Abstract
Introduction
Vulvar squamous cell carcinoma (VSCC) is a rare but increasingly prevalent gynecological malignancy. This study aimed to identify risk factors for stage I VSCC, which accounts for approximately 70% of VSCC patients, and to develop a nomogram to predict cancer-specific survival (CSS) for this large subgroup.
Methods
This study analyzed the datasets consisting of public training and independent external validation sets of patients diagnosed with stage I VSCC between 2010 and 2019. Prognostic factors were discerned through Cox regression analyses and the least absolute shrinkage and selection operator (LASSO) method. A nomogram for CSS was developed and evaluated using the C-index, Kaplan-Meier curves, and decision curve analysis (DCA) plots.
Results
Our analysis revealed variations in predictors of CSS and overall survival (OS) in stage I VSCC cases from the Surveillance, Epidemiology, and End Results (SEER) database. The multivariate Cox model suggested associations between CSS and age, grade, and number of tumors (NMT), while the LASSO model indicated potential roles for age, stage, invasion depth, NMT, and surgical method. The nomogram showed reasonable discriminative ability in the training (C-index: 0.785) and validation cohorts (C-index: 0.729), with supporting Kaplan-Meier and DCA analyses.
Conclusion
This study proposes a prognostic model for CSS in stage I VSCC, identifying exploratory associations with multifocal tumors and surgical extent. Further prospective studies are needed to validate these findings and clarify their clinical implications.
Introduction
Vulvar squamous cell carcinoma (VSCC) constitutes over 90% of vulvar cancers, 1 and although rare, it is the fourth most prevalent gynecological malignancy. 2 High-income countries, mainly Europe and the US, account for more than half (50.1%) of new cases of vulvar cancer diagnosed each year, 3 despite the higher rates of human papillomavirus (HPV) vaccine coverage in these countries.4,5 In response, former leaders from the International Society for the Study of Vulvovaginal Disease (ISSVD) and the European Society of Gynaecological Oncology (ESGO) have urged healthcare providers, including gynecologists, pathologists, and dermatologists, to pay closer attention to patients with vulvar cancer and to make more significant contributions to their care. 6
It is noteworthy that the 5 year overall survival rate of 69.6% 7 for vulvar cancer is even higher than the survival rate of stage II (64.5%) and is not significantly impacted by the low survival rates of advanced stages such as stage IV (below 26%) of this disease, since most vulvar cancer cases (about 70%) are concentrated in stage I. 8 Staging surgery such as radical local excision is the standard of care for VSCC, but even patients with stage I tumors may require additional radiotherapy.1,3,8 Currently, no research has focused on the predictors for clinical outcomes within the largest distribution group of VSCC patients, namely those with stage I VSCC. Hence, this study aims to identify risk factors and develop a predictive model for the forecast of the cancer-specific survival (CSS) of stage I VSCC.
Methods
Patients and Database
To establish the predictive model, data on patients with stage I VSCC were gathered from two sources: the SEER database and our hospital (WHZJU), between 2010 and 2019. Inclusion criteria for the SEER database were the following: (1) tumor confined to the vulva, using International Classification of Diseases for Oncology, third Edition (ICD-O-3) codes C51.0 (Labium majus), C51.1 (Labium minus), C51.2 (Clitoris), C51.8 (Overlapping lesion of vulva), or C51.9 (Vulva, NOS); (2) squamous cell carcinoma, using ICD-O-3 histology codes 8051-8076, 8078, 8081, and 8084-8086; and (3) stage I tumor, according to the FIGO staging system.
9
Exclusion criteria included: (1) no surgery performed; and (2) other causes of death in the cancer-specific survival (CSS) cohort. Inclusion criteria for VSCC patients at our hospital were: (1) stage I VSCC confirmed by pathological diagnosis; (2) surgery performed at our hospital; and (3) complete follow-up information available. Exclusion criteria were: (1) combined with other tumors; and (2) the patient died of causes other than VSCC. A total of 863 patients diagnosed with stage I VSCC from the SEER database were included in the overall survival (OS) cohort. Of these, 655 patients were incorporated into the cancer-specific survival (CSS) cohort, which consisted of individuals who either succumbed to VSCC or survived. Additionally, 52 stage I VSCC patients from our hospital were enrolled in the CSS cohort for external validation based on the inclusion/exclusion criteria. The selection process for the SEER database was detailed in Figure 1 and Supplementary File (Table S1). The study was approved by the Institutional Review Board at Zhejiang University School of Medicine Women’s Hospital, China (IRB No. 20200158-R), and informed consent was obtained from all patients before using their records for research. The Workflow Chart of This Study. VSCC: Vulvar Squamous Cell Carcinoma; SEER: Surveillance, Epidemiology, and End Results; WHZJU: Women’s Hospital, School of Medicine, Zhejiang University; OS: Overall Survival; CSS: Cancer-Specific Survival; COD: Causes of Death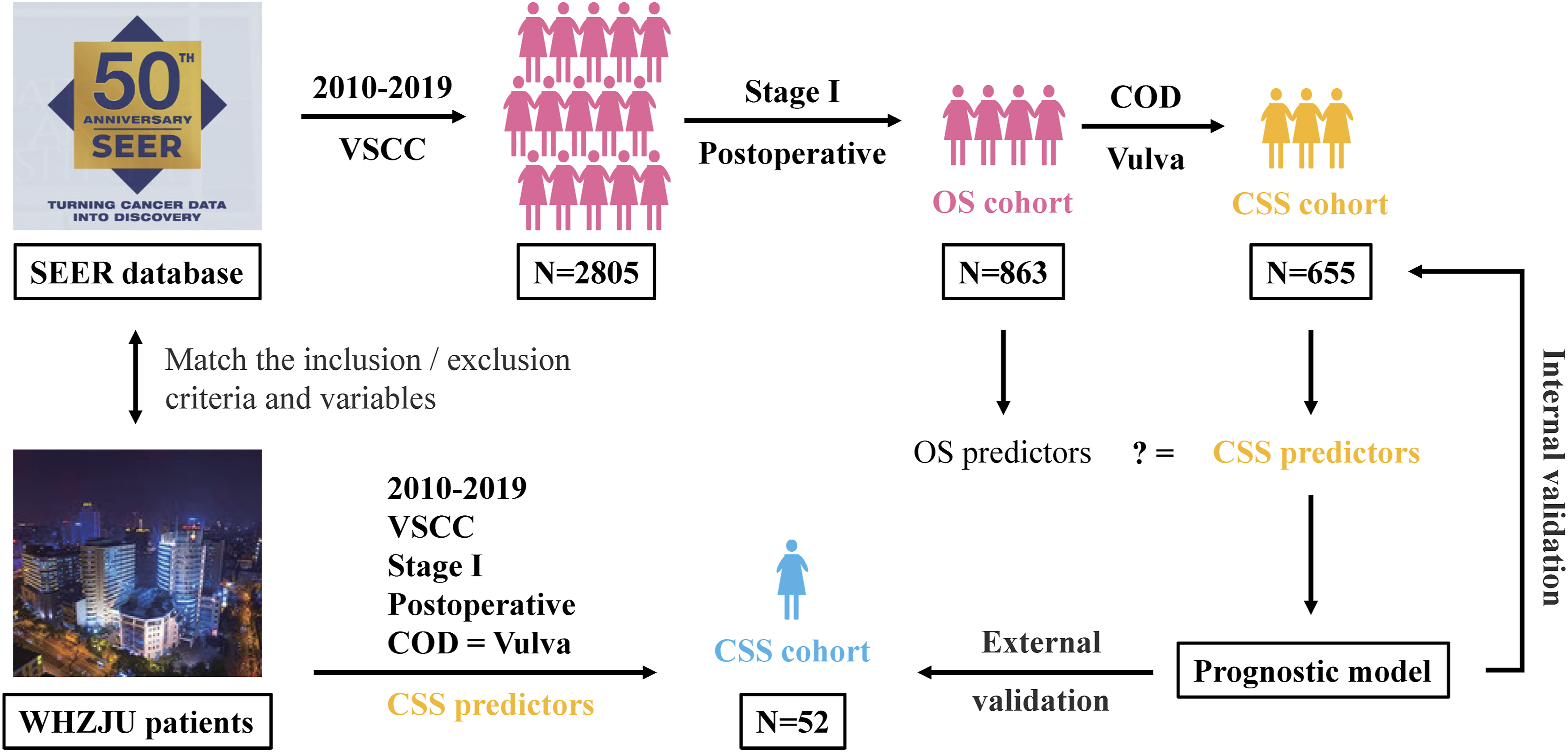
Prognostic Variables and Modeling Process
The study utilized data from the SEER database to examine various risk factors, including age, race, marital status, tumor stage, grade, tumor site, tumor size, invasion depth, number of tumors, surgical method, lymph node management, and the National Comprehensive Cancer Network (NCCN) guidelines compliance status. 10 Noncompliance with NCCN guidelines was characterized by a surgical approach that was less extensive than radical or local excision, as well as by the absence of lymph node assessment in stage IB cases. Firstly, through analysis of large samples from the SEER database, it was necessary to establish whether the risk factors were consistent across both the CSS cohort and OS cohort in stage I VSCC. Secondly, the predictors extracted by SEER’s CSS cohort were collected for analysis in our hospital (WHZJU) CSS cohort, where the risk factors were compared between the two cohorts. Finally, the prognostic model was constructed using the predictive factors identified above and validated using both internal (SEER’s CSS cohort, n = 655) and external data (WHZJU’s CSS cohort, n = 52). The modeling process is illustrated in Figure 1. The reporting of this study conforms to TRIPOD guidelines. 11
Statistical Analysis
In this study, categorical variables were reported as frequencies (proportions). Chi-square tests were conducted to analyze associations between variables using cross-tables. For the analysis of prognostic factors, both univariate and multivariate Cox regression models were employed. The prognostic factors for CSS were compared and selected using a combination of multivariate Cox regression and the least absolute shrinkage and selection operator (LASSO) method. 12 The selection of the tuning parameter (λ) in the LASSO model was performed using 10-fold cross-validation. The optimal predictor variables were determined and selected based on the concordance index (C-index) using the one standard error of the minimum criterion (1-SE criterion). To visualize the results, Venn plots were utilized. Nomogram model building was performed following a prospectively defined process. The performance and discrimination of the nomogram model were assessed using various techniques, including the (C-index), Kaplan-Meier curves, calibration curves, and decision curve analysis (DCA). Kaplan-Meier curves were used to stratify the internal and external validation cohorts based on different cut-off values derived from the nomogram. The calibration curves were conducted in the internal and external validation cohort, consisting of 1000 bootstrap samples. These curves were used to depict the relationship between the actual outcome frequencies and the predicted probabilities. DCA plots were employed to demonstrate the net benefit of the nomogram model compared to the FIGO staging system, thus illustrating its performance. A two-sided P-value of less than 0.05 was considered statistically significant. All statistical analyses were performed using R software (version 4.1.0). The R packages were used for detailed statistical analyses and data visualization, as stated in Supplementary File (Table S1).
Results
Baseline Information of SEER’s Cohorts
Univariate and Multivariate Analysis of the Prognostic Variables and Overall Survival in Stage I VSCC

*P < 0.05; **P < 0.01; ***P < 0.001.
aOther: American Indian/Alaskan native, Asian/Pacific islander, or unknown.
bMarital status: married group included the domestic partners; divorced group included the separated.
cFIGO 2009 staging system.
dOther surgery included laser excision or electrocautery.
CI, confidence interval; FIGO, International Federation of Gynecology and Obstetrics; LNE, lymph node excision; SLNB, sentinel lymph node biopsy; NCCN, National Comprehensive Cancer Network; GCS, guidelines compliance status.
Univariate and Multivariate Analysis of the Prognostic Variables and Cancer-Specific Survival in Stage I VSCC

*P < 0.05; **P < 0.01; ***P < 0.001.
aOther: American Indian/Alaskan native, Asian/Pacific islander, or unknown.
bMarital status: married group included the domestic partners; divorced group included the separated.
cFIGO 2009 staging system.
dOther surgery included laser excision or electrocautery.
CI, confidence interval; FIGO, International Federation of Gynecology and Obstetrics; LNE, lymph node excision; SLNB, sentinel lymph node biopsy; NCCN, National Comprehensive Cancer Network; GCS, guidelines compliance status.
Univariate and Multivariate Analysis of OS in Stage I VSCC Patients from SEER Database
Table 1 presents the results of a study investigating the relationship between various prognostic factors and overall survival (OS) in patients of the SEER’s OS cohort. The univariate analysis revealed that several factors were significantly associated with poor OS, including older age (≥70), being widowed, having stage IB disease, G3 grade, larger tumor size (20-39 mm or ≥40 mm), deeper invasion depth (>1 mm or unknown), having multiple tumors, and receiving adjuvant therapy (all P < 0.01). After adjusting for other confounding variables in the multivariate Cox model, age ≥ 70 (HR: 3.925; P < 0.001), stage IB (HR: 2.112; P = 0.019), G3 grade (HR: 1.681, P = 0.003), and multiple tumors (HR: 1.683; P < 0.001) were found to be independent prognostic factors of worse OS. Moreover, an unknown tumor size (HR: 0.432, P = 0.018) was identified as an independent prognostic factor associated with better OS.
Univariate and Multivariate Analysis of CSS in Stage I VSCC Patients from SEER Database
As shown in Table 2, univariate analysis revealed that several factors were significantly associated with poor CSS, including age ≥ 70, being widowed, having stage IB, high grade (G3 or G4), larger tumor size (20-39 mm or ≥40 mm), deeper invasion depth (>1 mm or unknown), multiple tumors, and receiving adjuvant therapy were related to poor CSS (all P < 0.05). In contrast, the Black race, unknown grade, and local excision or other treatments such as biopsy, laser excision, and electrocautery were identified as positive factors (HR < 1 and P < 0.05) for CSS using univariate Cox models. Multivariate analysis further indicated that age ≥ 70 (HR: 4.066; P < 0.001), G4 grade (HR: 9.872; P = 0.010), and multiple tumors (HR: 1.902; P = 0.001) were negative factors significantly associated with worse CSS.
Comparison of Potential Predictors for CSS and OS in Stage I VSCC
To address issues of collinearity and high-dimensionality, we employed the Cox regression models with LASSO penalty to select predictors for CSS and OS (Figure 2A–D). For CSS, the LASSO model identified five prognostic factors: age, invasion depth, stage, number of tumors (NMT), and surgical method (Figure 2A and B). In contrast, the LASSO model identified only three predictors for OS: age, stage, and NMT (Figure 2C and D). To show the different and intersecting predictors, we applied the Venn plots for CSS and OS respectively from the multivariate and LASSO models (Figure 2E and F). Notably, two models highlighted that age and number of tumors were independent and significant predictors for both CSS and OS. In stage I VSCC, CSS predictors were more varied and slightly differed from OS predictors. The Predictors Selection for CSS and OS Using the Cox Regression Model With LASSO Penalty (10-Fold Cross-Validation). The Optimal Values (λ) Using the 1-SE Criteria. (A, B) CSS Model: 1-SE log(λ) Selected 5 Predictors (Age, Invasion Depth, Stage, NMT, Surgical Method. (C, D) OS Model: 1-SE log(λ) Selected 3 Predictors (Age, Stage, NMT). Venn Plots Illustrating the Selection of Predictors Using Multivariate Cox Regression (M-Cox) and Lasso Model for CSS (E) and OS (F). SE: standard Error; CSS: Cancer-specific Survival; OS: Overall Survival; LASSO: Least Absolute Shrinkage and Selection Operator; C-Index, Concordance Index; NMT, Number of Tumors
Construction and Validation of the Predictive Nomogram for CSS in Stage I VSCC
Based on the LASSO and multivariate analysis results (Figure 2E), the prognostic nomogram was formulated to predict the 3- and 5-year CSS in the SEER cohort (Figure 3A). The predictive outcome of CSS was determined by calculating the total score of this nomogram, which encompassed six factors: age, stage, grade, invasion depth, NMT, and surgery. For instance, consider a 70-year-old patient diagnosed with early-stage VSCC, classified as stage IB (100 points), G3 (60 points), with an invasion depth greater than 1 mm (12 points), multiple tumors (43 points), and undergoing radical excision (31 points). This patient would accumulate a total of 246 points. Based on these parameters, the predicted 3-year cancer-specific survival (CSS) rate is 82%, while the predicted 5-year CSS rate is 74%. Prognostic Nomogram for CSS in Stage I VSCC Patient and Its Assessment. (A) The Nomogram to Predict 3-Year CSS and 5-Year CSS was Created Based on 5 Independent Prognostic Factors. Kaplan-Meier Curves Demonstrating CSS in Stage I VSCC Patients With (B) Internal Validation Cohort and (C) External Validation Cohort. Calibration Curves of the Nomogram for Predicting 3-Year CSS (D), 5-Year CSS (E) in the Internal Validation Cohort, and 3-Year CSS (F) and 5-Year CSS (G) in the External Validation Cohort. The DCA of the 3- and 5-Year CSS for the Internal Validation Cohort. (H) And the External Validation Cohort. (I). VSCC: Vulvar Squamous Cell Carcinoma; CSS: Cancer-specific Survival; SEER: Surveillance, Epidemiology, and End Results; DCA: Decision Curve Analysis; FIGO, International Federation of Gynecology and Obstetrics
Furthermore, the chi-square test was utilized to examine the disparities of all predictors between the internal and external validation groups, as illustrated in Supplementary File (Table S1). The total score for each patient in the CSS/internal cohort (n = 655) and WHZJU/external cohort (n = 52) was computed. In the internal cohort, the median total score for CSS patients was 158, while in the external cohort, it was 162. Based on these median total scores, patients were categorized into two groups. Kaplan-Meier curves were utilized to illustrate the CSS outcome for these two cohorts (Figure 3B and C). Patients with scores above the median total score exhibited significantly poorer CSS compared to those with lower scores (P < 0.0001 and P = 0.023, respectively).
To further evaluate the discrimination and performance of the nomogram, calibration curves and DCAs were also carried out in this study (Figure 3D–I). The VSCC patients from the CSS cohort in the predictive nomogram exhibited a C-index of 0.785 (95% CI: 0.748-0.821). The adjusted C-index for CSS was 0.772 for the internal validation cohort and 0.729 for the external validation cohort. The calibration curves demonstrated that the predicted 3 year CSS models displayed greater consistency with the actual survival probability compared to the calibration plots for 5 year CSS (Figure 3D–G). The DCA showed that the nomogram was more effective than the FIGO staging system in predicting CSS for stage I VSCC patients, offering higher positive net gains (Figure 3H and I).
Discussion
We first focus on the staging subgroup with the most significant number of VSCC patients (stage I). To identify crucial prognostic factors, this study utilized a multivariate Cox regression analysis and employed a machine learning algorithm (Lasso regression). Furthermore, the external validation cohort established by our medical center only collects the screened characteristic parameters, which enhances the efficiency of follow-up procedures and improves the accuracy of data. Subsequently, a robust nomogram model with CSS as the predictive endpoint for stage I VSCC patients was developed and validated through testing with both internal and external validation cohorts.
Here, we summarize our main results. By employing the LASSO model, we successfully ascertained supplementary predictors for CSS, encompassing invasion depth, stage, and surgical method. Conversely, in the evaluation of OS, the LASSO model exhibited a decrease in variables, such as tumor grade and size, in comparison to the multivariate model. Our final nomogram assigned higher scores to factors such as elder age (≥70), higher stage (IB), high grade (≥G3), deeper invasion depth (>1 mm), multiple tumors, and a wider surgical method called radical excision. A higher total score in the nomogram indicated a shorter 3 year or 5 year CSS. The robustness of our prediction model was confirmed through validation with both internal and external cohorts, demonstrating good accuracy.
Six searched studies utilized the publicly accessible SEER database to develop nomogram models for VSCC.13-18 Age and staging consistently emerged as the primary predictors, albeit with variations in the staging systems employed across the studies. Specifically, Lei et al 18 utilized FIGO staging, Zhou and Yue 17 and Wei et al 13 employed TNM staging, Liu and Wang 16 utilized SEER staging, and Mao et al 15 and Qiu et al 14 employed a combination of AJCC, SEER, and TNM staging. This study utilized the widely recognized FIGO staging system for vulvar cancer, with a specific focus on patients classified as stage I, as this subgroup represents the largest patient population. Similar to prior findings, our study revealed a significant correlation between aging (≥70) and FIGO stage IB with unfavorable CSS outcomes. Zhang et al innovatively developed an integrated model that incorporated clinicopathological parameters and immune biomarkers to assess prognosis in VSCC. 19 Their findings consistently indicated that certain predictive factors, such as aging, exhibited a comparatively lesser influence on progression-free survival (PFS) in contrast to OS. It may be more pertinent to focus on factors identified by the PFS nomogram models, such as immune destruction.
In this study, NMT and surgical approach were not parameters for tumor staging compared with other prognostic factors such as stage, grade, and depth of invasion. These factors (NMT and surgery method) possess the potential to modify current preventive measures and treatment programs. NMT and surgical approach were rarely reported in past prediction models.
In the case of NMT, multiple tumors of VSCC have been reported to be associated with HPV-positive tumors or multifocal precancerous lesions,10,20 primarily HPV16 and HPV33 positive neoplasia. 21 There is substantial evidence supporting the effectiveness of HPV vaccination, particularly the nine-valent vaccine, as a crucial preventive measure against vulvar cancer. 22 Nevertheless, the primary challenge in preventing VSCC lies not in vaccination, but in screening for HPV. Currently, there are only HPV screening strategies available for cervical cancer, but not for vulvar cancer, particularly for women aged 70 and older.
Regarding surgical management, in line with our findings, an increasing body of evidence suggested that, when compared to downsizing operations such as wide local excision, modified vulvectomy or sentinel lymph node biopsy, a broader surgical approach such as radical vulvectomy and bilateral lymphadenectomy does not confer any additional benefits, particularly in cases of early-stage disease.23-28 However, due to the risk of recurrence, a significant number of surgeons have not embraced the concept of early VSCC downsizing surgery, as exemplified in our hospital. In conjunction with the nomogram developed in this study and previous literature, we aim to address the question of how to individualize surgical approaches (downsizing operations) in stage I VSCC. For stage I tumors with invasion ≤1 mm, including IA and IB without midline vulvar tumors, the surgical approach consists of a wide local excision (limited to the tumor) based on the concept of minimal resection margins, 29 which is adequate if margins are negative (1-2 cm). 30 For Stage IB tumors with invasion >1 mm, treatment consists of a modified radical vulvectomy (preserving more skin tissue)23,31 with surgical lymph node assessment. Furthermore, sentinel lymph node (SLN) biopsy, which is one of the lymph node assessments, should be advocated to avoid the complications associated with routine bilateral lymphadenectomy, such as lymphedema and nerve injury. 28 Because stage I tumors do not exhibit lymph node metastasis, the cutoff values for unilateral (≥10 nodes) and bilateral lymphadenectomy counts (≥15 nodes) presented in the Panici et al 32 review article may assist in reducing the false-negative rate of SLN biopsies.
These findings suggest that potential strategies such as implementing HPV screening protocols tailored for vulvar cancer and revising surgical concepts may offer pathways to improve the prognosis of early-stage VSCC; however, these implications should be interpreted with caution and require further rigorous clinical investigation for validation.
This study has some limitations: (1) The utilization of CSS as an endpoint offers notable benefits in comparison to OS; however, its assessment in this particular population characterized by advanced age and the presence of multiple comorbidities frequently poses challenges; (2) Statistical bias may arise due to partial disparities in baseline data between the SEER study cohort and the external validation cohort; (3) During validation with the external cohort, the performance and discrimination of the prediction model were compromised due to the fact that all stage I VSCC patients at our medical center underwent radical vulvectomy; (4) In the latest 2021 version of the FIGO staging system, the stage I vulvar cancer classification remained unchanged from the 2009 FIGO staging except for the depth of invasion definition. The use of the 2009 FIGO staging system in this study could potentially introduce bias; (5) Sample size of the internal and external validation cohort was small. Nomograms developed under conditions of low incidence of positive events, such as mortality, within a sample may exhibit suboptimal predictive performance; and (6) Being a retrospective analysis, the clinical implications of the identified risk factors for CSS are exploratory. Future large-scale prospective research is essential to validate their utility in guiding clinical interventions.
Conclusion
In summary, we have constructed a precise prediction model for CSS in stage I VSCC. Our research has uncovered novel insights by identifying distinct predictors for CSS and OS, highlighting less emphasized risk factors, such as multifocal tumors and excessive surgical interventions that are not justified. Moving forward, it is crucial for forthcoming studies to delve into elucidating the underlying mechanisms that govern these identified risk factors. This in-depth exploration is essential to enhance our comprehension and facilitate the development of more targeted and efficacious interventions for the management of stage I VSCC patients.
Supplemental Material
Supplemental Material - A Nomogram for Predicting Cancer-Specific Survival in Patients With Stage I Vulvar Squamous Cell Carcinoma: A Study Based on the SEER Database and External Validation
Supplemental Material for A Nomogram for Predicting Cancer-Specific Survival in Patients With Stage I Vulvar Squamous Cell Carcinoma: A Study Based on the SEER Database and External Validation by Yingfan Zhu, Tao Zhang, Jiali Xu, Zhuoqun Lin, Fenfen Wang, Xiaodong Cheng in Cancer Control.
Footnotes
Acknowledgment
Thanks to the SEER website for providing us with the data download.
Ethical Approval
The study received approval from the Institutional Review Board at Zhejiang University School of Medicine Women’s Hospital, China No. IRB-20200158-R. Prior to using their records for scientific research, informed consent was obtained from each patient.
Author Contributions
YZ and TZ contributed equally. YZ: Conceptualization, data curation, investigation, visualization, methodology, writing–original draft, writing–review and editing. TZ: Conceptualization, formal analysis, investigation, visualization, methodology, writing–review and editing. JX: Data curation, investigation, methodology, writing–review and editing. ZL: Data curation, methodology, writing–review and editing. FW: Supervision, project administration, writing–review and editing. XC: Conceptualization, supervision, investigation, visualization, methodology, project administration, writing–review and editing. All authors have read and approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Scientific Research Fund of Zhejiang Provincial Education Department (No. Y202454712) and the Education Reform Project of the School of Obstetrics and Gynecology, Zhejiang University School of Medicine (ZDFY202404).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
