Abstract
Objectives
The incidence of pancreatic ductal adenocarcinoma (PDAC) increases with age and is frequently diagnosed at an inoperable stage, which limits treatment options. There is limited evidence concerning patients over 75 years old, and clinical practice often lacks clear guidance regarding the choice of first-line therapy. The primary objective of this retrospective study was to assess overall survival (OS) in elderly patients receiving first-line monochemotherapy vs combination therapy. Secondary objectives included evaluating progression-free survival (PFS) and safety.
Methods
This retrospective study analyzed the records of 150 patients aged 75 or older with confirmed PDAC treated with first-line chemotherapy at Piacenza General Hospital, Italy. Of these, 72 patients received monotherapy, while 78 underwent polychemotherapy. The majority of patients (93.3%) were administered reduced doses, and within this group, 67.9% had their doses reduced by more than 20%. Most patients (80%) presented with comorbidities, predominantly hypertension and diabetes.
Results
The median overall survival was significantly higher in the polychemotherapy group (8.2 months) compared to the monotherapy group (4.7 months), with a P-value of 0.0022. The median PFS was 5.7 months for polychemotherapy and 2.8 months for monotherapy, showing a statistically significant difference (P = 0.004). In the multivariate analysis, poor performance status, high CA19.9 levels, and monotherapy were significantly associated with worse OS. Patients treated with polychemotherapy had a 37% lower likelihood of death within the year compared to those treated with monotherapy (HR 0.58, P = 0.009).
Conclusion
Polychemotherapy may provide a survival advantage over monotherapy in the late-elderly population, although considerations for dose adjustments due to comorbidities and polypharmacy are necessary. These findings suggest that, when feasible, polychemotherapy could offer a balance between effectiveness and tolerability, potentially improving outcomes in this age group.
Introduction
The incidence of pancreatic cancer increases with age. The average age of diagnosis of pancreatic cancer is 70 years, of which a substantial proportion are older than 75 years; in fact, 24.7% of patients arrive at diagnosis from 75–84 years of age, and 12.1% are older than 80 years. 1
Pancreatic adenocarcinoma is typically diagnosed at a late, or advanced, stage of disease (stage III or IV) by virtue of the clinical features of the disease and relative asymptomaticity: up to 80%–90% of patients have unresectable cancer due to advanced stage at diagnosis. 2
Prior to 2010, the gold standard of management of unresectable or metastatic pancreatic adenocarcinoma (mPDAC) was gemcitabine monotherapy, with a clinical benefit rate of 23.8% and a median survival of only 5.65 months. 3
Significant improvement in first-line treatment of mPDAC was achieved in 2011 with the FOLFIRINOX drug combination (5-fluorouracil (5-FU), leucovorin (LV), irinotecan (CPT11), and oxaliplatin (Oxa)), which showed better survival benefits in patients with mPDAC than gemcitabine alone in the randomized phase III PRODIGE4/ACCORD11 clinical trial. 4 In 2013, another randomized phase III clinical trial, MPACT, evaluated the efficacy and safety of the Nab-paclitaxel plus gemcitabine (AG) regimen in the same disease setting, showing that the AG combination gave a significant survival benefit over gemcitabine monotherapy. 5
Therefore, based on the above 2 clinical trials, these 2 regimens have been recommended as standard first-line treatment for metastatic pancreatic cancer by several national and international guidelines. The most recent NAPLES 3 trial compared for the first time head-to-head 2 combination regimens such as NALIRIFOX (liposomal CPT11, 5-FU, LV and Oxa) and AG and demonstrated a statistically significant median survival for NALIRIFOX (11.1 months vs 9.2) with a good tolerability profile likely to make it the best future candidate in first-line treatment; median age in the 2 regimens was 64 (range 20-85; IQR 57-70) and 65 years (range 36-82; IQR 59-70), respectively. 6
Although there is more robust data supporting monotherapy, as well as capecitabine and gemcitabine, as the standard first-line treatment for unfit patients, 7 the two-drug combinations FOLFOX and FOLFIRI are not part of routine clinical practice. However, these combinations may be considered as optional approaches for patients who are not candidates for gemcitabine-based therapy or more established multi-drug regimens like FOLFIRINOX.
“Elderly” (or late-elderly) patients, defined as over 75 years old, 8 represent a peculiar group due to the high presence of comorbidities, polypharmacy, and related pharmacokinetic and pharmacodynamic alterations compared with a younger population.
Unfortunately, clinical trial data on the elderly are scarce; advanced age is an exclusion criterion in some clinical trials, 9 and ad hoc observational studies are few and fragmentary, in some cases focusing on dose modifications, 10 arguing for further prospective research to define the optimal treatment approach for the elderly with mPDAC. Rare retrospective observational studies compare monochemotherapy to combination in patients over 75 years of age, however, combination regimens do not always seem to add efficacy to monochemotherapy in the face of a higher incidence of adverse events. 11
The elderly population is in fact subject to age-related bodily and physiological changes 12 that lead to an increased risk in the use of active ingredients (drugs) with a low therapeutic index, such as chemotherapeutics. The main changes that may occur in the elderly population concern pharmacokinetic parameters, such as absorption, volume of distribution, hepatic metabolism, and excretion 13 : these parameters are affected and complicated by the possibility of pharmacokinetic or pharmacodynamic interactions. Therapies used in pancreatic cancer, including for elderly patients, involve the use of active ingredients that may give potentially toxic interactions with different drugs taken as part of polypharmacy and, often, are used in combination contributing to the potential interaction with concomitant therapies. 14
A recent systematic review 15 showed statistical significance in favor of prolonged OS for polychemotherapy in both elderly and young patient subgroups in first-line studies,4,5 confirmed by meta-analysis of first-line studies that compared single vs multi-agent chemotherapy in both elderly and young patients.
In clinical practice, the elderly and very elderly patient actually represents a high percentage of patients with pancreatic cancer accessing oncology outpatient clinics; however, there are not enough data in the literature to clearly define the benefit of first-line combination therapy vs treatment with a single chemotherapy, effectively leaving the choice of regimen to the clinician. The cut-off considered in this retrospective observational study is 75 years of age in view of the high proportion of patients in this age group in pancreatic cancer in the face of the few efficacy data of combination therapy regimens in the literature. The objective of the study is to evaluate in real world the impact of polychemotherapy (or combination therapy) vs monochemotherapy in elderly patients with inoperable pancreatic cancer treated with first-line therapy in terms of efficacy and safety.
Materials and Methods
Study Population and Data Collection
In this study, a retrospective cohort single-center analysis, we reviewed the medical records of 175 consecutive ≥75 years-old patients with a histologically confirmed diagnosis of PDAC who received first-line chemotherapy between December 2011 and January 2023 at the Oncology Unit, Piacenza General Hospital, Italy.
The study was approved by the Institutional Review Board of Piacenza Hospital and Ethical Committee Area Vasta Emilia Nord (code 746/2023/OSS/AUSLPC) and was conducted in accordance with the Helsinki Declaration. Written informed consent was obtained from each patient if possible and covered information on the study’s purpose, risks, benefits, and data confidentiality. The reporting of this study conforms to STROBE guidelines. 16 All patient details have been de-identified to ensure anonymity.
Primary study objective was overall survival (OS) in elderly patients (≥75 years) with advanced pancreatic adenocarcinoma treated with first-line monochemotherapy vs combination therapy, defined as the time from the start of first-line treatment until death from any cause or last follow-up visit. Secondary objectives were progression-free survival (PFS), defined as the time from the start of first-line treatment until first-line treatment failure, and the safety of each regimen with toxicity assessment according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. Treatment failure was considered at the time of clinical or radiological progression of the disease or death from any cause. Inclusion Criteria were patients with established diagnosis of inoperable, locally advanced or metastatic pancreatic adenocarcinoma of the pancreas on a first-line treatment regimen, patients aged ≥75 years, capable of expressing informed consent, if there is an opportunity to do so.
Pre-treatment evaluation included assessment of comorbidities (cardiac/pulmonary/hepatic, diabetes mellitus, hypertension), usual medications, ECOG performance status, tumor marker level (CA 19-9) at the time of initiation of first-line therapy, metastatic burden (1, 2 or ≥3 organs affected by metastasis), site of metastasis (liver, lung, peritoneal carcinosis, other), site of primary tumor (head, body, tail), presence of previous surgery (previous resection of primary tumor), previous therapy (adjuvant chemotherapy, radiotherapy). Tumor response was assessed by CT scan according to RECIST criteria (version 1.1). Follow-up was set at 12 months. Follow-up evaluation included tumor assessment by CT of the chest, abdomen, and pelvis with contrast medium, levels of tumor markers (CA 19-9), and toxicities regarding chemotherapy regimens used. Treatment was continued until disease progression, unacceptable toxicities, patient refusal, or medical decision. Dose reductions during treatment were not applied randomly but were based on clinical criteria, reflecting the observational nature of this study. Adjustments were typically made in steps of approximately 20% to better manage side effects, with internal guidelines prohibiting reductions beyond 50% to ensure that patients continued to receive a minimally effective dose.
The data to be recorded included: patient characteristics (age, sex, ethnicity, BMI, performance status, comorbidities), tumor characteristics (location of primary tumor in the pancreas, stage of disease at diagnosis, sites of metastasis, CA 19-9), previous treatment (surgery, radiotherapy, adjuvant chemotherapy received), toxicity assessment according to the CTCAE 5.0 and survival (overall survival and PFS from the time of development of metastatic disease).
Chemotherapeutic regimens are described in the Supplementary Material.
Mortality was identified from medical records, and living patients were censored at the time of data cutoff.
Statistical Analysis
Clinical and demographic characteristics of patients will be described by mean ± standard deviation or median and interquartile range, after checking for normality; frequencies and percentages will be used for categorical variables. Pearson’s X2 test or Fisher’s exact test will be used to compare categorical variables, and t test or Mann-Whitney test will be used to compare continuous variables. Statistical tests will be 2-tailed, and a P < 0.05 will be considered statistically significant for all analyses. The survival plot will be constructed by Kaplan-Meier analysis, and comparison between the 2 treatment arms will be done by the log-rank test. To identify potential prognostic factors of survival, Cox proportional hazards model will be used in univariate and multivariate analyses, the result will be expressed as Hazard Ratio and 95% confidence interval. The statistical software RStudio version 6.0 will be used for statistical analysis. To handle missing data, a comprehensive approach was undertaken to maintain the robustness of the retrospective analysis. The dataset was carefully inspected to identify variables with missing values. For cases where data were absent, potential reasons were systematically recorded. The extent of missingness across variables was quantified to assess its impact on the overall dataset. Regarding the calculation of the sample size, using a Risk Ratio value of 0.86 in patients treated with monotherapy and 0.69 in patients treated with combination therapy, 4 to achieve 90% power and detect a Hazard Ratio of 0.48 in the combination therapy group using a two-sided log-rank test with α = 0.05, the minimally required sample size for the monotherapy group is 51 and for the combination therapy group is 51, resulting in a total of 102 patients.
Results
175 patients were identified as eligible for the study, of whom 16 did not give consent (Figure 1). Of the 159 patients who were enrolled, 6 were excluded due to the inability to collect the required data, and 3 were excluded because their diagnoses could not be confirmed. Lastly, a total of 150 patients aged 75 years or older with pancreatic cancer were enrolled, receiving first-line therapy for locally advanced or metastatic disease. Patient demographics and baseline characteristics are summarized in Table 1. Seventy-two patients (48%) received monotherapy, while seventy-eight (52%) received polychemotherapy. The therapeutic regimens used are summarized in Table 2; among patients treated with monotherapy, the most commonly used therapy was gemcitabine (88.89%); among patients treated with polychemotherapy, the majority were treated with AG (57.69%) and FOLFIRINOX (20.51%). The most represented age group was 75-79 years (54%), with the >85 years group being the least represented (10%). Approximately 50% of the cohort was female, and the majority of patients had a performance status (PS) of 0-1 at the start of first-line therapy (86%). Most patients presented with a single metastatic site (66.7%), with the liver being the predominant site (52%), followed by peritoneal (23.3%) and lung metastasis (20%). The majority of patients (93.3%) received reduced doses, primarily owing to advanced age and secondarily to comorbidities and performance status, as detailed in Table 1. Of this group, 67.9% had their doses reduced by more than 20%. There was a statistically significant imbalance in dose reductions, with a greater proportion of patients in the polychemotherapy group receiving reductions of 20% or more compared to the monotherapy group (76% vs 58.5%, P = 0.04). Comorbidities were present in 80% of the patients, primarily hypertension (62.5%) and diabetes (50.8%). A significant difference in the frequency of diabetes was observed between the 2 treatment groups (62.3% in the polychemotherapy group vs 41.8% in the monotherapy group), while other variables were evenly distributed between the groups. Flowchart of the patient enrollment process of study cohort. Demographics and Clinical Characteristics of Patients. BMI, Body Mass Index; PS, Performance Status. The values in bold represent the results that have reached a threshold of statistical significance (e.g., p < 0.05). Therapeutic Regimens. Cap-gem, Capecitabine plus gemcitabine; AG, Nab-paclitaxel plus gemcitabine.


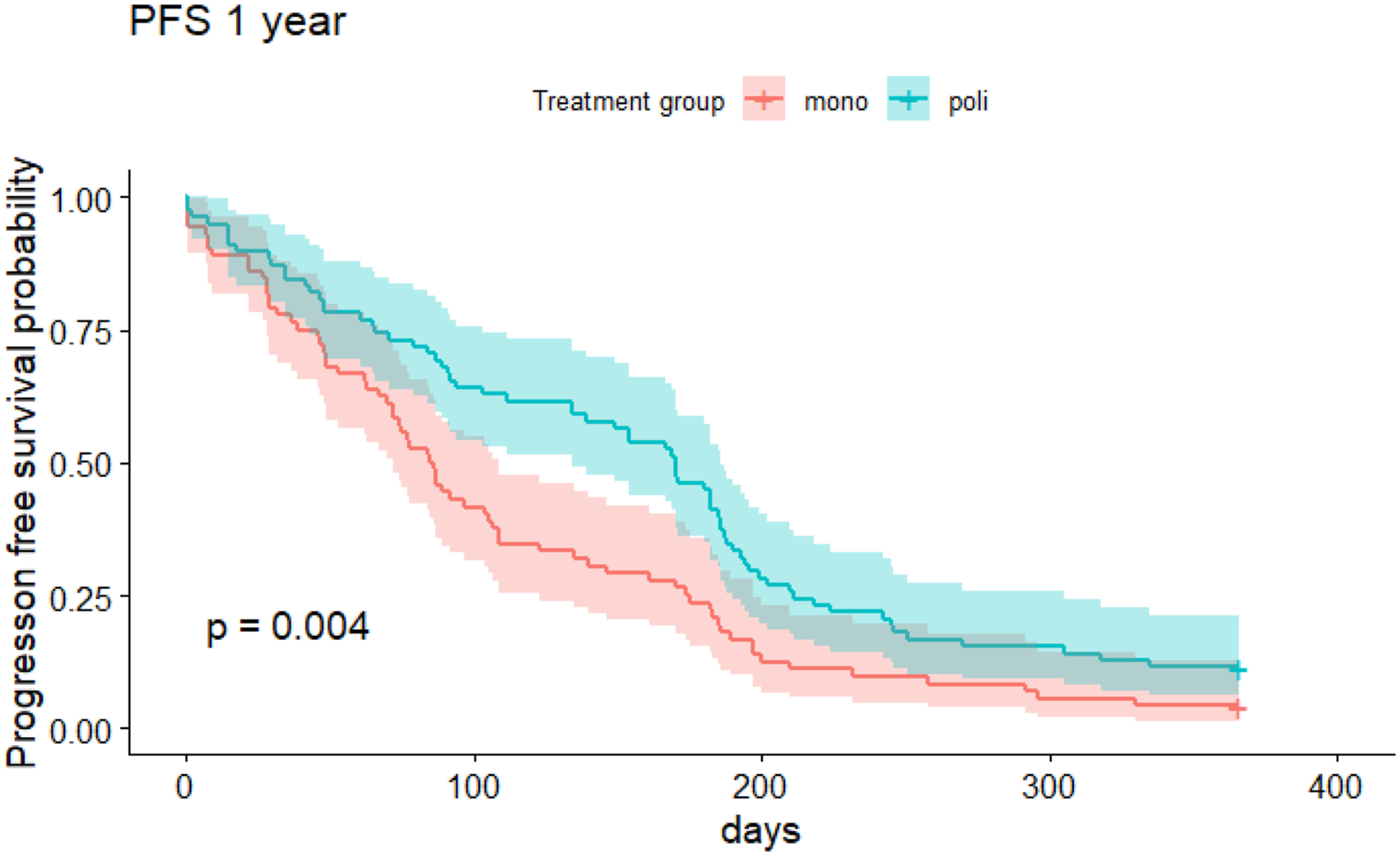
The median overall survival (OS) from the start of therapy was approximately 4.7 months (95% CI 3.6-6.2) in the monotherapy group and 8.2 months (95% CI 6.3-10.4) in the polychemotherapy group (Figure 2). The median progression-free survival (PFS) was approximately 2.8 months (95% CI 2.4-3.6) in the monotherapy group and 5.7 months (95% CI 4.5-6.2) in the polychemotherapy group (Figure 3). Kaplan-Meier curves for 1-year overall survival for patients receiving monochemotherapy vs polychemotherapy. Kaplan-Meier curves for 1-year progression free survival for patients receiving monochemotherapy vs polychemotherapy.
Univariate and Multivariate Analysis of One-Year Overall Survival.

BMI, Body Mass Index; PS, Performance Status; Ref, reference. The values in bold represent the results that have reached a threshold of statistical significance (e.g., p < 0.05).
Table of Toxicities According to CTCAE 5.0.

Discussion
Our investigation uniquely contributes to the literature by focusing on the “late-elderly” population, a group often underrepresented in pancreatic cancer trials. The findings underscore the necessity of developing tailored therapeutic strategies that consider both the potential benefits and the heightened risk profile associated with older patients.
Polychemotherapy demonstrated a marked improvement in both overall survival and progression-free survival compared to monotherapy. This aligns with the advancements in treatment protocols, as corroborated by the PRODIGE4/ACCORD11 and MPACT trials, which have established the superiority of combination regimens in younger patient cohorts.4,5 However, the translation of these benefits to the late-elderly demographic is not straightforward due to their unique clinical profile, characterized by increased comorbidities and polypharmacy, which necessitate cautious dose adjustments and vigilant management of therapy-related toxicities.12,14
A recent review examined several studies, although primarily sourced from retrospective small case series, unplanned subset analyses, and case reports, demonstrating that the aging process alone does not eliminate the possibility of utilizing chemotherapy; older patients appear to derive similar benefits from anticancer treatments as their younger counterparts, albeit with a frequent need for reduced-dose chemotherapy and enhanced supportive therapy to maintain tolerability. 17 In a Dutch nationwide study on metastatic pancreatic cancer patients, examining chemotherapy use and survival across various elderly age groups, those aged 70-74 receiving chemotherapy showed similar tumor verification rates, timing, early mortality, and overall survival as patients under 70. However, the limited and highly selected group of elderly patients aged over 75 treated with chemotherapy exhibited notably poor survival outcomes. 18 Our survival data are slightly below those reported in the real-life literature on first-line chemotherapy, 19 considering that most studies focus on a younger population and data limited to this subpopulation are extremely scarce and thus too heterogeneous to be adequately compared.18,20
The association of higher CA19.9 levels with poorer survival outcomes further supports the use of this biomarker as a prognostic tool in clinical settings. Our data are in line with the literature, which has shown that elevated CA19-9 levels predict poor prognosis in PDAC patients, correlating not only with lower resectability but additionally survival rates even in advanced stages of the disease.21,22 This is confirmed by a meta-analysis highlighting its robustness as a prognostic marker before and after treatment. For late-elderly patients with inoperable pancreatic cancer, CA19-9 levels could help identify those with potentially less unfavorable prognoses who might benefit more from aggressive polychemotherapy. 23
The significant variance in diabetes prevalence between the treatment groups could suggest a possible interaction between metabolic disorders and the efficacy of chemotherapy, meriting further exploration. Notably, this condition is more prevalent among patients treated with monotherapy; however, it does not assume significance in affecting outcomes in both univariate and multivariate analyses. This suggests that while metabolic conditions may influence treatment choices, their direct impact on clinical outcomes requires further investigation to understand fully.
The high percentage of patients receiving dose reductions, especially those with dose reductions greater than 20%, suggests that tolerability is a critical concern in this age group. It is important to note that the significant imbalance in dose reductions between the polychemotherapy and monotherapy groups did not appear to negatively impact the overall survival outcomes observed in our study. Specifically, despite the higher rate of substantial dose reductions in the polychemotherapy group, these patients still demonstrated superior survival outcomes compared to the monotherapy group. This suggests that the therapeutic benefit of polychemotherapy may persist even when administered at significantly reduced doses. Consequently, we did not find it necessary to perform a propensity score analysis to rebalance the population, as the observed survival benefit in the polychemotherapy group remained robust despite the dose reductions. This finding underscores the importance of personalized treatment plans that consider both the efficacy and the tolerability of chemotherapy to optimize patient outcomes. The toxicities observed during the study show a higher prevalence in the polychemotherapy treatment group. This finding is consistent with the literature evidence. 15 The most prevalent grade 3-4 event was febrile neutropenia, accounting for 19.23% of the severe toxicities observed in the polychemotherapy treatment group, particularly in the GA-treated population where it represented the 26.6% of grade 3-4 toxicities for this therapeutic regimen. The data shows a higher incidence of this toxicity compared to the literature data, 24 likely due to the advanced age of the study population. The main treatment regimens in which toxicities occurred were gemcitabine, FOLFIRINOX, and GA. Compared to literature data, our population showed a higher incidence of diarrhea for the 3 treatment regimens, while a lower incidence of neutropenia and peripheral neuropathy, particularly in the GA-treated group. The other adverse events are in line with what has been shown in the literature. 11 These data confirm the complexity of managing elderly patients, also due to their frailty.
The data from this study notably underscore the practical challenges in treating late-elderly patients. While the NAPLES 3 trial suggested potential for newer regimens like NALIRIFOX to become preferred first-line treatments due to their tolerability and survival benefits, unfortunately, the average age in these trials often does not reflect the upper age spectrum of patients seen in routine clinical practice. 6 It would be advisable to consider this subgroup in clinical trials, as they represent a significant percentage of patients with inoperable pancreatic pathology, or to implement real-life studies in the late-elderly.
One of the primary limitations of our retrospective study is the lack of a proper geriatric tool assessment to identify the frailest patients and guide the clinician in selecting the chemotherapy regimen. The insufficient focus on conducting geriatric assessments for elderly patients diagnosed with pancreatic cancer is evident in the literature. This oversight could lead to instances of overtreatment, as indicated by the results of a prospective phase IV clinical trial that explores the consequences and implications of insufficient attention to the specific needs and considerations of older individuals with pancreatic cancer, highlighting potential areas for improvement in their medical care. 25 The significance of employing a geriatric assessment is also investigated in other studies,26,27 yet the findings remain limited. Therefore, as there is no established tool in the literature, we did not explore this possibility. Although this study suffers from limitations typical of retrospective designs, efforts were made to mitigate these issues through the use of multivariate analysis, which helps to control for potential confounders. While a statistically significant imbalance existed in dose reductions and diabetes between the mono and polychemotherapy groups, these factors did not significantly affect overall survival outcomes. The survival benefit observed in the polychemotherapy group remained robust, reinforcing the validity of our findings despite these limitations. Due to the retrospective design of the study, it was not possible to accurately determine the number of patients who discontinued treatment due to toxicity, as this information was not consistently available in the medical records
Nonetheless, the absence of randomization might have allowed for additional, unidentified confounders to affect the results. These structural limitations highlight the necessity for more rigorous, prospective studies to confirm our observations.
Conclusion
The findings suggest that the lower risk of progression and mortality associated with polychemotherapy may indicate its potential as a more effective therapeutic strategy for managing advanced pancreatic cancer in elderly populations. However, these results highlight the need for prospective studies focusing exclusively on this age group to better understand the balance between efficacy and tolerability of various chemotherapeutic regimens. Future oncological care for the elderly should aim for personalized, patient-centered approaches that consider the complexities and unique challenges of treating this vulnerable population.
Supplemental Material
Supplemental Material - Comparing Combination vs. Monochemotherapy in Late-Elderly Patients with Advanced Pancreatic Cancer: Insights from a Single-Center Study in China
Supplemental Material for Comparing Combination vsvs. Monochemotherapy in Late-Elderly Patients with Advanced Pancreatic Cancer: Insights from a Single-Center Study by Elena Orlandi, Chiara Citterio, Rebecca Chinelli, Ilaria Dotti, Elena Zaffignani, Claudia Biasini, Elisa Anselmi, Gabriele Cremona, and Stefano Vecchia in Cancer Control.
Abbreviations
pancreatic ductal adenocarcinoma
overall survival
progression-free survival
performance status
metastatic pancreatic ductal adenocarcinoma
5-fluorouracil
leucovorin
irinotecan
oxaliplatin
Nab-paclitaxel plus gemcitabine
Common Terminology Criteria for Adverse Events
Footnotes
Authors Contributions
EO, CC, RC, ID, EZ, CB, GC, EA and SV, assessed the conception and design of the manuscript, and were involved in acquisition, analysis and interpretation of data. SV, ID and RC contributed to the investigation and data curation. EZ, CB and EA drafted the article. EO, CC, RC, ID, EZ, CB, GC, EA and SV critically revised the manuscript for important intellectual contents. EO and SV reviewed and edited the final version of the manuscript. EO and SV confirm the authenticity of all the raw data. All authors have read and approved the final version of the manuscript, and participated sufficiently in the work to take public responsibility for appropriate portions of the content. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study is supported by Associazione Malato Oncologico Piacentino (AMOP). We also extend our gratitude to Poisetti Piergiorgio, MD, for his valuable contributions.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
