Abstract
Introduction
The influence of iodine on papillary thyroid carcinoma (PTC) remains a subject of debate. This meta-analysis was conducted to evaluate the risk association between varying levels of iodine intake and the occurrence of PTC and different subtypes of thyroid carcinoma (TC), particularly papillary thyroid microcarcinoma (PTMC).
Methods
Four databases—the Cochrane Library, Embase, PubMed, and Web of Science—were systematically searched for relevant studies published up until May 30, 2024. An updated search was conducted on November 20, 2025. Literature screening and information collection were performed according to predefined eligibility criteria. The Newcastle-Ottawa Scale (NOS) was used to appraise the quality of the eligible literature. Statistical analysis was performed using Stata 17.
Results
This meta-analysis encompassed 17 studies involving 273 651 individuals. The findings revealed a correlation between high urinary iodine concentrations and an increased risk of TC (odds ratio [OR]: 6.43, 95% confidence interval [CI]: 2.72-15.22, P < .05). The elevated risk was observed for both PTC (OR: 7.56, 95% CI: 1.6-35.78, P < .001) and PTMC (OR: 8.96, 95% CI: 5.89-13.64, P < .001). These results suggested that greater urinary iodine concentrations were associated with a higher risk of TC. However, there was no significant association between dietary iodine intake and TC risk (OR: 0.75, 95% CI: 0.37-1.52, P > .05).
Conclusion
This meta-analysis demonstrated a definitive link between high urinary iodine excretion and an increased risk of TC. The relationship between dietary iodine intake and TC requires further investigation. Considering the current limitations, future large-scale, multicenter, prospective investigations are anticipated to provide further validation.
Background
Thyroid carcinoma (TC) originates from the thyroid gland’s malignant neoplasm and is primarily categorized into four types: differentiated TC (encompassing papillary and follicular carcinomas, which constitute the majority and possess a favorable prognosis), medullary carcinoma (genetically linked), and undifferentiated carcinoma (rare but highly aggressive). TC’s incidence has exhibited an upward trend in recent years. According to estimates, there were approximately 44 280 new cases of TC in the United States in 2020. Females accounted for 71.3% of these cases. Projections for 2022 indicated an estimated 43 800 new cases, with females comprising 71.2%. 1 Although the 2022 figure was slightly lower, the National Cancer Institute predicts a rebound to 44 020 new cases in 2024, with females accounting for 71.6% of cases. This suggests an overall fluctuating yet persistently increasing incidence, with female cases consistently constituting over 70% of the total. 2 This phenomenon may be attributed to various factors. Established risk factors include childhood head and neck radiation exposure, specific gene mutations, a family history of the disease, and female sex. Furthermore, the rise in TC incidence might be linked to advancements in diagnostic technologies and alterations in environmental factors. However, the precise pathogenic mechanisms remain incompletely elucidated.
Among numerous potential environmental contributors, the nexus between iodine intake and the risk of TC development has been a focal point of both research and contention. Iodine serves as a crucial precursor for the synthesis of thyroid hormones in the human body, yet its intake exhibits a complex relationship with thyroid disease pathogenesis. A 2024 meta-analysis indicated a positive correlation between excessive iodine intake (urinary iodine concentration [UIC] ≥ 300 μg/L) and TC occurrence. However, this analysis focused solely on UIC and did not account for other exposure routes such as dietary iodine intake. A systematic review and meta-analysis from 2025 also found that iodine deficiency (UIC <100 μg/L) could moderately increase the risk of developing nodules, though excessive iodine intake (>200 μg/L) did not reveal a consistent association. However, this study primarily addressed the relationship between iodine levels and thyroid nodules, rather than TC itself.3,4 Consequently, a definitive conclusion regarding a significant association between iodine intake and TC risk has yet to be established. Moreover, these studies employed inconsistent UIC cutoff values and failed to systematically compare different forms of iodine exposure, such as dietary vs urinary iodine. Additionally, they did not conduct in-depth analyses of different histological subtypes of TC, including PTC and papillary thyroid microcarcinoma (PTMC). Therefore, we believe a new, comprehensive analysis is necessary to further explore this topic.
Considering the continuous emergence of new findings and research data in this field, this meta-analysis aims to comprehensively and accurately evaluate the association between iodine intake levels (including dietary iodine intake and urinary iodine excretion) and the risk of TC development. The objective is to provide more robust evidence-based support for future clinical prevention, treatment, and the design of related research concerning TC.
Methods
This study’s reporting conformed to PRISMA guidelines. 5 This investigation strictly adhered to the Population, Intervention, Comparison, Outcome, Study Design (PICOS) criteria and was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42024584715).
Literature Retrieval
Up to May 30, 2024, a comprehensive literature retrieval was conducted across the Cochrane Library, Embase, PubMed, and Web of Science, incorporating both controlled vocabulary and keyword terms such as “Thyroid Neoplasms”, “Thyroid Cancer”, “Iodine” and their corresponding variations. Detailed strategies are provided in the supplementary materials. An updated search was performed on November 20, 2025. To mitigate the risk of omission, the reference lists of retrieved articles were also meticulously examined.
Eligibility Criteria
This meta-analysis strictly adhered to the Population, Exposure, Comparison, Outcome, Study Design (PECOS) principles for establishing its eligibility criteria.
Inclusion criteria: (i) Individuals diagnosed with TC, including PTC, PTMC, FTC, and TC. (ii) Dietary and urinary iodine levels were considered, with data reported in various units, including time/week, µg/gCr, and µg/L, as presented in the original articles. (iii) Studies that reported differences in urinary iodine levels between individuals with and without TC, and the risk association between varying urinary iodine concentrations and TC incidence. (iv) Observational study designs, encompassing cohort, case-control, and cross-sectional studies.
Exclusion criteria: (i) Studies that did not report or independently differentiate TC populations. (ii) Studies lacking clear reporting of urinary iodine levels or specific urinary iodine statistical data. (iii) Studies where relevant data could not be obtained or collected. (iv) Publications not written in English. (v) Duplicate literature or studies reporting repeated results; the most recent and comprehensive data were retained for statistical analysis.
Literature Screening
Two researchers independently imported the retrieved literature into the EndNote X21 database for data screening. Duplicate articles were initially removed. The titles and abstracts of the de-duplicated literature were then reviewed for preliminary screening. Full texts of potentially relevant articles were obtained and read for a thorough re-screening to confirm final eligibility. Any discrepancies were addressed through discussion with a third reviewer.
Information Collection
Two researchers independently collected the following information from eligible studies: country, publication year, first author, source institution of the population, study design, iodine source, iodine value, and outcome measures. Cross-checking of collected information was performed. Any disagreements were settled through discussion with a third reviewer.
Quality Appraisal
The quality of the eligible studies was evaluated independently by two researchers utilizing the Newcastle–Ottawa Scale (NOS). This tool rates study quality across three domains, comprising a total of eight items. With the exception of the comparability domain, which allows a maximum of two points, all other items have a maximum score of one. Studies with total scores ranging from 7 to 9 were classified as high quality, while those scoring between 4 and 6 were deemed moderate. Following the initial assessment, the two evaluators cross-checked their results. Any discrepancies were resolved through consultation with a third researcher.
Statistical Analysis
This meta-analysis was performed using Stata 17. Heterogeneity was quantified with Cochran’s Q test and Higgins’ I2 statistic. A P-value <.10 or an I2 > 50% indicated significant heterogeneity, prompting the adoption of a random-effects model (REM). 6 Otherwise, a fixed-effects model was employed. When substantial heterogeneity was present, sensitivity, subgroup, and meta-regression analyses were conducted to identify potential sources of variation. A funnel plot was used to visually assess publication bias. Egger’s and Begg’s tests were used for statistical evaluation. When publication bias was detected, the trim-and-fill method was applied to evaluate its effect on the results of the meta-analysis. A P-value below 0.05 was considered statistically significant for pooled effect estimates.
Results
Literature Screening results
The literature search yielded 12 432 articles: 4637 from PubMed, 284 from Embase, 4328 from the Cochrane Library, and 3451 from Web of Science. On November 20, 2025, an update was conducted that supplemented these results with additional records from PubMed (n = 141), Embase (n = 390), the Cochrane Library (n = 23), and Web of Science (n = 343). After deduplication, 4885 articles were removed. Preliminary screening excluded 8662 articles for the following reasons: 851 published before 2000, 131 meta-analyses, 546 reviews, 3527 with incompatible exposure factors, 196 case reports, 42 animal experiments, 504 non-English publications, 1459 with incompatible populations, 1348 conference abstracts, and 58 letters and guidelines. Subsequently, 33 articles were excluded during re-screening (five due to unclear population, 24 due to unextractable data, and four due to inappropriate study design). Ultimately, 17 articles were incorporated.7–23 The detailed screening process is depicted in Figure 1. Literature screening flowchart
Basic Characteristics and Quality of Eligible Literature
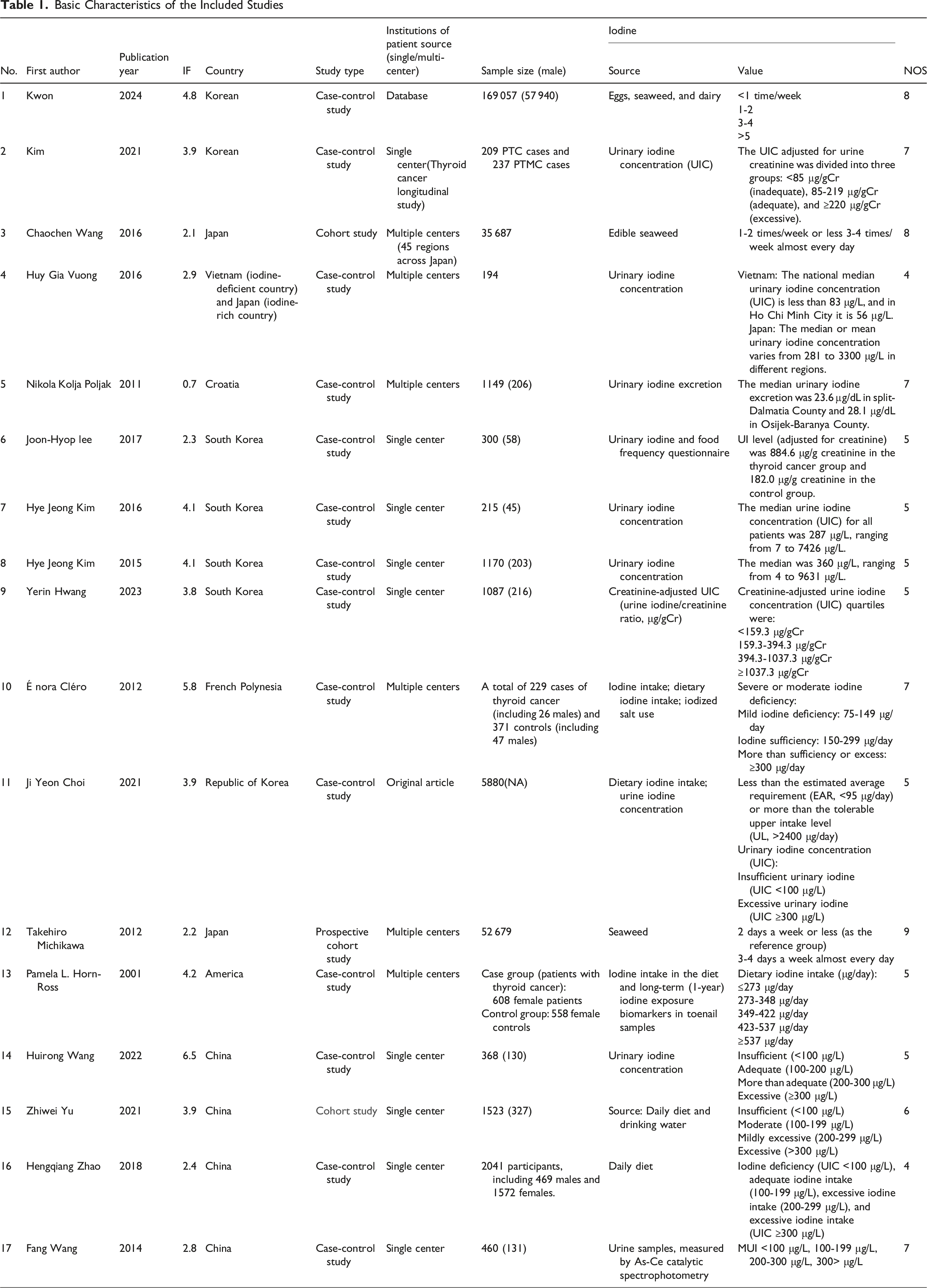
Basic Characteristics of the Included Studies
Meta-Analysis
Correlation Analysis
Urinary Iodine Source
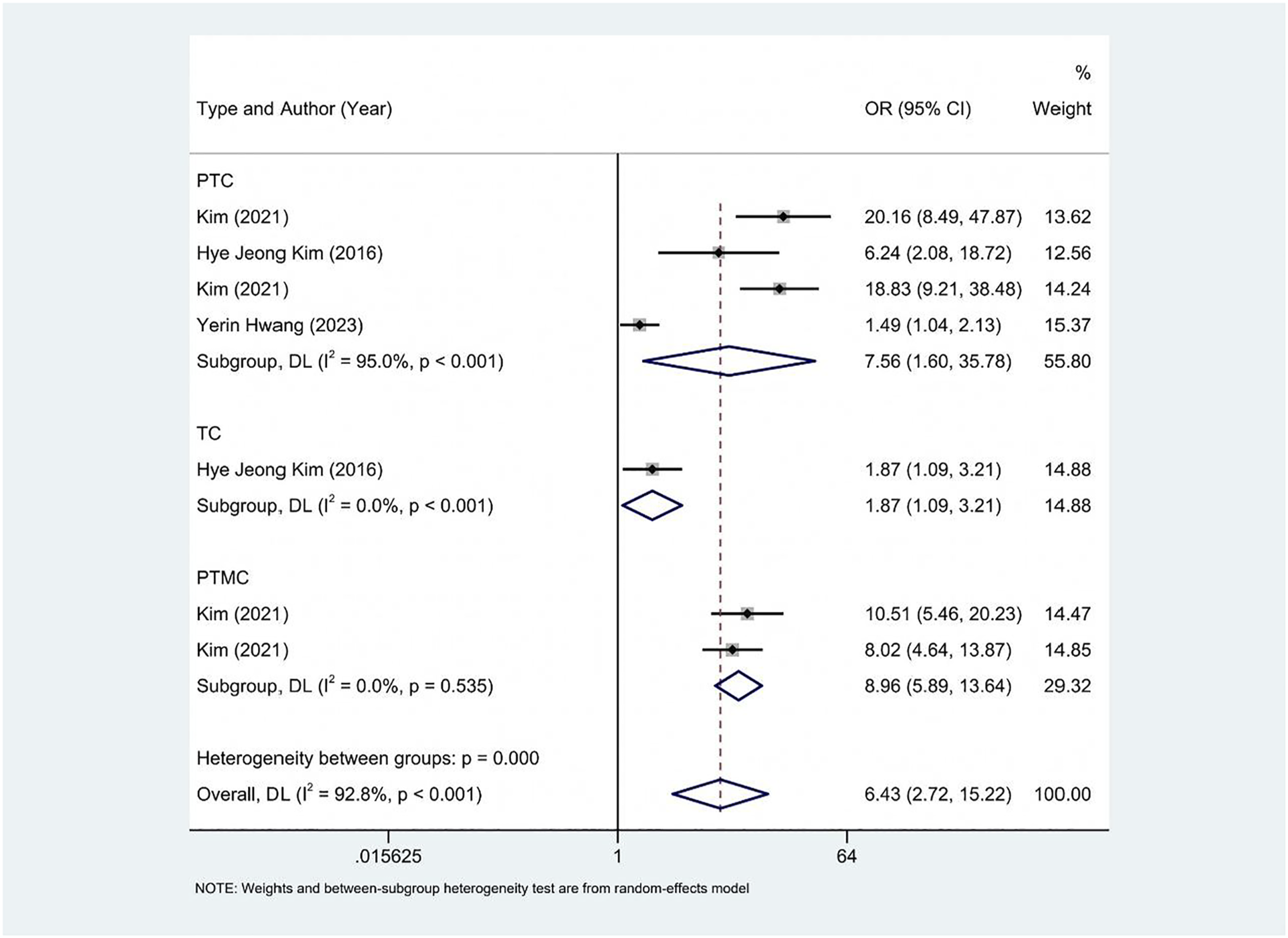
Seven studies (based on units of µg/L or µg/gCr) investigated the association between urinary iodine and the risk of TC. Significant heterogeneity was detected (I2 = 92.8%), leading to the application of an REM for pooling results. The analysis indicated that high urinary iodine concentrations were associated with an increased TC risk (odds ratio [OR]: 6.43, 95% confidence interval [CI]: 2.72-15.22, P < .05) (Figure 2). Further stratification by TC subtypes revealed that high urinary iodine concentrations were associated with elevated risks for both PTC (OR: 7.56, 95% CI:1.6-35.78, P < .001) and PTMC (OR: 8.96, 95% CI: 5.89-13.64, P < .001) (Figure 3). Forest plot illustrating the association between urinary iodine levels and the risk of TC Forest plot illustrating the association between urinary iodine levels and the risk of different subtypes of TC
A subgroup analysis based on different units further demonstrated that four studies using µg/L (OR: 6.90, 95% CI: 2.21-21.56, P < .05) and three studies using µg/gCr (OR: 5.95, 95% CI: 1.27-27.80, P < .05) all indicated an increased TC risk with high urinary iodine concentrations (Figure 4). Forest plot illustrating the association between urinary iodine levels (based on different units) and the risk of TC
Dietary Iodine Source
Three studies that reported dietary iodine intake based on times/week units examined the association between dietary iodine and the risk of TC. A significant heterogeneity was observed (I2 = 86.3%). Utilizing an REM for pooling, no significant association was found between iodine intake and TC risk (OR: 0.75, 95% CI: 0.37-1.52, P > .05) (Figure 5). Forest plot illustrating the association between different levels of dietary iodine intake (based on times/week) and the risk of TC
Sensitivity Analysis
Sensitivity analyses were performed for both urinary iodine and dietary iodine by sequentially removing each study. The results showed minimal change upon sequential removal, indicating the stability and reliability of the findings (Figure 6). Sensitivity analysis of the association between different iodine intake levels and the risk of TC. (A) Urinary iodine; (B) Dietary iodine
Publication Bias
A funnel plot was employed to assess publication bias for the results (Figure 7). Furthermore, Egger’s test revealed no significant publication bias for dietary iodine (P = .238). However, significant publication bias was detected for the urinary iodine-related outcome (P = .041). Funnel plot illustrating the association between urinary iodine levels and the risk of TC
Discussion
The current study focused on the intricate relationship between iodine intake and TC. Through a systematic and rigorous meta-analysis of 17 representative and scientifically valuable recent studies, we have comprehensively reviewed findings to deeply analyze the risk factors for TC and the association between iodine intake levels and this risk. Our analysis revealed a significant positive correlation between urinary iodine concentration, regardless of the unit of measurement, and the risk of TC. This implies that as urinary iodine concentration progressively increases, so does the risk of TC. However, a similar association was not observed with dietary iodine intake. This relationship was relatively consistently validated across different population samples, providing a crucial starting point for further in-depth research into the underlying mechanisms linking iodine and TC, as well as for clinical prevention and control strategies.
Two previous studies have investigated the relationship between iodine and TC risk. Compared to our results, the first meta-analysis from 2024 3 primarily relied on urinary iodine levels, which reflect actual internal exposure after iodine metabolism. Our findings indicate that high UIC is significantly associated with TC risk, particularly with PTC and PTMC. This further supports the sensitivity and stability of urinary iodine as an indicator of iodine exposure in TC risk assessment. A second study from 2025 4 suggested that iodine deficiency is associated with an increased risk of thyroid nodules. That study also used urinary iodine as the main exposure metric. However, we posit that high UIC is associated with TC. This discrepancy may arise because the 2025 study focused on thyroid nodules, which are precursor lesions to cancer, though not all nodules progress to malignancy. Our study specifically targets TC, especially PTC and PTMC, and considers urinary iodine levels and dietary iodine intake. We identified a positive correlation between UIC and TC risk. No similar association was observed for dietary iodine intake.
In a deeper analysis of TC subtype differences, we further explored the potential mechanisms of iodine’s role in disease initiation and progression.
Excess Iodine and PTC: RET/PTC Rearrangement and Sustained MAPK Pathway Activation
In environments with excessive iodine, thyroid follicular epithelial cells are chronically subjected to high oxidative stress. Elevated levels of reactive oxygen species (ROS) can directly induce DNA double-strand breaks. Multiple in vitro and animal experiments have confirmed that ROS can trigger RET/PTC rearrangement, one of the most common initiating events in PTC. Following RET/PTC rearrangement, the MAPK signaling pathway becomes constitutively activated, driving unlimited cell proliferation and inhibiting apoptosis, thereby promoting PTC development. Furthermore, high iodine intake can upregulate the expression of NIS (sodium-iodide symporter), which exacerbates iodine accumulation in the thyroid. 14 This forms a ‘positive feedback’ loop that amplifies oxidative damage and leads to more pronounced activation of the RET/PTC-MAPK axis. Consequently, coastal high-iodine regions exhibit a significantly higher incidence of PTC compared to inland areas, with a particularly prominent increase in the proportion of micro-papillary carcinomas (≤1 cm). 13
Iodine Deficiency and FTC: RAS Mutations and Predominant PI3K/AKT Pathway Activation
In contrast, FTC is uncommon in high-iodine areas but is relatively prevalent in mild to moderate iodine-deficient regions. Mechanistically, iodine deficiency leads to chronically elevated compensatory TSH levels. 22 Sustained TSH stimulation results in overactivation of the PI3K/AKT/mTOR pathway, which in turn induces RAS point mutations (NRAS61, HRAS61) (Cancer Genome Atlas Research Network, 2014). RAS mutations, on the one hand, promote cell cycle progression. On the other hand, they enhance invasion and metastatic potential by downregulating E-cadherin and upregulating matrix metalloproteinases (MMPs), aligning with the clinical characteristic of FTC’s propensity for hematogenous metastasis. Animal models have also substantiated that a low-iodine diet for 6 months can increase the incidence of FTC in rat thyroids from 2% to 12%.2 24
In light of clinical treatment strategies, the findings of this investigation hold significant clinical guidance. Firstly, for TC individuals, understanding the association between iodine intake and disease risk can aid in optimizing postoperative management strategies. For instance, during differentiated therapy for TC, judicious control of iodine intake may contribute to improved efficacy of 131I treatment while concurrently avoiding adverse effects on thyroid function and disease recurrence due to excessive or insufficient iodine intake.
This work also offers valuable insights into the screening and early diagnosis of TC. For populations residing in high iodine intake regions, particularly those with a family history of TC or other risk factors, enhanced TC screening efforts are warranted to facilitate early detection and timely intervention. Concurrently, clinicians evaluating individuals with thyroid nodules should thoroughly consider their iodine intake status, in conjunction with other clinical indicators such as thyroid function and thyroid antibody levels, to improve the accuracy of early TC diagnosis.
In addition to the aforementioned clinical implications, it is crucial to investigate potential reasons for the discrepancies in urinary and dietary iodine levels in relation to TC risk. Dietary iodine is an exogenous source that reflects short-term or habitual intake patterns. Common dietary sources of iodine include eggs, seaweed, and dairy products. However, dietary iodine intake is often quantified by intake frequency (times per week), which is susceptible to recall bias, regional variations in food iodine content, and losses during cooking. Additionally, not all ingested iodine is fully absorbed. Its bioavailability is influenced by factors such as the gastrointestinal environment (eg, gastric pH), goitrogenic substances, the concurrent intake of other nutrients (eg, calcium and iron), and the composition of the gut microbiota. These factors contribute to variability and uncertainty in the association between dietary iodine and TC. In contrast, urinary iodine reflects the overall outcome of iodine metabolism, including gastrointestinal absorption, systemic distribution, thyroid utilization, and renal excretion. Its level is influenced by dietary iodine intake, individual metabolic capacity, thyroid function, renal clearance, and iodine reabsorption in the enterohepatic circulation. Furthermore, while both urinary and dietary iodine analyses exhibited high heterogeneity (UIC: I2 = 92.8%; dietary iodine: I2 = 86.3%), the effect size for urinary iodine was significantly greater than that for dietary iodine (UIC: OR: 6.43, 95% CI: 2.72-15.22; dietary iodine: OR: 0.75, 95% CI: 0.37-1.52). These results suggest that UIC shows a more stable association with TC risk and more sensitively reflects the carcinogenic potential related to iodine. This supports the hypothesis that internal iodine exposure is a better indicator of iodine-associated TC than external intake.
The strengths of this research are primarily evident in several aspects. Firstly, including multiple iodine intake assessment metrics, such as dietary and urinary iodine, provides a relatively comprehensive reflection of population iodine intake status, thereby enabling a more precise analysis of its association with TC risk. This offers a richer and more complete perspective for understanding the impact of iodine intake from different sources and forms on TC. Secondly, this investigation yields results with strong representativeness and generalizability through a systematic meta-analysis of 17 high-quality studies encompassing a broad range of geographical areas and population samples. These results provide a valuable reference for developing global TC prevention and control strategies.
However, this research is also subject to certain unavoidable limitations. Firstly, the small number of eligible studies impacted the stability and statistical power of some analytical results. Particularly in the in-depth analysis of the relationship between different TC subtypes and iodine intake, sample size limitations may prevent a full elucidation of potential subtle differences and complex associations. Secondly, the heterogeneity of our findings was quite pronounced. Although various methods were employed to identify sources of heterogeneity, the limited number of eligible studies precluded more detailed subgroup analyses to further investigate the specific causes of heterogeneity. Finally, while we acknowledge the potential for a dose-response relationship between iodine intake and TC risk, significant variations in reporting units and iodine intake ranges across eligible studies prevented us from accurately depicting this dose-response curve. Since all of the included studies were retrospective in design, they may be subject to recall and selection biases. These biases restricted our ability to elaborate on the relationship between specific iodine intake thresholds and TC risk. This underscores the need for future research: more studies with unified units and finer stratification of iodine intake reporting to supplement and validate this issue.
Conclusion
In summary, this investigation, through a systematic meta-analysis of 17 relevant studies, has elucidated the strong association between iodine intake levels and the risk of TC. We found that as iodine intake increased, so did the risk of TC. Nevertheless, the findings of this study necessitated further confirmation and supplementation by more high-quality, multicenter, large-sample prospective investigations. Future research should focus on unifying the measurement standards and reporting units for iodine intake, thoroughly investigating the dose-response relationship between iodine intake and TC risk, and comprehensively considering other potential influencing factors such as genetic background, environmental exposures, and lifestyle. This approach will contribute to the development of a more comprehensive and precise TC risk assessment model.
Supplemental Material
Suppplemental Material - Dietary and Urinary Iodine in Relation to Thyroid Cancer Risk: A Meta-Analysis
Suppplemental Material for Dietary and Urinary Iodine in Relation to Thyroid Cancer Risk: A Meta-Analysis by Shitao Li, Hengxu Yu in Cancer Control
Footnotes
Ethical Considerations
As this study is a meta-analysis review and not human or animal experiments, it does not require ethical approval from the ethics committee.
Author Contributions
All authors contributed to the study conception and design. Writing - original draft preparation: Shitao Li; Writing - review and editing: Shitao Li; Conceptualization: Shitao Li; Methodology: Shitao Li; Formal analysis and investigation: Shitao Li; Funding acquisition: Shitao Li; Resources: Shitao Li; Supervision: Hengxu Yu, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analysed during this study are included in this published article and its supplementary information files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
