Abstract
Introduction
Colorectal cancer (CRC) is common among older patients, who face an increased risk of postoperative complications from malnutrition and systemic inflammation. The serum Albumin-Neutrophil-to-Lymphocyte Ratio Score (ANS), indicative of nutritional and inflammatory status, is a potential predictive tool in various cancers. This study evaluated the predictive value of preoperative ANS for postoperative survival and complications in older CRC patients.
Methods
We retrospectively analyzed 182 patients aged 65 and over who underwent curative-intent CRC surgery between 2018 and 2020 at a Taiwanese medical center. Based on pretreatment-ANS, patients were categorized into three groups: ANS 0, high albumin and low neutrophil-to-lymphocyte ratio (NLR); ANS 1, low albumin or high NLR; and ANS 2, low albumin and high NLR. Overall survival (OS), length of hospital stay (LOS), postoperative complications, and health-related quality of life (HRQoL) were analyzed and compared across groups.
Results
Patients in the ANS 2 group exhibited significantly poorer OS, with an adjusted hazard ratio of 2.90 (95% confidence interval [CI]:1.15-7.30, P = 0.024), compared to ANS 0. Additionally, they experienced a longer median LOS (14 vs 9 days, P < 0.001) and a higher risk of major postoperative complications (Odds ratio [OR] 3.74, 95% CI: 1.59-8.83, P = 0.003) and 30-day readmission rate (OR 5.04, 95% CI: 1.32-19.3, P = 0.018) than the ANS 0 group. ANS 2 patients reported worse HRQoL, particularly in mobility and maintaining purpose (P < 0.001 and P = 0.020, respectively), compared to ANS 0 patients.
Conclusion
Preoperative ANS significantly predicts survival, postoperative complications, and HRQoL in older CRC patients undergoing surgery, potentially guiding perioperative management.
Introduction
Colorectal cancer (CRC) ranks as the third most common malignancy and the second leading cause of cancer-related mortality worldwide. 1 In the United States, over 50% of CRC cases occur in older patients aged 65 years and above, resulting in approximately 35 000 deaths annually in this demographic. 2 In Taiwan, the median age at CRC diagnosis ranges from 66 to 68 years, indicating that half of the newly diagnosed patients are older. 3
Among older patients with CRC, 70% are diagnosed at a localized or regional stage, 4 where surgical resection remains the primary curative treatment. 2 However, postoperative morbidity following colorectal resection in older patients is estimated at around 30%, with complications including bleeding, infection, poor wound healing, bowel obstruction, and anastomotic leakage.5,6 These complications significantly impact overall survival.6,7
Old age in cancer patients is associated with increased morbidities, including higher treatment-related toxicity, more frequent hospitalizations, and a greater number of surgical complications.8,9 Malnutrition plays a significant role in the aging process of these patients. Studies suggest that approximately one-third of patients with CRC are at least moderately malnourished, 10 a condition exacerbated by gastrointestinal (GI) dysfunction, which is more prevalent in CRC patients than in those with non-GI tract cancers. 11 The prevalence of malnutrition is even more pronounced in older patients with GI tract cancers, reaching about 45%, roughly 1.5 times higher than in younger patients. 12 Malnutrition is linked to a 1.5 to 2-fold increase in cancer mortality.9,13 Older patients, particularly those with poor nutritional status such as sarcopenia, face higher rates of postoperative complications and mortality. 14 Therefore, preventing postoperative complications in older patients with CRC is crucial for improving survival and quality of life, particularly in those who are malnourished.
Systemic inflammation and malnutrition interact synergistically to drive frailty, impair wound healing, and shorten survival in older surgical patients. A host of blood-based indices, including the Neutrophil-to-lymphocyte ratio (NLR), 15 lymphocyte-to-monocyte ratio (LMR), 15 platelet-to-lymphocyte ratio (PLR), 15 Prognostic Nutritional Index (PNI), 16 Controlling Nutritional Status (CONUT) score, 17 and Glasgow Prognostic Score (GPS), 16 have been proposed for colorectal cancer. However, several practical limitations have curtailed their clinical adoption: (i) many require three or more laboratory parameters, 17 adding complexity and potential for missing data; (ii) some incorporate cholesterol or C-reactive protein that are not universally measured in preoperative work-ups16,17; and (iii) cut-off values vary widely across studies, reducing generalizability. 16 Serum albumin and the NLR stand out because they are inexpensive, routinely obtained, and reflect overlapping but biologically relevant processes. Albumin is traditionally viewed as a marker of nutritional status, but its levels are also influenced by systemic inflammation. In contrast, NLR reflects acute immune/inflammatory activation. Together, the composite ANS provides an integrated snapshot of vulnerability in older surgical patients.18,19 The composite Albumin–NLR Score (ANS) therefore provides an integrated snapshot of nutritional and inflammatory status with minimal additional testing burden. Evidence from gastric, esophageal, pancreatic, and breast cancers suggests that ANS yields prognostic discrimination.20–22 Nevertheless, no study has specifically validated ANS in older patients (≥65 y) undergoing curative colorectal resection, a population characterized by heightened vulnerability to both malnutrition and systemic inflammation. Furthermore, the association of ANS with quality of life in cancer patients has yet to be elucidated. This study aims to evaluate the effectiveness of preoperative ANS in predicting postoperative survival outcomes, complication rates, and quality of life in patients over 65 years old undergoing curative intent CRC surgery.
Methods
Patient Selection
We retrospectively enrolled 182 consecutive older patients from a prospective dataset who underwent radical surgery for CRC treatment between August 2018 and December 2020 at a medical center in Taiwan. The inclusion criteria were: (1) age ≥65 years; (2) histologically confirmed adenocarcinoma of the colorectum; and (3) receipt of radical surgery as a curative-intent treatment. Exclusion criteria were (1) prior CRC surgery, (2) emergency surgery, (3) simultaneous non-colorectal malignancy, (4) pre-existing chronic inflammatory or autoimmune disease, and (5) receipt of intravenous or oral albumin supplementation within 30 days before surgery.
All patients underwent radical colectomy with regional lymphadenectomy. Concurrent metastectomy was permitted for patients with M1a disease. Adjuvant chemotherapy for patients with poor prognostic factors (tumor rupture, positive surgical margin, regional lymph node metastasis, or M1a disease) was determined based on multidisciplinary tumor board discussion. All participants provided written informed consent. The study was approved by the Institutional Review Board of Chang Gung Memorial Hospital, Taiwan (IRB No. 201600916B0). This IRB approval covers a prospective institutional project (2016-2020) designed to enroll cancer patients receiving frailty assessment before the initiation of antitumor treatment. The current study represents a secondary analysis of that project, focusing on the prognostic impact of albumin and neutrophil-to-lymphocyte ratio in patients with colorectal cancer undergoing surgery.
Data Collection
Patient demographics (age, gender, marital status, educational level, preexisting comorbidities, Eastern Cooperative Oncology Group [ECOG] performance status, and American Society of Anesthesiologists [ASA] score) and laboratory data (albumin, NLR, and carcinoembryonic antigen [CEA]) were collected within 1 week before surgery. The reference ranges at our institution during the study period were: serum albumin 3.5 - 5.2 g/dL; absolute neutrophil count 1.5 - 7.0 × 109/L; absolute lymphocyte count 1.0 - 3.0 × 109/L; and CEA < 5 ng/mL. The Charlson Comorbidity Index (CCI) was calculated based on the patients’ preexisting comorbidities. Due to the exclusive focus on cancer patients and their age in this study, a modified CCI excluding the scores for patient age and cancer type was used.
Surgical information, including operative methods (open or laparoscopic), operative times, and intraoperative blood loss, was recorded by a well-trained research assistant immediately after surgery. Tumor characteristics, including tumor stage (according to the eighth edition of the pathological staging system issued by the American Joint Committee on Cancer), differentiation grade, and microscopic margins, were obtained from the final pathological report. Adjuvant chemotherapy prescription was recorded by retrospectively reviewing medical records.
Albumin and Neutrophil-Lymphocyte-Ratio Score
An albumin value higher or lower than the median (4.1 g/dL in this study) was assigned a score of 0 or 1 point, respectively.20–22 Similarly, a NLR value lower or higher than the median (2.6 in this study) was assigned a score of 0 or 1 point, respectively. Based on their albumin and NLR values, patients were categorized into three groups: ANS 0 (albumin ≥ 4.1 g/dL and NLR < 2.6), ANS 1 (albumin < 4.1 g/dL or NLR ≥ 2.6), and ANS 2 (albumin < 4.1 g/dL and NLR ≥ 2.6). The reporting of this study conforms to REMARK guidelines. 23
Health-Related Quality of Life (HRQoL)
All eligible patients were assessed for health-related quality of life (HRQoL) by a trained clinical assistant within 7 days prior to surgery and three months post-surgery. HRQoL was evaluated using the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire-ELD14 (QLQ-ELD14). 24 Patient responses were rated on a 4-point Likert scale and converted to a scale ranging from 0 (least symptoms) to 100 (most symptoms), with higher scores indicating poorer HRQoL. The QLQ-ELD14 comprises seven scales: mobility, worries about others, future worries, maintaining purpose, burden of illness, joint stiffness, and family support.
Survival Outcome and Postoperative Event
The primary outcome measure was the comparison of overall survival (OS) among the ANS groups. Overall survival time was calculated from the date of surgery until death or the last known date the patient was alive. All included patients were followed until their date of death or until December 31, 2023.
The secondary outcome measures included HRQoL and adverse surgical outcomes such as prolonged hospital stay, postoperative intensive care unit (ICU) stay, 30-day readmission, 30-day emergency room visit rate, and major surgical complications (Accordion severity grade 2 or higher). 25 The decision for postoperative ICU stay was made by the surgeon based on the patient’s intraoperative or postoperative condition.
Statistical Analysis
Basic patient demographic data are presented as frequencies (%) for categorical variables and as medians with ranges for continuous variables. The ANS groups were compared using the Kruskal-Wallis test for continuous and ordinal variables and the Chi-square or Fisher’s exact tests for categorical variables. A binary logistic regression model was used to estimate the odds ratio (OR) and 95% confidence interval (CI) for variables associated with postoperative events. Univariate Cox regression was employed to estimate the hazard ratio (HR) and 95% CIs for variables associated with overall survival (OS). The multivariate model was also adjusted for the same factors. For each outcome we fitted univariate models for every baseline variable and multivariate models including all covariates (age, Body mass index, ECOG performance, CCI, CEA, and tumor stage) with P < 0.10 in univariate testing plus clinically essential factors. Backward elimination (stay P < 0.10) yielded the final parsimonious model. Multicollinearity was assessed with variance-inflation factors and proportional-hazards assumptions were tested with Schoenfeld residuals. All statistical analyses were conducted using SPSS software (version 17.0; SPSS Inc., Chicago, IL, USA). Statistical assessments were two-sided, with a P-value of < 0.05 considered statistically significant.
Results
Patient Characteristics
Patient Characteristics
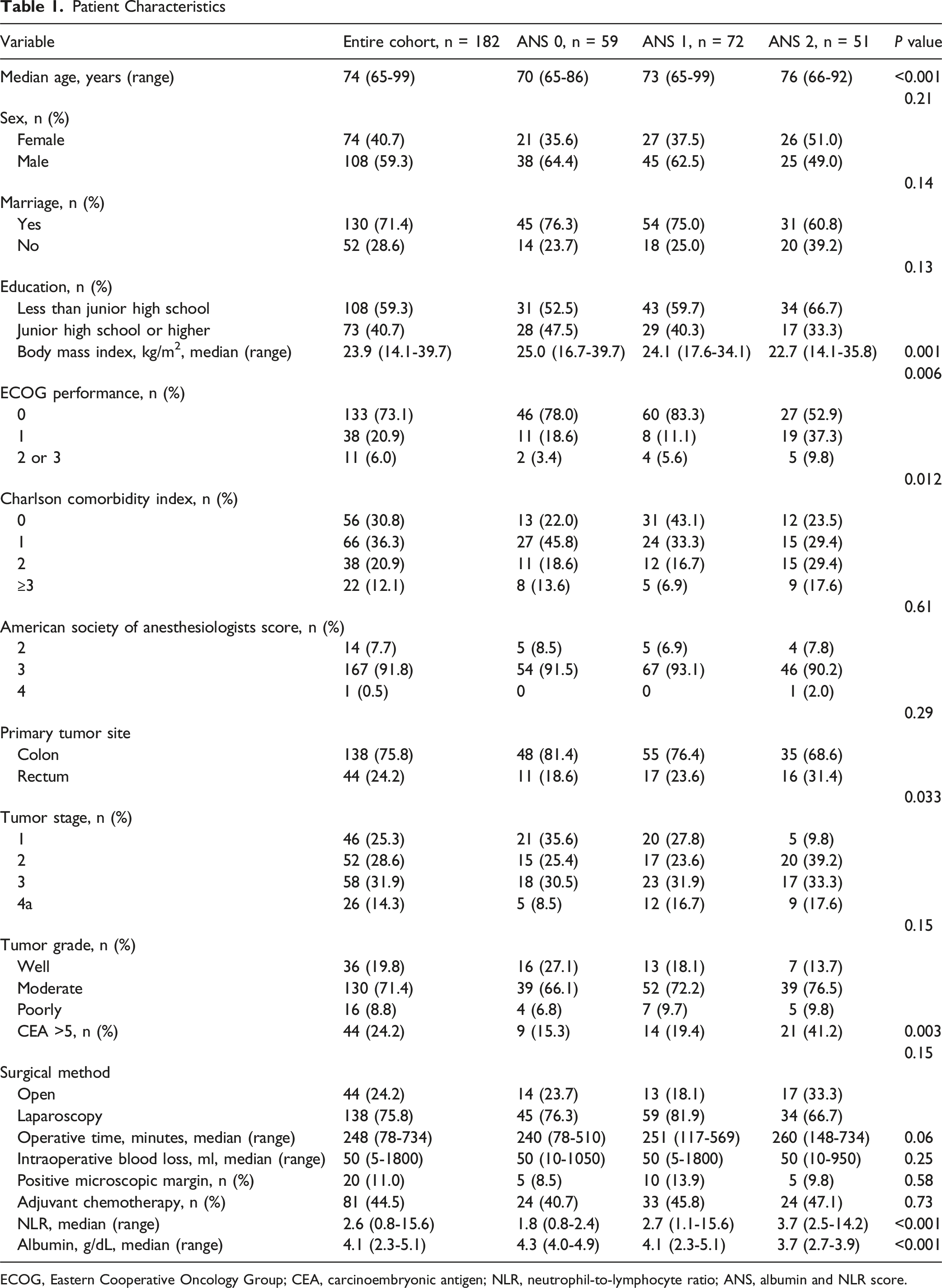
ECOG, Eastern Cooperative Oncology Group; CEA, carcinoembryonic antigen; NLR, neutrophil-to-lymphocyte ratio; ANS, albumin and NLR score.
Overall, 75.8% of the patients underwent laparoscopic surgery. The median operative time was 248 min (range: 78-734), with a median intraoperative blood loss of 50 mL (range: 5-1800).
Patients were allocated into ANS 0, 1, and 2 groups as follows: 59 (32.4%), 72 (39.6%), and 51 (28.0%), respectively. No statistical differences were observed among the ANS groups regarding sex, marital status, education, ASA score, primary tumor site, tumor grade, surgical method, operative time, intraoperative blood loss, microscopic margins, and the prescription of adjuvant chemotherapy. However, patients in the ANS 2 group were older, had lower body mass indices, poorer ECOG performance statuses, higher comorbidities, more advanced tumor stages, and a higher percentage of abnormal CEA values compared to the other two ANS groups.
Overall Survival
The median follow-up period was 40. 1 months (range: 2.4-59.2), during which 42 patients (23.1%) died. Mortality rates were 11.9%, 22.2%, and 37.3% for the ANS 0, 1, and 2 groups, and the corresponding 3-year survival rates were 89.2%, 76.8%, and 63.4%, respectively (Figure 1). The hazard ratio (HR) for overall survival (OS) was 2.04 (95% CI: 0.84-4.97, P = 0.12) when comparing the ANS 1 group to the ANS 0 group, and 3.96 (95% CI: 1.66-9.43, P = 0.002) when comparing the ANS 2 group to the ANS 0 group. In the multivariate analysis, the adjusted HR for OS was 1.63 (95% CI: 0.64-4.16, P = 0.31) for the ANS 1 group and 2.90 (95% CI: 1.15-7.30, P = 0.024) for the ANS 2 group, compared to the ANS 0 group. The full model C-index for OS was 0.78 and the C-index without ANS was 0.75. Overall Survival According to Different ANS Groups
Length of Stay
The median LOS in our patient cohort was 11 days (range: 4-84). The median LOS for the ANS 0, ANS 1, and ANS 2 groups was 9 days (range: 4-29), 11 days (range: 4-44), and 14 days (range: 7-84), respectively (Figure 2). There was no significant difference in LOS between patients in the ANS 0 and ANS 1 groups (P = 0.14). However, patients in the ANS 2 group had a significantly longer LOS compared to those in the ANS 1 group (P = 0.031) and those in the ANS 0 group (P < 0.001). Length of Stay According to Different ANS Groups
A total of 45 patients (24.7%) had an LOS greater than 14 days. The rates of an LOS greater than 14 days were 8.5%, 23.6%, and 45.1% for the ANS 0, ANS 1, and ANS 2 groups, respectively (Figure 3A). The ANS 2 group had a significantly higher risk of an LOS greater than 14 days compared to the ANS 0 group in both univariate analysis (OR 7.17, 95% CI: 2.84-18.1, P < 0.001) and multivariate analysis (adjusted OR 6.29, 95% CI: 2.40-16.5, P < 0.001). Univariate and Multivariate Analysis for Length of Stay (A), Grade 2 or Higher Postoperative Complications (B), 30-Days Readmission Rate (C), and Emergency Room Visiting Rate (D) Among Different ANS Groups
Postoperative Complications
Among the 182 patients, 68 (37.4%) experienced at least one major postoperative complication. The incidence of any complication increased with higher ANS scores: 23.7% in the ANS 0 group, 33.3% in the ANS 1 group, and 58.8% in the ANS 2 group (Figure 3B). Patients in the ANS 2 group had a 4.59-fold increased risk (95% CI: 2.02-10.4, P < 0.001) of major surgical complications in univariate analysis and a 3.74-fold increased risk in multivariate analysis (95% CI: 1.59-8.83, P = 0.003) compared to those in the ANS 0 group.
Major Postoperative Complications

aIncludes pulmonary edema, respiratory distress, or re-intubation.
bNew onset atrial fibrillation or supraventricular tachycardia that requiring treatment.
cIncluded pulmonary embolism or deep vein thrombosis.
The 30-day readmission rate (Figure 3C) and emergency room visiting rate (Figure 3D) were 7.1% and 13.2% in the entire cohort, respectively. The ANS 2 and ANS 1 groups had higher risks for 30-day readmission and emergency room visits compared to the ANS 0 group. The differences in the 30-day readmission and emergency room visit rates between the ANS 0 and ANS 2 groups were significant in both univariate and multivariate analyses.
Based on the distribution of the cohort, the sample medians (albumin 4.1 g/dL; NLR 2.6) were selected a-priori as pragmatic cut-points to stratify patients into ANS 0, 1, and 2. To test the robustness of this strategy, we performed a post-hoc time-dependent receiver-operating-characteristic (ROC) analysis for 3-year overall survival and grade ≥ 2 complications. Optimal thresholds were identified by the Youden index and by the maximal χ2 method; re-analyses using these thresholds are presented in Supplemental Table 1. Time-dependent ROC analysis yielded optimal cut-offs of 3.9-4.0 g/dL for albumin and 2.7-2.9 for NLR. The close agreement between our median-based cut-offs and the ROC-derived thresholds for albumin and NLR confirms that the results are not artifacts of the median-split approach.
HRQoL Outcomes
QLQ-ELD 14 Before and After Colectomy Based on ANS Group

n.s., not significance.
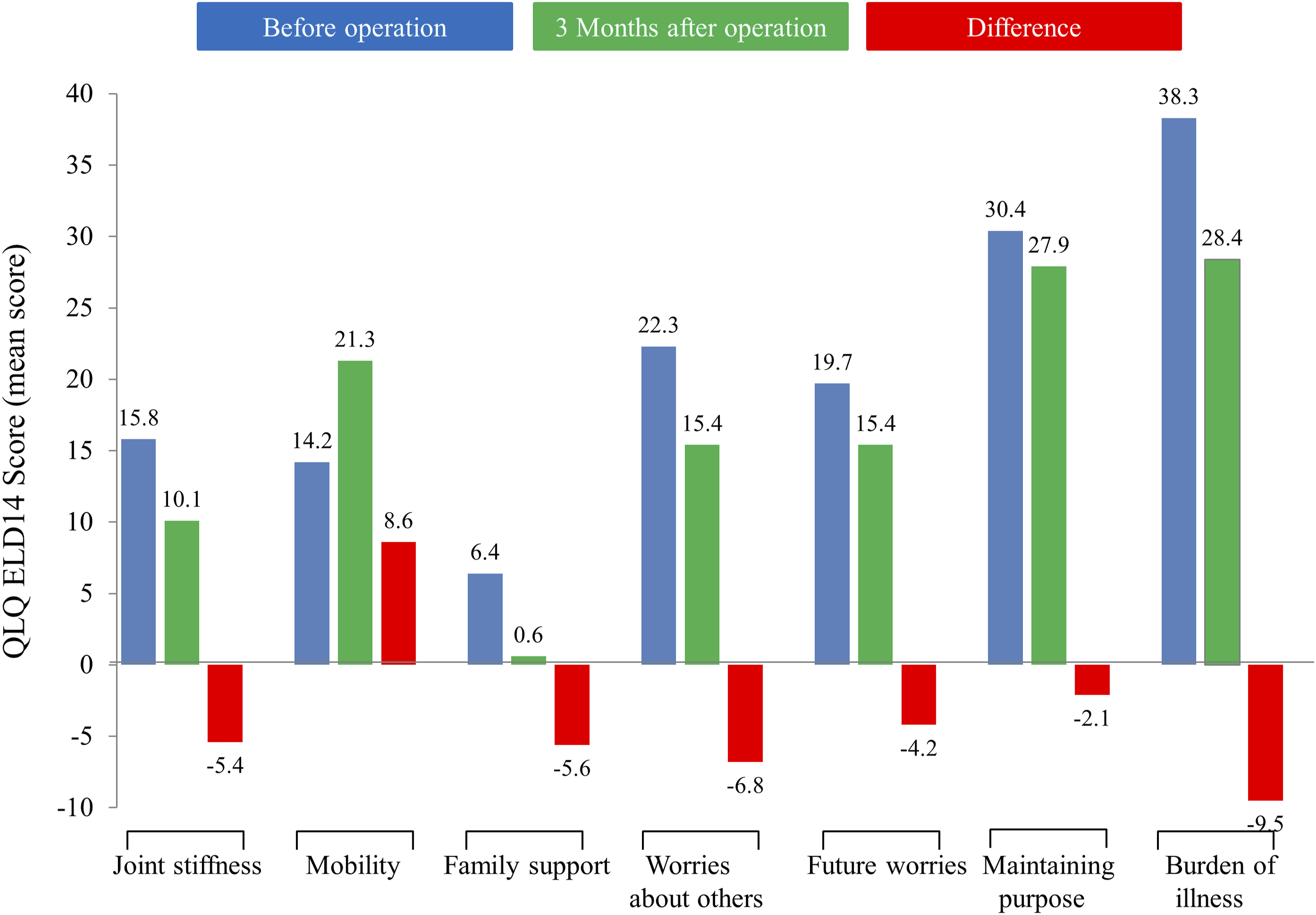
Severity of Each Symptom on the QLQ-ELD14 before the Operation and 3 months after the Operation
In terms of changes in severity for each QLQ-ELD14 scale before and after surgery among the entire cohort, six symptoms showed an improvement in HRQoL: burden of illness (decrease in mean score: −9.5), worries about others (decrease in mean score: −6.8), family support (decrease in mean score: −5.6), joint stiffness (decrease in mean score: −5.4), future worries (decrease in mean score: −4.2), and maintaining purpose (decrease in mean score: −2.1). However, the mobility symptom worsened (increase in mean score: +8.6) (Figure 5). Differences in the Severity of Each QLQ-ELD14 Scale before and after the Operation Among Different ANS Groups *P < 0.05; **P < 0.001
Preoperatively, patients in the ANS 2 group had significantly poorer QoL in terms of mobility and maintaining purpose compared to the other two ANS groups. At three months post-operation, patients in the ANS 0 group continued to exhibit better QoL in mobility compared to the other two ANS groups.
Comparative Performance of ANS, Albumin, NLR, and PNI
Albumin < 4.1 g/dL and NLR ≥ 2.6 each showed modest discrimination for 3-year overall survival (AUC 0.63 and 0.61, respectively), but both were significantly inferior to the composite ANS (AUC 0.71; DeLong P = 0.017 and 0.009, respectively, Supplemental Table 2).
For head-to-head comparison we also calculated the Prognostic Nutritional Index (PNI = 10 × albumin [g/dL] + 0.005 × absolute lymphocyte count [/mm3]). 16 ANS demonstrated superior discrimination for 3-year OS (AUC 0.71, 95 % CI 0.63-0.79) compared with PNI (AUC 0.66, 0.57-0.75; P = 0.041).
Multivariate Analysis of Independent Predictors
In multivariate analysis, ANS 2 remained independently associated OS (Supplemental Table 3) and every tested end-point (Supplemental Table 4). In particular, patients with ANS = 2 experienced a 178 % higher mortality risk and more than a three-fold increase in major complications compared with ANS = 0.
Discussion
In this single-center cohort of 182 adults aged ≥ 65 years, a high pre-operative Albumin–NLR Score (ANS = 2) was independently associated with a near-threefold increase in mortality, a six-fold rise in prolonged hospitalization, and markedly higher rates of major postoperative complications compared with ANS = 0. Our findings strengthen emerging evidence that coupling albumin, a marker influenced by both nutritional status and inflammation, with NLR, a barometer of acute systemic immune activation, captures the dual biological forces that underlie poor surgical resilience in older patients. While other composite indices such as GPS, PLR, and CONUT incorporate similar constructs,16,17 ANS relies on only two universally available analyses, making it uniquely scalable for routine geriatric pre-operative assessment. Even after accounting for pathological stage and pre-operative CEA, a high ANS independently doubled the risk of death and improved the model C-index from 0.75 to 0.78. This suggests that ANS captures a dimension of vulnerability, malnutrition-inflammation synergy, not reflected by tumor burden or tumor-specific markers, and does so at a time-point when treatment intensity can still be adjusted.
The results of our study align with findings from similar research in other cancer types, such as head and neck, esophageal, and pancreatic cancers, where high ANS scores have been linked to poor survival outcomes and increased complication rates. In head and neck cancer, patients with high ANS scores were more likely to experience poor treatment tolerance, with incomplete chemoradiotherapy. 26 In esophageal cancer, patients with high ANS scores were more likely to suffer from treatment-related adverse events, such as infections, which ultimately led to worse survival rates. 27 Similarly, in pancreatic cancer, high ANS scores were predictive of shorter overall survival, higher rates of severe treatment-related toxicities, and postoperative complications, further emphasizing the ANS’s role as a powerful prognostic marker in these aggressive cancer types. 28 Our study contributes to this body of evidence by demonstrating that high ANS scores are similarly associated with a high incidence of postoperative complications and poor survival outcomes in CRC.
The ability of the ANS to predict postoperative outcomes is particularly relevant in older patients who are more vulnerable to malnutrition and systemic inflammation. The high prevalence of sarcopenia and malnutrition in this population exacerbates their susceptibility to adverse events following surgery.14,29,30 Consistent with previous research, our study confirms that poor nutritional status, as indicated by low albumin levels, correlates with poor survival and increased morbidity. Furthermore, an elevated NLR reflects heightened systemic inflammation, which is associated with immune suppression and poorer tumor control, further contributing to unfavorable postoperative outcomes.18,31 The stratified analysis of postoperative complications revealed that patients in the ANS 2 group had disproportionately higher rates of several serious complications, particularly infectious complications such as sepsis and surgical wound infections, as well as organ dysfunctions including respiratory failure and acute renal failure. These findings highlight the specific vulnerability of malnourished and inflamed older CRC patients to infections and multi-organ complications after surgery, underscoring the clinical significance of combined hypoalbuminemia and elevated NLR as markers of malnutrition and systemic inflammation, which in turn contribute to increased postoperative morbidity and hindered recovery.
Our study also found that higher ANS scores were linked to poorer HRQoL in older CRC patients. Patients in the ANS 2 group reported greater difficulties in mobility and maintaining purpose postoperatively, suggesting that ANS can predict declines in physical function and overall well-being. Gupta et al reported that well-nourished patients with CRC had statistically significantly better QoL scores on multiple symptom scales compared to malnourished patients. 32 Similarly, Gillis et al found that CRC patients with malnutrition experienced worse HRQoL, particularly in terms of walking and mental health, compared to well-nourished patients prior to surgery. 33 In line with these reports, our study demonstrates that malnutrition and systemic inflammation are closely tied to diminish HRQoL in CRC patients. The interaction between ANS and HRQoL in CRC patients illustrates how malnutrition and inflammation impair physical and mental health, leading to a decline in postoperative recovery and survival outcomes. Maisey et al reported that baseline HRQoL is an independent predictor of survival in CRC patients. 34 Given that ANS is associated with worse HRQoL, this suggests that ANS not only serves as a predictor of surgical complications and survival but also interacts with preoperative and postoperative quality of life, reinforcing its role as a comprehensive prognostic tool in managing CRC patients.
Head-to-head within the present cohort, ANS achieved an AUC of 0.71 for 3-year survival vs 0.66 for the PNI; ANS retained independent prognostic value for every surgical and patient-reported outcome after multivariable adjustment. The Glasgow Prognostic Score (GPS)1 16 could not be replicated here because C-reactive protein is not part of our routine pre-operative panel—an omission common in many elective colorectal centers and a practical reminder that GPS, although informative, requires an additional assay. Meta-analyses in colorectal cancer generally report GPS AUCs in the 0.60-0.65 range,35,36 suggesting that the discriminatory power of ANS observed in our study is at least comparable and may be higher, while demanding no extra laboratory resources. Crucially, neither PNI nor GPS has been associated with early post-operative health-related quality of life. By demonstrating that ANS predicts persistent mobility limitations in older adults, our study extends prognostic relevance beyond traditional clinical outcomes to what matters most to patients—functional recovery. Taken together, these findings support ANS as a pragmatic, biologically grounded, and patient-centered tool for peri-operative risk stratification in geriatric colorectal oncology.
Based on our findings, we recommend integrating ANS into routine peri-operative care for older CRC patients. Because serum albumin and differential counts are already part of standard pre-admission labs, calculating ANS is cost-free and quickly pinpoints high-risk individuals. When ANS is high (score = 2), clinicians can launch a targeted prehabilitation bundle that has strong evidence: (1) dietitian-led high-protein nutrition plus 5-7 days of arginine/ω-3–enriched immunonutrition, which meta-analyses show cuts infectious complications by roughly 50 % and shortens hospital stay by ≈ 1.5 days 37 ; (2) concurrent aerobic and resistance exercise three times weekly together with a daily goal of ≥7500 steps, an approach linked to improved functional capacity and ≈50 % fewer post-operative complications 38 ; and (3) rapid correction of modifiable risks such as occult infection, anemia, smoking and unnecessary medications, as endorsed by current multimodal prehabilitation guidelines. 38 In addition, an elevated ANS can steer operative strategy (eg, minimally invasive techniques) and personalize Enhanced Recovery After Surgery pathways, 39 giving patients and families clearer expectations about length of stay, complication risk and post-discharge support.
Our study showed clinical outcomes for ANS 0 and ANS 1 were indistinguishable. Three factors likely account for this observation. First, the adverse biology of malnutrition and systemic inflammation appears synergistic. Only a ‘double-hit’ (low albumin and high NLR, ANS 2) consistently overwhelmed the physiological reserve of older patients, whereas a single abnormality (ANS 1) was often offset by compensatory mechanisms in nutrition, immunity, or wound healing. Second, the albumin (<4.1 g/dL) and NLR (>2.6) thresholds we applied represent mild deviations from normal; either alteration alone may fall below a biological threshold needed to influence surgical resilience. Third, ANS 1 is inherently heterogeneous, some patients are inflamed but well-nourished, others malnourished but non-inflamed, so risk estimates are averaged across two contrasting phenotypes, diluting statistical power in our 182-patient cohort. Together, these considerations explain why marked differences emerged only when both insults were present (ANS 2), reinforcing the clinical relevance of combining albumin and NLR rather than evaluating either marker in isolation.
This is the first study to evaluate the predictive value of the ANS on postoperative prognosis, complications, and quality of life in older patients with metastatic CRC undergoing curative surgery. Importantly, when we incorporated all baseline variables that differed across ANS sub-groups into multivariate models, ANS remained an independent determinant of postoperative morbidity, prolonged LOS, readmission, ER use, and overall survival. This confirms that the prognostic value of ANS is not merely a surrogate for age, comorbidity, tumor stage, or other established risk factors. This study had some limitations. First, there was patient selection bias, as the study population was accrued from only one medical center in northern Taiwan. Second, the median albumin (4.1 g/dL) and NLR (2.6) were arbitrary cutoff points for ANS calculation, facilitating the classification of patients into three observable groups. These may differ from albumin and NLR cutoff points used in past studies.19,22,31 In fact, there is not yet a consensus on the optimal albumin and NLR cutoff points for CRC patients undergoing curative surgery. Although albumin and NLR thresholds can vary across studies, our sensitivity analyses using ROC-derived cut-offs (Albumin ≈4.0 g/dL; NLR ≈2.8) produced results nearly identical to the median-based analysis. These data support the external validity of a simple, median-anchored ANS classification, which is easily reproducible in routine practice where laboratory calibration and patient mix differ. Further studies are needed to reach a consensus on optimal albumin and NLR cutoff points for predicting surgical survival outcomes and complications.
Additionally, we used the QLQ-ELD14 scale to evaluate quality of life before and after surgery. Compared to the QLQ-C30 scale, 32 the QLQ-ELD14 scale lacks assessment power related to disease burden (eg, fatigue, nausea, pain, and insomnia). Further studies are necessary to integrate physical, mental, and disease burden scales for quality of life evaluation before and after surgery.
Conclusion
Our study suggests that ANS may be a valuable tool for predicting postoperative survival, complications, and quality of life in older patients with CRC. As the population ages and the incidence of CRC continues to rise, the use of a simple, reliable prognostic tool like ANS will become increasingly important for optimizing treatment strategies and improving outcomes for vulnerable older patients with cancer. Further studies are needed to refine and validate the use of ANS in clinical practice, but our findings highlight its potential to guide preoperative decision-making and postoperative care.
Supplemental Material
Supplemental Material - The Predictive Role of Albumin and Neutrophil-to-Lymphocyte Ratio Score on Postoperative Outcomes and Survival in Older Patients With Colorectal Cancer
Supplemental Material for The Predictive Role of Albumin and Neutrophil-to-Lymphocyte Ratio Score on Postoperative Outcomes and Survival in Older Patients With Colorectal Cancer by Jhih-Sheng Wang, MD, Chieh-Ying Chang, MD, Cheng-Chou Lai, MD, Yu-Shin Hung, MD, and Wen-Chi Chou, MD, PhD in Cancer Control
Footnotes
Acknowledgments
The authors gratefully acknowledge the assistance of the patients who participated in this study.
Ethical Consideration
All participants provided written informed consent. The study was approved by the Institutional Review Board of Chang Gung Memorial Hospital, Taiwan (IRB No. 201600916B0). This IRB approval covers a prospective institutional project (2016-2020) designed to enroll cancer patients receiving frailty assessment before the initiation of antitumor treatment. The current study represents a secondary analysis of that project, focusing on the prognostic impact of albumin and neutrophil-to-lymphocyte ratio in patients with colorectal cancer undergoing surgery.
Author Contributions
Conception and design of study: JSW, CYC, CCL, YSH, WCC; Acquisition of data: JSW, CYC, CCL, YSH; Analysis and interpretation of data: JSW, CYC, CCL; Drafting of the manuscript: JSW, CYC, CCL, YSH, WCC.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants from the Chang Gung Memorial Hospital at Linkou, Taoyuan, Taiwan, R.O.C. (CMRPG3L1611) and the National Science and Technology Council, Taiwan, R.O.C. (NSTC 111-2314-B-182A-162 and NSTC 112-2314-B-182A-152).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. WCC is a Section Editor of Cancer Control but did not participate in the editorial handling or peer review process of the article.
Data Availability Statement
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
