Abstract
Background
The incidence and prevalence of early-onset colorectal cancer (EO-CRC), defined as colorectal cancer diagnosed before the age of 50, are increasing globally. However, the current status and trends of the disease burden of EO-CRC in China, including incidence, prevalence, mortality, and disability-adjusted life-years (DALYs), are not well understood. This study aimed to analyze the epidemiological trends of EO-CRC in China from 1990 to 2021 and to project its future burden.
Methods
We analyzed data from the Global Burden of Disease (GBD) 2021 study to assess the trends in incidence, prevalence, mortality, and DALYs of EO-CRC in China from 1990 to 2021. Joinpoint regression analysis was used to identify significant changes in trends. Age-period-cohort (APC) analysis was conducted to disentangle the effects of age, period, and birth cohort. The Bayesian APC model was employed to project the burden of EO-CRC up to 2036.
Results
From 1990 to 2021, the absolute number of EO-CRC incident and prevalent cases in China increased substantially. The age-standardized incidence rate (ASIR) and age-standardized prevalence rate (ASPR) also rose significantly, with an accelerated increase after 2007 in men and after 2015 in women. In contrast, the age-standardized mortality rate (ASMR) and age-standardized DALYs rate (AS-DALYs) generally declined; however, a concerning reversal of this trend has been observed in recent years. Incidence, prevalence, mortality and DALYs rates all showed significant age, period, and cohort effects. Projections indicate that ASIR and ASPR will continue to rise until 2036, especially in males, and the disparity in disease burden between men and women is expected to widen.
Conclusion
The disease burden of EO-CRC in China has increased significantly and is rising rapidly, particularly among males. Further research is essential to fully understand the factors contributing to the increased incidence of EO-CRC and to develop effective mitigation strategies.
Plain Language Summary
Purpose
This study aimed to analyze the epidemiological trends of EO-CRC in China from 1990 to 2021 and to project its future burden.
Methods
We analyzed data from the Global Burden of Disease (GBD) 2021 study to assess the trends in incidence, prevalence, mortality, and DALYs of EO-CRC in China from 1990 to 2021. Joinpoint regression analysis was used to identify significant changes in trends. Age-period-cohort (APC) analysis was conducted to disentangle the effects of age, period, and birth cohort. The Bayesian APC model was employed to project the burden of EO-CRC up to 2036.
Results
From 1990 to 2021, the absolute number of EO-CRC incident and prevalent cases in China increased substantially. The age-standardized incidence rate (ASIR) and age-standardized prevalence rate (ASPR) also rose significantly. In contrast, the age-standardized mortality rate (ASMR) and age-standardized DALYs rate (AS-DALYs) generally declined; however, a concerning reversal of this trend has been observed in recent years. Incidence, prevalence, mortality and DALYs rates all showed significant age, period, and cohort effects. Projections indicate that ASIR and ASPR will continue to rise until 2036, especially in males, and the disparity in disease burden between men and women is expected to widen.
Conclusion
The disease burden of EO-CRC in China has increased significantly and is rising rapidly, particularly among males. Further research is essential to fully understand the factors contributing to the increased incidence of EO-CRC and to develop effective mitigation strategies.
Introduction
In recent years, the overall incidence of colorectal cancer (CRC) has declined, primarily due to the widespread implementation of CRC screening programs and advancements in treatment modalities. 1 However, in contrast to this trend, the incidence of early-onset colorectal cancer (EO-CRC) has been increasing, drawing significant attention. 2 EO-CRC defined as CRC diagnosed before the age of 50. 3 A study encompassing 1 922 167 CRC patients demonstrated an increasing trend in EO-CRC incidence across all five continents. 4
EO-CRC may exhibit distinct etiological factors, molecular characteristics, clinical presentations, and prognoses compared to late-onset colorectal cancer (LO-CRC). 5 EO-CRC is more frequently located in the distal colon and rectum and is more likely to present with aggressive histological subtypes, such as poorly differentiated, mucinous, and signet ring cell carcinomas. 6 The etiology of EO-CRC remains incompletely understood but likely involves complex interactions between genetic predisposition, environmental factors, and the gut microbiota. Familial history is recognized as a substantial risk factor for EO-CRC. 7 Approximately 30% of individuals with EO-CRC have at least one first-degree relative with a prior diagnosis of CRC. 8 Hereditary CRC syndromes, such as Lynch syndrome, are strongly associated with EO-CRC development.9,10 However, genetic factors alone cannot fully explain the rapidly rising trend of EO-CRC incidence, which has prompted researchers to turn greater attention towards the widespread influence of environmental and lifestyle factors. Westernized dietary patterns, characterized by high fat, low fiber intake, and consumption of red and processed meats, are considered risk factors.11-13 Furthermore, gut dysbiosis may play a role in EO-CRC pathogenesis.14,15 Consequently, EO-CRC represents a substantial cancer burden for young adults.
China has experienced rapid economic growth over the past three decades, driven by policies such as the Reform and Opening-up and its accession to the World Trade Organization. While this economic development has led to improvements in living standards, it has also been accompanied by significant lifestyle changes, including shifts in dietary habits and reduced physical activity, which may contribute to the rising incidence of EO-CRC. 16 Previous research has shown a substantial increase in the burden of EO-CRC in China from 1990 to 2019. In 2019, the age-standardized incidence rate (ASIR) of EO-CRC in China was 7.30 (95% UI: 6.02, 8.71) per 100 000 population, and the age-standardized prevalence rate (ASPR) was 49.54 (95% UI: 41.06, 58.91) per 100 000 population. 17
Accurate epidemiological data and trend analyses are essential to address the rapidly growing disease burden of EO-CRC. However, the lack of recent epidemiological data on EO-CRC in China poses significant challenges to prevention and screening efforts. Therefore, utilizing the latest data from the Global Burden of Disease (GBD) 2021 study, we present the long-term trends in the burden of EO-CRC in China from 1990 to 2021. Key indicators examined include incidence, prevalence, mortality, and disability-adjusted life-years (DALYs). DALYs, a key metric used in the GBD study, quantify the health loss caused by diseases and injuries. One DALY represents the loss of one year of healthy life due to premature mortality or disability. 18 DALYs offer several advantages for assessing disease burden: they go beyond simply counting deaths (mortality) to capture the overall impact of a disease on population health, and they allow for standardized comparisons across diseases and regions. DALYs is particularly well-suited for evaluating the disease burden of EO-CRC. Because EO-CRC patients are often diagnosed at a relatively young age, they tend to lose more potential years of healthy life compared to those with LO-CRC. Furthermore, we project the burden of EO-CRC in China for the next 15 years (2022-2036) to inform future policy decisions aimed at addressing the evolving burden of EO-CRC and meeting the healthcare needs of the population.
Methods
Data Sources
Data for this study were sourced from the Global Burden of Disease (GBD) 2021 database (https://ghdx.healthdata.org/gbd-results-tool). The GBD study employs a systematic, standardized approach to collect, analyze, and synthesize data on disease and injury burden worldwide, providing estimates at the global, regional, and national levels. 19 We obtained estimates of incidence, prevalence, mortality, and DALYs, along with their 95% uncertainty intervals (UIs), for China, stratified by year, location, age group, and sex. This study complies with the GATHER guidelines. 20
Definition of Early-Onset Colorectal Cancer (EO-CRC)
In this study, EO-CRC was defined as CRC diagnosed before the age of 50. 21 As the GBD 2021 database did not include CRC cases diagnosed before the age of 15 (data not shown), we used data for individuals aged 15 to 49 for our analysis.
Joinpoint Regression Analysis
To quantify and assess temporal trends in EO-CRC burden indicators, joinpoint regression analysis was performed. This statistical method identifies points in time where a significant change in the trend occurs, dividing the time series into segments with linear trends. The optimal number of joinpoints was determined using the grid search method. 22 First, a range for the number of joinpoints was pre-specified (typically 0-5). Beginning with the model with the fewest joinpoints, the sum of squared errors (SSE) was calculated for each model and compared. The optimal model was selected based on the SSE between models. If two models did not differ significantly, the model with fewer joinpoints was chosen. Subsequently, a permutation test was used to assess whether adding joinpoints significantly improved model fit. 23 Joinpoint software (version 4.9.1.0; National Cancer Institute, Rockville, MD, USA) was used for this analysis. After determining the optimal number of joinpoints, the software calculated the annual percentage change (APC) for each linear segment. A positive APC indicates an increasing trend, while a negative APC indicates a decreasing trend. Statistical significance of the APCs was assessed using a two-sided t-test, with a P-value less than .05 considered statistically significant. 24
Age-Period-Cohort Analysis
We used the Age-Period-Cohort (APC) web-based analysis tool developed by the National Cancer Institute (NCI) (https://analysistools.cancer.gov/apc/) to analyze trends in EO-CRC disease burden. 25 The core of the NCI’s APC web tool is the use of a Poisson regression model to estimate and interpret the relative contributions of age, period, and cohort effects. 26 Age effects refer to changes in disease risk with increasing age. Period effects reflect changes in risk affecting all age groups at a specific time due to events or environmental factors. Cohort effects refer to similar risk patterns among individuals born in the same period. The model is specified as follows: log(E(Rate ijk )) = μ + αi + βj + γk, where log(E(Rate ijk )) represents the logarithm of the expected value, μ represents the overall level, αi represents the age effect for the ith age group, βj represents the period effect for the jth period, and γk represents the cohort effect for the kth birth cohort. Both age and period intervals were set at 5 years in this study. The overall linear trend in disease burden, after accounting for age, period, and cohort effects, was estimated as the net drift. We assessed the statistical significance of the net drift by examining its 95% confidence interval (CI). A CI excluding zero indicated a statistically significant net drift. A positive net drift signified an increasing trend in the outcome, while a negative net drift indicated a decreasing trend. Local drifts represented fluctuations in disease burden within specific age groups. 27 Subsequently, we fitted a Bayesian Age-Period-Cohort (BAPC) model to the existing data to project estimates of EO-CRC disease burden in China, along with their 95% CIs, for the period 2022-2036. The BAPC model represents a sophisticated statistical approach for forecasting future disease trends. This model incorporates the age, period, and cohort effects estimated from the APC analysis, extrapolating these effects to project future patterns. 28 The assumptions of the BAPC model include the data distribution assumption and the Prior Structure Assumption. The BAPC model assumes that the observed case counts follow a Poisson distribution. The most critical assumption for BAPC is the Prior Structure Assumption. To address the inherent non-identifiability problem in APC models, BAPC does not introduce arbitrary equality constraints. Instead, it introduces structured prior distributions (second-order random walk priors) for the age, period, and cohort effects. This imposes an assumption that the effects vary smoothly over time (or across age/cohort), which is a standard approach in BAPC modeling. 29 BAPC modeling was performed using the BAPC package in R 4.3.1. 30
Results
Descriptive Analysis
Number of Cases and Age-Standardized Incidence, Prevalence, Mortality, and DALYs Rates for EO-CRC in China in 1990 and 2021.
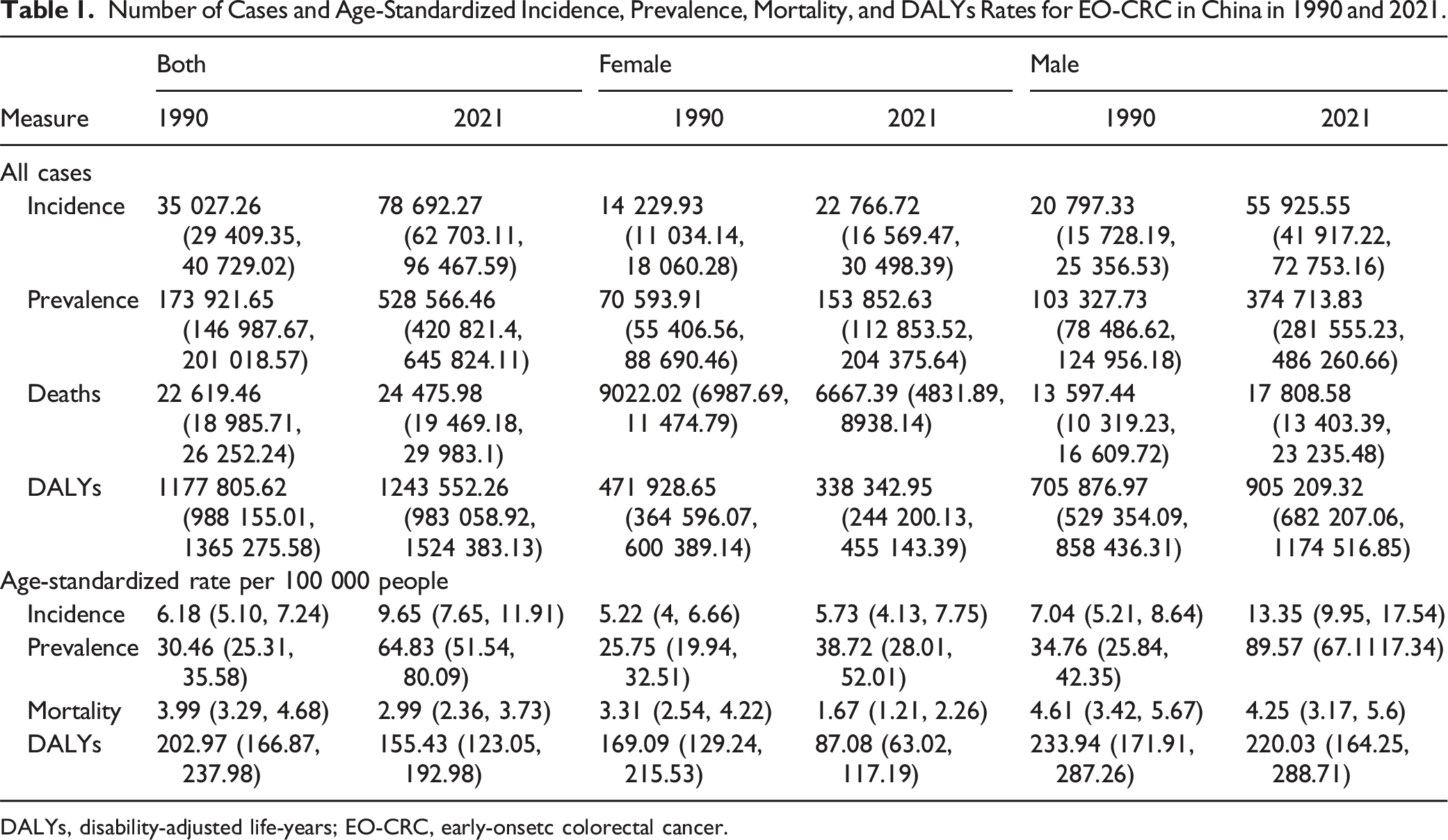
DALYs, disability-adjusted life-years; EO-CRC, early-onsetc colorectal cancer.
Figure 1 illustrates the age and sex distribution of EO-CRC incidence and prevalence in China in 1990 and 2021. From 1990 to 2021, the number of incident and prevalent cases increased significantly in age groups 25 and older, with the magnitude of increase escalating with age (Figure 1A and B). For incidence, the change across age groups was relatively stable in females, while a substantial increase was observed in males after age 25. Prevalence trends mirrored those of incidence (Figure 1C and D). For mortality and DALYs, trends differed between males and females. From 1990 to 2021, the number of deaths and DALYs decreased in most age groups for females, while in males, these numbers decreased before age 30 and increased thereafter. ASMR and AS-DALYs decreased across all age groups in females, with little change observed in males (Figure 2). Age and Sex Distribution of Incidence and Prevalence of EO-CRC in China, 1990-2021. (A) Number of Incident Cases by Age Group. (B) Number of Prevalent Cases by Age Group. (C) Incidence Rates by Age Group. (D) Prevalence Rates by Age Group. EO-CRC, Early-Onset Colorectal Cancer. Age and Sex Distribution of Mortality and DALYs Rates for EO-CRC in China, 1990-2021. (A) Number of Deaths by Age Group. (B) Number of DALYs by Age Group. (C) Mortality Rates by Age Group. (D) DALYs Rates by Age Group. DALYs, Disability-Adjusted Life-Years; EO-CRC, Early-Onset Colorectal Cancer.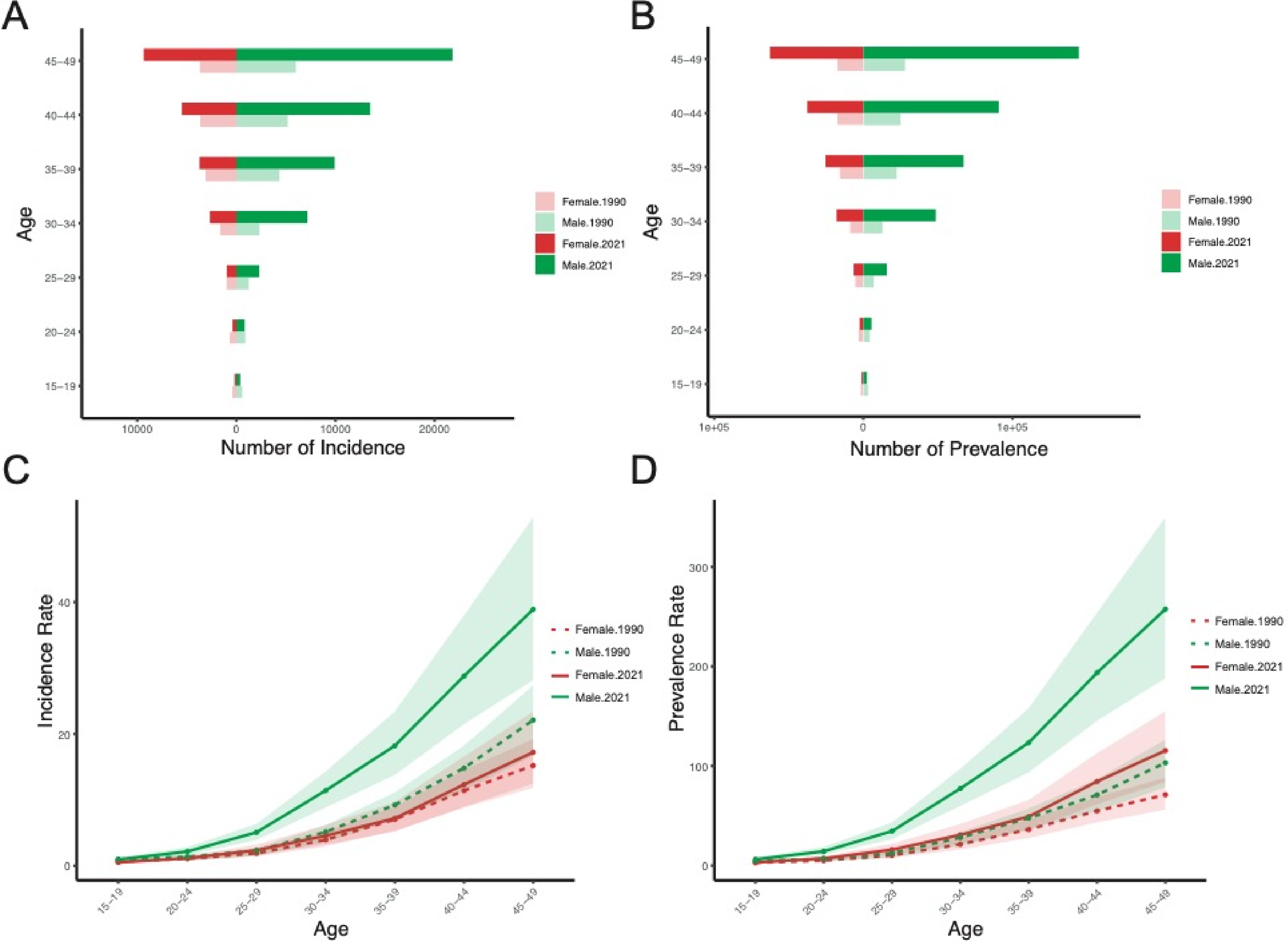

Temporal Trends in EO-CRC Burden
Joinpoint regression analysis was conducted to investigate temporal trends in ASRs of EO-CRC in China. Female ASIR exhibited a trend of initial decline followed by an increase, with the fastest annual percentage change (APC) observed from 2015 to 2021 (APC = 2.82%). Female ASPR also rose rapidly after 2015 (APC = 3.41%). In males, both ASIR and ASPR showed a continuous upward trend, accelerating notably after 2007. The fastest increase in ASIR was observed from 2007 to 2010 (APC = 4.61%), while ASPR showed an APC of 4.40% from 2007 to 2021 (Figure 3A and B). Female ASMR and AS-DALYs exhibited a continuous decline, although AS-DALYs showed a slight increase after 2015 (APC = 0.57%). While ASMR in females showed a positive APC, it was not statistically significant. Male ASMR and AS-DALYs displayed a pattern of initial decline followed by an increase, with the most rapid rise occurring from 2007 to 2010 (APC = 2.50% for ASMR and 2.44% for AS-DALYs) (Figure 4A and B). Temporal Trends in Age-Standardized Incidence and Prevalence Rates of EO-CRC in China, 1990-2021. (A) Age-Standardized Incidence Rate by Year. (B) Age-Standardized Prevalence Rate by Year. EO-CRC, Early-Onset Colorectal Cancer. Temporal Trends in Age-Standardized Mortality and DALYs Rates of EO-CRC in China, 1990-2021. (A) Age-Standardized Mortality Rate by Year. (B) Age-Standardized DALYs Rate by Year. DALYs, Disability-Adjusted Life-Years; EO-CRC, Early-Onset Colorectal Cancer.

Net and Local Drifts
Figure 4 examines the net and local drifts of incidence, prevalence, mortality, and DALYs rates across different age groups. Net drift represents the overall annual percentage change, while local drift refers to the annual percentage change within specific age groups. For incidence (Figure 5A-C) and prevalence (Figure 5D-F), the net drifts were 1.47 (95% CI: 1.17, 1.77) and 2.49 (95% CI: 2.22, 2.76), respectively, both indicating increasing trends. For mortality (Figure 5G-I) and DALYs (Figures 5J-L), the net drifts were −1.27 (95% CI: −1.56, −0.97) and −1.22 (95% CI: −1.5, −0.93), respectively, both indicating decreasing trends. Regarding local drifts, incidence and prevalence in individuals under 20 years old were significantly lower than the net drift. The decline in mortality and DALYs rates was significantly greater than the net drift. The remaining age groups generally fluctuated around the net drift, suggesting consistent overall trends across age groups. All numerical values of net and local drifts are presented in Table S1. Net and Local Drifts of Incidence, Prevalence, Mortality, and DALYs Rates for EO-CRC in China. (A) Net and Local Drifts for All Age Incidence Rates. (B) Net and Local Drifts for Female Incidence Rates. (C) Net and Local Drifts for Male Incidence Rate. (D) Net and Local Drifts for All Age Prevalence Rates. (E) Net and Local Drifts for Female Prevalence Rates. (F) Net and Local Drifts for Male Prevalence Rates. (G) Net and Local Drifts for All Age Mortality Rates. (H) Net and Local Drifts for Female Mortality Rates. (I) Net and Local Drifts for Male Mortality Rates. (J) Net and Local Drifts for All Age DALYs Rates. (K) Net and Local Drifts for Female DALYs Rates. (L) Net and Local Drifts for Male DALYs Rates. DALYs, Disability-Adjusted Life-Years; EO-CRC, Early-Onset Colorectal Cancer.
Age-Period-Cohort (APC) Analysis of EO-CRC Burden
To investigate the contributions of age, period, and cohort to EO-CRC burden, we conducted APC analysis on prevalence, incidence, mortality, and DALYs rates of EO-CRC in China (Figure 6). As shown, incidence, prevalence, mortality, and DALYs rates all increased with age, corroborating the age-related burden increase observed in Figures 1 and 2. Figure 6B, E, H and K illustrate period effects. Incidence and prevalence rapidly increased after 2005. Although mortality and DALYs rates continuously declined from 1990 to 2015, an upward trend emerged after 2015. A clear cohort effect was also observed for EO-CRC burden, with later birth cohorts exhibiting higher incidence and prevalence and lower mortality and DALYs rates. While incidence and prevalence decreased slightly in cohorts born after 1997, this is likely attributable to the younger age of these populations. Age, period, and cohort effects were similar in males and females (Figures S1 and S2). Age-Period-Cohort (APC) Effects on EO-CRC Burden in China. (A) Age Effect on Incidence. (B) Period Effect on Incidence. (C) Cohort Effect on Incidence. (D) Age Effect on Prevalence. (E) Period Effect on Prevalence. (F) Cohort Effect on Prevalence. (G) Age Effect on Mortality. (H) Period Effect on Mortality. (I) Cohort Effect on Mortality. (J) Age Effect on DALYs Rate. (K) Period Effect on DALYs Rate. (L) Cohort Effect on DALYs Rate. DALYs, Disability-Adjusted Life-Years; EO-CRC, Early-Onset Colorectal Cancer.
Prediction of EO-CRC Burden
To project future EO-CRC burden, we stratified patients by sex and employed Bayesian APC (BAPC) modeling to predict changes in EO-CRC burden. ASIR and ASPR are projected to continuously increase in both males and females over the next 15 years (Figure 7), with a faster increase observed in males (steeper slope). By 2036, the projected ASIR for females is 7.93 (95% CI: 2.35, 13.50) per 100 000 population, 1.38 times the 2021 rate. For males, the projected ASIR is 24.98 (95% CI: 9.97, 40.00) per 100 000 population, 1.87 times the 2021 rate. The projected ASPR for females in 2036 is 57.72 (95% CI: 14.90, 100.51) per 100 000 population, 1.49 times the 2021 rate. For males, the projected ASPR is 180.43 (95% CI: 67.11, 293.75) per 100 000 population, 2.01 times the 2021 rate. For ASMR and AS-DALYs, a slight decrease is projected for females by 2036, while an upward trend is anticipated for males (Figure S3). Evidently, the disease burden in males is significantly higher than in females, and this disparity is projected to widen further. Observed and Projected Age-Standardized Incidence and Prevalence Rates by Sex in China, 1990-2036. (A) Observed and Projected ASIR for Females. (B) Observed and Projected ASIR for Males. (C) Observed and Projected ASPR for Females. (D) Observed and Projected ASPR for Males. ASIR, Age-Standardized Incidence Rate; ASPR, Age-Standardized Prevalence Rate.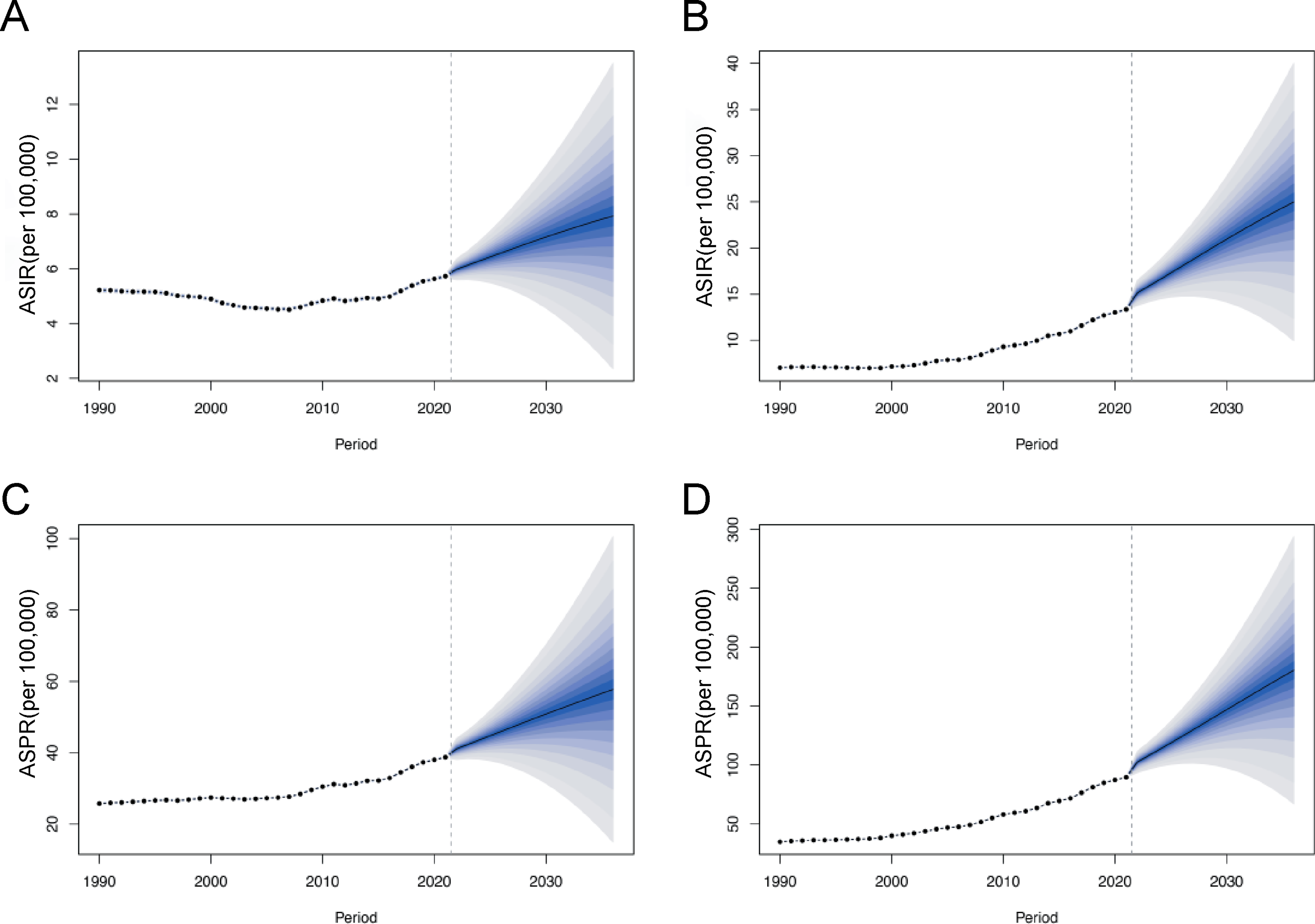
Discussion
This study leverages data from the Global Burden of Disease (GBD) 2021 study to provide a comprehensive analysis of the disease burden of EO-CRC in China from 1990 to 2021, with projections to 2036. Our findings reveal a concerning trend: a substantial increase in the burden of EO-CRC in China over the past three decades. Specifically, the ASIR and ASPR of EO-CRC have shown a consistent upward trend overall, while ASMR and AS-DALYs rates initially declined but have subsequently exhibited an upward trend. This signifies a growing public health concern that necessitates increased attention and targeted interventions.
Previous literature has reported that, as of 2019, the ASIR for EO-CRC in males was 10.6 (95% CI: 10.5, 10.7) per 100 000 population and 4.62 (95% CI: 4.56, 4.68) per 100 000 population in females. 31 Our study indicates that the ASIR and ASPR of EO-CRC have continued to rise after 2019. The rise in ASIR and ASPR of EO-CRC in China may be linked to multiple factors, including Westernized diets, reduced physical activity, rising obesity rates, smoking, and alterations in gut microbiota. Westernized diets, characterized by high fat, low fiber, and high intake of red and processed meat, are a recognized risk factor for EO-CRC. Traditional Chinese diets, typically rich in plant-based foods and lower in animal fats, may have provided a protective effect that is being eroded by these dietary changes. 32 Obesity may be an independent risk factor for EO-CRC; the exact mechanism of this effect is unknown but may involve insulin resistance and chronic inflammatory states. 33 The relationship between the gut microbiome and EO-CRC is a complex and evolving area of research. While the exact mechanisms remain unclear, growing evidence suggests a close association between gut microbiota alterations and the development of EO-CRC. 34 Imbalances in the gut microbial community can lead to chronic inflammation in the gut. This persistent inflammation can damage the intestinal lining and create an environment conducive to cancer development. Certain bacteria, such as Fusobacterium nucleatum, are known to promote inflammation and are enriched in colorectal tumors, including those in EO-CRC patients, leading to poorer prognoses. 35 Furthermore, decreased physical activity, increased sedentary lifestyles, and rising obesity rates associated with modernization may have also contributed to the observed trends.36,37
While EO-CRC and LO-CRC share some common risk factors, there are key distinctions in the potential drivers behind their increasing incidence. EO-CRC demonstrates a stronger influence of genetic predisposition. Specific gene mutations and inherited syndromes, such as Lynch syndrome, are more prevalent in EO-CRC patients compared to those with LO-CRC. 38 Early life exposures, such as antibiotic use and ultra-processed food consumption, have a greater impact, and even a mother’s diet and weight during pregnancy may influence offspring’s risk of developing EO-CRC. 39
The ASMR and AS-DALYs rates initially declined but have subsequently shown an upward trend, similar to trends reported in previous studies.31,40 Advancements in medical technology may be a contributing factor to the initial decline in ASMR and AS-DALYs among EO-CRC patients. However, the reversal of the declining trends in ASMR and AS-DALYs after 2007, with a subsequent increase, necessitates close monitoring and potential reevaluation of current prevention and treatment strategies. Surgical treatment for EO-CRC follows the same stage-adaptive oncological principles as for CRC but should minimize long-term complications from medical and surgical treatments while optimizing cure rates, including emphasizing systemic therapy and judicious use of radiotherapy. Patient medical condition, clinical stage of disease, and tumor molecular profiling are key factors in treatment planning. 41 Traditional sequential drug regimens may not yield optimal outcomes, and personalized treatment approaches based on patient and tumor characteristics should be preferred. International treatment guidelines developed by a consensus process involving 69 experts provide guidance for the management of EO-CRC. These guidelines advocate for germline genetic testing of multiple genes (including APC, BMPR1A, EPCAM, MLH1, MSH2, MSH6, MUTYH, POLD1, POLE, PMS2, PTEM, SMAD4, STK11, and TP53) in all newly diagnosed EO-CRC patients, preferably before initiating any treatment. 42
APC analysis reveals a complex interplay of age, period, and cohort effects on the EO-CRC burden in China. The strong age effect, with increasing disease burden with advancing age, is consistent with the known biology of CRC. The period effect, characterized by a sharp rise in incidence and prevalence after 2005 and increasing mortality and DALYs after 2015, underscores the influence of time-specific factors such as environmental exposures and lifestyle changes. Cohort effects show that later birth cohorts have higher incidence and prevalence (except for those born after 1997, likely due to their young age). This suggests that something about the environment or shared lifestyle of younger individuals increases their susceptibility to EO-CRC, with possible explanations being changes in dietary habits and unhealthy lifestyles.
Our BAPC model projections reveal concerning trends in disease burden. The projected continued rise in ASIR and ASPR, particularly in men, underscores the importance and urgency of disease prevention, early screening, early diagnosis, and early intervention. The first priority is to strengthen primary prevention. Public health campaigns should promote healthy lifestyles, including balanced diets rich in vegetables and grains, reduced intake of red and processed meat, regular physical activity, and maintaining a healthy weight. The second priority is early screening. Currently, multiple guidelines recommend advancing the screening age for colorectal cancer. The American Cancer Society (ACS) recommends screening for average-risk individuals beginning at age 45, based on disease burden, microsimulation modeling results, and the reasonable expectation that screening in adults aged 45-49 will have similar effectiveness as in older adults.43,44 In 2021, the US Preventive Services Task Force (USPSTF) guidelines also added a B recommendation for screening individuals aged 45-49 to its recommendations on screening start and end ages. 45 A study predicted that starting screening earlier would prevent one additional CRC death per 1000 people screened regularly starting at age 45 (rather than 50). 46
The Chinese Expert Consensus on Early Diagnosis and Treatment of Colorectal Cancer (2023 Edition) provides specific screening recommendations for individuals at high risk of EO-CRC. 47 These recommendations include: (1) Individuals with two first-degree relatives diagnosed with CRC (or one first-degree relative diagnosed before age 60) should undergo colonoscopy every 5 years. (2) Individuals with adenomatous polyposis syndromes or those carrying pathogenic mutations should undergo annual colonoscopy. (3) Carriers of pathogenic mutations in Lynch syndrome families should begin colonoscopy every 2 years starting at age 20. (4) Patients with ulcerative colitis should undergo colonoscopic screening no later than 8 years after the onset of symptoms. However, based on our clinical experience, adherence to these recommendations among young adults appears to be low. 48 Raising risk awareness among young high-risk individuals with a family history of advanced adenomas or CRC is an effective way to improve screening adherence in this high-risk population and curb the rising incidence of EO-CRC. 8 To maximize screening awareness among young adults, a multi-pronged approach is needed. Offline, efforts should leverage primary healthcare institutions to conduct health education lectures, free medical consultations, and similar outreach activities. Online, the widespread use of social media platforms in China (such as WeChat, Weibo, Douyin, and Xiaohongshu) should be utilized for sustained educational campaigns, emphasizing warning signs such as rectal bleeding, changes in bowel habits (diarrhea or constipation), persistent abdominal pain or bloating, and unexplained weight loss.
Although ideally screening should be tailored to individual risk, personalized risk assessment has not yet been adopted for CRC screening. Assessment of genetic polymorphisms, early life exposures, and their environmental interactions might improve the efficiency of screening and early detection of EO-CRC. On the other hand, although the incidence of CRC is increasing in young people, the absolute incidence of CRC in young people is still much lower than in older people. Therefore, there is very limited evidence on whether current screening methods are suitable for young people. For screening young people, using risk scores to identify high-risk individuals can reduce the burden of colonoscopy and is a more recommended screening approach. However, existing risk prediction models for colorectal cancer need further optimization and validation with local data before they can be better translated into application. 49
Limitations
Admittedly, this study has some limitations. First, our research is subject to the general limitations of the GBD study, such as the availability and quality of raw data. Second, GBD does not provide detailed information differentiating cancer locations (proximal colon, distal colon, or rectum), which prevents us from conducting sub-analyses of EO-CRC disease burden. Finally, due to China’s vast territory, diverse ethnic groups, and significant differences in socio-economic development between the eastern and western regions, the burden of colorectal cancer may vary considerably across different regions. Therefore, we call for the establishment of a dedicated EO-CRC database in China. This would allow for a clear observation of disease burden disparities across different regions and ethnic groups, enabling targeted solutions to address the specific challenges and difficulties faced by various populations.
Conclusions
This study provides a comprehensive and systematic analysis of the EO-CRC disease burden in China using high-quality data from GBD 2021 and multiple analytical methods. It offers valuable insights into the current status and trends of EO-CRC burden in China, providing a scientific basis for developing effective disease prevention and control strategies. Our findings reveal that from 1990 to 2021, both ASIR and ASPR increased substantially, while the declining trend in ASMR and AS-DALYs has been reversed in recent years. There are significant sex differences in EO-CRC burden, with a substantially higher burden in males. Furthermore, ASIR and ASPR are projected to continue rising rapidly over the next 15 years. Our study reinforces the call for improved early detection strategies, increased public awareness, and expanded research into the genetic and lifestyle factors contributing to the increasing burden of EO-CRC.
Supplemental Material
Supplemental Material - Trends and Projections of Early-Onset Colorectal Cancer Burden in China, 1990-2036: Findings From the Global Burden of Disease 2021 Study
Supplemental Material for Trends and Projections of Early-Onset Colorectal Cancer Burden in China, 1990-2036: Findings From the Global Burden of Disease 2021 Study by Tianze Huang, Jianfu Qiu, Changhao Wang, Xiang Ma, Duo Liu, and Jian Cai in Cancer Control
Footnotes
Acknowledgements
We thank the Institute for Health Metrics and Evaluation at the University of Washington for contributing to this study.
Ethical Statement
Author Contributions
Tianze Huang and Jian Cai contributed to conception and design of the study. Tianze Huang, Jianfu Qiu and Changhao Wang participate in acquisition of data, analysis and interpretation of data. Xiang Ma and Duo Liu participate in acquisition of data. All authors participate in manuscript writing and revision. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Shenzhen Second People’s Hospital Clinical Research Fund of Shenzhen High-level Hospital Construction Project (Grant No. 20243357016).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
