Abstract
Objectives
Not all patients with stage III and IV osteosarcoma who undergo surgery to remove the primary tumor will benefit from surgery; therefore, we developed a nomogram model to test the hypothesis that only a subset of patients will benefit from surgery.
Methods
412 patients were screened from the Surveillance, Epidemiology and End Results (SEER) database. Subsequently, 1:1 propensity score matching (PSM) was used to screen and balance confounders. We first made the hypothesis that patients who underwent the procedure would benefit more. A multivariate Cox model was used to explore the independent influencing factors of CSS in two groups (benefit group and non-benefit group) and constructed nomograms with predicted prognosis. Finally, receiver operating characteristic (ROC) curves, calibration curves, and decision curve analysis (DCA) were used to verify the performance of the nomogram.
Results
Of these patients, approximately 110 did not undergo primary tumour resection. After passing PSM, they were divided into a surgical group and a non-surgical group. Age, primary site and chemotherapy as calculated independent factors were used to construct a nomogra. The predicted nomogram showed good consistency in terms of the ROC curve and the calibration curve, and the DCA curve showed a certain clinical utility. Finally, dividing the surgical patients into surgical beneficiaries and surgical non-beneficiaries, a Kaplan–Meier analysis showed that the nomogram can identify patients with osteosarcoma who can benefit from surgery.
Conclusion
A practical predictive model was established to determine whether patients with stage III or IV osteosarcoma would benefit from surgery.
Introduction
Osteosarcoma is the most common bone cancer,1,2 typically with the first peak during adolescence (under the age of 20)3,4 and the second peak during older adulthood (over the age of 60).3,5 The incidence of osteosarcoma in adolescent patients is mainly in the extremities, and the incidence of the disease in the spine increases with age. As osteosarcoma is a highly malignant tumour, the prognosis of patients is poor. According to a recent study, the 3-year and 5-year overall survival (OS) rates of osteosarcoma patients are 67.2% and 58.0%, respectively. 6 With the development of medical technology, more and more treatments are devoted to improving the prognosis of patients with osteosarcoma.
With advances in science and technology, the best treatment for osteosarcoma has changed. It is now generally accepted that systemic chemotherapy combined with extensive surgical resection is the best option.7,8 Surgical treatment is also the cornerstone of many tumour treatment programs. For patients with osteosarcoma, amputation surgery, limb salvage surgery and other surgeries have emerged as the times require. Amputation is the surgical removal of a diseased limb. In principle, this operation can minimize the possibility of tumour recurrence or metastasis, but it causes patients great inconvenience. In their study, Daniel et al found that most patients who chose amputation were affected by factors such as older age, advanced tumour progression, an overly large tumour, comorbidities and lower income. 9 Limb salvage surgery is performed to preserve a patient’s diseased limb as much as possible under the premise of ensuring the safety of the patient’s life. Surgical methods include joint replacement after the removal of adjacent joint tumours. At the same time, there are also boiled, inactivated and replanted tumour segments with preserved epiphysis, large-segment allogeneic bone reconstruction, and autologous bone reconstruction with vascular pedicle. At present, the OS of osteosarcoma tends to be stable, and surgeons are increasingly advocating limb salvage surgery. 10 In a retrospective analysis by Han et al, 934 patients underwent limb salvage surgery and 662 underwent amputation. The amputation group had a significantly lower 5-year survival rate compared with the limb salvage group (OR .628; 95% CI 0.431-.913, P = .015), and the limb salvage group had better limb function. 11
However, surgery does not benefit every patient. For patients with early-stage osteosarcoma, a corresponding individualized treatment plan is formulated. After fully evaluating the overall condition of the patient, the doctor will choose to perform surgical treatment at a suitable time. However, for patients with stage III and IV osteosarcoma aggressive surgical treatment may be counterproductive, and non-surgical treatment may provide patients with a better quality of life at the end of their lives. This is also the purpose of our study, and a personalized predictive model will help provide a reference for surgeons in this situation to identify candidates who can benefit from primary osteosarcoma resection.
Materials and Methods
Population Cohort
The included data were downloaded from the US public database seer (version 8.4.0.1; National Cancer Institute, USA). We selected a total of 412 patients with osteosarcoma during the period 2004-2015. Inclusion criteria were as follows: (1) confirmed histological type of osteosarcoma, ICD-O-3/WHO 2008 morphological codes are 9180-9187 and 9192-9195, including osteosarcoma; chondroblastic osteosarcoma; fibroblast osteosarcoma; telangiectatic osteosarcoma; osteosarcoma in Paget’s disease; and paraosseous osteosarcoma, etc; (2) confirmed as the first tumour; and (3) with complete follow-up data. Exclusion criteria were as follows: (1) age, gender, primary site, pathological type, American Joint Committee on Cancer (AJCC) sixth edition staging, TNM classification, radiotherapy, chemotherapy, surgery information was incomplete; and (2) survival time <1 month. It is particularly important to emphasize that ethical approval was not required for this study as the SEER database is a public database.
Data Analysis
To clarify whether surgery or not will have an impact on the patient’s prognosis, we divided the population into a surgical group and a non-surgical group. We used X-tile software (Yale University, New Haven, CT, USA) to obtain the cut-off value of age. At the same time, to minimize the influence of confounding factors on the results, we calculated using propensity score matching (PSM) to match the closest propensity score on the logit scale 1:1 and callipers set to .01. After PSM, a chi-square test was used to analyse the differences between the variables in the surgery group and the non-surgery group. Variables included age, sex, race, primary tumour site, pathological type, sixth edition staging, TNM, radiotherapy and chemotherapy.
Cancer-specific survival (CSS) for patients in the surgical group was calculated by Kaplan-Meier and calibrated by log-rank test. Independent prognostic factors for patients with osteosarcoma were determined by Cox proportional risk regression with a hazard ratio (HR) of 95%.
Establishment and Verification of Nomogram
After exploring the effect of primary tumour resection on the prognosis of osteosarcoma patients using the above methods, we assumed that the median CSS of patients undergoing primary tumour resection was longer than that of the non-surgical group based on the characteristics of patients in the surgical group. Therefore, we divided the patients who underwent surgery into two groups based on the difference in median CSS—the benefit group (median CSS >10 months) and the non-benefit group (media CSS ≤10 months).
Next, we randomly divided the patients who received surgical treatment and benefited into a training cohort and a validation cohort in a 7:3 ratio. First, the univariate proportional hazards models were used to screen for factors that were significant in the training cohort (P < .2). Then the selected variables were included in multivariate Cox proportional hazards regression to identify independent risk factors (P < .05) that affect the CSS of patients, and finally these factors were drawn into a nomogram picture. According to the calculation results of the nomogram, we defined surgical patients with total predicted probability >.5 as ‘surgical benefit candidates’, and surgical patients with total predicted probability ≤.5 as ‘non-surgical benefit candidates’. Then, receiver operating characteristic (ROC) curves to verify discriminative ability, calibration curves to verify the nomogram predictions, and exploring the net clinical benefit through decision curve analysis (DCA), respectively.All data and analysis procedures were implemented through the statistical software R (version 4.1.2, https://www.r-projectorg/). Finally, we again used Kaplan-Meier analysis to test whether nomogram could distinguish from all patients who could benefit from surgical treatment.
Results
Demographic Characteristics
This study included 412 patients with stage III and IV osteosarcoma. We divided the age into 03-18,18-50, >50 according to the overall age difference performance, as shown in Figure 1. Of these patients, 302 (73.7%) underwent surgery, while 110 (27.3%) opted for non-surgical treatment. The basic data of all patients (age, gender, primary site, pathological type, sixth edition staging, TNM, radiotherapy, chemotherapy) and the differences before and after PSM are shown in Table 1. Before PSM, there were significant differences between the two groups in age of first onset site, disease stage, radiotherapy and chemotherapy, indicating that the baseline characteristics of the two groups were unbalanced. After PSM, 78 osteosarcoma patients in each group were generated. All baseline characteristics of patients in the surgical and non-surgical groups were well balanced (all P > .05). At the same time, the standardized differences in baseline variables between the two groups after matching were all <10%, as shown in Figure 2. (A) histogram (B) Kaplan-Meier curve are constructed based on the determined critical values. Demographic Information of Patients With Stage Ⅲ and Ⅳ Osteosarcoma Before and After PSM. PSM: propensity score matching. Standardized differences of baseline variables between patients with and without surgery before and after PSM. PSM: propensity score matching.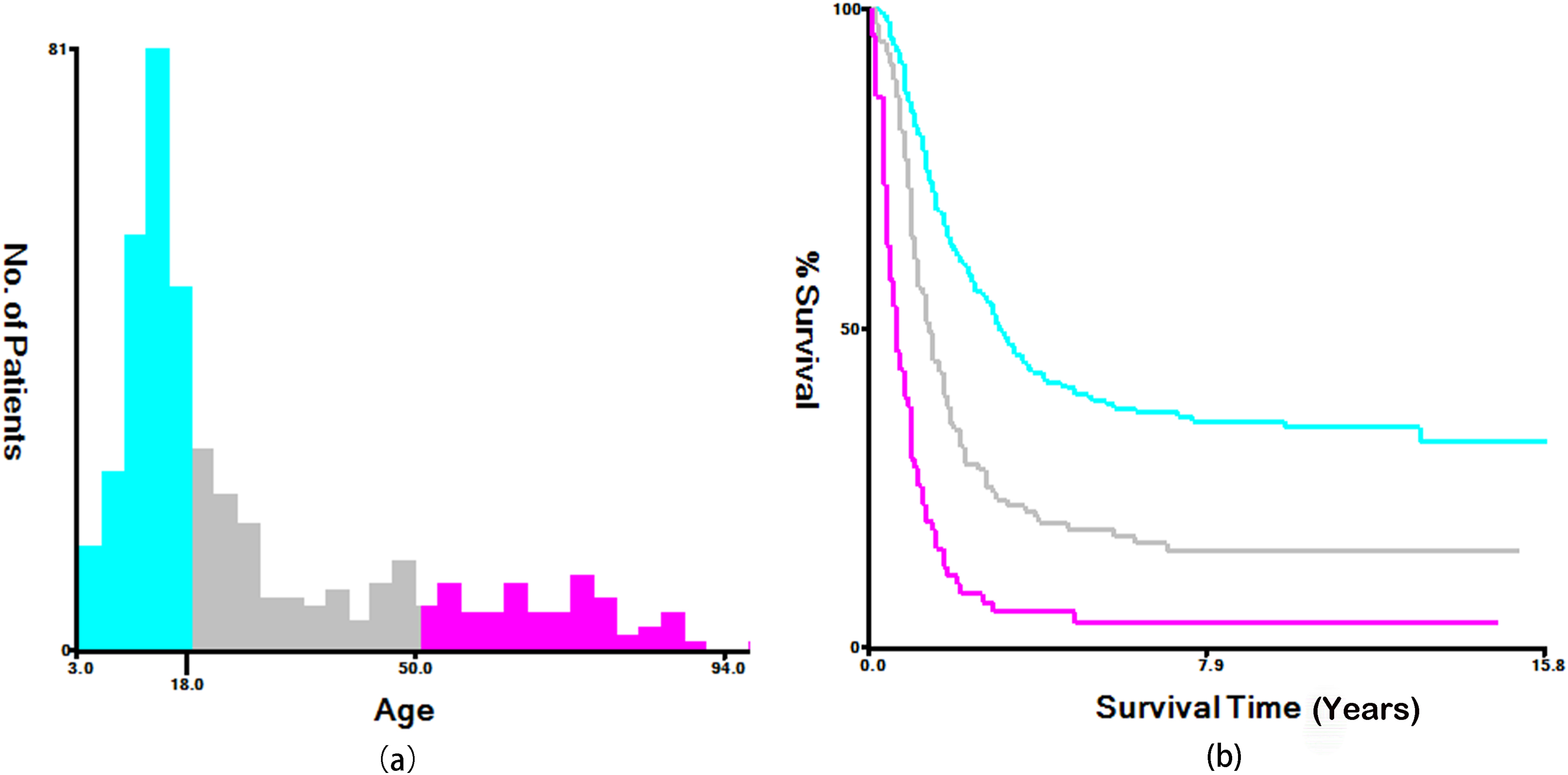


Correlation Between surgery and Survival in Patients with Osteosarcoma
In the following analyses, the information of the patients after PSM was used. As shown in Figure 3, a Kaplan–Meier analysis of the patients showed a significant difference in survival outcomes between patients who underwent surgery (median CSS: 17 months) and non-surgical patients (median CSS: 10 months) (HR = 1.834, 95% CI: 1.284-2.619, P < .001). At the same time, as shown in Figure 4, multivariate logistic regression analysis shows that patients with osteosarcoma who receive surgical treatment have a better prognosis than those who do not receive surgical treatment. Kaplan–Meier curve to evaluate the effect of surgery on CSS in patients with osteosarcoma after PSM. CSS: cancer-specific survival. Cancer-specific hazard ratios for CSS in surgical vs non-surgical patients under different variables. Squares represent effect size (HR) calculated by primary tumour surgery vs no primary tumour surgery in different subgroups; horizontal bars (error bars) represent 95% CIs. CSS: cancer-specific survival; HR: hazards ratio; CI: confidence interval.

Construction and Verification of Nomogram
With the appeal analysis, it is not difficult to come to the conclusion that the prognosis of osteosarcoma patients who underwent primary tumour resection was improved. To distinguish Chinese osteosarcoma patients as candidates who could benefit from surgery, we hypothesized that patients who underwent surgery and survived longer than the median CSS (10 months) in the non-surgical group could benefit from primary tumour resection. We divided the participants in the surgical group into two categories. Fifty-five patients (70.5%) survived for more than 10 months and were divided into the benefit group, and 23 (29.5%) patients with survival time ≤10 months were divided into the non-benefit group of surgery. Univariate and multivariate logistic analyses of the patients in the benefit group concluded that age, primary site and chemotherapeutic factors were all significant influencing factors (P < .05). These were used to construct a predictive nomogram (Figure 5). Nomogram predicts candidates for primary tumour benefit from osteosarcoma surgery. When the calculated predicted probability of a surgical benefit candidate is > .5, the patient will be classified as a benefit candidate. SBCPP: surgery benefit candidate predictive probability.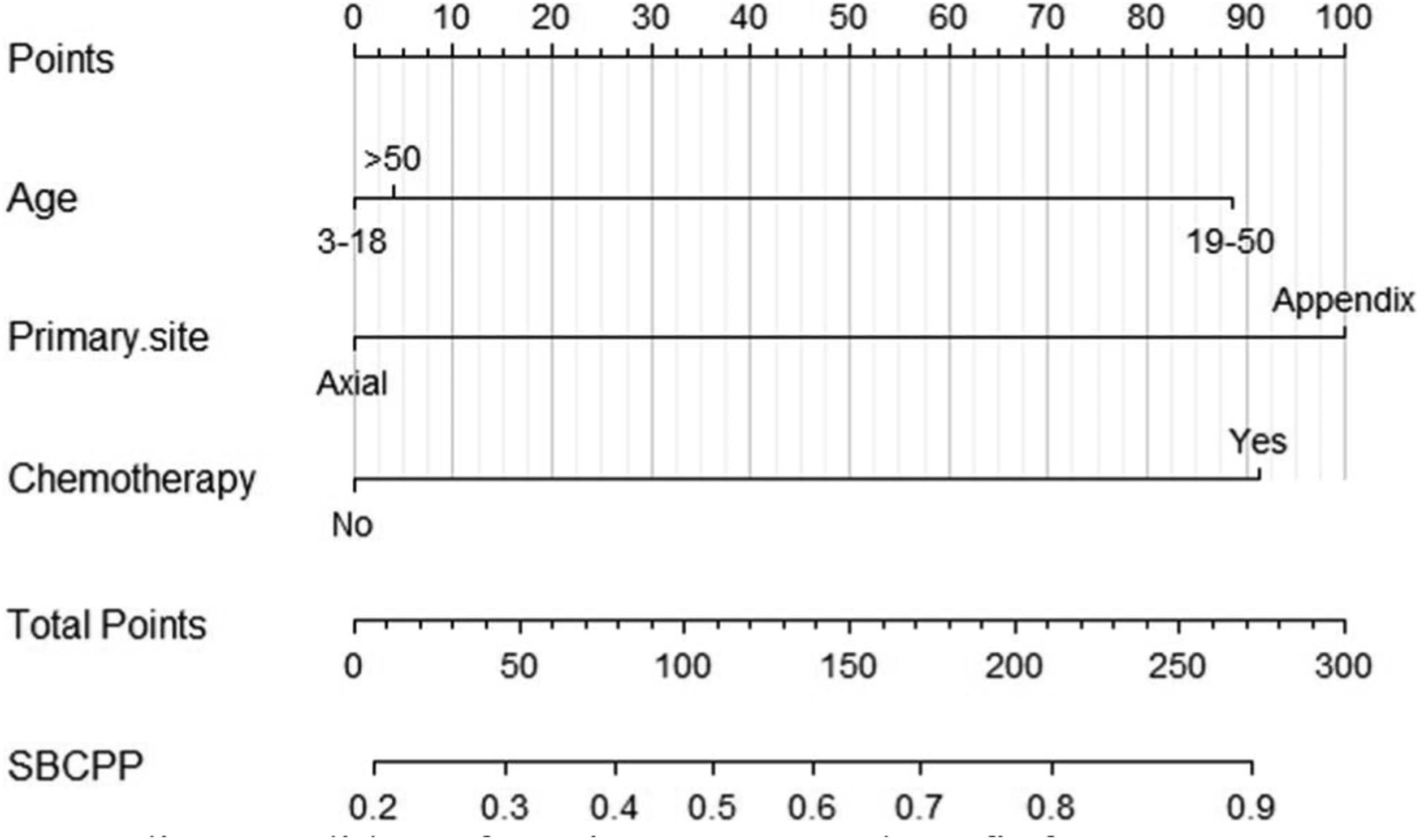
Next, we validated the predicted nomogram. The ROC curve shows that the nomogram has good discriminative ability (Figure 6); the training cohort AUC = .677 (.527-.828), and the validation cohort AUC = .848 (.689-1.000). The calibration curve demonstrated that the nomogram possessed good prediction accuracy (Figure 7). Subsequently, we plotted DCA curves and confirmed that surgical treatment could benefit patients and improve patient outcomes (Figure 8). In the final Kaplan–Meier analysis, we can see that the survival outcomes of the different groups are accurately distinguished in both the training and validation cohort (Figure 9). In the training cohort, the CSS of the surgery benefit group was significantly higher than that of the surgery non-benefit and non-surgery groups (HR = .3.420, 95% CI: .296-13.046, P < .001); the same results were obtained in the validation cohort (HR = 3.657, 95% CI: .627-21.312, P = .029), indicating that the nomogram can identify patients with osteosarcoma who can benefit from surgery and can identify some patients who cannot benefit from surgery. ROC curve of predicted nomogram. A: training cohort ROC curve, AUC = .677 (.527-.828); B: validation cohort ROC curve, AUC = .848 (.689-1.000). ROC: receiver operating characteristic; AUC: area under the curve. Calibration curve for predicted nomogram. A: training cohort; B: validation cohort. Decision curve of the nomogram. The area under the curve represents net income. Kaplan–Meier survival curves of osteosarcoma patients with different benefit classifications according to nomogram (surgical benefit group and surgery non-benefit group) and non-surgical group.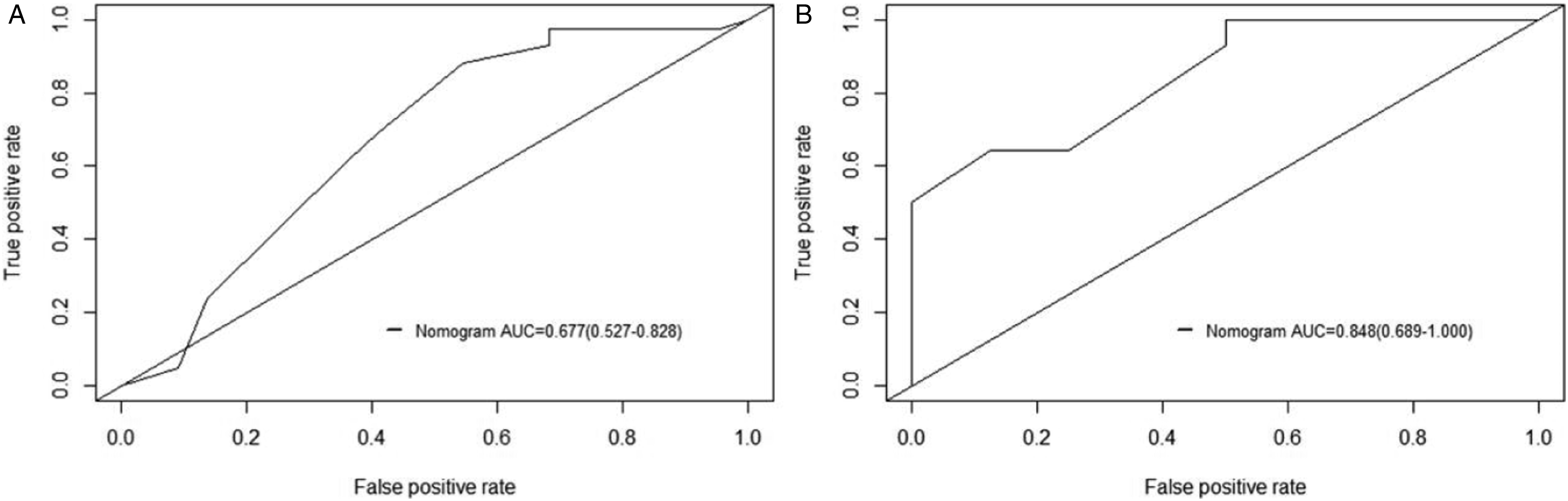



Discussion
Osteosarcoma typically occurs in the metaphysis of the proximal humerus, distal femur and proximal tibia and is rarely seen in the axial bone. It is characterized by rapid progression, early pulmonary metastasis, poor prognosis and easy recurrence . 12 Factors affecting the prognosis of patients with osteosarcoma include tumour growth site, tumour size, Enneking stage, whether the osteosarcoma is combined with pathological fractures and whether neoadjuvant chemotherapy is performed. In the past 30 years, the 5-year OS of osteosarcoma patients increased from 20% to 60%, 6 and multidisciplinary assistance in diagnosis and treatment is inseparable. There is no research report on whether patients with stage III and IV osteosarcoma also need to actively consider surgical treatment. The question is if surgery will benefit the patient.
The nomogram is a tool that can combine multiple clinical risk factors, perform predictive assessments, and ultimately visualize the results, 13 thus giving clinicians the ability to make more accurate survival assessments and treatment decisions. 14 As shown in Figures 3 and 4, surgery is an important factor affecting the prognosis of patients with osteosarcoma, and patients with osteosarcoma who receive surgical treatment have a better prognosis. Subsequently, Cox regression analysis was performed on all variables, and it was determined that age, tumour primary site and chemotherapy were important factors affecting the prognosis of patients with stage III and IV osteosarcoma. Using these three factors, we constructed nomograms that predicted whether a patient might benefit from surgery. The ROC curves and calibration graphs of the training cohort and the validation cohort show that the nomogram has good discriminative ability, and the DCA curve shows that the nomogram has certain clinical utility.
The primary tumour site is an important factor in determining the prognosis of patients. The incidence of osteosarcoma in adolescent patients is mainly concentrated in the extremities, and the incidence of osteosarcoma in the spine increases with age. 3 In a study by Pan et al, the survival rate of patients with osteosarcoma of the extremities was significantly higher than that of patients with a first occurrence in the spine. 5 Age is also an important factor, with first episodes mainly occurring in children and adolescents. 15 A study by Xu et al showed that the first peak incidence of osteosarcoma is typically before 30 years of age. 16 The prognosis of younger children and older people is not good, which may be related to physical reasons and resistance.
Chemotherapy is an indispensable treatment before and after surgery, but after many years of research there is no standard for drug regimens and doses. At present, most regimens are based on MAP (methotrexate, doxorubicin/doxorubicin, cisplatin). However, in clinical use it is empirical medication.17,18 Adequate preoperative chemotherapy can improve the success rate of surgery. 19 Currently, 8-10 weeks of neoadjuvant chemotherapy is recommended before surgery to improve the survival rate of patients17,20; the stronger the tumour response to preoperative chemotherapy, the better the patient’s prognosis. 21
Surgery is a vital part of many treatments. Thirty years ago, amputation was the mainstay surgical treatment for osteosarcoma, but it could lead to deformity, disability and dysfunction, with a high recurrence rate and a low survival rate. 22 Currently, traditional amputation has been gradually abandoned, and clinicians will try to choose limb-preserving surgery, which is also the first choice for the surgical treatment of osteosarcoma. The advantage of amputation surgery is that it can remove the primary tumour lesions to the greatest extent. Combined chemotherapy before and after surgery can prevent tumour metastasis, significantly reduce the recurrence rate and may even have a radical effect. 23 The shortcomings of amputation surgery are very clear. The loss of limbs can lead to dysfunction, can seriously affect the quality of life of patients and can cause great psychological trauma to patients. 11 Although limb salvage surgery is currently highly praised, it is mainly suitable for patients with early osteosarcoma. The contraindications are also clear, including age of onset of osteosarcoma <8 years old, if the condition of soft tissue is extremely poor or accompanied by infection, if the tumour is overly large and if the tumour is not sensitive to chemotherapy or is invalid. 24 Of course, surgical methods should be selected according to the individual differences of patients. The nature of the surrounding tissue of the primary tumour should be fully considered when deciding to employ the tumour resection method. For example, fat removal should be more extensive compared to the removal of fascia. At the same time, limb salvage surgery is not recommended for patients with significant disease progression or those on neoadjuvant chemotherapy. The surgical method also depends on the location of the tumour in the bone, as the articular surface and normal bone tissue should be preserved as much as possible to improve postoperative function. 10 In conclusion, the choice of type of surgery is affected by many factors, including age, advanced tumour stage, larger tumour size, comorbidities and lower income. 9
In the previous analysis, we found that the survival period of patients who received surgery was 7 months longer than that of patients who did not undergo surgery, based on Kaplan–Meier curve analysis. This is in agreement with the research results of other scholars. Through the SEER database, we analysed the survival data of all patients with stage I-IV osteosarcoma. Almost all patients with stage I and II received surgical treatment, and a small number of patients with stage III and IV osteosarcoma did not receive surgical treatment. This is the purpose of our study. Do all patients with advanced osteosarcoma benefit from surgery? In the final analysis, the Kaplan–Meier curve showed a significant difference in survival between patients who benefited from surgery and those who did not benefit from surgery. This also verifies our conjecture that surgery cannot benefit every stage III and IV osteosarcoma patient. Therefore, we first hypothesized and then built a predictive model to screen candidates who would benefit from primary tumour surgery.
Of course, this study has limitations. First, although the SEER database contains a large number of samples, some important variables are screened out. At the same time, because it is a retrospective study data analysis is inevitably biased. Second, to ensure at least 5 years of follow-up, we included observations from 2004-2015. Because the AJCC staging of osteosarcoma is different from other tumours and the staging standards for spinal osteosarcoma and extremity osteosarcoma are different, we can only use the sixth edition of the AJCC staging. Finally, even after careful consideration and with the expectation of including more variables, we found that there were only 110 non-surgical patients registered and only 78 patients who met the criteria after PSM. This is also an imperfect point. In future studies, it is necessary to examine more cases and to improve the predictive nomogram. Although there are related deficiencies, there is no denying the results of this study. We evaluated the impact of multiple factors on the survival of patients with advanced osteosarcoma who opted for surgical treatment or not, and the final results also suggest that nomogram can help clinicians understand who benefits from surgery.
Conclusion
Nomogram, as an effective prediction model, has been used in previous studies . 25 Through the initially proposed hypothesis and validation, we constructed a predictive model to screen out patients with stage III and IV osteosarcoma who would benefit from surgery. Primary tumour resection has undeniable benefits for patients, but it is not suitable for every patient with stage III and IV osteosarcoma. Our prediction model can serve as reference for surgeons to a certain extent and can help in avoiding unnecessary surgical risk to patients.
Footnotes
Acknowledgments
Thanks to the National Cancer Institute for providing the SEER data.
Author Contributions
All authors were responsible for the experimental design. ZJ is mainly responsible for data analysis and article writing. XY is involved in data analysis, SX is responsible for experimental design guidance. XY and ZJ contributed to the work equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Our data is available from the Surveillance, Epidemiology, and End Results (SEER) research database. This is a public research database.
