Abstract
Introduction
Cancer is a significant worldwide health problem, and targeted drugs like Cabozantinib and Atezolizumab offer new therapeutic options. These drugs enhance patient survival by specifically targeting cancer cells while minimizing damage to healthy tissues. The current research investigates the effectiveness of employing the combination of Cabozantinib and Atezolizumab vs single-agent Cabozantinib.
Methods
In this study, we searched databases including PubMed/Medline, Scopus, Embase, Cochrane, and Web of Science up to the end of January 2025. The results were categorized into two groups: the intervention group (Cabozantinib plus Atezolizumab) and the control group (Cabozantinib monotherapy) in cancer patients. We evaluated variables such as efficacy outcomes, including Objective Response Rate (ORR) and Disease Control Rate (DCR), as well as adverse events (safety). Data analysis was performed using random-effects models, and Relative Risk (RR) was reported as the effect size.
Results
Three clinical trials were included in this study. Regarding efficacy outcomes, there was no statistically significant difference in ORR between the intervention group and the control group (RR = 1.11, 95% CI: 0.76-1.61). Similarly, DCR did not differ significantly between groups (RR = 0.98, 95% CI: 0.94-1.03). In terms of safety, the combination therapy was associated with a statistically significant reduction in four treatment-related adverse events: diarrhea (RR = 0.91, 95% CI: 0.83-0.99), nausea (RR = 0.79, 95% CI: 0.65-0.95), vomiting (RR = 0.70, 95% CI: 0.52-0.96), and hypokalemia (RR = 0.28, 95% CI: 0.19-0.40).
Conclusion
The findings suggest that the combination of Cabozantinib and Atezolizumab does not offer significant improvement in treatment efficacy compared to Cabozantinib alone. However, the combination may be associated with a lower incidence of certain chemotherapy-related adverse events. These exploratory findings may inform future research on treatment strategies for patients who experience intolerance to Cabozantinib monotherapy.
Plain Language Summary
Cancer treatment often involves using drugs that target the cancer cells, but these treatments can come with side effects. One such treatment is Cabozantinib, which is approved for several types of cancer. Recently, it has been tested with a second drug, Atezolizumab, to see if the combination could be more effective in treating cancer and also reduce side effects. In our study, we looked at research from three clinical trials that compared Cabozantinib alone to the combination of Cabozantinib and Atezolizumab. We wanted to understand if adding Atezolizumab would improve treatment results, like how many patients' cancer shrank or stopped growing (this is called the Objective Response Rate, or ORR) and how well the disease was controlled (the Disease Control Rate, or DCR). We also looked at the side effects of these treatments. The combination therapy did not show a clear improvement in shrinking or controlling the cancer compared to Cabozantinib alone. However, the combination therapy did lead to fewer side effects, such as nausea, vomiting, diarrhea, and low potassium levels, making it a better option for patients who struggle with these side effects from Cabozantinib alone. Our findings suggest that while the combination treatment may not be better at treating cancer, it could be a good choice for patients who cannot tolerate Cabozantinib on its own. More research is needed to better understand who might benefit the most from this combination therapy, especially for patients with certain types of tumors.
Introduction
Cancer arises from the uncontrolled growth of cells in various organs of the body and their ability to spread to other parts. 1 According to World Health Organization (WHO) estimates, 20 million new cancer cases and 9.7 million cancer deaths were registered in 2022, with one in every five individuals developing cancer during a lifetime. 2 Treating cancer has been a challenging issue for centuries. For many years, the therapeutic options were only surgical resection of tumor tissues, accompanied by radiotherapy and chemotherapy. 3 Nonetheless, these therapeutic strategies possess tremendous adverse events. Surgical interventions are invasive in nature, whereas radiotherapy is able to damage neighboring healthy tissues. On the other hand, chemotherapy targets healthy cells, especially those with high growth and divisional rates, and can ultimately result in drug resistance. 4 Because of these limitations, researchers have continuously looked for other cancer treatments and new therapeutic agents. One such approach is targeted drug therapy, also known as molecularly targeted drugs, precision medicines, or molecularly targeted therapies. 5 Unlike conventional chemotherapy, targeted therapies specifically attack cancer cells, distinguishing them from normal cells and interfering with cellular growth processes. By minimizing damage to healthy tissues, these treatments inhibit tumor progression and significantly improve survival rates in certain cancers.6,7 Targeted agents fall into different categories, with monoclonal antibodies and small molecule inhibitors being the most commonly used types.
Cabozantinib, classified as a small molecule inhibitor, is a multi-kinase inhibitor with a broad spectrum of anti-tumor activity. 8 It primarily targets MET (Mesenchymal Epithelial Transition factor) and VEGFR (Vascular Endothelial Growth Factor Receptor). The U.S. Food and Drug Administration (FDA) has approved Cabozantinib since 2012 for the treatment of advanced renal cell carcinoma (RCC), hepatocellular carcinoma (HCC), and progressive metastatic medullary thyroid cancer (MTC). 9 On the other hand, Atezolizumab is a monoclonal anti-PD-L1 (Programmed Death-Ligand 1) antibody that restores anti-tumor immune responses and has been approved for treating HCC, melanoma, and lung cancer.10,11 Recently, its combination with Cabozantinib has shown promising anti-cancer effects, suggesting its potential as a therapeutic regimen for specific cancers. 12
A study on NSCLC by Rittmeyer et al. confirmed that Atezolizumab improved patient survival and reduced toxicities related to treatment when compared to Docetaxel. 13 Similarly, research by Gregor et al on RCC established that Atezolizumab in combination with Cabozantinib improved treatment response, disease control, and survival. 14 Also, El-Khoueir et al showed that the combination of Cabozantinib and Atezolizumab had synergistic effects in modulating the tumor microenvironment and enhancing immune responses in multiple cancers, such as hepatocellular carcinoma (HCC), thus making it a potential first-line treatment for this neoplasm. 15
Overall, early-phase trials demonstrate that the pairing of Cabozantinib with Atezolizumab can enhance clinical outcomes across various tumor types. However, the existing literature is fragmented, with individual studies (eg, COSMIC-021, 16 COSMIC-312, 17 ) focusing on specific cancer types, and no prior meta-analysis has synthesized this evidence across multiple malignancies. Given the increasing interest in using this combination in broader clinical settings, a pooled analysis of its efficacy and safety across tumor types is warranted. Our study aims to address this gap by conducting a systematic review and meta-analysis that compares Cabozantinib monotherapy with its combination with Atezolizumab in terms of both therapeutic efficacy and adverse events. This approach provides a more integrated understanding of its clinical impact and informs future research and clinical decision-making.
Materials and Methods
Study Design
This study was conducted as a systematic review and meta-analysis. PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines were followed in the reporting of this study. 18 We have included the completed PRISMA checklist in Appendix 1. Additionally, the AMSTAR (Assessment of Multiple Systematic Reviews) criteria were utilized to ensure methodological rigor in the study design. 19 PROSPERO 2025 registration code: CRD420251017051. This systematic review and meta-analysis was approved by the Ethics Committee of Babol University of Medical Sciences, Iran, under the approval code IR.MUBABOL.HRI.REC.1403.334. Since this study exclusively involved analysis of data from previously published clinical trials and did not involve direct experimentation on human subjects or the collection of human tissue or samples, informed consent and additional ethical approvals were not required.
Search Strategy and Data Sources
Several databases were comprehensively searched for literature, including PubMed/Medline, Scopus, Embase, Cochrane Library, Web of Science, and Google Scholar, as well as clinical trial platforms such as ClinicalTrials.gov and the International Clinical Trials Registry Platform (ICTRP). Searches covered the period from the start of these databases to January 31, 2025. To identify additional relevant studies, gray literature was also reviewed.
The search was conducted without any language or time restrictions. The key search terms included: “Cabozantinib,” “Atezolizumab,” “Neoplasms,” and “Cancer.” Furthermore, a multi-stage process was implemented to refine the search terms and design the search syntax. As a result of this strategy, free-text keywords and Medical Subject Headings (MeSH) terms were utilized to ensure that relevant literature was extensively covered (Appendix 2).
Study Selection Process
In the following steps, two independent researchers generated search syntax for each database and imported search results into EndNote (version 2019). EndNote software enabled the detection and removal of duplicate records. The remaining articles then underwent independent screening by the two reviewers to determine the eligible studies. Any disagreements in judgment between the two reviewers were settled by a third investigator who had the responsibility of overseeing the study design.
The inclusion criteria for this study consisted of interventional studies, specifically published clinical trials with full-text availability. The selected studies had to include two groups of patients: one receiving a combination therapy of Cabozantinib and Atezolizumab, and the other receiving Cabozantinib monotherapy. In this study, patients treated with Cabozantinib and Atezolizumab were classified as the intervention group, while those receiving Cabozantinib alone were considered the control group.
Studies were excluded if they were review articles, clinical trials without a control group, book chapters, conference abstracts, or in vivo/in vitro studies. Additionally, cross-sectional, case-control, and cohort studies were not included. Research that involved a control group treated with medications other than Cabozantinib or Atezolizumab was also excluded. Furthermore, studies that assessed a broader category of drugs rather than specifically evaluating Cabozantinib and Atezolizumab were not considered. 20
Data Extraction and Variables
After identifying the final set of articles, data extraction was conducted independently by two researchers. In case of any discrepancies, the responsible investigator resolved the differences. The extracted data were recorded using Microsoft Excel 2019 (Version 2019; Microsoft, Redmond, WA, USA).
The study variables were categorized into two main groups: efficacy outcomes and adverse events (safety). In analyzing efficacy outcomes, we considered the objective response rate (ORR) and disease control rate (DCR) as the primary variables. These variables were extracted from the included studies based on RECIST v1.1 (Response Evaluation Criteria in Solid Tumors) guidelines as assessed by the investigators. RECIST v1.1 is the most recent guideline for evaluating treatment response in solid tumors. 21 Other efficacy-related variables, such as Duration of Response (DOR), Progression-Free Survival (PFS), and Overall Survival (OS), were not included due to incomplete primary data or lack of final reports.
For adverse events (safety), we extracted data on 20 types of Treatment-Emergent Adverse Events (TEAEs), including fatigue, hypertension, decreased appetite, nausea, vomiting, diarrhea, increased ALT, stomatitis, constipation, abdominal pain, increased AST, hypothyroidism, asthenia, rash, proteinuria, Palmar-Plantar Erythrodysesthesia Syndrome (PPES), anemia, pyrexia, hypokalemia, and hypomagnesemia. The extracted adverse events were classified as TEAEs, as these were the most commonly reported across the included studies. According to the ICH E9 guideline, TEAEs are defined as adverse events that emerge during treatment and were either not present before treatment or worsened compared to the pre-treatment condition. 22 Additionally, adverse events such as increased bilirubin, increased amylase, and ascites were excluded from the analysis due to insufficient data.
The final Excel dataset included information on author name, publication date, country, trial name, year of data collection, age of intervention and control groups, number of male patients in each group, dosage information, cancer type, number of patients in each group, efficacy outcome variables, and adverse events. This structured dataset facilitated a comprehensive analysis of treatment efficacy and safety.
Quality Appraisal (Risk of Bias Assessment)
For quality assessment, we used the revised Cochrane tool for assessing randomized trials (Cochrane RoB 2.0). It examines the risk of bias in a systematic way in five fundamental domains, ie, the randomization process, deviations from intended interventions, missing outcome data, outcome measurement methods, and selection of reported results. Five domains are rated as low, high, or some concern based on pre-established criteria. By applying the same three-tier grading scale to all five domains, an overall score is also calculated. 23
Statistical Analyses
The data were analyzed using a statistical program, and STATA version 17.0 (Stata Corporation, College Station, TX) was utilized for the analysis. For managing heterogeneity between studies, the random effects model was applied. In order to compare the outcome and variables, relative risk (RR) was used, along with a 95% confidence interval (CI). Furthermore, forest plots were employed to display the effect sizes in each of the studies and the pooled estimates. A P-value below 0.05 was taken as significant statistically. Heterogeneity among studies was assessed using Cochran’s test and Higgins’ I2 test. In this evaluation, I2 values between 25% and 50% indicate moderate heterogeneity. Values below and above this level are considered low and high heterogeneity, respectively. 24 Given the inherent qualitative differences between studies, we generally applied the random effects model for all analyses when heterogeneity was present. The leave-one-out method was also used for sensitivity analysis, in which individual studies were systematically removed to assess their impact on the overall effect size. Additionally, a subgroup analysis was conducted based on cancer grade for the reported adverse event variables. In order to determine publication bias, funnel plots and Egger’s weighted regression tests were used. 25 The certainty of evidence for each primary outcome (ORR and DCR) was assessed using the GRADE approach, considering domains such as risk of bias, inconsistency, indirectness, imprecision, and publication bias. 26 It was assumed that no significant publication bias existed if the P-value was greater than 0.05.
Results
Study Selection
A total of 1975 articles were identified through searches in international databases. After removing duplicates, 124 articles remained for title and abstract screening. Following this step, 17 articles were selected for full-text review. After thoroughly assessing the full texts, three articles (comprising nine records) were included in the final analysis. Furthermore, we reviewed the reference lists of the included studies to determine whether any additional studies were relevant. The study selection process is illustrated in Figure 1. Preferred Reporting Items for a Systematic Review and Meta-Analysis (PRISMA) 2020 Flow Diagram for New Systematic Reviews, Including Database and Other Source Searches
Study Characteristics and Quality Appraisal (Risk of Bias Assessment)
Demographic Characteristics of the Included Studies in the Systematic Review and Meta-Analysis

NA; Not mentioned, SD; Standard deviation.
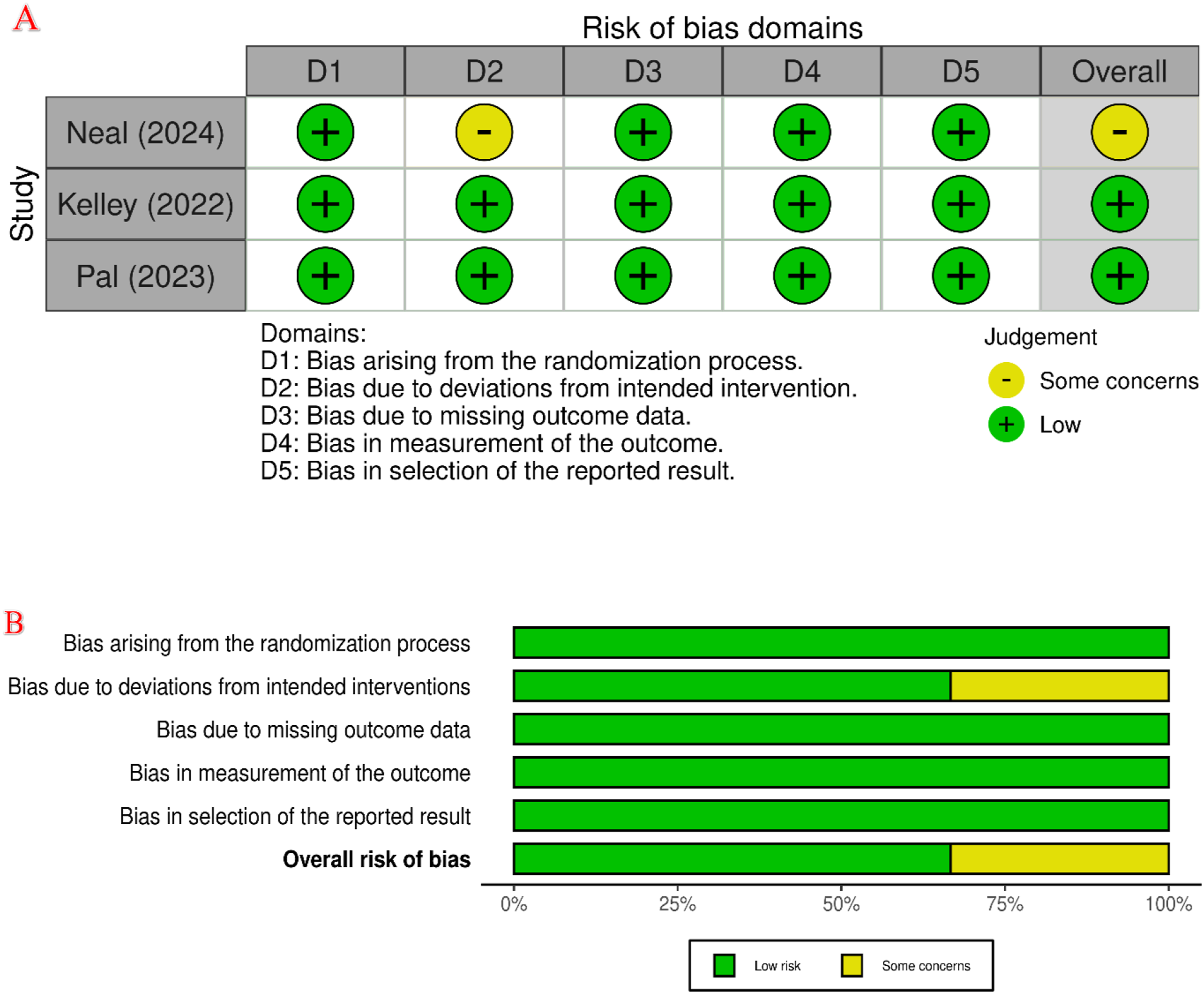
Risk of Bias Assessment; Risk of Bias Assessment of Included Randomized Control Trials (RCTs). (A) Risk of Bias Summary of all Included RCTs; (B) Risk of Bias
Results of the Meta-Analysis
Intervention vs Control Group Outcomes
Meta-Analysis Results and Heterogeneity of the Efficacy of Cabozantinib Monotherapy vs Its Combination with Atezolizumab in Cancer Patients


Forest Plot of the Efficacy of Cabozantinib Monotherapy vs Cabozantinib in Combination With Atezolizumab in Cancer Patients
Intervention and Control Group Adverse Events
In terms of safety outcomes, which involved the assessment of 20 different adverse events across the two groups, most adverse events were less frequent in the intervention group (Cabozantinib + Atezolizumab) compared to the control group (Cabozantinib monotherapy). Among these, four adverse events were statistically significantly reduced in the intervention group. The rates of diarrhea, nausea, vomiting, and hypokalemia were significantly lower in the intervention group compared to the control group, as follows: Diarrhea: 9% lower (RR = 0.91, 95% CI: 0.83-0.99, P = 0.043); Nausea: 21% lower (RR = 0.79, 95% CI: 0.65-0.95, P = 0.012); Vomiting: 30% lower (RR = 0.70, 95% CI: 0.52-0.96, P = 0.026); Hypokalemia: 72% lower (RR = 0.28, 95% CI: 0.19-0.40, P < 0.001).
Meta-Analysis Results and Heterogeneity of Adverse Events (Safety) of Cabozantinib Monotherapy vs Its Combination With Atezolizumab in Cancer Patients

PPES; Palmar-Plantar Erythrodysesthesia Syndrome, CI; Confidence interval.
*Bolded values indicate statistically significant findings (P < 0.05).
Publication Bias
Finally, we constructed funnel plots to assess the presence of publication bias for the primary outcomes of the study, namely ORR and DCR. The results of Egger’s test indicated evidence of publication bias for ORR (bias: 2.16, SE = 1.06, P = 0.042), whereas no significant bias was detected for DCR (bias: 2.15, SE = 1.33, P = 0.107) (Appendix 6).
Certainty of Evidence
Based on the GRADE assessment (Appendix 7), the certainty of evidence was rated as very low for ORR and low for DCR, primarily due to imprecision, limited number of studies, and potential publication bias.
Discussion
This study investigated the effects of adding Atezolizumab to Cabozantinib in the treatment of various cancers, including RCC, HCC, and lung cancer. According to the meta-analysis of three studies, the addition of Atezolizumab to Cabozantinib did not result in a significant improvement in ORR or DCR compared to Cabozantinib monotherapy. However, in terms of safety outcomes, the Atezolizumab-containing regimen (intervention group) demonstrated fewer adverse events, particularly in common chemotherapy-related adverse events. Specifically, nausea (21% reduction), vomiting (30% reduction), diarrhea (9% reduction), and hypokalemia (72% reduction) were significantly lower in the intervention group. Although hypertension did not show an overall significant reduction, high-grade hypertension was reduced by 38% in the Atezolizumab group. Pyrexia was the only adverse event that occurred more frequently in the Atezolizumab group, though it was not statistically significant. These results suggest that Atezolizumab was helpful in managing patients who experienced chemotherapy toxicity and had difficulty tolerating Cabozantinib monotherapy. Moreover, the lack of significant improvement in ORR and DCR highlights the need for further investigation of Atezolizumab in specific patient subgroups, particularly those with PD-L1-positive tumors. This approach may assist in generating hypotheses for identifying patient subgroups who could potentially respond differently to immune checkpoint inhibition, particularly based on biomarker status or underlying tumor biology. The findings also underscore the clinical importance of managing treatment-related adverse events, suggesting that Atezolizumab may offer a more tolerable profile for patients who struggle with Cabozantinib-related adverse events.
Atezolizumab, a monoclonal antibody inhibitor of PD-L1, is effective against a wide variety of cancers. This drug has been extensively studied in urothelial carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer, RCC, triple-negative breast cancer (TNBC), melanoma, and colorectal cancer.28-38 PD-L1 plays a key role in immune evasion, allowing cancer cells to escape immune surveillance by increasing PD-L1 expression.28,39 Consequently, Atezolizumab has shown greater activity in tumors with PD-L1 expression ≥1% on tumor-infiltrating immune cells. In several landmark trials, Atezolizumab improved key clinical outcomes. In metastatic NSCLC, it significantly prolonged overall survival (OS) compared to platinum-based chemotherapy. 40 Similarly, in unresectable HCC, the IMbrave150 study showed that Atezolizumab plus Bevacizumab improved ORR, DCR, OS, and PFS relative to Sorafenib. 41 The IMpower150 trial also reported enhanced ORR, OS, and PFS with the addition of Atezolizumab to Bevacizumab, Carboplatin, and Paclitaxel in metastatic non-squamous NSCLC. 42 Comparable survival advantages were observed in extensive-stage small cell lung cancer in the IMpower133 trial, 32 and improved PFS and ORR were noted in TNBC when Atezolizumab was added to nab-Paclitaxel. 33 Despite promising findings in these settings, our meta-analysis did not demonstrate statistically significant improvements in ORR or DCR with the Atezolizumab-Cabozantinib combination compared to Cabozantinib alone. The lack of efficacy signal in our analysis may reflect several factors, including limited sample size, pharmacokinetic interactions, or tumor heterogeneity across the included studies. Notably, while our pooled analysis showed a reduction in specific chemotherapy-related adverse events, these findings must be interpreted cautiously in the absence of corresponding efficacy improvements. The differential results across studies may also be influenced by PD-L1 expression status. Prior evidence suggests that PD-L1-positive tumors may be more likely to respond to immune checkpoint inhibitors.43,44 However, PD-L1 assessment was inconsistently reported across the included trials, precluding stratified analyses. Therefore, further research is needed to evaluate whether the combination of Atezolizumab and Cabozantinib may confer clinical benefit in biomarker-defined populations, such as patients with PD-L1-positive tumors or immune-resistant phenotypes.
A significant finding in our study was the reduction in adverse events, including nausea, vomiting, diarrhea, and high-grade hypertension, when Atezolizumab was combined with Cabozantinib. In contrast, most previous studies examining the addition of Atezolizumab to cancer treatment regimens have reported similar rates of adverse events between the intervention and control groups. For instance, in the IMvigor130 trial, which evaluated the addition of Atezolizumab to platinum-based chemotherapy in first-line treatment of metastatic urothelial carcinoma, although the Complete Response (CR) rate doubled in the Atezolizumab-containing group, the adverse event profile remained similar to the control group without Atezolizumab. 30 Cabozantinib has been associated with multiple adverse events, including diarrhea, loss of appetite, fatigue, nausea, vomiting, and hypertension, in various clinical trials.45,46 In the CELESTIAL trial, which compared Cabozantinib to placebo in advanced HCC, the Cabozantinib group exhibited a higher incidence of nausea, vomiting, hypertension, and palmar-plantar erythrodysesthesia syndrome (PPES). 47 It has been demonstrated that Cabozantinib-induced VEGF inhibition can lead to increased blood pressure as a compensatory response. 48
In our study, the addition of Atezolizumab to Cabozantinib resulted in a significant reduction in adverse events such as nausea, vomiting, diarrhea, and high-grade hypertension. This observation highlights the potential of combining immune checkpoint inhibitors (ICIs) like Atezolizumab with targeted therapies such as Cabozantinib. Such combinations may not only enhance treatment efficacy but also reduce treatment-related toxicity, providing a more favorable safety profile for patients who might otherwise struggle with the adverse events of Cabozantinib monotherapy. The observed reduction in treatment-related toxicities without a corresponding improvement in efficacy may be attributable to several underlying pharmacological and immunological mechanisms. Atezolizumab, a PD-L1 inhibitor, modulates T-cell activity without directly targeting tumor cells, thereby potentially reducing the collateral damage to non-cancerous tissues often observed with conventional chemotherapy. Additionally, immunotherapy may alter the tumor microenvironment in a way that dampens pro-inflammatory cytokine cascades responsible for gastrointestinal toxicities such as nausea and diarrhea.39,49 It is also possible that Cabozantinib’s inhibition of VEGFR and MET pathways may synergize with immune checkpoint blockade to selectively affect tumor cells while minimizing off-target toxicities. 8 While these hypotheses require validation in mechanistic studies, they provide a plausible rationale for the clinical observations in our analysis. This study, focusing on Atezolizumab and Cabozantinib combined in cancer treatment, holds significant importance, particularly in the context of reducing chemotherapy-related adverse events. While the results did not indicate a substantial improvement in treatment response or disease control, a statistically significant reduction was observed in common chemotherapy-related adverse events, including nausea, vomiting, and diarrhea. This finding is especially relevant for patients who struggle to continue standard treatments due to adverse events.
Previous studies have generally reported that Atezolizumab enhances treatment response and overall survival in various cancers compared to control groups. However, the differences observed in our study may be attributed to the unique pharmacological interactions of the Cabozantinib-Atezolizumab combination, which may yield variable results across different cancer types. A key strength of this study is its multicenter design conducted across different countries, significantly enhancing the external validity of the findings. Furthermore, the detailed analysis of treatment-related adverse events based on severity (grade) allowed for a more precise evaluation of the therapeutic impact of Atezolizumab and Cabozantinib combination therapy. While previous research has mainly focused on evaluating Atezolizumab in combination with chemotherapy, this study effectively highlights its role in mitigating specific adverse events such as diarrhea and nausea when combined with Cabozantinib. Another notable strength is the use of meta-analysis for data synthesis, providing a comprehensive and robust assessment of the therapeutic impact. This approach not only offers a better understanding of the clinical effects of the combination therapy but also paves the way for future investigations into its broader applicability. Based on our findings, future studies should focus on evaluating the combination of Atezolizumab and Cabozantinib in PD-L1-positive tumors and in patients who exhibit chemotherapy resistance. This direction may assist in identifying patient subgroups, particularly those with PD-L1-positive or chemotherapy-resistant tumors, who could potentially exhibit differential responses to this combination, warranting further investigation in stratified clinical trials.
Study Limitations and Future Directions
As we interpret the results of this study, we must acknowledge its limitations. First, the findings of this meta-analysis should be interpreted with caution due to the limited number of included trials (n = 3), which restricts the statistical power and may affect the generalizability of the results, particularly in the subgroup analyses of adverse events. This limitation is further reflected in the GRADE assessment, which rated the certainty of evidence as very low for ORR and low for DCR due to imprecision and publication bias. Although we employed a random-effects model and none of the I2 values exceeded 80%, the small number of studies and variability in event rates and sample sizes may still limit the robustness of pooled estimates. Subgroup analyses based on toxicity severity (Grades 1-2 vs 3-4) were conducted to address this variability, but additional trials are needed to validate these findings. Second, our analysis pooled data from heterogeneous cancer types, including RCC, HCC, and NSCLC. While this approach was necessary due to the limited number of available studies, such clinical variability may obscure cancer-specific treatment effects. Moreover, due to the scarcity of trials and insufficient subgroup data, stratified meta-analyses based on cancer type or PD-L1 status could not be performed. This limitation underscores the need for future studies focusing on more homogeneous cancer populations with biomarker-based stratification. Also, differences in treatment settings, such as dosing regimens and follow-up duration, introduce further variability that complicates the interpretation of both efficacy and safety outcomes. These inconsistencies limit the external validity and comparability of the results. Third, the majority of included studies were relatively recent and lacked long-term follow-up data, limiting the ability to assess sustained treatment outcomes and late-onset toxicities. Short intervention periods may affect conclusions regarding long-term efficacy and safety. Finally, PD-L1 assessment remains a significant challenge, as variations in evaluation methods across studies may lead to inconsistent interpretations. Standardizing PD-L1 testing protocols in future trials would improve the reliability and comparability of results. To address these limitations, future research should involve larger, cancer-specific patient cohorts, uniform methodologies, longer follow-up durations, and standardized biomarker assessments to enhance both the validity and applicability of findings.
Further analysis stratifying patients into PD-L1-positive or chemotherapy-resistant subgroups may help determine whether the Atezolizumab-Cabozantinib combination elicits differential clinical responses in specific populations, which could guide future biomarker-driven trials. Moreover, future studies may explore the potential of Atezolizumab in combination with other targeted agents or immunotherapies, in order to determine optimal strategies for patients with diverse molecular and clinical profiles. However, such applications were beyond the scope of this study and are based solely on prior literature hypotheses.
Conclusion
This research indicated that Atezolizumab and Cabozantinib together did not significantly affect ORRs or DCRs as compared to Cabozantinib alone. The Atezolizumab and Cabozantinib combination together, however, reduced some common cytotoxic chemotherapy-related toxicities - nausea, vomiting, and diarrhea. Although no significant improvement in ORR or DCR was observed, the combination of Atezolizumab and Cabozantinib may warrant further investigation in patients who are unable to tolerate Cabozantinib monotherapy, particularly in light of its potentially favorable toxicity profile. Future Studies are warranted to evaluate the combination of Atezolizumab and Cabozantinib in patients with PD-L1-positive or chemotherapy-resistant tumors, using extended follow-up periods, in order to investigate its potential long-term safety and efficacy.
Supplemental Material
Supplemental Material - Analysis of the Efficacy and Safety of Cabozantinib Monotherapy versus Its Combination with Atezolizumab in Cancer Patients: A Systematic Review and Meta-Analysis
Supplemental Material for Analysis of the Efficacy and Safety of Cabozantinib Monotherapy versus Its Combination with Atezolizumab in Cancer Patients: A Systematic Review and Meta-Analysis by Kimia Pakdaman, Hossein-Ali Nikbakht, Amin Esmaeilnia Shirvani, Saba Sahraian, Layla Shojaie and Pouyan Ebrahimi in Cancer Control.
Footnotes
Acknowledgement
We are grateful to the Health Research Institute at the Babol University of Medical Sciences for its support in preparing this manuscript.
Ethical Approval
This systematic review and meta-analysis was approved by the Ethics Committee of Babol University of Medical Sciences, Iran, under the approval code IR.MUBABOL.HRI.REC.1403.334. Since this study exclusively involved analysis of data from previously published clinical trials and did not involve direct experimentation on human subjects or the collection of human tissue or samples, informed consent and additional ethical approvals were not required.
Author Contributions
This study was conceptualized by K. P., P. E., and H. A. N. Also, K. P, P. E., and A. E. S. conducted the searches and extracted data. H. A. N. and P. E. analyzed the data sets and interpreted the results. K. P, P. E., A. E. S., S. S and L. S. drafted and edited the manuscript. The final version of the manuscript was drafted by all authors after comments, or edits, were made. The final manuscript was read and approved by all authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Detailed data are included in the manuscript and supplementary files. Upon reasonable request, the corresponding author can provide additional data.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
