS and in 80.6% of cases after DPS. The results of the statistical analysis showed that complete resection of all macroscopic disease was the strongest independent variable in predicting survival so that NACT followed by DPS resulted to be not inferior to IPS followed by chemotherapy. Complete resection of all macroscopic disease, whether performed as primary treatment or after NACT, remains the objective whenever cytoreductive surgery is performed. Another trial aimed to establishing whether NACT followed by DPS is superior to IPS. 87 This is a phase III randomized study enrolling 110 women who were randomly assigned (1:1 ratio) to undergo either IPS followed by adjuvant chemotherapy (standard arm), or NACT followed by DPS (experimental arm). Co-primary objectives were postoperative complications and PFS; secondary objectives were OS and quality of life (QoL). Despite different extension of surgery, rates of complete residual disease were similar between the groups (45.5% vs 57.7%; p: .206) but data analysis are still uncomplete. The CHORUS trial is a phase III, non-inferiority, randomized, controlled study which included women with stage III or IV ovarian cancer. 88 It aimed to establish whether the use of platinum-based NACT followed by DPS was an effective and safe alternative treatment regimen. A total of 550 women were randomly assigned (ratio 1:1) either to undergo primary surgery followed by 6 cycles of chemotherapy, or to 3 cycles of NACT then surgery followed by 3 more cycles of chemotherapy. Each 3-week cycle consisted of carboplatin plus paclitaxel, or an alternative carboplatin combination regimen, or carboplatin monotherapy. The primary endpoint was OS and it resulted 22.6 months in the primary-surgery group vs 24.1 months in primary chemotherapy 1. Unfortunately, no data on pCR are reported. Author concluded that in women with stage III or IV ovarian cancer, survival with primary chemotherapy is non-inferior to primary surgery. The ICON 8 was an international, multicentre, randomized, phase III trial that included 1566 women with newly diagnosed FIGO stage IC–IV epithelial cancer of the ovary, fallopian tube, or primary peritoneum. 89 Patients were randomly assigned (ratio 1:1:1) to receive intravenous carboplatin and paclitaxel (control group); intravenous carboplatin and dose-fractionated paclitaxel (group 2) or intravenous dose-fractionated carboplatin and paclitaxel (group 3) followed by DPS. The co-primary objectives were PFS and OS. The primary PFS analysis showed no significant differences between standard arm, group 2 and 3. A post-hoc analysis evaluated the RECIST and Gynecologic Cancer InterGroup (GCIG) CA125 responses. 90 Median PFS was 14.4 months for patients with a RECIST complete or partial response and 13.3 months for those with RECIST stable disease. Median PFS for women with a GCIG CA125 response was 13.8 months and 9.7 months for those without a GCIG CA125 response. Complete cytoreduction was achieved in 187/335 (56%) with a RECIST complete or partial response and 73/172 (42%) women with RECIST stable disease and in 290/576 (50%) women with a GCIG CA125 response and 30/101 (30%) women without a GCIG CA125 response. Authors concluded that RECIST and GCIG CA125 responses to NACT for epithelial ovarian cancer should not be used as individual predictive markers to stratify patients who are likely to benefit from DPS, and a patient should not be denied surgery based solely on the lack of a RECIST or a CA125 response. Recently, the results of the HIPEC ovaio trial were published. 91 This is a phase II study where 6 rather than 3 cycles of NACT with three-weekly paclitaxel plus cisplatin or carboplatin, followed by DPS and HIPEC, were adopted. A total of 56 patients with stage III primary ovarian cancer and peritoneal carcinomatosis were assigned to 6 cycles of platinum and taxane-based NACT. Of these, two had progression, 8 underwent palliative surgery, and 46 had DPS and HIPEC. A pCR was observed in 9 patients. Among the 46 patients who completed the treatment protocol, 29 had no macroscopic residual tumor. The 5-year median OS was 36 months. In 46 patients who received DPS and HIPEC, the 5-year median OS was 53 months, and the 5-year median PFS was 23 months. Completeness of cytoreduction, peritoneal cancer index and FIGO stage resulted significant prognostic factors. As concerning the proper number of NACT cycles that should be performed, an ongoing study will try to evaluate the efficacy and safety of reducing 1 cycle of NACT. 92 This study is a prospective, multicentre, open-label, randomized phase III trial which will included a total of 298 patients with advanced ovarian, fallopian or primary peritoneal cancer and will randomly assigned women to either 3 (control group) or 2 cycles of NACT (experimental group). After the NACT, DPS with maximal cytoreduction will be performed and then adjuvant CT for 3 or 4 cycles for a total of 6 cycles. The primary objective is PFS, and among the secondary endpoints, tumour response after NACT, tumour response by positron emission tomography-computed tomography after NACT and success rate of optimal cytoreduction. Authors hypothesize that reducing 1 cycle of NACT will contribute to more resection of visible tumours despite 10% reduction of optimal cytoreduction, which could improve survival.
Immunotherapy had a strong growth in recent years: ovarian cancer has long been considered a poorly immunogenic neoplasm, but evidence of mechanisms of immune evasion, spontaneous tumor regressions and some responses to immunotherapy have been described in early phase clinical trials. 93 Data on single agent immunotherapy in platinum-sensitive ovarian cancer are lacking so, in the last few years, more attention has been paid to new strategies with a special focus on combination therapies with anti-angiogenic agents, PARP-inhibitors, chemotherapy or unconventional immunotherapeutic agents, such as adoptive cell therapy or therapeutic vaccine. However, also in this context, robust data on early stage disease are lacking with early phase trials focusing on relapse or platinum refractory stage with contradictory results.
In conclusion, ACT is not inferior to primary cytoreductive surgery for patients with stage IIIC or IV ovarian carcinoma. No significant advantages of NACT or primary debulking surgery have been observed in clinical trial with respect to survival, adverse effects, QoL, or postoperative morbidity or mortality. The administration of immunotherapy is possible only in the context of clinical trials waiting for the results of the ongoing phase II–III trials.
The Role of TILs in Early Ovarian Cancer
Relatively few data exists regarding the effects of chemotherapy on anti-tumor immunity in ovarian cancer patients. A work by Polcher and colleagues reported that NACT with carboplatin and docetaxel induced an increase of CD4þ, CD8þ, and Granzyme Bþ TIL in post-NACT tumors without any change in FoxP3þ TIL. 94 Peng et al reported that CD8þ TIL may have an increase in patients undergoing NACT. 95 Finally, Bohm et al postulated that a significant decline in stromal FoxP3þ cells and an increase in Th1/cytolytic T cell gene signatures were be evident in patients with good responses to NACT. 96 Collectively, these studies seem to indicate that chemotherapy enhance CD8/Th1/cytolytic TIL and reduce Tregs. However, it is unclear whether these changes may result in complete, prognostically favorable TIL patterns with effects on major immunosuppressive pathways. It is well known that BRCA 1/2 mutated high grade serous ovarian cancer shows a higher mutational load and increased expression of TILs, PD-1 and its ligand, PD-L1. Furthermore, patients with T-cell-rich tumors experience longer progression-free and OS, while immune evasion mechanisms are associated with poor survival. 97 One I/II study by Konstantinopoulos et al evaluated niraparib and pembrolizumab. 98 Interestingly, ORR was 23% without any significant difference in biomarkers selected populations (25% in BRCA-mutated and 24% in BRCA wild-type respectively). The second study combined olaparib and durvalumab in platinum-sensitive relapsed BRCA mutated ovarian cancer. 99 Overall, 23/32 patients had objective responses with olaparib plus durvalumab, including 6 (19%) complete responses and a 12-week DCR of 81%. Furthermore, 6 patients had complete responses, 17 a partial response resulting in an ORR of 72%; 3 patients had SD, bringing the DCR to 81%.
Pathologic Complete Response in Urothelial Bladder Cancer
Bladder cancer is the ninth most frequent cancer worldwide, with annual incidence of 430000 new cases. About 25% of patients are diagnosed in advanced stage as muscle-invasive bladder cancer (MIBC), while 20-30% of non MIBC (NMIBC) ones will become invasive during the follow up period. 100 Radical cystectomy (RC), bilateral pelvic lymph node dissection, and cisplatin-based NACT have become the standard of care for the treatment of MIBC. 101 One of the most active chemotherapy regimen is the combination of methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC); clinical trials showed similar efficacy results also for gemcitabine plus cisplatin (GC) doublet. 102 RC alone for MIBC is associated with a 50% risk of developing distant metastases within 2 years and up to a 60% risk of death within 5 years. Cisplatin-based NAC has been shown to increase OS with an absolute benefit of 5% and decrease bladder cancer–specific mortality. 103 The residual tumor regression on surgical specimen is evaluated as reported in Table 1. Several randomized clinical trials and meta-analyses demonstrated a survival benefit for patients with MIBCs who received NACT prior to cystectomy compared with surgery alone with, in particular, better outcomes for patients who had pCR. 104 In this context, the use of NACT may allow the downstaging of the primary tumor with the evaluation of the response and the patient’s prognosis. 105 In the SWOG 8710 study, a pivotal neoadjuvant phase III trial, Grossmann and colleagues demonstrated that, in the MVAC arm, a pCR rate of 38% was recorded at the time of RC vs 15% of pT0 in the RC group at surgery. 106 In addition, the 5 year-OS rate was higher in the pT0 patients compared with the no-pT0 cases, regardless of chemotherapy received. A meta-analysis by Petrelli et al tried to determine whether pCR after NACT is associated with a better outcome in MIBCs. 107 All patients underwent cisplatin-based NACT and RC. A total of 13 trials were included (886 patients analyzed) with a pCR rate of 28.6%. Moreover, patients who achieved pCR had a relative risk (RR) for OS of .45 (P < .00001). Authors concluded that patients who achieved pCR after NACT have a better OS and RFS than patients without pCR. Recently, the results of a phase III trial to compare the efficacy of dose-dense (dd)-MVAC or GC in perioperative (neoadjuvant or adjuvant) setting were published. 108 A total of 500 patients were randomized to receive either 6 cycles of dd-MVAC or 4 cycles of GC. The primary endpoint was PFS and results are expected in mid-2021. As secondary endpoints, pCR was observed in 84 (42%) and 71 (36%) patients, respectively (P = .2). An organ-confined status (<ypT3pN0) was obtained in 154 (77%) and 124 (63%) patients, respectively (P = .001). Authors concluded that a higher local control rate was observed in the dd-MVAC arm (P = .021). An alternative treatment to RC for selected patients is represented by the trimodal treatment (TMT), an organ preservation strategy based on trans urethral bladder resection (TURB) followed by radical RT and concomitant chemotherapy. 109 Results of clinical trials showed that oncological outcomes after TMT and RC may be similar so that a salvage surgery approach should be performed only when partial response or local recurrence are evident. 110 A complete response to induction therapy with concurrent chemotherapy/RT has typically been defined as negative urine cytology, absence of visible tumor and negative biopsies at cystoscopy. Achieving a complete response to induction therapy is required to avoid salvage cystectomy and has been associated with improved DFS and OS after TMT. A complete response rate of 70% for patients with T2-T4a disease treated with TMT has been described. 111 Factors that may impact the likelihood of achieving a complete response after TMT and that should be considered include: completeness of TURBT, tumor stage, hydronephrosis, multifocality, presence CIS, and baseline bladder function. A recent Italian study by Francolini et al including 10 clinical trials compares TMT vs RC. The work developed GRADE (Grades of Recommendation, Assessment, Development and Evaluation) Recommendation by the Italian Association of Radiotherapy and Clinical Oncology (AIRO) for treatment of MIBC. 112 Results showed a salvage cystectomy rate and a pCR rate after TMT of 12% and 72-77.5%, respectively.
In this context, several variables are connected with the probability of pCR. A recent study prospectively evaluated the impact of smoking. 113 A total of 167 patients were enrolled and smoking was significantly associated with advanced age, worse ECOG performance status, and decreased pCR to NACT.
Immune checkpoint inhibitors (ICI) belong to the therapeutic armamentarium for patients with metastatic urothelial carcinoma. Recently, clinical trials have reported results of neoadjuvant ICI alone or in combination before radical cystectomy in patients with resectable bladder cancer. 114 Necchi et al reported the results of THE PURE-01 trial, a single-arm phase II study evaluating pembrolizumab as neoadjuvant treatment before cystectomy in patients with MIBC. 115 Results showed that pCR rate was 37% (42/114 patients). Updated results in a larger cohort, which included also histology variants, showed a lower activity of pembrolizumab with a reported pCR of 16%. 116 The prognosis was favorable across all subgroups, with the exception of ypN + cases (n = 21), in which a 24-month RFS of 39.3% was evident. Moreover, a statistically significant event-free survival (EFS) benefit was observed in those who reached pCR. Powles et al reported the results of THE ABACUS trial, a single-arm phase II study, investigating 2 cycles of atezolizumab before RC in patients with operable MIBC not candidate for NACT. 117 The pCR rate was 31% (37% in PD-L1 positive cases) and 1-year RFS was 79%. An association with pCR was also seen in presence of preoperative activated CD8+ T cells within the tumor and cytotoxic T cell gene signature. By contrast, preoperative TMB and the high number of DDR gene alterations was not associated with oncological outcomes and no significant correlations between preoperative PD-L1 expression on immune or tumor cells and pCR were evident. Gao et al evaluated the combination of neoadjuvant durvalumab plus tremelimumab in cisplatin ineligible patients or in those refusing cisplatin-based NACT. 118 The recorded pCR rate was 37.5% and the downstaging rate was 58.3%. Kaimakliotis et al reported the results of a phase Ib/II trial evaluating neoadjuvant GC plus pembrolizumab. 119 The primary endpoint was pathological non–muscle-invasive rate that resulted 60% with no correlations with baseline PD-L1 score. Final analysis reported a 2-year disease specific survival of 86% and a OS of 84%. Recently, Gupta et al reported the results from the Bladder Cancer Signal Seeking Trial-1 (BLASST-1), a single-arm phase II trial evaluating the combination of GC plus nivolumab in the neoadjuvant setting. 120 The primary endpoint was pathological response defined as non–muscle-invasive rate (<pT2N0). Results showed that 21/41 (66%) patients reached pathological response, while 14/40 (35%) had a pCR (pT0). No influence of preoperative PD-L1 status was seen. Moreover, Cathomas et al reported the results of an interim analysis of a single-arm phase II trial evaluating 4 cycles of neoadjuvant durvalumab combined with GC before RC, followed by 10 cycles of adjuvant durvalumab. 121 Authors observed a pCR rate of 33% (10/30 cases). Furthermore, Rodriguez-Moreno et al recently reported the preliminary results of the trial evaluating the impact of neoadjuvant durvalumab plus olaparib prior to RC. 122 The study reported a pCR rate of 50% (10/20 patients). Currently, several phase III randomized trials are investigating perioperative immunotherapy in patients with localized MIBC. 114
In conclusion, in the context con urothelial bladder cancer, an increasing role in neoadjuvant setting will be played by ICIs. Investigators from early-phase trials have shown comparable rate of pCR to historic NACT series in one-third of the patients after 2 or 3 injections of single-agent ICI with also satisfactory results with the combinations ICI plus chemotherapy.
The Role of TILs in Early Urothelial Bladder Cancer
Identifying validated biomarkers to predict responses to neoadjuvant treatment prior to RC should be important for treatment decisions. In the ABACUS trial, activated CD8+ T cells in the tumor have been described to be correlated with the immune response in patients who received neoadjuvant atezolizumab. 117 Additionally, a strong correlation with pCR was seen in presence of high CD8 expression within the tumor and with the eight-gene cytotoxic T cell transcriptional signature (IFN, CXCL9, CD8A, GZMA, GZMB, CXCL10, PRF1, and TBX21). Similarly, other transcriptional immune signatures (such as IFNg, IFNa, and inflammatory response) resulted significantly associated with pCR in the PURE01 study in which pCR was reached in 19 patients (54.3%) with a PD-L1 CPS ≥10% and in only 2 patients (13.3%) with a PD-L1 CPS <10%. 116 Moreover, recent gene expression profiling studies revealed that tumors with higher levels of pre-existing immune infiltration had good responses to neoadjuvant pembrolizumab but not to platinum-based NACT. In contrast with clinical trials with anti-PD1/PD- L1 monotherapy, complete response to ipilimumab plus nivolumab in the NABUCCO trial was independent of baseline CD8+ presence or T-effector signatures. 123 However, results from the DUTRENEO trial did not demonstrate enhanced efficacy in patients treated with durvalumab plus tremelimumab on the basis of tumor inflammation signature. 124
Pathological Complete Response in Lung Cancer
Lung cancer is the second most common cancer worldwide and the leading cause of cancer death. 9
At diagnosis about 20-25% of patients have a resectable disease, and 30-55% who undergo curative surgery have recurrence. 125 The absolute difference in 5-years RFS and OS between NACT and surgery alone is about 5-6%. A small part of patients has a pCR rate (about 4%). Checkmate 816 is an open-label, phase III trial in which 505 patients with a stage Ib-III resectable Non-Small-Cell-Lung Cancer (NSCLC), were randomized in 1:1 ratio to receive nivolumab plus platinum-doublet or platinum-doublet chemotherapy alone before surgery. A third group received nivolumab plus ipilimumab closed enrollement during the trial. After surgery, patients received up to 4 cycles of adjuvant chemotherapy, radiotherapy, or both. There were 2 primary end-points: EFS and pCR (0% residual viable tumor cells in the primary tumor and sampled lymph nodes). The median EFS was 31.6 months with nivolumab plus chemotherapy vs 20.8 months with chemotherapy alone (HR = .63, P= .005). pCR rate was higher in nivolumab plus chemotherapy arm than in the chemotherapy alone one, 36.9% vs 8.9%, respectively anf EFS was higher in patients with a pCR with a median EFS not reached among patients with pCR. In patients without a pCR, median EFS was 26.6 months with nivolumab plus chemotherapy and 18.4 months with chemotherapy alone (HR = .84). Nadim trial is an open-label phase II study in which 46 patients with stage IIIa NSCLC received, as NACT, nivolumab, paclitaxel, and carboplatin AUC6, for 3 cycles. 126 Primary endpoint was PFS, while pCR was recorded as secondary endpoint. At the median follow-up of 24 months, 35/41 were progression free (PFS = 77.1%); 26 (63%) had pCR and 96.2% were progression free at 18-24 months.
Neostar study is a phase II trial in which 44 patients affected by resectable NSCLC were randomized to receive neoadjuvant nivolumab or nivolumab plus ipilimumab, followed by surgical and adjuvant standard of care therapy. 127 The primary endpoint was major pathological response (MPR), defined as ≤10% viable tumor in resected tumor specimens.
The nivolumab plus ipilimumab arm met the primary endpoint, recording 38% MPR rate (8/21), while 22% MPR rate (5/23) in the nivolumab arm. The combination arm reached a higher pCR rate than mono-immunotherapy arm, 38% vs 10%, respectively. Waser NA et al published a systematic literature review and meta-analysis including 33 studies in which patients received platinum-based chemotherapy regimen ± radiotherapy. 128 For OS by pCR status, HR was .49; for EFS by pCR status, the HR was .50. Another meta-analysis by Cao C et al analyzed the immunotherapy neoadjuvant approach (durvalumab, ipilimumab, nivolumab, pembrolizumab, atezolizumab, sintilimab, camrelizumab) in patients affected by resectable NSCLC. 129 548 patients received immunotherapy, of whom 507 underwent surgery: 52% achieved a major pathological response, 24% a complete pathological response, and 20% reported a complete pathological response of both primary lesion as well as the sampled lymph nodes.
In conclusion, in the context of lung cancer treatment, pCR remains poor with traditional chemotherapy-based treatment. ICIs combined with chemotherapy seem to improve pCR but the impact on prognosis remains to be evaluated and many phase III clinical trial are ongoing.
The Role of TILs in Lung Cancer
To date, no studies assess the relationship between TILs and prognosis of lung cancer nor the association between TILs and pCR.
Conclusions
In different solid tumor, the survival of patients with pCR after neoadjuvant treatment is significantly longer than unresponsive patients. In the next years an increasing role in neoadjuvant setting will be played by ICIs and in this context several clinical trials are ongoing with possible practice changing results. However, the role of the improvement in pCR on long-term outcomes must always be demonstrated in well-conducted clinical trial. Even if we have identified precise inclusion criteria of the studies for this review and we selected only studies that met the inclusion criteria, a limitation of this review remains its narrative nature with a possible subjective method in the selection of the studies and influences in drawing conclusions due to selection bias of the studies and subjective weighing of the studies chosen.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ricerca Corrente Funding scheme of the Italian Ministry of Health.
