Abstract
Background
Previous studies have established that higher baseline quality of life (QOL) scores are associated with improved survival in patients with metastatic colorectal cancer (mCRC). We examined the relationship between overall survival (OS) and baseline QOL.
Patients and Methods
A total of 1 247 patients with mCRC participating in N9741 (comparing bolus 5-FU/LV, irinotecan [IFL] vs infusional 5-FU/leucovorin [LV]/oxaliplatin [FOLFOX] vs. irinotecan/oxaliplatin [IROX]) provided data at baseline on overall QOL using a single-item linear analogue self-assessment (LASA) 0–100 point scale. The association of OS according to clinically deficient (defined as CD-QOL, score 0–50) vs not clinically deficient (nCD-QOL, score 51–100) baseline QOL scores was tested. A multivariable analysis using Cox proportional hazards modeling was performed to adjust for the effects of multiple baseline factors. An exploratory analysis was performed evaluating OS according to baseline QOL status among patients who did or did not receive second-line therapy.
Results
Baseline QOL was a strong predictor of OS for the whole cohort (CD-QOL vs nCD-QOL: 11.2 months vs 18.4 months, P < .0001), and in each arm IFL 12.4 vs 15.1 months, FOLFOX 11.1 months vs 20.6 months, and IROX 8.9 months vs 18.1 months. Baseline QOL was associated with baseline performance status (PS) (P < .0001). After adjusting for PS and treatment arm, baseline QOL was still associated with OS (P = .017).
Conclusions
Baseline QOL is an independent prognostic factor for OS in patients with mCRC. The demonstration that patient-assessed QOL and PS are independent prognostic indicators suggests that these assessments provide important complementary prognostic information.
Keywords
Introduction
As the trend for individualization of cancer therapy increases, independent prognostic factors are becoming increasingly utilized, particularly in the palliative disease setting. Patient-reported outcomes (PROs) provide additional information regarding prognosis beyond what clinicians can identify. 1 Quality of life (QOL), one of the most commonly measured PROs, can be easily measured with techniques that have been well validated. 2 There is a growing body of the literature demonstrating associations between QOL and survival, and QOL outcome has become a frequent endpoint in clinical trials.3,4 Furthermore, QOL both during and after cancer treatment can be monitored and improved with structured, regular assessment and prompt intervention.5,6
The prognostic ability of baseline QOL assessment has been demonstrated across a broad spectrum of tumor types with considerable consistency, 7 although there have been exceptions. Specifically, early-stage breast cancer patient survival did not demonstrate a strong linkage with baseline QOL. 8 Also, Bauer and others have found weak relationships between baseline QOL and survival in subsets of brain cancer patients. 9 In colorectal cancer (CRC), several studies have investigated the impact of baseline QOL on overall survival (OS) in metastatic CRC (mCRC). Maisey et al 10 established that patient-reported baseline QOL scores were prognostic for one-year survival in advanced CRC. Efficace et al11,12 demonstrated the PRO of social functioning as an independent predictor of survival. Likewise, Mol et al 13 showed that baseline patient-reported physical functioning was more prognostic than clinician-assessed performance status (PS) in patients with advanced CRC. In each of these studies, the PRO outperformed clinician-rated PS as a prognostic factor.
QOL has not only shown prognostic value in clinical trials, but it has also further shown durable predictive power for all-cause mortality in cohorts of CRC survivors’ years after initial diagnosis. 14 Several other population-based analyses have confirmed that “real-world” QOL predicts OS independently of routine social and clinical assessments. 15
The aforementioned trials were performed using single-agent 5-fluorouracil (5-FU) regimens or capecitabine/oxaliplatin (CAPOX) backbones with or without irinotecan or cetuximab and bevacizumab. None of these trials included the commonly used FOLFOX regimen. The phase III Intergroup trial N9741 established the superiority of FOLFOX over the prior standard of care IFL for all key oncologic efficacy parameters, including OS. 16 We sought to determine the effect of baseline QOL on OS in patients with mCRC in N9741 as a potential independent prognostic marker after accounting for PS and study treatment.
Methods
QOL assessment was embedded in the North Central Cancer Treatment Group (NCCTG) trial N9741, which compared IFL, FOLFOX, and IROX in patients with mCRC. This protocol was reviewed and approved by the institutional review board of each participating institution. Each patient provided written informed consent to participate in the study, including providing the necessary QOL data. Patients provided data at baseline on overall QOL using a single-item linear analogue self-assessment (LASA) Uniscale that was routinely included in NCCTG clinical trials.2,17 This measure has shown to provide comparable psychometric properties to longer, more arduous QOL assessments.18,19 All QOL assessment scores were transformed onto a 0 to 100 point scale where 0 represented the worst QOL and 100 represented the best QOL achievable. Clinically deficient quality of life (CD-QOL) was pre-defined as a score of 0–49, and non-clinically deficient quality of life (nCD-QOL) was defined as a score of 50–100. This cutoff has been previously validated.2,17,19–22
Statistical Considerations
Median OS (whole study group and by study arm) was tested for association with CD-QOL vs nCD-QOL baseline scores using standard Kaplan–Meier estimation and the log-rank test. A multivariable analysis using Cox proportional hazards modeling was performed to adjust for demographics and variables previously demonstrated to be prognostic for OS. 23 This model adjusted for PS, treatment arm, age, gender, disease site, number of sites involved, hemoglobin (Hgb) levels, absolute neutrophil count (ANC), alkaline phosphatase (ALK) level, and aspartate transaminase (AST) level. Proportionality assumptions were validated using plots of Schoenfeld residuals.
The influence of missing values for ANC, ALK, AST, and Hgb for 208 patients (missing at random due to study logistics) was examined using multiple imputation. For this multiple imputation sensitivity analysis, missing laboratory values (ANC, ALK, AST, and Hgb) were imputed using linear regression estimation with age, sex, cancer site, and prior adjuvant chemotherapy as independent variables. These missing laboratory values were all modeled based on their logarithmic transformations and imputed with their predicted values based on the regression models. 24
The relationship between PS and QOL was estimated using simple correlation coefficients. In a secondary analysis, OS was compared among patients with CD-QOL vs nCD-QOL when patients were grouped by good PS (Eastern Cooperative Oncology Group [ECOG] 0) and patients with impaired PS (ECOG 1-2).
Logistic regression analysis was used to assess the effect of baseline QOL on the likelihood of receiving second-line therapy. This model was adjusted for PS, treatment arm, age, gender, disease site, number of sites involved, Hgb, ANC, ALK, and AST levels. An exploratory analysis was performed evaluating OS according to baseline QOL status, stratified by those who did or did not receive second-line therapy. All P-values reported are two-sided.
Results
Baseline characteristics of patients enrolled on clinical trial N9741.

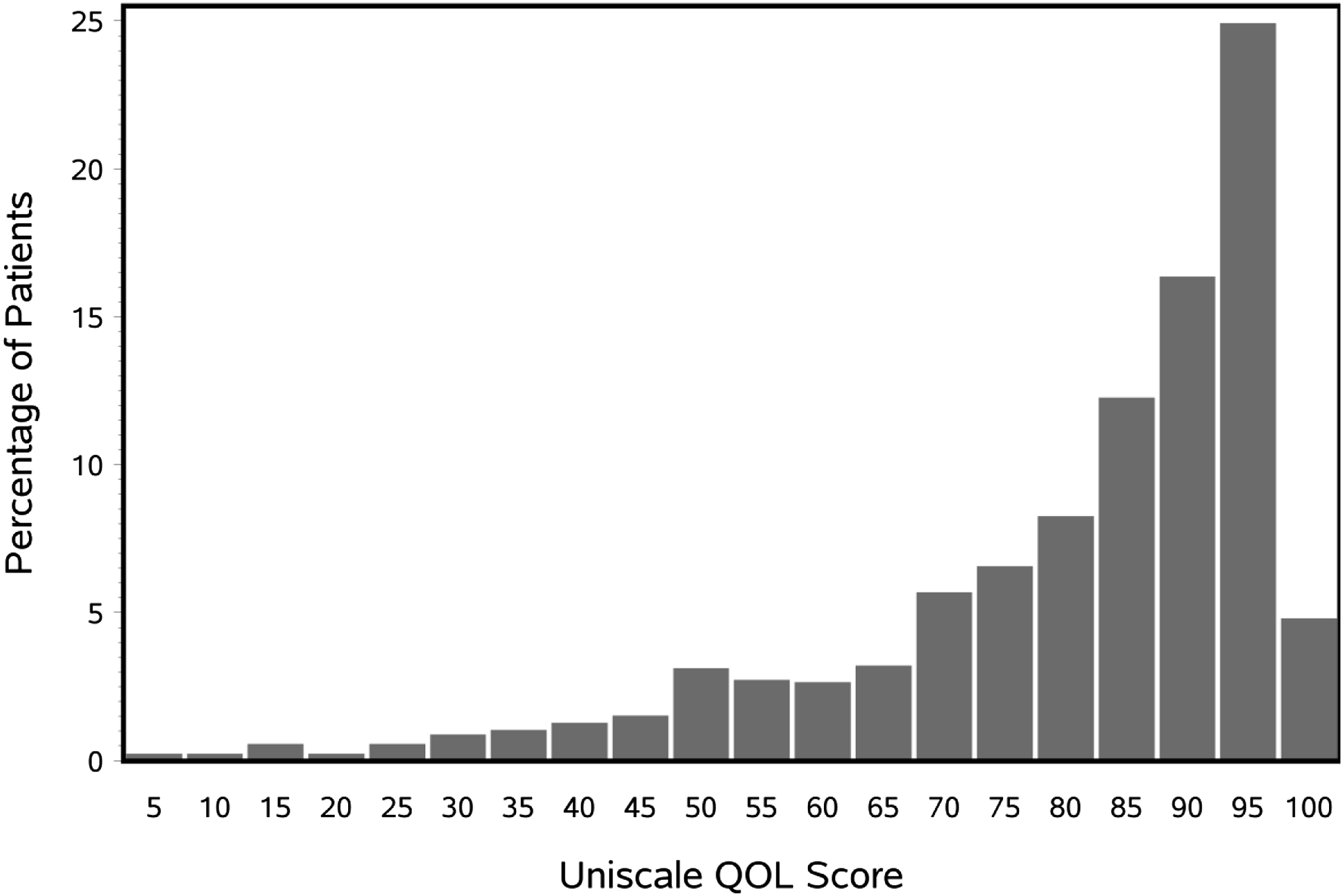
Distribution of baseline QOL scores in the entire cohort.
Baseline QOL was a strong predictor of OS for the whole study group (CD-QOL vs nCD-QOL: 11.2 months vs 18.4 months, HR 1.53, 95% CI 1.27–1.85, P < .001, Figure 2). This effect was consistently seen in all treatment arms. Median survival in patients with CD-QOL and nCD-QOL was 12.4 months vs 15.1 months in the IFL arm (P = .2389), 11.1 months vs 20.6 months in the FOLFOX arm (P = .0013), and 8.9 months vs 18.1 months in the IROX arm (P = .0009). Overall survival based on CD-QOL vs nCD-QOL. Median OS was increased in the nCD-QOL group.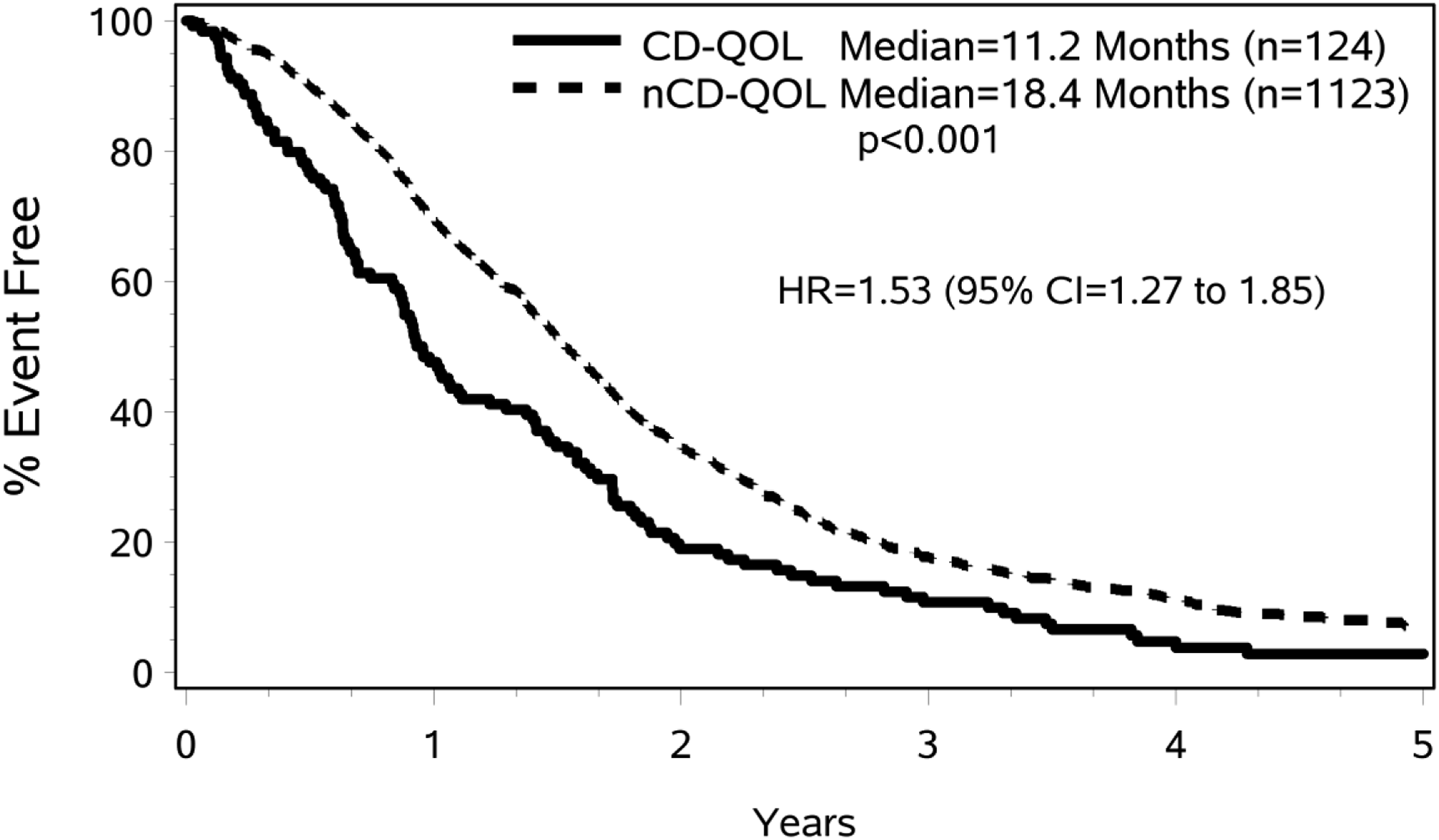
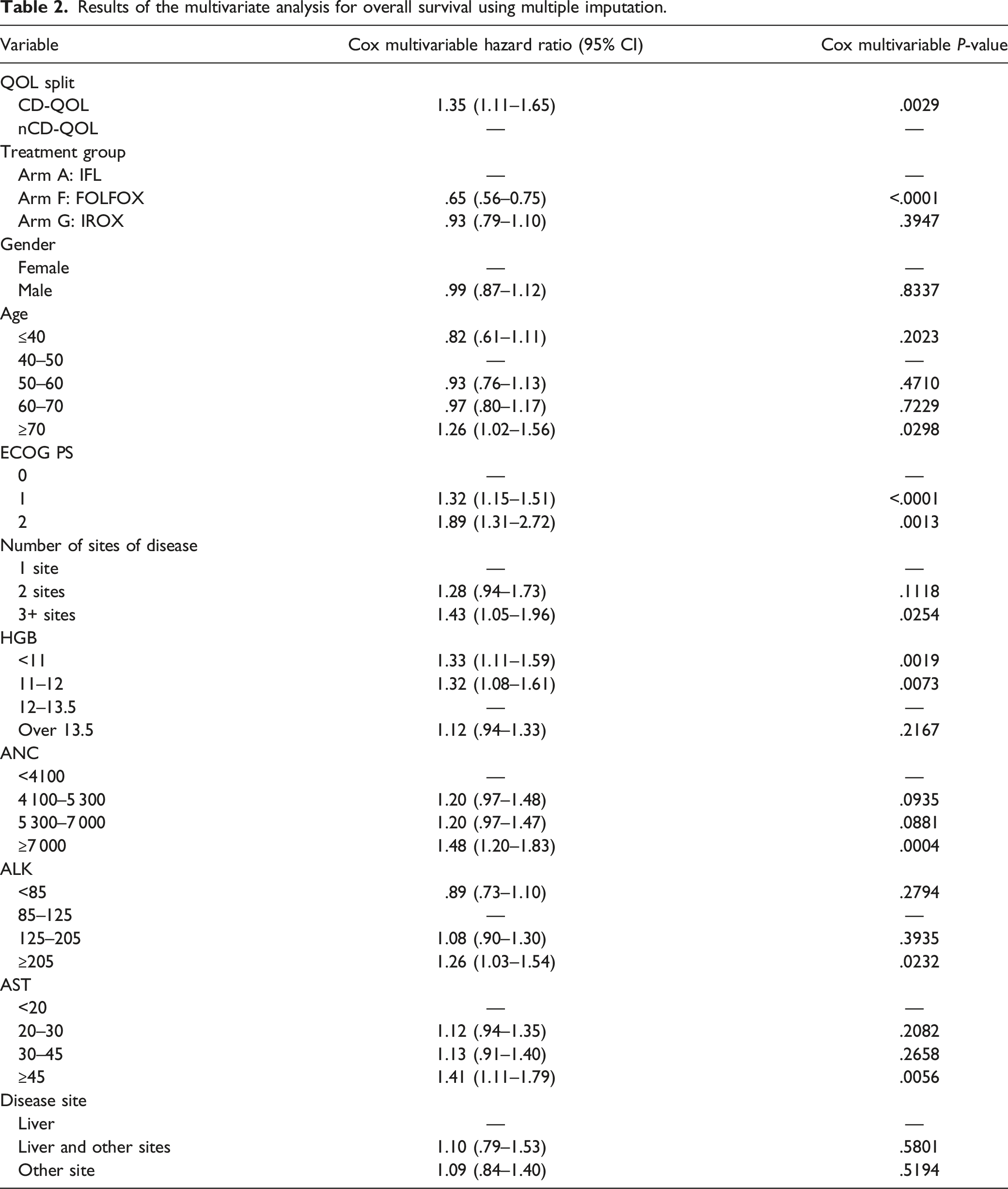
As shown in Figure 3, OS was strongly associated with PS (log-rank P < .001). PS and QOL were also highly associated. Among patients with baseline PS of 1 or 2, 14% had CD-QOL, compared to only 6% of patients with CD-QOL among the patients with a PS of 0 (P < .001). After adjusting for PS and treatment arm, baseline QOL was still associated with OS (HR 1.29, 95% CI 1.05–1.59, P = .0169). Results of the multivariate analysis using multiple imputation are shown in Table 2. In the saturated Cox model, the effect of CD-QOL remained significant (HR 1.35, 95% CI 1.11–1.65, P = .0029). Association between OS and PS. Those with inferior ECOG PS had significantly decreased median OS. Results of the multivariate analysis for overall survival using multiple imputation.
CD-QOL differentiated survival to a greater degree for those patients with poorer PS (Figure 4). Among patients with PS 1 or 2, patients with nCD-QOL survived significantly longer than those patients with CD-QOL, with a median OS of 13.8 months and 10.5 months, respectively (HR 1.32, 95% CI 1.03–1.71, P = .0314). Among patients with PS of 0, patients with nCD-QOL had a median OS of 21.0 months vs 15.5 months in patients with CD-QOL, which was not statistically significant (HR 1.26, 95% CI 0.88–1.82, P = .21). Survival curves according to performance status. Patients with ECOG PS of 1 or 2 had improved median OS when baseline QOL was higher.
Seventy-six percent (957) of patients received second-line therapy. Although patients with nCD-QOL scores were more likely to receive second-line therapy (P = .0020), higher baseline QOL scores were associated with improved survival even when adjusting for having received second-line therapy in the regression model. The improved survival was appreciated in both subsets of those who did and did not receive second-line therapy. In those patients who did go on to receive second-line therapy, patients with nCD-QOL scores had significantly improved survival over those with CD-QOL, with median OS times of 20.5 months and 17.6 months, respectively (HR 1.33, 95% CI 1.05–1.67, P = .0173). In the subset of patients who did not receive second-line therapy, this effect was even more pronounced. Here, patients with nCD-QOL had improved OS compared to patients with CD-QOL with a median OS of 10.9 months and 4.9 months, respectively (HR 2.02, 95% CI 1.44–2.82, P < .0001).
Discussion
Our study demonstrates that baseline QOL is a strong and independent prognostic factor for OS in patients with mCRC treated with a modern combination chemotherapy regimen (FOLFOX) in the phase III trial N9741. This effect was consistently found in all study treatment arms and was independent of multiple other factors including PS, age, and race. Improved QOL scores at baseline were associated with an increased likelihood of receiving second-line therapy; however, nCD-QOL was associated with improved OS regardless of whether second-line therapy was received. This provides further evidence that PROs provide information prognostic for survival beyond what is currently clinically measurable and recorded by clinicians during routine office visits and support the use of baseline QOL as a prognostic tool in mCRC.
With recent advances in PRO assessment, information on global QOL can be easily obtained in a clinical practice setting using the single-item linear analogue self-assessment (LASA) Uniscale questionnaire, a well-validated measure of global QOL. 25 In oncology studies, categorizing QOL scores according to CD-QOL and nCD-QOL (as opposed to median scores) provides a more sensitive measure of identifying patients with clinically significant deficiencies in their QOL at higher risk of poor clinical outcomes. 20
The patients’ perspective may more accurately reflect overall functional status than physicians’ perceptions. 26 Patient self-assessment of PS has a strong correlation with survival and is independent of physician-determined PS assessments in predicting outcomes in various advanced malignancies. 1 This study clearly shows that while QOL is associated with PS, it provides independent prognostic information and therefore is not solely a surrogate for PS.
Increasing evidence demonstrates that simple PRO assessments can identify patients at risk for adverse events and in need of supportive care. This study confirms the findings of other analyses27–29 that PROs can identify patients who are at the highest risk of mortality. A next logical step is to incorporate PROs into clinical practice in a way such that vulnerable patients can be identified. Specific risk factors for poor QOL in patients with CRC have been described. With identification of patients most likely to benefit from intensive PRO monitoring, prophylactic actions and supportive care can be implemented. This will allow for medical resources to be applied more effectively through the use of a PRO-based screening and triaging system.
Previous studies have demonstrated that there are effective interventions for improving QOL among patients in the clinical setting. Interventions during radiation therapy treatment, for example, can prevent a decrease of symptoms during treatment, which would be of optimal concern in the palliative setting. 30 Social work interventions on emotional, spiritual, physical, and social domains have also been shown to improve QOL during radiation therapy for advanced malignancies. 31 To explore the best way to utilize patient-reported QOL, a number of “real-time” QOL data collection and feedback systems have been developed and have been tested clinically.5,32 This approach to PRO assessment resulted in improved QOL and treatment tolerance, and it was strongly endorsed by both patients and clinical staff. 33
Whether interventions to improve QOL or maintain optimal QOL would translate into improved survival has been explored. Previous attempts to answer this question in the setting of breast cancer trials have yielded conflicting results.30,34,35 However, Basch et al demonstrated a survival benefit for patients randomized to a real-time electronic PRO monitoring system for patients with metastatic solid tumors. These are encouraging results, though it should be noted that this trial did not include patients with gastrointestinal malignancies.
Over the past two decades, there has been a growing body of the literature recognizing the prognostic value of PROs on OS. In a comprehensive review of clinical trials that included at least one PRO (including baseline PRO), data on survival, and multivariate analyses, 36 of the 39 identified studies identified at least one PRO that was independently associated with OS in multivariate analysis. 36 Global QOL and physical functioning were the most common PROs associated with survival. In a similar review of studies involving QOL and survival outcomes, Montazeri 37 reported almost all of the 104 relevant studies demonstrated QOL, or some aspect of QOL such as appetite loss, fatigue, and pain, was independently correlated with survival duration.
There are several limitations to the present study. Firstly, QOL is an inherently complex concept and there is likely wide variability in how individuals rate this parameter. There are many potential inputs such as cultural identity, socioeconomic status, race and ethnicity, and family background. While the QOL measurement in aggregate is prognostic, applying this finding when counseling individual patients in clinical practice is presumably much less straightforward given the myriad factors mentioned. Another limitation is that the analysis was conducted using regimens that are no longer used with much frequency, namely, IFL and IROX. Therefore, the findings may be less applicable to present standard of care clinical practice.
Conclusion
The demonstration that patient-assessed QOL and PS are independent prognostic factors suggests that these assessments provide important complementary prognostic information. This association is clear, consistent, and easily identified. The next step is to capitalize on this information to improve the well-being and survival of cancer patients. Incorporating these simple QOL assessments into clinical practice and associating the identification of clinically significant deficits in QOL with clinical pathways will ultimately allow for the integration of QOL assessments as part of the routinely assessed clinical indicators. This will not only augment patient well-being but also improve the quality of the care delivered by readily incorporating key information from the perspective of the patient.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute of the National Institutes of Health under Award Numbers U10CA180821 and U10CA180882 (to the Alliance for Clinical Trials in Oncology) and U10CA180888 (SWOG).
Ethical Approval
This trial was approved by the Intergroup/Alliance IRB N9741, and consent was obtained from all trial subjects.
