Abstract
Introduction
For patients with advanced gastric cancer (AGC) who are HER2-negative and ineligible for targeted therapies, chemotherapy remains the cornerstone of treatment. The modified docetaxel, cisplatin, and fluorouracil (mDCF) regimen has been proposed as an effective and tolerable triplet regimen in this setting. This study aimed to evaluate the efficacy and safety of first-line mDCF in Vietnamese patients with HER2-negative AGC.
Methods
We conducted a retrospective study of 64 patients with histologically confirmed HER2-negative advanced or recurrent gastric adenocarcinoma treated with mDCF at K Hospital, Vietnam, between January 2017 and January 2022. The regimen was administered every 14 days, consisting of docetaxel 40 mg/m2, cisplatin 40 mg/m2, leucovorin 400 mg/m2, 5-FU 400 mg/m2 IV bolus, and 5-FU 2000 mg/m2 as a 48-hour continuous infusion. The primary endpoints were disease control rate (DCR) and progression-free survival (PFS). Safety was assessed as a secondary endpoint.
Results
The overall DCR was 87.5%, including complete response in 9.4%, partial response in 59.4%, and stable disease in 18.7%. Median PFS was 8.7 months. Grade 3-4 hematologic toxicities occurred in 21.9% of patients, including neutropenia (15.6%) and thrombocytopenia (3.1%). No grade 3-4 non-hematologic toxicities were observed. Primary prophylactic G-CSF use was associated with a significantly lower incidence of grade 3-4 neutropenia (P < .05). In multivariate analysis, poor ECOG performance status (≥2) and poor histological differentiation were independently associated with shorter PFS (P < .05).
Conclusions
mDCF is an effective and tolerable first-line chemotherapy regimen for HER2-negative AGC patients in Vietnam. These findings support mDCF as a practical first-line option for HER2-negative AGC in real-world settings with limited access to targeted therapies.
Plain Language Summary
Gastric cancer is a major health concern in Vietnam, with many patients diagnosed at an advanced stage when surgery is no longer possible. For patients whose tumors do not have HER2 overexpression—a condition that limits the use of targeted treatments—chemotherapy remains the mainstay of care. This study evaluated the effectiveness and safety of a chemotherapy regimen called modified DCF (mDCF), which includes docetaxel, cisplatin, and 5-fluorouracil. We reviewed the treatment outcomes of 64 patients with HER2-negative advanced or recurrent gastric cancer who received mDCF as their initial therapy at a major oncology hospital in Vietnam. The findings showed that nearly 87.5% of patients experienced disease control, meaning the tumor either shrank or stopped growing. The time before the disease worsened was about 8.7 months. Most side effects were manageable. The most common serious issue was low white blood cell count (neutropenia), but this was significantly reduced in patients who received preventive treatment with G-CSF. Overall, this study suggests that mDCF is an effective and tolerable first-line treatment option for patients with HER2-negative advanced gastric cancer in Vietnam. In settings where access to other treatment options may be limited, mDCF continues to serve as a valuable and practical regimen that can help improve patient outcomes.
Keywords
Introduction
Gastric cancer (GC) continues to represent a substantial global public health burden. According to the most recent data from GLOBOCAN 2022, 1 gastric cancer remains highly prevalent, standing as the third most common cancer among men and the fourth most common among women in Vietnam, with a significant proportion of patients presenting at an advanced stage due to the absence of nationwide screening programs and the disease’s insidious onset. 2
In recent years, substantial advances have been made in systemic therapy for advanced gastric cancer (AGC).3-7 These innovations, enabled by improved diagnostics and molecular profiling, have transformed treatment paradigms in high-income countries. However, in low- and middle-income countries like Vietnam, access to such therapies remains limited due to high costs and regulatory constraints. As a result, cytotoxic chemotherapy remains the mainstay of first-line treatment for most patients with AGC. This is particularly true for HER2-negative individuals, who account for approximately 75%-85% of gastric cancer cases in Vietnam. A recent study reported HER2 overexpression in only 23.2% of patients with gastric adenocarcinoma, supporting the predominance of HER2-negative disease in Vietnam. 8
In Vietnam, HER2-negative patients represent the vast majority of gastric cancer cases. 8 However, access to HER2-targeted agents such as trastuzumab remains limited due to high treatment costs, lack of reimbursement, and restricted availability in public healthcare settings. For patients with unresectable, locally advanced, or metastatic gastric cancer (GC), systemic chemotherapy remains the cornerstone of first-line treatment, with the primary goals of extending survival, reducing tumor-related symptoms, and maintaining quality of life. Platinum-based doublets combined with fluoropyrimidines, such as cisplatin/oxaliplatin plus 5-fluorouracil/capecitabine, are standard regimens recommended by major international guidelines. 9 In patients with preserved performance status, triplet regimens incorporating a taxane, such as docetaxel, may offer enhanced antitumor efficacy. 10 However, the original DCF (docetaxel, cisplatin, 5-FU) regimen has demonstrated considerable toxicity, particularly myelosuppression, which limits its feasibility in routine clinical practice. 11
To overcome these limitations, a modified DCF (mDCF) regimen has been developed, incorporating dose adjustments and altered schedules to improve tolerability while maintaining efficacy. Several studies have shown that mDCF achieves comparable outcomes with reduced grade 3-4 toxicity, making it a practical option in daily clinical settings, especially in resource-constrained environments.12,13
In Vietnam, HER2-negative gastric cancer accounts for the vast majority of cases. However, patients in this group remain largely dependent on conventional cytotoxic chemotherapy, as access to HER2-targeted agents such as trastuzumab is restricted by high costs and limited insurance reimbursement. Although trastuzumab-based regimens have improved outcomes for HER2-positive patients, this subgroup represents only a minority (10%-20%) of gastric cancers, and treatment availability is further constrained in resource-limited settings. This disparity underscores the need to optimize conventional chemotherapy strategies—particularly mDCF—for the HER2-negative majority. The molecular classification of GC has introduced new treatment options for patients with HER2-positive tumors, who may benefit from trastuzumab-based chemotherapy. However, HER2 overexpression is only observed in approximately 10%-20% of cases, and in resource-limited settings, access to anti-HER2 therapies is often restricted by cost and availability. 14 Consequently, HER2-negative patients—who comprise the majority—continue to rely primarily on conventional chemotherapy, highlighting the need to optimize cytotoxic regimens such as mDCF in this subgroup.
Despite the increasing use of mDCF across oncology centers in Vietnam, local evidence on its efficacy and safety in HER2-negative GC remains limited. Moreover, real-world studies from low- and middle-income countries (LMICs) are scarce, although they provide critical insight into treatment applicability under resource constraints. Therefore, we conducted a retrospective cohort study to evaluate the clinical outcomes and toxicity profile of mDCF as first-line chemotherapy in Vietnamese patients with HER2-negative advanced gastric adenocarcinoma. This study aims to generate context-specific evidence to guide therapeutic decisions in Vietnam and other LMICs facing similar constraints.
Materials and Methods
Study Population
This retrospective cohort study was conducted at K Hospital, Hanoi, Vietnam, from January 2017 to January 2022. Medical records were reviewed to identify patients who met the eligibility criteria and received first-line chemotherapy with the modified docetaxel, cisplatin, and 5-fluorouracil (mDCF) regimen. A total of 64 patients with histologically confirmed gastric adenocarcinoma were included. This study was approved by the Ethics Committees of both K Hospital (Decision No. 922/QĐ-BVK) and Hanoi Medical University (Decision No. 5622/QĐ-ĐHYHN). Given the retrospective design and use of de-identified medical records, written informed consent was waived by both committees.
Inclusion/exclusion criteria
Eligible patients were aged ≥18 years, had histologically confirmed HER2-negative gastric adenocarcinoma (HER2-negative status was confirmed by immunohistochemistry (IHC), with a score of 0 or 1+ classified as negative, following standard criteria), and had locally advanced, recurrent, or metastatic disease not amenable to curative surgery. All patients had an ECOG performance status of 0 to 2 and adequate hematologic, renal, and hepatic function. Patients who were HER2-positive or received trastuzumab were excluded from this study. All consecutive eligible patients initiating mDCF during the study period were included; no patient was excluded due to missing or incomplete data.
All patient data were anonymized and de-identified prior to analysis.
Treatment
Patients received the mDCF regimen every 14 days. On day 1 of each cycle, docetaxel at a dose of 40 mg/m2 was administered intravenously over 60 min, followed by cisplatin 40 mg/m2 infused over 60 min. Subsequently, leucovorin 400 mg/m2 was given intravenously over 2 hours, followed by a bolus injection of 5-fluorouracil (5-FU) 400 mg/m2. This was immediately followed by a continuous infusion of 5-FU 2000 mg/m2 over 48 h. Chemotherapy cycles were repeated every 14 days and continued until disease progression or the emergence of unacceptable toxicity. Prior to each cycle, patients underwent clinical evaluations and laboratory tests to determine eligibility for continued treatment or necessary dose adjustments.
Toxicity Management
Primary prophylaxis with granulocyte-colony stimulating factor (G-CSF) was administered at the discretion of the treating physician, particularly in patients deemed at high risk for developing neutropenia. In patients who experienced febrile neutropenia or prolonged grade 3-4 neutropenia during treatment, secondary prophylaxis with G-CSF was introduced in subsequent cycles. Dose modifications and delays were permitted based on hematologic and non-hematologic toxicity as evaluated prior to each cycle. Supportive care—including antiemetics, hydration, hepatoprotective agents, nutritional support, and antidiarrheals—was provided according to institutional protocols. Patients were closely monitored for clinical symptoms and laboratory abnormalities, and hospitalization was indicated in cases of febrile neutropenia, significant hepatic enzyme elevation, or grade ≥3 toxicity requiring medical intervention.
Outcomes
The primary outcomes were disease control rate (DCR) and progression-free survival (PFS). DCR was defined as the proportion of patients achieving complete response (CR), partial response (PR), or stable disease (SD) according to RECIST version 1.1. PFS was defined as the interval from the start of mDCF treatment to the date of radiologically confirmed progression or death from any cause. Secondary outcomes included the incidence and severity of adverse events, which were graded according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, rates of hospitalization due to treatment-related toxicities, and the association between primary G-CSF prophylaxis and the occurrence of grade 3-4 neutropenia. Radiological evaluation was performed using contrast-enhanced computed tomography (CT) before treatment initiation and after every two to three cycles. Radiological response assessments were based on the original CT scan reports available in the patients’ medical records. No independent re-evaluation of imaging was performed. The reporting of this study conforms to STROBE guidelines. 15
Statistical Analysis
All data were analyzed using SPSS version 20.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were used to summarize patient characteristics and treatment outcomes. Categorical variables were compared using the chi-square test or Fisher’s exact test as appropriate. Kaplan–Meier survival analysis was used to estimate median PFS, and differences between groups were assessed using the log-rank test. All P-values were two-sided, and a P-value <.05 was considered statistically significant.
Results
Patient Characteristics
Patient Demographic and Clinical Characteristics

Tumor Response and Survival
All 64 patients were evaluable for treatment response. The disease control rate (DCR) was 87.5%, including complete response (CR) in 9.4% (n = 6), partial response (PR) in 59.4% (n = 38), and stable disease (SD) in 18.7% (n = 12). Progressive disease (PD) was observed in 12.5% (n = 8). The median progression-free survival (PFS) was 8.7 months (95% CI, 6.9-10.5) (Figure 1A). Kaplan-Meier Survival Curves for (A) Progression-Free (PFS) in the Entire Cohort of Patients With HER2-Negative Advanced Gastric Cancer Treated With the mDCF Regimen (n = 64). (B) Progression-Free Survival (PFS) Stratified by Histological Tumor Differentiation. Patients With Well or Moderately Differentiated Adenocarcinoma Showed Significantly Longer PFS Compared to Those With Poorly Differentiated Tumors (P = .016)
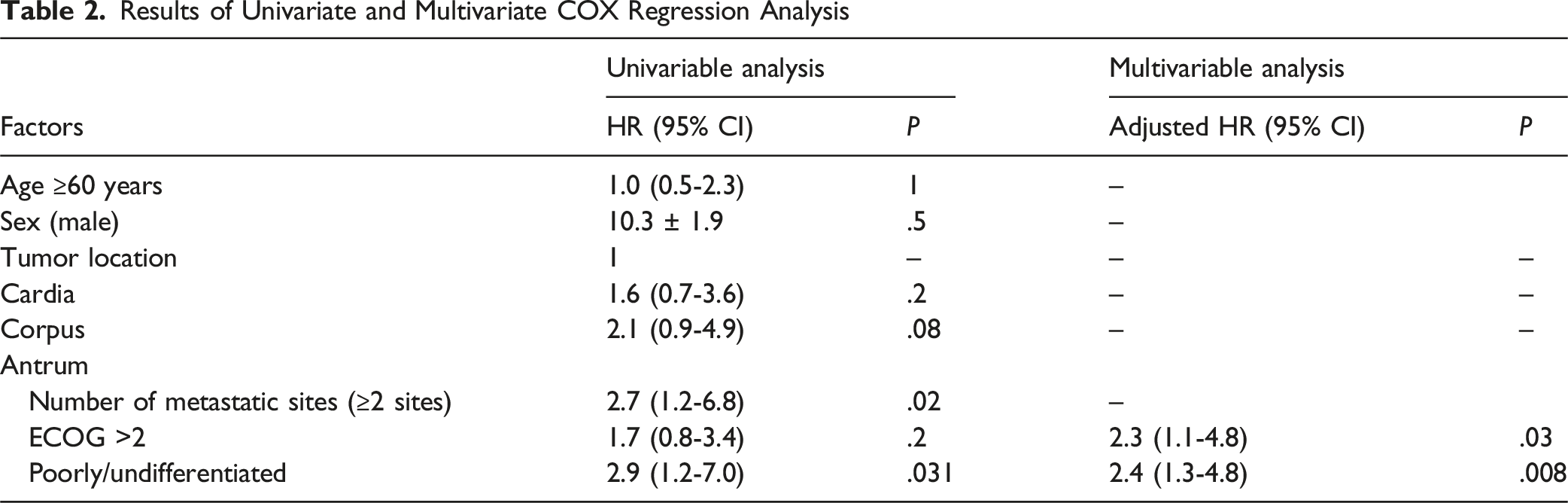
Results of Univariate and Multivariate COX Regression Analysis
Toxicity
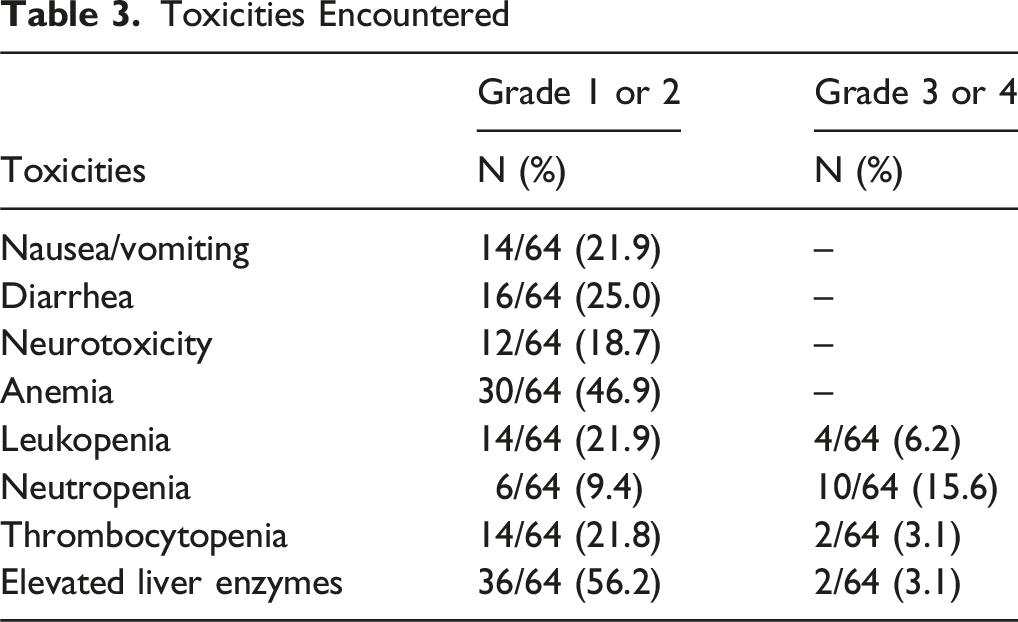
The most common treatment-related adverse events were mild to moderate. Grade 1-2 toxicities included elevated liver enzymes (56.2%), anemia (46.9%), diarrhea (25.0%), leukopenia (21.9%), nausea/vomiting (21.9%), thrombocytopenia (21.8%), neurotoxicity (18.7%), and neutropenia (9.4%). No grade 1-2 renal toxicity was observed.
Toxicities Encountered
G-CSF Prophylaxis
Primary G-CSF prophylaxis was administered in 78.1% (n = 50) of patients. Among the remaining 14 patients who did not receive primary prophylaxis, 12 developed neutropenia and required secondary G-CSF.
The incidence of grade 3-4 neutropenia was significantly lower in the group receiving primary G-CSF prophylaxis compared to those who did not (4.0% vs 57.1%; P < .05), highlighting the protective role of G-CSF prophylaxis in maintaining chemotherapy tolerance.
Discussion
This retrospective study assessed the efficacy and tolerability of the modified docetaxel, cisplatin, and 5-fluorouracil (mDCF) regimen in 64 patients with HER2-negative advanced gastric cancer (AGC) treated at K hospital, with a disease control rate (DCR) of 87.5% and a median progression-free survival (PFS) of 8.7 months. On multivariate analysis, poor Eastern Cooperative Oncology Group (ECOG) performance status (≥2) and poor histological differentiation emerged as independent prognostic factors for shorter PFS, highlighting the dual importance of baseline functional reserve and tumor biology in determining clinical outcomes.
Our median PFS compares favorably with prior real-world studies of mDCF regimens. In a Western cohort reported by Shah et al, a 6-month PFS rate of 63% and overall response rate (ORR) of 49% were observed with dose-modified DCF 12 while Keskin and colleagues 16 reported an 82.4% DCR and median PFS of 6.5 months in a Turkish real-world population. These findings align with our results and support the reproducibility of mDCF’s efficacy across diverse ethnic and clinical practice settings. Moreover, when compared with other triplet regimens in East Asian trials—such as SOX (S-1 plus oxaliplatin), which achieved a median PFS of 5.5-5.6 months, and ECX (epirubicin, cisplatin, capecitabine), with a median PFS of approximately 6.5 months— mDCF demonstrates at least equivalent, if not superior, disease control.17,18 In our Vietnamese cohort, the DCR and PFS achieved were comparable or superior to those reported in other local studies. To Nhu Hanh et al reported a median PFS of 6.04 months using ECX (epirubicin, cisplatin, capecitabine), while Nguyen Khanh Ha et al found a DCR of 67% and a median PFS of 5.4 months for TCX (docetaxel, cisplatin, capecitabine).19,20 Our findings suggest that mDCF may outperform ECX, TCX, SOX, and FOLFIRI in the Vietnamese context, all of which have demonstrated generally lower PFS and response rates.21,22
Among newer regimens, FLOT (5-fluorouracil, leucovorin, oxaliplatin, and docetaxel) has shown superior efficacy compared to ECF/ECX in randomized trials. 23 However, the four-drug schedule requires central venous access, intensive supportive care, and carries a substantial toxicity burden, potentially limiting feasibility in low-resource settings. By contrast, mDCF offers a more pragmatic alternative with comparable disease control and a simpler biweekly regimen—particularly when G-CSF is available. Formal cost-effectiveness studies are needed to directly compare mDCF and FLOT in LMICs.
Toxicity remains a central concern in any triplet regimen. In our study, grade 3-4 neutropenia occurred in 15.6% of patients and thrombocytopenia in 3.1%. Notably, no grade 3-4 non-hematologic toxicities were observed. These outcomes are more favorable than those seen in the V325 trial and comparable to those reported by Shah et al, 12 who emphasized the role of G-CSF in minimizing hematologic adverse events.
We observed that 78.1% of patients received primary prophylactic G-CSF, which significantly reduced the incidence of severe neutropenia (P < .05). This approach is supported by international clinical guidelines, such as those from ESMO and ASCO, which recommend G-CSF prophylaxis when the risk of febrile neutropenia exceeds 20%.24,25 Proactive G-CSF support likely contributed to treatment adherence, dose intensity, and outpatient feasibility in our cohort.
Beyond efficacy and safety, our findings highlight the importance of tailored patient selection. The independent prognostic significance of ECOG ≥2 and poor tumor differentiation suggests that intensive regimens like mDCF may be suboptimal for frail patients or those with aggressive histology. Alternative approaches, such as dose-adjusted regimens or integration with immunotherapy, may be more appropriate for these subgroups.
Several limitations warrant consideration. First, the relatively small sample size (n = 64) may have limited statistical power, particularly for subgroup analyses. Second, the single-center, retrospective design introduces risks of selection and information bias, though consecutive case inclusion and electronic records aimed to mitigate this. Third, the absence of a control arm prevents formal comparison with alternative regimens such as FLOT, FOLFOX, or DCF. Fourth, overall survival (OS) data were immature at the time of analysis. Lastly, lack of centralized HER2 testing and biomarker profiling (eg, MSI, PD-L1, EBV) constrained subgroup analysis and precluded exploration of chemo-immunotherapy strategies.
Nonetheless, our findings provide valuable real-world evidence in a purely HER2-negative population—an underrepresented group in clinical trials—and support mDCF as a highly effective and tolerable option for first-line therapy in AGC. This is particularly relevant in low-resource settings where access to trastuzumab, immune checkpoint inhibitors, or clinical trials is limited.
From a public health perspective, these findings support the incorporation of mDCF into national treatment guidelines for HER2-negative AGC in LMICs. Given its manageable toxicity, simplified administration, and compatibility with existing chemotherapy infrastructure, mDCF represents a scalable, equitable, and accessible standard of care. Broader implementation of such regimens may help reduce disparities in gastric cancer outcomes across regions.
Future research should validate our findings through prospective, multicenter studies, ideally incorporating biomarker stratification to identify subgroups most likely to benefit from chemo-immunotherapy combinations.
Conclusion
For patients with HER2-negative advanced gastric cancer, mDCF provides a rational, chemotherapy-based option with both efficacy and tolerability confirmed in clinical practice. Its applicability to patients with poor performance status and aggressive disease biology supports its continued use when targeted therapies are not available. These findings reinforce the role of mDCF as a core component of first-line systemic treatment in this population.
Footnotes
Acknowledgements
The authors would like to thank the physicians, nurses, and medical record staff at K Hospital for their support and dedication in the management of patients and in facilitating data collection for this study.
Ethical Considerations
This study was approved by the Ethics Committees of both K Hospital (Decision No. 922/QĐ-BVK) and Hanoi Medical University (Decision No. 5622/QĐ-ĐHYHN).
Consent for Publication
Requirement for obtaining informed consent was waived by the Ethics Committees due to the retrospective nature of this study.
Author Contributions
Conceived and designed the analysis: Dinh TLA, Le TY, Nguyen TTH. Collected the data: Dinh TLA, Bui TO, Nguyen VT, Nguyen VC, Pham TH. Contributed data or analysis tools: Dinh TLA, Bui TO. Performed the analysis: Dinh TLA, Bui TO. Wrote the paper: Le TY, Dinh TLA, Nguyen TTH.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
