Abstract
Introduction
Papillary thyroid carcinoma (PTC) generally has a favorable prognosis, yet advanced PTC has higher recurrence and mortality risks. This study constructs and validates a competing risk model for cause-specific mortality (CSM) in advanced PTC.
Methods
Stage III-IV PTC cases (AJCC 7th edition) from the SEER database (2010-2015) were analyzed. Patients were split into training and validation sets (7:3). Univariate and multivariate analyses identified independent CSM predictors, forming the basis of a risk prediction nomogram. Model accuracy was evaluated via the C-index and calibration curve.
Results
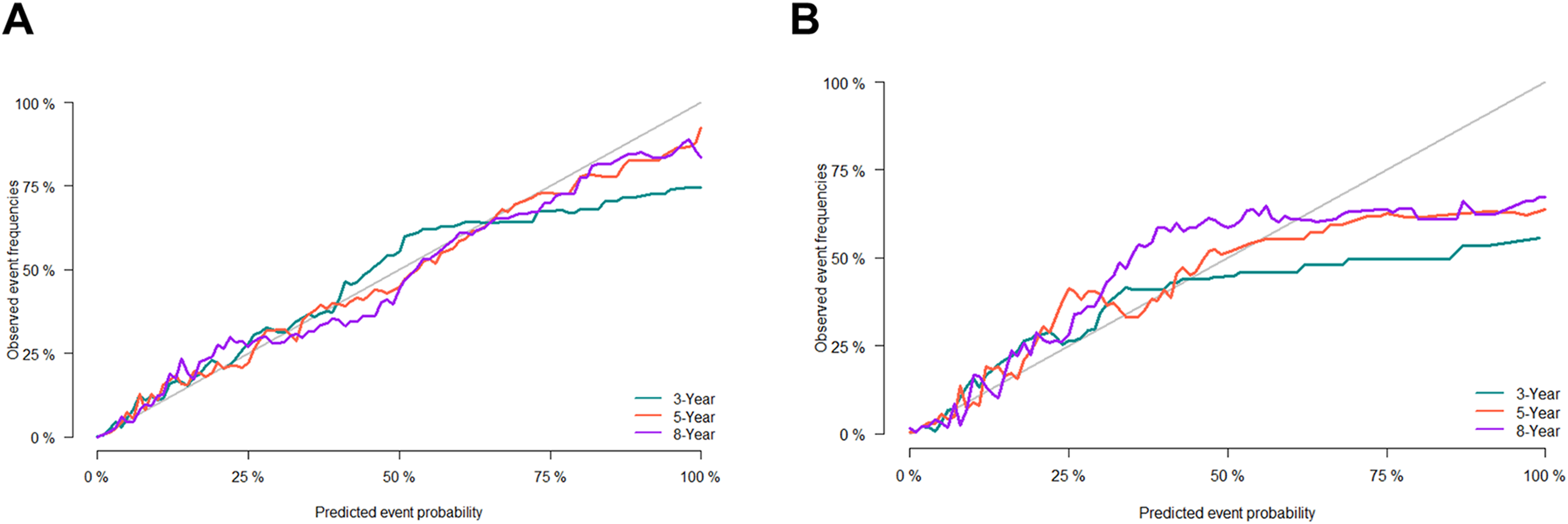
A total of 11 913 advanced PTC cases were analyzed. Competing risk model analysis unraveled that age, race, sex, grade, stage, T stage, M stage, surgery, chemotherapy, and tumor size were risk factors for CSM in advanced PTC. The AUC values of the constructed nomogram in predicting 3-, 5-, and 8-year survival were 0.931 (95%CI 0.909-0.953), 0.915 (95%CI 0.897-0.933), and 0.902 (95%CI 0.883-0.92) in the training set, and 0.948 (95%CI 0.916-0.981), 0.93 (95 % CI 0.903-0.957), and 0.917 (95%CI 0.891-0.943) in the validation cohort, respectively. The C-index of the nomogram for advanced PTC was 0.908 and 0.921 in the training and validation cohorts, respectively. The calibration curve unveiled that the predicted estimates by the model were basically congruent with the observed values, suggesting a high degree of calibration.
Conclusion
The competing risk model offers a reliable tool for assessing prognosis in advanced PTC, supporting personalized treatment and risk management in clinical practice.
Plain Language Summary
Papillary thyroid cancer (PTC) is the most common type of thyroid cancer, and it usually has a good outlook. However, for patients with advanced stages of the disease, the risks of complications and death are higher. This study aimed to identify the factors that contribute to death caused specifically by advanced PTC and to develop a tool to predict these risks. We analyzed data from over 11 000 patients with advanced PTC collected between 2010 and 2015 from a large database in the United States. By using advanced statistical methods, we identified key factors that affect the risk of death from advanced PTC, such as age, tumor size, cancer stage, and whether the patient underwent surgery or chemotherapy. We used this information to create a prediction tool called a “nomogram,” which can estimate the chances of survival for three, five, and eight years after diagnosis. The tool was tested and found to be highly accurate in predicting outcomes, making it a valuable resource for doctors. It helps them assess risks and make more personalized treatment decisions for patients with advanced PTC. For example, doctors can use the nomogram to identify patients who may benefit from surgery or other targeted treatments.
Introduction
Papillary thyroid carcinoma (PTC) is the most frequent thyroid malignancy, representing about 80%-85% of all thyroid cancer cases. 1 The incidence of PTC is on the rise worldwide, and the increase in the incidence of thyroid cancer is mainly attributable to an elevating incidence of PTC.2,3 According to epidemiological data, the incidence of PTC in females is approximately three times that in males, and is the highest in individuals aged between 30 and 50 years old.4,5 While the prognosis of most patients is favorable with a relatively high survival rate, the risk of death in individuals with advanced PTC remains high owing to disease progression and complications. 6
PTC originates from thyroid follicular cells and is one of the most frequent differentiated thyroid cancers. 7 For patients with advanced PTC, common clinical manifestations include local invasion, distant metastasis, or recurrence, which complicate the prediction of their prognosis. 8 Generally, PTC patients would receive surgical treatment combined with postoperative thyroid-stimulating hormone (TSH) suppression. 9 Despite a high 5-year survival rate of over 90%, 10 the risk of recurrence is significantly increased in patients with lymph node metastasis or distant metastasis, leading to poor prognosis. 11 Advanced PTC is typically characterized by local invasion, lymph node metastasis, and distant metastasis, which considerably compromise the prognosis of patients. In recent years, radioiodine therapy, targeted therapy (such as BRAF inhibitors), and immunotherapy have been gradually used in the management of advanced PTC. However, the prognosis of advanced PTC is still uncertain. 12 The long-term survival rate of individuals with advanced PTC is considerably lower than that of those with early-stage PTC. Furthermore, the response to different treatments is significantly distinct between individuals. Therefore, it is crucial to develop more accurate risk assessment models and personalized treatment plans. Because advanced PTC is more intractable than localized PTC, the disease progression to advanced PTC may result in increased mortality. The causes of death in individuals with advanced PTC are diverse. Except for tumor-related factors, non-tumor factors such as age and treatment-related side effects may also lead to mortality.13,14 Due to such diverse causes of death, it is difficult to use traditional survival analysis methods to accurately assess the likelihood of mortality in individuals with advanced PTC. In contrast, competing risk models provide new methods and ideas for exploring such complex situations. 15
The Surveillance, Epidemiology, and End Results (SEER) database is a cancer surveillance program established by the National Cancer Institute (NCI) of the United States. It covers specific clinical information on numerous cancer patients across the United States and is an important data source for studying cancer survival and prognosis. 16 Based on the SEER database, the current study intended to deeply analyze the cause-specific mortality (CSM) in individuals with advanced PTC and to evaluate the influence of different factors on the risk of CSM by constructing a competing risk model. The current study is expected to elucidate the causes of death in advanced PTC and optimize risk assessment methods, thereby offering a solid basis for clinical decision-making.
Materials and Methods
Data Sources and Study Population
The data utilized in the current study were collected from the SEER database. This database encompasses detailed information on plentiful cancer patients in different regions of the United States, including patient demographic characteristics, tumor characteristics, treatment methods, and survival. The SEER*Stat software (version 8.3.5) was leveraged to extract data on individuals diagnosed with advanced PTC between 2010 and 2015. The patients who met the following criteria were incorporated into the current study: individuals diagnosed with advanced PTC, defined as cases with local invasion, lymph node metastasis, or distant metastasis; individuals aged ≥18 years; individuals with TNM (American Joint Committee on Cancer, AJCC 6th) stage III-IV; individuals with complete clinical and follow-up data, including diagnosis time, treatment information, survival status, and cause of death. The following patients were removed: patients with non-primary thyroid cancer; individuals with missing or incomplete data; and patients with a survival time of < one month after diagnosis, to avoid the influence of extreme data on the results. This study was reported following the TRIPOD guidelines.17,18
Data Collection
The clinicopathological information on individuals with PTC collected in the current study encompassed age, marital status, race, sex, grade, stage, T stage, N stage, M stage, surgery, lymph node metastasis surgery, radiation, chemotherapy, regional nodes positive, bone metastasis, brain metastasis, liver metastasis, lung metastasis, tumor size, multifocal tumor, status, and survival months.
Model Construction
At a ratio of 7:3, the data were randomized into a training cohort and a validation cohort. In the training set, the variables with P < .1 in the univariate competing risk model were incorporated into the multivariate competing risk model to determine independent risk factors for CSM in PTC. Then, a prediction nomogram was constructed based on the risk factors. The constructed model was validated utilizing the validation cohort. The C-index and area under the receiver operating characteristic (ROC) curve (AUC) were leveraged to appraise the predictive performance of the model, and a calibration curve was utilized to examine the calibration of the model.
Statistical Analysis
Descriptive statistics were executed for the demographic and clinical characteristics of the patients with advanced PTC. Continuous variables were displayed as mean ± standard deviation (SD), and categorical variables were described as frequencies and percentages. The Competing Risks Model proposed by Fine and Gray et al. 19 was leveraged to analyze the specific causes of death in patients with advanced PTC. This model is suitable for survival data analysis where there are multiple competing endpoints. The consistency index (C-index), AUC values at different time points, and calibration curve were utilized to verify the applicability and predictive performance of the model. In addition, the Wald test was used to test the significance of each variable in the model, and P values <.05 suggested statistical significance. All data analyses were executed utilizing R software (4.2.0) and SPSS software (25.0).
Result
Clinical Characteristics of Patients with Advanced PTC
Clinicopathological Characteristics of Patients With Advanced PTC.

Note: Surg1 (Lobectomy and/or isthmectomy; Removal of less than a lobe, NOS); Surg2 (Removal of a lobe and partial removal of the contralateral lobe; Subtotal or near total thyroidectomy; Thyroidectomy, NOS; Surgery, NOS); Surg3 (Total thyroidectomy).
Variable Screening Based on Univariate and Multivariate Cox Regression Analysis
Univariate and Multivariate Analysis.
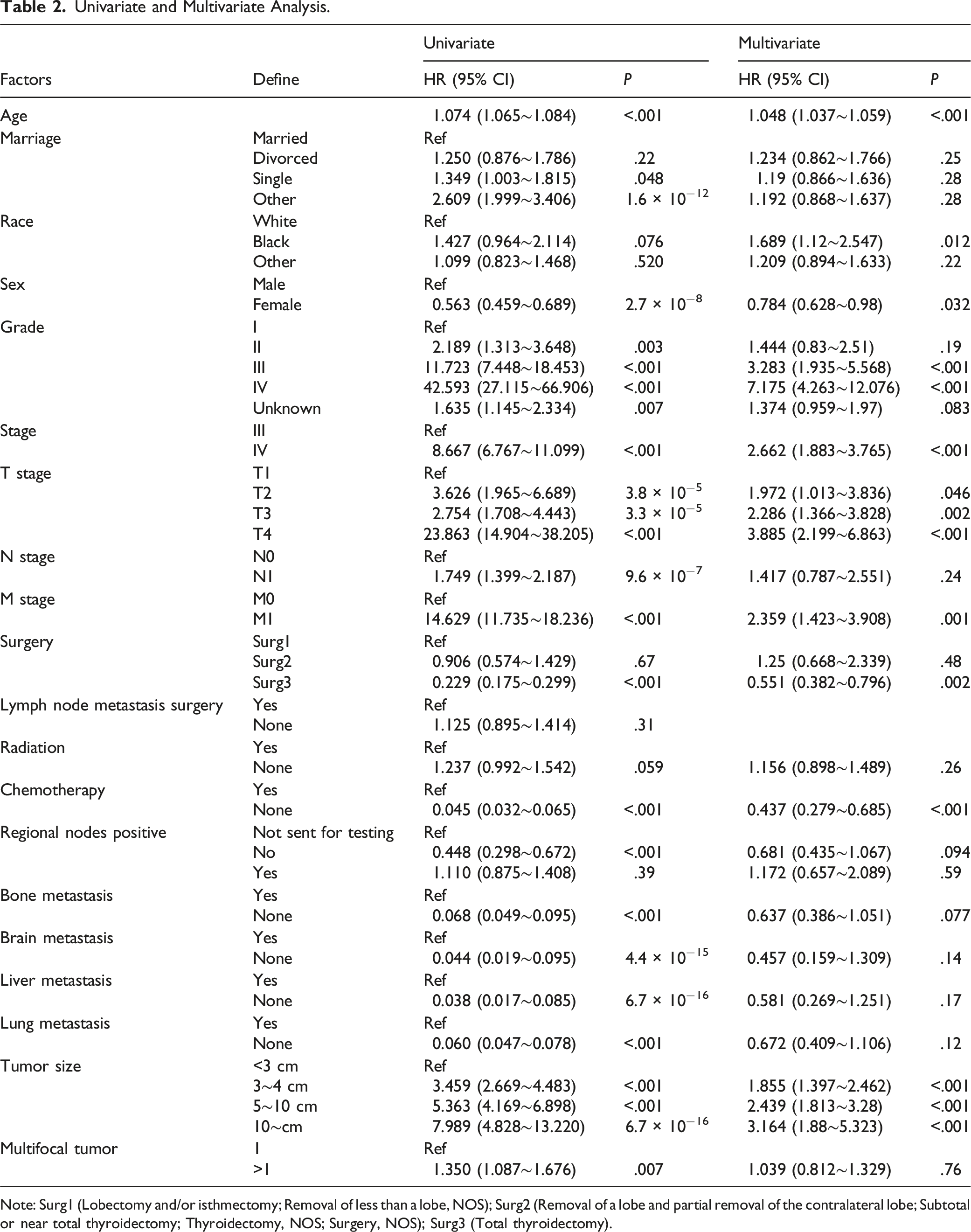
Note: Surg1 (Lobectomy and/or isthmectomy; Removal of less than a lobe, NOS); Surg2 (Removal of a lobe and partial removal of the contralateral lobe; Subtotal or near total thyroidectomy; Thyroidectomy, NOS; Surgery, NOS); Surg3 (Total thyroidectomy).
Nomogram Construction
Based on the variables identified by Cox proportional hazard regression, a prediction nomogram for patients with advanced PTC was constructed. As shown in Figure 1, each variable corresponded to a score, and the total score was the sum of all the scores of all variables. The nomogram can be used to forecast 3-, 5-, and 8-year survival probabilities for a given patient. As illustrated in the nomogram, age, stage, and grade significantly affected the prognosis of advanced PTC (Figure 1). Nomogram for Forecasting 3-, 5-, and 8-Year Disease-specific Survival in Patients with Advanced PTC in the Training Cohort.
Nomogram Validation
The predictive performance of the nomogram was appraised by several methods. First, the discrimination ability of the nomogram was evaluated using the C-index and AUC. The C-index of the nomogram was 0.908 (0.001) and 0.921 (0.002) in the training and validation cohorts. The AUC values for forecasting 3-, 5-, and 8-year survival were 0.931 (95%CI 0.909-0.953), 0.915 (95%CI 0.897-0.933) and 0.902 (95%CI 0.883-0.92) in the training cohort, respectively, and 0.948 (95%CI 0.916-0.981), 0.93 (95%CI 0.903-0.957), and 0.917 (95%CI 0.891-0.943) in the validation cohort (Figure 2). Second, the calibration curve (Figure 3) was plotted to examine the consistency between the predicted and actual survival outcomes in patients with advanced PTC. The results unraveled that the predicted survival rates were highly congruent with actual survival outcomes in the training and validation cohorts, unraveling that the discrimination performance of the nomogram was satisfactory. The distribution of cumulative mortality of independent predictors is shown in Figure S1. ROC Curve of the Nomogram for Cause-specific Survival in Advanced PTC. (A) Training Cohort; (B) Validation Cohort. Calibration Curve for Forecasting Survival Probability in Patients with Advanced PTC. (A) Training Cohort; (B) Validation Cohort.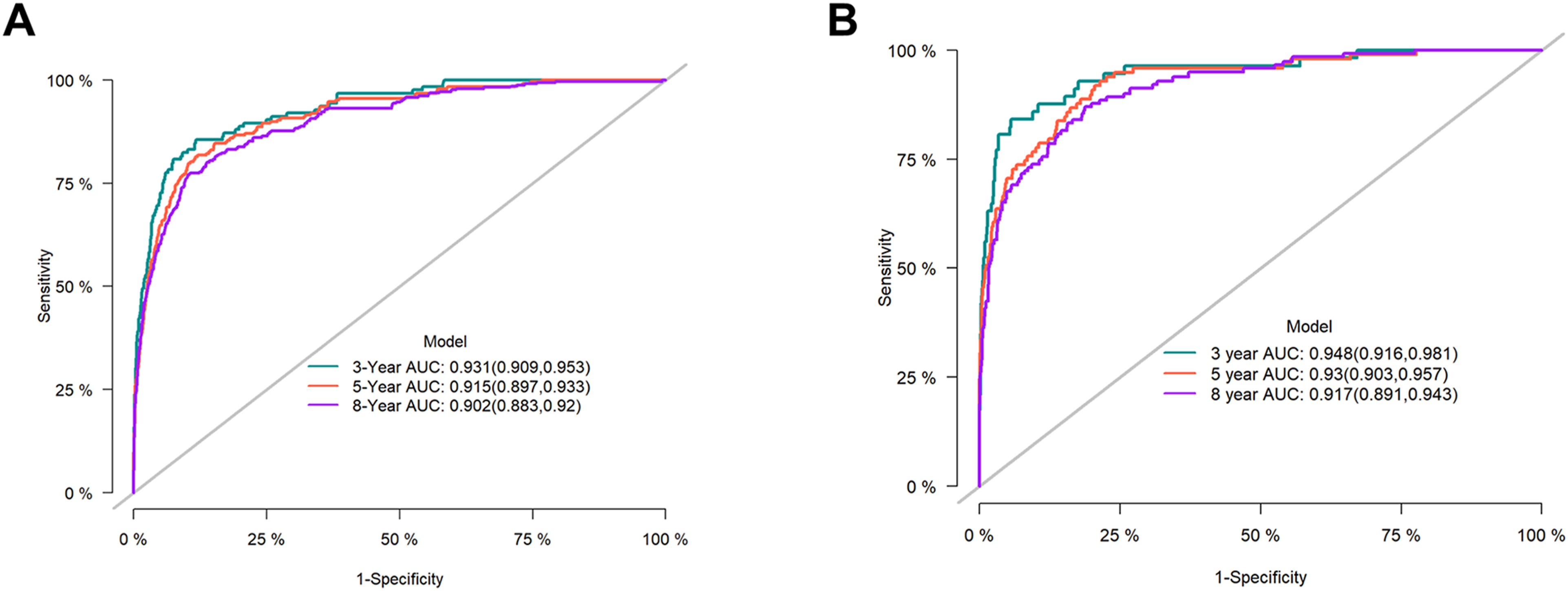
Discussion
By utilizing competing risk regression, several key factors significantly associated with CSM in individuals with advanced PTC were found. Multivariate competing risk regression analysis unveiled that age, race, sex, grade, stage, T stage, M stage, tumor size, chemotherapy, and surgery were risk factors for CSM in advanced PTC. In the training set, the AUC values of the nomogram for forecasting 3-, 5-, and 8-year survival were 0.931 (95%CI 0.909-0.953), 0.915 (95%CI 0.897-0.933), and 0.902 (95%CI 0.883-0.92), respectively, and 0.948 (95%CI 0.916-0.981), 0.93 (95%CI 0.903-0.957) and 0.917 (95%CI 0.891-0.943) in the validation cohort, respectively. These findings highlight the complex interplay of clinical and treatment-related variables and demographics in determining patient outcomes. This study represents the first study to develop a competing risk model specifically for advanced PTC, providing a novel approach to predicting the risk of CSM in individuals with advanced PTC.
The global burden of PTC is increasing, and age is one of the key factors in the occurrence of all cancers. The risk of genetic mutations rises with age, and thus age serves as one of the strongest predictors of cancer death. 20 The study by Deng, Y. et al 21 showed that the age-standardized incidence rate of thyroid cancer was on the rise; thyroid cancer-related death cases mainly occurred in individuals aged 70 and older; and the mortality rate increased significantly with age. Shi et al 22 Have established a Kaplan-Meier survival curve and performed multivariate Cox model analysis. They suggested that both thyroid cancer-specific survival rate and overall survival rate decreased with increasing age. Their study unraveled that age over 60 years was a predictor of poor prognosis and high risk of thyroid cancer-specific death. Our competing risk model confirmed age as an independent risk factor for CSM in advanced PTC, consistent with previous studies. Kaliszewski K et al. 23 successfully stratified PTC patients with poorer prognoses by using the age of 55 years as a threshold. Our competing risk model further validated age as an independent risk factor for PTC at advanced stages. In addition to age, tumor aggressiveness (reflected in characteristics such as local invasion and metastatic potential) is influenced by other factors, including gender, tumor stage, chemotherapy, and metastasis. Ruiz et al. 24 supported this multifactorial interaction. Their univariate analyses demonstrated that age, gender (specific to thyroid carcinoma), histologic type, pathological stage, and cervical lymph node status had a profound impact on the prognosis of thyroid cancer. Their results reveal that stage plays a critical role in PTC outcomes, which is consistent with our findings. Notably, mortality is found to be significantly higher in advanced recurrent thyroid cancer compared to early stage. 25 As a result, the clinical stage is a reliable predictor of tumor behavior (e.g., recurrence or progression). 26 The research by Li et al. 27 also unravels that stage is an independent prognostic factor for thyroid cancer, and both stage and tumor size are important risk factors. 28 These findings are consistent with our findings, further emphasizing the critical role of stage and tumor size in the prognostic evaluation of advanced PTC.
In our study, multivariate competing risk regression analysis identified tumor grade as a significant risk factor for CSM in patients with advanced PTC. This finding is particularly noteworthy, as PTC is typically regarded as a well-differentiated malignancy with a relatively favorable prognosis. Historically, tumor grade is less noted compared to factors such as tumor stage or size in clinical risk assessments. However, our model demonstrated that a higher tumor grade, particularly in poorly differentiated tumors, was independently associated with an increased risk of CSM. This result suggests that tumor grade may reflect biological features and aggressiveness of tumors in advanced PTC, which directly impact patient prognosis. While PTC is generally classified as well-differentiated, it is important to recognize that a subset of cases may be dedifferentiated and progress into poorly differentiated or even mesenchymal thyroid cancers. These subtypes are known to be more aggressive with a higher risk of metastasis and are associated with poorer outcomes. Similar findings have been reported in some existing studies. For instance, Rodríguez-Cuevas et al. 29 emphasized the prognostic value of histological features, and Wang et al. 30 highlighted the impact of clinical stage and tumor size on patient outcomes. Our results suggest that tumor grade complements traditional risk factors, such as stage and tumor size, providing a more comprehensive approach to risk stratification in advanced PTC. This result underscores the potential of tumor grade as a valuable marker for identifying high-risk patients. Therefore, we propose that tumor grade should be considered an important supplementary indicator in clinical assessments, helping predict cancer-specific mortality in PTC. Future research is needed to explore the molecular basis of tumor grade, including potential mutations and epigenetic changes, and assess its role in guiding clinical decision-making. Specifically, investigating how tumor grade influences the efficacy of therapeutic interventions in advanced cases could enhance our understanding of its clinical utility.
In addition to clinicopathological factors like age, tumor stage, tumor grade and tumor size, complete surgical resection and postoperative radiotherapy have a significant influence on the prognosis of PTC. Complete tumor resection has been demonstrated to be the most effective technique for patients with TNMI PTC. However, incomplete surgical resection is associated with higher mortality, especially in high-risk. 31 Based on the SEER database, Ho et al 32 conducted stratified analysis according to surgical and non-surgical treatment and found that as age and tumor size increased, the risk of death in patients who did not undergo surgery gradually elevated, but this increase was only significant at certain points above thresholds. In their study, non-surgical treatment may be an appropriate option in some cases; however, once a certain critical point is exceeded, the survival benefit of surgical treatment becomes significant. Su et al. 33 applied the Cox proportional hazard model to compute the hazard ratio of prognostic factors. The multivariate analysis in their study unraveled that distant metastasis, local recurrence, and positive surgical margins were important independent risk factors for overall survival, death, and cumulative mortality in individuals with thyroid cancer. Therefore, identifying the risk factors for distant metastasis and local recurrence is crucial for implementing early prevention and surgical treatment. In fact, although many patients survive after receiving incomplete surgical resection, the resection of the primary tumor is still a crucial prognostic factor. Our study also unravels that surgical treatment may be appropriate for individuals with PTC regardless of their age. Currently, with rapid advances in the treatment of thyroid cancer, individuals with well-differentiated thyroid cancer, especially PTC, are treated by surgical removal of the thyroid gland. 34 Besides, it has been confirmed that total thyroidectomy can lower the recurrence rate of PTC with a diameter of ≥1.0 cm and improve the survival rate. 35 For cervical lymph node metastasis, therapeutic neck dissection of the central and lateral neck is necessary. Nonetheless, the use of preventive central neck dissection remains controversial and is determined by risk factors in individuals. 36 In conclusion, our results support the National Comprehensive Cancer Network guidelines for the management of PTC, which recommend thyroid lobectomy for low-risk patients. 37
Although some investigations on thyroid cancer have assessed deaths from other causes, they mostly rely on data from a single center 38 and rarely focus on the comprehensive prediction of the risk of CSM in patients with advanced PTC. These studies mainly focus on postoperative risk assessment or Cox regression model analysis, and the main risk factors include age, tumor size, multifocality, metastasis, and extrathyroidal invasion.20,39,40 However, these studies cannot fully consider potential factors that lead to non-thyroid cancer-related deaths, which may weaken the overall prediction performance of the model. In addition, the difference in risk factors is subtle between our competing risk model and the traditional Cox regression model, possibly due to a lower risk of death from thyroid cancer compared with other cancers, which is closely related to the advances in the early diagnosis and management of thyroid cancer. Our results showed that the C-index was 0.908 in the training set and 0.921 in the validation set, indicating that the model is highly accurate in predicting the risk of CSM. In the highly developed medical environment, follow-up strategies are developed in accordance with patient preferences and clinical criteria (e.g., family history, history of neck radiation, hypothyroidism, or thyroid nodules), and thus most PTC patients undergo thyroidectomy in the early or middle stages of the disease. However, in the presence of competing events, a competing risk model was more accurate in analyzing the prognostic factors of advanced PTC.
Strengths and Limitations
Our competing risk model is advantageous because it can account for the likelihood of different causes of death, thereby providing a more nuanced understanding of the contributors to mortality in patients with advanced PTC. Unlike the traditional AJCC TNM staging system, which focuses primarily on tumor characteristics, our model incorporates a wider range of clinical and demographic factors, and can comprehensively assess the risk of death. Our model can predict the risk of CSM at specific time points, such as 3, 5, and 8 years. This capability is particularly valuable for patients with advanced PTC, who are often caught in a dilemma between aggressive treatment options and quality of life. By providing personalized risk assessments, our model may assist clinicians in making more informed decisions about treatment strategies and end-of-life care. On the contrary, several limitations in this study should be acknowledged. First, this is a retrospective study based on SEER data, which may introduce selection bias, and fail to establish causal relationships. Second, the SEER database lacked several key variables, such as detailed co-morbid history, genetic information, and specific treatment details, such as chemotherapy regimen (drug type and dose), radiotherapy parameters, or use of postoperative adjuvant therapy. Consequently, we cannot comprehensively evaluate all the factors affecting the prognosis of advanced PTC. Second, as a generalized retrospective cancer registry, the SEER database is not a validated database designed specifically for this study, and its data quality is dependent on the accuracy and consistency of the reporting institution, which may introduce potential bias. In addition, SEER data are primarily collected from populations in specific regions of the United States. Their racial composition, medical practices, and disease management patterns may differ from those in other countries and regions. Thus, the generalizability of the results of this study may be limited. To overcome these limitations, future studies should incorporate multicenter, internationalized prospective data, include a wider range of variables and more representative populations, and integrate databases containing detailed treatment data to externally validate our findings and promote the generalization of the model.
Although our competing risk model is relatively accurate in predicting the risk of CSM in patients with advanced PTC (C-index is 0.908 in the training set and 0.921 in the validation set), its clinical application is limited since it is complex and is not externally validated. The model integrates multiple variables (e.g., age, gender, grading, staging, etc.), is computationally complex, may not be easy to implement in clinical practice, and is less intuitive than the simplified AJCC TNM staging system. In addition, its generalizability is limited as SEER is primarily representative of a specific population in the United States. Future studies should focus on external validation using multicenter prospective data and explore strategies to simplify the model, such as reducing the number of variables or developing user-friendly digital tools to facilitate its integration into everyday clinical practice.
The duration of follow-up in this study ranged from 1 to 107 months, reflecting real-world data on patients with advanced PTC from the SEER database. We did not deliberately exclude patients with longer follow-up periods. Rather, we included all eligible individuals and incorporated their data comprehensively into the competing risk models. The model effectively assessed long-term prognosis by predicting the cumulative risk of CSM at 3, 5, and 8 years. However, due to the upper limit of follow-up in the SEER database (107 months), it is infeasible to analyze outcomes in patients with ultra-long-term follow-up. Future research could explore the ultra-long-term prognosis of PTC by utilizing extended prospective data over a longer time frame. Conducting multicenter, prospective studies is essential to validate our competing risk model, helping to refine its accuracy and generalizability. In addition, incorporating more detailed patient-level data, such as molecular markers and quality-of-life measures, could enhance the predictive power and clinical relevance of the model. Ultimately, our goal is to provide a powerful, personalized tool that can help clinicians tailor treatment strategies to the specific demands of patients with advanced PTC, thereby improving their outcomes and quality of life.
In summary, our study highlights that managing advanced PTC is complicated, and it is imperative to consider tumor-specific and patient-related factors in clinical decision-making. A competing risk model based on these factors may more accurately predict patient outcomes, thereby providing more personalized care. With the advances in research on thyroid cancer research, our findings provide a foundation for future studies aimed at improving outcomes and quality of life for individuals with advanced PTC.
Conclusion
The current study revealed that age, race, sex, grade, stage, T stage, M stage, tumor size, chemotherapy, and surgery are risk factors for CSM in individuals with advanced PTC. Undergoing surgery is an effective protective factor for mortality in this population. The nomogram for advanced PTC constructed based on the competing risk model can help clinicians identify risk factors and predict the probability of death in advanced PTC patients, thereby guiding individualized treatment and improving their prognosis.
Supplemental Material
Supplemental Material - Identification of Risk Factors for Cause-specific Mortality in Advanced Papillary Thyroid Cancer and Construction of a Competing Risk Model: A SEER-Based Study
Supplemental Material for Identification of Risk Factors for Cause-specific Mortality in Advanced Papillary Thyroid Cancer and Construction of a Competing Risk Model: A SEER-Based Study by Yunyi Yang, Xiaoli He, Xiaoxiao Qu, Shufa Tan, Xinyi Fu, Jiawen You, Weijin Huang, Jiayuan Cai, Yanming He, and Hongjie Yang in Cancer Control
Footnotes
Author contributions
All authors contributed to the study conception and design. Writing - original draft preparation: [Yunyi Yang]; Writing - review and editing: [Yunyi Yang and Xiaoli He]; Conceptualization: [Yunyi Yang, Xiaoli He and Xiaoxiao Qu]; Methodology: [Xiaoxiao Qu, Shufa Tan and Xinyi Fu]; Formal analysis and investigation: [Xinyi Fu, Jiawen You, Weijin Huang and Jiayuan Cai]; Funding acquisition: [Xiaoli He, Hongjie Yang, Yanming He]; Resources: [Yanming He]; Supervision: [Yanming He and Hongjie Yang], and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Yunyi Yang and Xiaoli He are co-first authors, and Hongjie Yang and Yanming He are co-corresponding authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partially supported by the National Natural Science Foundation of China (No. 82205009), Shanghai Hospital Collaborative Innovation Centre for Industrial Transformation of Chinese Pharmaceutical Preparations project, the Shanghai Municipal Health Commission Clinical Research Project (No. 20214Y0178), and the Shanghai Shenkang Three-Year Action Plan for Promoting Clinical Skills and Clinical Innovation in Municipal Hospitals-Research Physician Training in Innovative and Translational Capabilities Project (No. SHDC2022CRD009).
Declarations Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data utilized in the current study were collected from the SEER database.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
