Abstract
Background
The purpose of this study is to employ a competing risk model based on the Surveillance, Epidemiology, and End Results (SEER) database to identify prognostic factors for elderly individuals with sigmoid colon adenocarcinoma (SCA) and compare them with the classic Cox proportional hazards model.
Methods
We extracted data from elderly patients diagnosed with SCA registered in the SEER database between 2010 and 2015. Univariate analysis was conducted using cumulative incidence functions and Gray’s test, while multivariate analysis was performed using both the Fine-Gray and Cox proportional hazards models.
Results
Among the 10,712 eligible elderly patients diagnosed with SCA, 5595 individuals passed away: 2987 due to sigmoid colon adenocarcinoma and 2608 from other causes. The results of one-way Gray’s test showed that age, race, marital status, AJCC stage, differentiation grade, tumor size, surgical status, liver metastasis status, lung metastasis status, brain metastasis status, radiotherapy status, and chemotherapy status all affected the prognosis of SCA (P < .05). Multivariate analysis showed that sex, age, race, marital status, and surgical status affected the prognosis of SCA (P < .05). Multifactorial Fine-Gray analysis revealed that key factors influencing the prognosis of SCA patients include age, race, marital status, AJCC stage, grade classification, surgical status, tumor size, liver metastasis, lung metastasis, and chemotherapy status (P < .05).
Conclusion
Data from the SEER database were used to more accurately estimate CIFs for sigmoid colon adenocarcinoma-specific mortality and prognostic factors using competing risk models.
Introduction
Colorectal cancer, ranking as the fourth most lethal malignancy globally, claims the lives of nearly 900,000 individuals annually. Risk factors associated with this condition encompass obesity, physical sedentariness, and tobacco consumption. Profound insights into the pathophysiological underpinnings have engendered a plethora of therapeutic modalities, including endoscopic and surgical resection, radiation therapy, systemic interventions, and palliative chemotherapy. Notably, patients harboring non-metastatic afflictions exhibit the most favorable prognosis, prompting the implementation of global screening initiatives to enhance early detection. Colorectal cancer contributes to approximately 10% of all cancer diagnoses and related fatalities on a worldwide scale. Notably, incidence and mortality rates exhibit geographical disparities, with the highest burden observed in industrialized nations. Projections suggest that by 2035, the incidence of colorectal cancer will escalate to a staggering 2.5 million new cases. 1
Sigmoid and rectal cancers typically manifest early symptoms and collectively constitute nearly two-thirds of colon malignancies. 2 The sigmoid colon stands as a frequently afflicted site by colorectal cancer, offering prospects for curability when detected at an incipient stage and preventability through the identification and removal of precursor adenomas. Risk determinants for the development of colorectal cancer, including the sigmoid colon, encompass dietary patterns, genetic predisposition, and environmental influences. The genesis of most sigmoid colon cancers is attributed to the direct evolution of adenomatous polyps. 3 Age emerges as a pivotal risk factor in the evolution and progression of colorectal cancer, with its incidence exhibiting a steady ascent between the ages of 40 and 80. 4 This ailment is notably prevalent in the elderly and its prevalence rises in tandem with age. Approximately 90% of novel colorectal cancer diagnoses occur in individuals aged 50 and above. In the United States, the median age at colorectal cancer diagnosis stands at 69 years, with an exponential surge in incidence to 176.1 cases per 100,000 individuals beyond the age of 65. 5 Predominantly, colorectal cancers (in excess of 90%) take the form of adenocarcinomas, representing malignant neoplasms originating from glandular epithelial cells within the colon and rectum. 6 Scientific studies have shown a significantly elevated incidence of adenomas and advanced adenomas in the demographic aged 70-75 years compared to those aged 40-49 years. 5 Younger populations typically exhibit more aggressive disease characteristics, such as poorly or undifferentiated adenocarcinomas, with a higher likelihood of receiving multi-agent chemotherapy regimens compared to older adults. 7 In addition, younger patients tend to have later tumor stage at diagnosis and a higher incidence of rectal cancer resection, which is associated with poorer survival outcomes. 8 In contrast, older adults, particularly those in their eighties, face different challenges when diagnosed with sigmoid carcinoma. This group is more likely to have a higher burden of comorbidities and postoperative complications, and therefore require careful consideration of surgical intervention. 9 However, overall survival in the elderly is influenced by factors other than cancer, with a large proportion of deaths attributed to non-cancer causes such as respiratory failure and cardiovascular disease. 10 Treatment of sigmoid cancer also varies by age, with non-invasive screening options crucial for older adults due to the higher risks associated with invasive procedures. 11 At the same time, younger people may benefit from more aggressive treatment strategies, as they generally have a better prognosis and are more tolerant of intensive therapy. 12 This nuanced approach to treatment highlights the importance of personalizing care based on age, comorbidities, and cancer-specific characteristics. 13 In addition, the advent of minimally invasive surgery (MIS) has provided encouraging outcomes for older adults, with reduced long-term functional dependence and a higher probability of staying at home postoperatively compared to patients undergoing open resection. 14 In conclusion, the risk factors and prognosis of sigmoid carcinoma vary widely between young and older adults, reflecting the need for tailored management strategies to optimize care across the lifespan. Consequently, the exploration of risk determinants impacting the prognosis of elderly patients with sigmoid colon adenocarcinoma (SCA) will facilitate the formulation of more efficacious preventive measures, resource allocation strategies, and health care policies aimed at reducing the incidence of sigmoid colon adenocarcinoma and augmenting patient survival.
Statistical methodologies, such as Kaplan-Meier (KM) survival analysis and Cox proportional hazards regression, are frequently applied in prospective investigations of disease outcomes to accommodate variations in follow-up durations. The most commonly employed approach for generating time-to-event and survival curves is the Kaplan-Meier (K-M) method. Nevertheless, this method typically pertains to the characterization of the time until a singular type of event transpires. When significant and pertinent competing risks, such as mortality, are in play, these techniques can produce biased outcomes. When conducting surveys pertaining to maladies in the elderly population, the presence of competing risks, particularly the risk of mortality, is substantially heightened due to advanced age and the influence of comorbidities. In investigations involving older adults, wherein a substantial portion of participants succumbs during extended follow-up periods, it becomes imperative to consider the likelihood of disease incidence alongside the competing risk of death. In such instances, the employment of cumulative incidence estimates for competing risks and competing risk regressions is indispensable for ascertaining both the incidence and the associated effect estimates. Competing risk methodologies provide a more precise means of gauging disease risk. The accurate determination of an elderly individual’s susceptibility to developing subsequent ailments can significantly impact the clinical decisions made by geriatricians and their patients. 15 At present, two primary competing risk models are prevalent: the Cause-Specific Risk Function (CS) model, 16 used predominantly in etiological investigations, and the Fine-Gray model, 17 employed for the assessment of disease risk and prognostic factors.
This study harnessed data from the Surveillance, Epidemiology, and End Results (SEER) database to perform a competing risk analysis on patients diagnosed with SCA. The primary aim was to juxtapose the outcomes of the competing risk model with those of the Cox proportional hazards model. This comparative analysis sought to pinpoint the factors that exert a more precise influence on the prognosis of elderly patients grappling with SCA.
Methods
Ethics Approval Statement
All patient data in the NCI SEER database is anonymized. Due to the retrospective nature of this study and the guaranteed privacy of patient information, the ethics committee granted a waiver for consent.
Data Collection and Patient Selection
Data pertaining to cases of sigmoid colon adenocarcinoma were amassed through the SEER database, utilizing the SEER*Stat 8.4.2 client, and drawing upon the Incidence - SEER Research Plus Data, 18 Registries, Nov 2020 Sub (2000-2018) data module. Inclusions criteria were as follows: (1) the primary tumor site was the sigmoid colon; (2) patients were aged 65 years or older at the time of diagnosis; (3) the diagnosis was grounded in a positively confirmed pathological prompt; (4) complete and effective follow-up data were available, with survival times exceeding 0 days; (5) the diagnoses were made between 2010 and 2015. Exclusion criteria encompassed: (1) cases reliant on autopsy or death reports for information; (2) instances with indeterminate survival times; (3) non-primary tumors. The study incorporated a range of indicators, including age, gender, race, marital status, AJCC stage, grade classification, surgical interventions, tumor size, liver metastasis, lung metastasis, bone metastasis, tumor laterality, and the utilization of radiotherapy. The analysis tracked patients for specific deaths attributed to cecum cancer, competing events, and deletions based on SEER’s etiology-specific death classifications, in conjunction with vital status record entries present in the SEER database. The application of these criteria led to the enrollment of 10,712 patients in this study.
Statistical Analyses
Numerical and percentage values were employed to elucidate baseline data. In our analysis of competing risks, we took into account deaths resulting from causes other than sigmoid adenocarcinoma as competing events.
The subdistribution hazard function, as conceived by Fine and Gray, delineates the instantaneous occurrence rate of a specific type of event in individuals who have not yet encountered that particular event. The Fine-Gray model is aptly tailored for the construction of clinical predictive models and prognosticating the risk associated with a solitary endpoint of interest. The mathematical formulation deployed for the Fine-Gray model was as follows
The subdistribution hazard for event k is defined as the probability of a subject experiencing event k in an infinitesimally small time interval ∆t, given that the subject has not experienced any events up to time t or has experienced events other than k prior to time t.
Fine-Gray models provide insight into the relative influence of covariates on the cumulative incidence function, leading to a nuanced understanding of event probability over time. 18 Fine-Gray models are key statistical tools designed for analyzing competitive risk data, where the cumulative incidence function (CIF) plays a central role in estimating the probability of an event occurring in the presence of competitive risk. The model is particularly useful in survival analysis, where it provides a more nuanced understanding of the probability of risk over time than traditional methods (eg, cox). 19 The use of COX modeling and Fine-Gray modeling allowed us to compare and contrast the impact of different variables on survival outcomes from two perspectives: one perspective focusing only on cancer-specific survival (Cox modeling), and the other considering a broader range of competing risks (Fine-Gray modeling). This dual approach enhances the robustness of our findings and gives us a more nuanced understanding of the prognosis of elderly patients with adenocarcinoma of the sigmoid colon.
The SEER database was used to extract the relevant data, but some incomplete data or instances of selection bias were found during data processing. To ensure the accuracy and reliability of the analysis, I chose to remove incomplete data. The data were subjected to analysis using R 4.2.3 software. For univariate analysis, the Fine-Gray competitive risk model was employed. Indicators exhibiting statistically significant differences in the univariate analysis were subsequently integrated into both the Fine-Gray multivariate analysis and the Cox multivariate analysis.
Result
Patient Characteristics
Baseline Characteristics of Patients.

Results of the Univariate Analysis
In the univariate analyses, Fine-Gray’s test and Cumulative Incidence Function (CIF) were applied. In the presence of competing risks, Gray’s test results indicated that several factors significantly influenced the prognosis of SCA (P < .05), including age,race, marital status, AJCC stage, grade of differentiation, surgical status, liver metastasis, lung metastasis, brain metastasis, radiotherapy status and chemotherapy status. Over time, CIF demonstrated an increase at 12, 36, and 72 months for nearly all variables.
Univariate Analysis of Prognostic Factors in Patients With SCA.

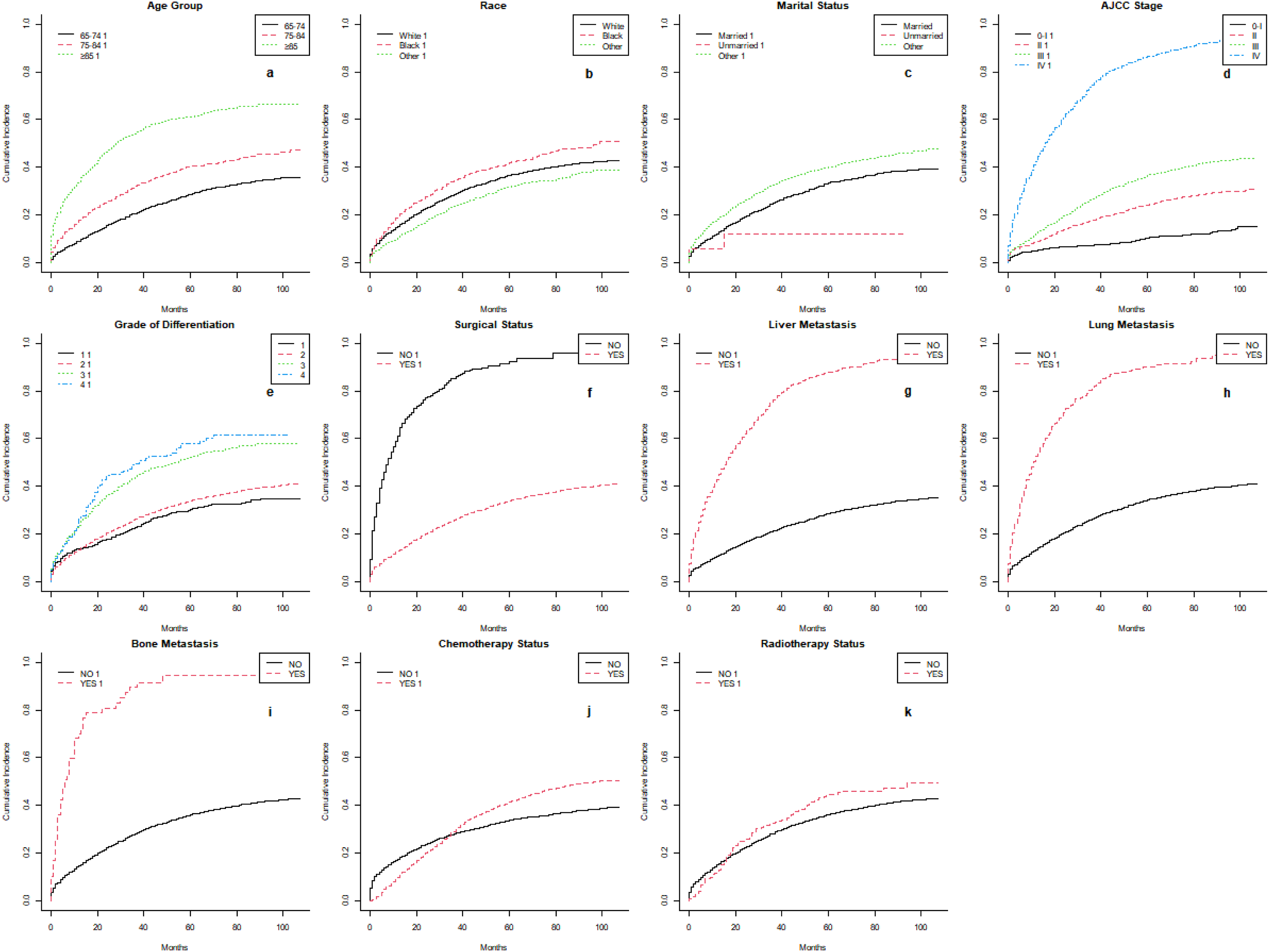
Using data from the Surveillance, Epidemiology, and End Results (SEER) database, cumulative incidence function (CIF) curves characterizing patients with adenocarcinoma of the sigmoid colon were plotted for the period 2010-2015. (a) Cumulative incidence function for age. (b) Cumulative incidence function for race. (c) Cumulative incidence function for marital status. (d) Cumulative incidence function for American Joint Committee on Cancer (AJCC) staging. (e) Cumulative incidence function for tumor grade. (f) Cumulative incidence function for surgical status. (g) Cumulative incidence function for liver metastasis status. (h) Cumulative incidence function for lung metastasis status. (i) Cumulative incidence function for bone metastasis status. (j) Cumulative incidence function for chemotherapy status. (k) Cumulative incidence function for radiotherapy status.
Results of the Multivariate Analysis
Multivariate Analysis of 2 Models of Prognostic Factors in Patients With SCA.

The effect on prognosis in the 75-84 age group compared with the ≥85 age group was significant in the Fine-Gray model (P < .0001), with a higher risk in the ≥85 age group (P < .0001,HR = 1.775, 95% CI 1.583 - 1.989). Patients from racial backgrounds other than white had a lower competitive risk compared to white patients (P = .0073, HR = .850, 95% CI: .755 - .957). Patients with marital status categorized as “other” had a relatively higher competitive risk compared to married patients (P = .0030, HR = 1.124, 95% CI: 1.041 - 1.215). AJCC stages II, III, and IV were all significant for prognosis compared with stage I in the Fine-Gray model (P < .0001), and the risk increased progressively with increasing disease stage. Grade 3 and 4 had a significant effect on prognosis in the Fine-Gray model compared to Grade 1 (P < .01), and Grade 2 was not significant compared to Grade 1. In the COX model, the relationship between AJCC stage, tumor grade and prognosis was not significant. Patients who underwent surgical treatment had a lower competing risk of death compared to those who did not (95% CI: .480 - .660). Tumors with a size between 1-3 mm exhibited a lower competing risk relative to those with a size of ≤1 mm, although this difference was statistically significant (HR = .001). Patients with hepatic metastasis had a higher competing risk of death compared to those without hepatic spread (P = .0005, HR = 1.308, 95% CI: 1.066 - 1.452). Patients with pulmonary metastasis also had a higher competing risk of death relative to those without pulmonary spread (P = .0056, HR = 1.244, 95% CI: 1.066 - 1.452). Additionally, chemotherapy, compared to no chemotherapy, significantly contributed to the improved prognosis of elderly patients diagnosed with SCA (HR = .747, 95% CI: .680-.819, P < .0001).
In a multifactorial COX regression analysis, factors affecting the prognosis of elderly SCA patients included sex, age, race, marital status, and surgical status (P < .05).
Discussion
In this study, both the Cox proportional hazards model and the Fine-Gray model were utilized. The Cox model revealed that gender was an independent risk factor for the prognosis of sigmoid colon cancer, with a risk ratio of 1.059 for males relative to females. This suggests that, all other factors being equal, males are at a 1.059-fold higher risk of developing sigmoid colon adenocarcinoma compared to females. Conversely, the Fine-Gray model demonstrated that sex was not an independent risk factor for prognosis of sigmoid colon adenocarcinoma (P = .1151). Previous research has shown that men tend to have a higher incidence rate than women in many regions, aligning with these findings.6,20 The global incidence of colon cancer in 2020 is estimated to be 14.7 cases per 100,000 people, and the incidence of sigmoid colon cancer is 5.9, suggesting that a higher incidence is observed in the male population. 21 Majek et al reported a survival advantage in female colorectal cancer (CRC) patients, which was particularly pronounced in younger and middle-aged patients, but was not observed in older subjects, suggesting that the effect of gender on survival may diminish with age. 22 Doria-Rose et al discuss the risk of inadequate sigmoidoscopy in older adults and women, which may indirectly affect survival by influencing the detection of sigmoid adenocarcinoma, although the primary focus of that study was on the adequacy of sigmoidoscopy rather than survival per se. 23 In conclusion, although some studies have suggested that gender may influence colorectal cancer (including sigmoid adenocarcinoma) survival, particularly in favor of women, the evidence is inconsistent, especially among older adults. The effect of gender on survival appears to diminish with age, and other factors such as disease stage, comorbidities, and treatment modalities may play a more important role in determining survival outcomes in this population. Wolbers et al. 24 proposed that the Fine-Gray model is better suited for the analysis of clinical prediction models. In the Fine-Gray model, the effects of other events (such as death from other causes) may have been taken into account, which may have somewhat weakened the effect of gender on the prognosis of sigmoid colon adenocarcinoma. In other words, gender may no longer be an independent risk factor for sigmoid colon adenocarcinoma prognosis once the effects of other events are taken into account. Therefore, when assessing survival data, it is crucial to consider the potential influence of competing risks and apply the appropriate statistical model to provide a more accurate estimation of disease risk.
Patients with adenocarcinoma of the sigmoid colon in the 75-84 and ≥85 age groups had a significantly increased risk of survival in the competing risks analysis (P < .0001). Whereas the risk tended to be significant for patients in the 75-84 age group compared with 65-74 years in the COX analysis (P = .0524), it was not significant for the ≥85 age group. These results are consistent with the study by Dalmat et al who found a higher risk of postoperative morbidity and mortality in the elderly population, with major problems including an increased incidence of pneumonia and thromboembolism. 25 This further supports the results observed in our competing risk analysis. In terms of racial differences, the results of the analysis of the two models showed that the Cox model showed a higher risk of death from adenocarcinoma of the sigmoid colon in patients of other races compared to Caucasians. In contrast, the competing risks model showed that patients from other racial had a lower risk of death than Caucasians. These variations may be attributed to differences in lifestyle, dietary habits, and genetics among various ethnic groups. 6 Arnold et al. 20 have previously shown that the incidence and mortality of colorectal cancer are linked to the adoption of a westernized lifestyle. The study also examined patients with different marital statuses, including married, unmarried, and other marital statuses. In Fine-Gray analysis, patients with marital status ‘other’ were at higher risk compared to married patients, while the difference was not significant for unmarried patients. The impact of marital status on survival outcomes in colorectal cancer patients has been established, with married patients having higher cause-specific survival (CSS) rates compared to patients with other marital statuses, including widowed patients. 26 This suggests that social support mechanisms may play a key role in the survival of colorectal cancer patients, including those with adenocarcinoma of the sigmoid colon, and that such mechanisms may be stronger in married individuals.
The Fine-Gray model revealed that Grade III (HR = 1.384, 95% CI = 1.152 - 1.664, P < .001) and Grade IV (HR = 1.517, 95% CI = 1.135-2.029, P < .001) stages were associated with a higher risk of death in patients with sigmoid colon adenocarcinoma compared to Grade I stages. In the COX model, the effect of tumor grade on prognosis was not significant. Previous studies have shown that Grade II is not a risk factor for death in patients with sigmoid colon adenocarcinoma.27,28 We believe that these findings may be the result of the interaction of multiple variables included in the model, and Yasmeen et al concluded that tumor border configuration (TBC) significantly affects overall and disease-free survival in stage II and III colon adenocarcinomas, so these relationships need to be further explored. 27
The study results suggested that tumor size had a differential impact on the competitive risk for patients with sigmoid colon cancer. In the Fine-Gray model, tumors sized ≤1 mm exhibited a significantly lower competitive risk. This could be attributed to the slower growth and reduced impact on patient survival associated with smaller tumors. However, the difference in risk for tumor sizes in the 1-3 mm and 3-5 mm groups did not reach statistical significance in the Cox model. It’s important to note that the analysis of basic population characteristics revealed an imbalance in the distribution of tumor sizes, with a substantial majority of patients having tumor sizes equal to or less than 1 mm, and a relatively small number of patients falling into the larger tumor size groups (1-3 mm and 3-5 mm). This distribution imbalance might have influenced the analysis results and might not fully represent the characteristics of different tumor size groups. Future studies should aim to include more samples to more accurately explore the relationship between tumor size and competitive risk.
Having liver metastases and having lung metastases compared with no metastases had a significant effect on prognosis in the Fine-Gray model (P < .01), whereas having bone metastases compared with no metastases was not significant. In contrast, Cox analysis showed that liver metastasis, lung metastasis, and bone metastasis did not have a significant effect on prognosis. Liver metastases have been identified as having the worst prognosis amongst major organ metastases in patients with stage IV lung cancer, suggesting a significant impact on survival, which is consistent with the findings of the Fine-Gray model, but contrasts with the results of Cox’s analysis.29,30 Similarly, lung metastases, particularly in patients with osteosarcoma, have been shown to significantly affect postmetastatic survival (PMS), and metastasectomy has the potential to improve prognosis. 30 This again supports the indication that Fine-Gray modeling suggests a significant impact on prognosis. On the other hand, bone metastases, although common and having an impact on quality of life, did not show a significant difference in progression-free survival in patients with intrahepatic cholangiocarcinoma compared to liver involvement only, which may explain their negligible impact in Cox analysis.31,32 However, the role of bone metastases in prognosis cannot be completely ignored, as their presence requires consideration of various therapeutic interventions, including radiation therapy specifically for painful bone metastases. 33 In addition, management of metastases, whether through radiotherapy, chemotherapy or surgical interventions such as metastasectomy or cytoreductive hepatectomy, plays a crucial role in influencing the prognosis of patients with liver, lung or bone metastases. 34 The development of a column-line graph for predicting overall survival in patients with bone metastases from lung cancer also highlights the complexity of prognostic factors including the site of metastasis. 35 In conclusion, although Fine-Gray modeling and Cox analysis provide different perspectives on the prognostic significance of liver, lung, and bone metastases, the combined data suggest that the impact of these metastases on survival is nuanced and influenced by a variety of patient characteristics, treatment choices, and disease-specific factors. 36
The effect of receiving chemotherapy on prognosis in elderly patients with adenocarcinoma of the sigmoid colon was significant in the Fine-Gray model (HR = .747, P < .0001), and the risk was lower in patients who received chemotherapy. In the COX analysis, the effect of receiving chemotherapy on prognosis was not significant. Regarding the impact of chemotherapy on the prognosis of elderly patients with adenocarcinoma of the sigmoid colon, differences between Fine-Gray models and Cox analyses highlight the complexity of assessing the impact of chemotherapy on survival outcomes. Fine-Gray models that take into account competing risks indicate a significant benefit of chemotherapy, suggesting that patients receiving chemotherapy have a lower risk of adverse outcomes. In contrast, Cox analyses, which may not adequately account for competing risks, did not find a significant effect of chemotherapy on prognosis. This discrepancy highlights the importance of selecting appropriate statistical models when assessing treatment effects in oncology studies. In the context of adenocarcinoma and the role of chemotherapy in older patients, several studies of different types of cancer provide insights. For example, in pancreatic ductal adenocarcinoma (PDAC), chemotherapy dose density was found to be a significant prognostic factor affecting survival, suggesting that receiving a higher proportion of the planned chemotherapy dose was associated with better outcomes. 37 Similarly, for primary pancreatic signet ring cell carcinoma, chemotherapy was identified as an independent prognostic factor for improving survival. 38 However, the impact of chemotherapy on older patients, particularly those with colorectal cancer, remains a nuanced issue. A study of colorectal cancer patients over 80 years of age found that chemotherapy improved overall survival and cancer-specific survival in stages III and IV, suggesting that there may be a benefit even in the very elderly. 39 However, the decision to administer chemotherapy to elderly patients must take into account a variety of factors, including the stage of the cancer, the patient’s overall health, and potential adverse effects. For example, the benefits of adjuvant chemotherapy appear to be less desirable in patients with stage II colon cancer aged 70 years or older, 40 and in sigmoid colon cancer, the presence of tumor deposits significantly affects survival outcomes, suggesting that personalized treatment decisions are critical. 41 In conclusion, although the Fine-Gray model suggests that chemotherapy is beneficial for older patients with sigmoid colon adenocarcinoma, the complexity of treatment outcomes, patient characteristics, and cancer stage must be carefully considered in clinical decision-making. Evidence from various studies suggests that chemotherapy can provide a survival benefit in some cases, but its use in the elderly population requires a nuanced approach to balance potential benefits and risks.42–44
However, all two models used in this study showed that age, race, marital status, and surgical status were independent factors influencing the prognosis of sigmoid colon adenocarcinoma in the elderly. Cumulative incidence and sigmoid colon adenocarcinoma-specific mortality were lower in older patients with early-stage tumors who received chemotherapy. However, the opposite was true for long-term cumulative incidence (Figure 1(j)), which may be related to hematologic toxicity of chemotherapeutic agents. Hematological toxicity is a significant side effect of chemotherapy in gastrointestinal cancers, including adenocarcinoma of the sigmoid colon, which can manifest as neutropenia, thrombocytopenia and anemia. 45 In addition, a study by Ngatali et al highlighted that hematological toxicity including anemia and neutropenia was prevalent in patients receiving chemotherapy, with a large number of patients experiencing grade 3 neutropenia. 46 This suggests the possibility of increased morbidity, which could lead to higher mortality. The study by Wang et al also identified chemotherapy cycle length, age, and body mass index as risk factors for fluoropyrimidine-induced cardiotoxicity, another serious side effect that may increase the overall risk of death in patients with colorectal cancer. 47 Hematological toxicity in chemotherapy may account for the increased long-term cumulative sigmoid colon adenocarcinoma-specific mortality in chemotherapy patients.
This study has some limitations. As a retrospective study, it may have suffered from selection bias and inaccuracy in recording information. The observational nature limits causal inferences and some key clinical variables may not have been fully captured. Over time, differences in data measures, diagnostic methods, and treatment regimens may affect consistency. Although multivariate models adjusted for confounders, unmeasured factors such as socioeconomic status and medical care were still not considered. In addition, varying lengths of follow-up may contribute to bias. External validation in different populations is necessary to confirm these findings.
Conclusion
In conclusion, this study was the first to develop a competing risk model using the SEER database for assessing prognostic risk factors in patients with sigmoid colon adenocarcinoma (SCA). Compared with the COX model, the competing risk model was able to estimate effect sizes more accurately. The findings suggest that age, race, marital status, AJCC staging, grade classification, surgical status, tumor size, liver metastasis, lung metastasis, and chemotherapy are independent prognostic factors for elderly patients with sigmoid colon adenocarcinoma. Although the retrospective analysis of the SEER database data has limitations, the results of this study provide clinicians with useful clinical information to improve clinical decision making.
Footnotes
Author Contributions
(I) Conception and design: Ruofei Du
(II) Collection and assembly of data: Ruofei Du, Jiayu Guo
(III) Data analysis and interpretation: Ruofei Du,Jiayu Guo, Jing Li, Jun Lyu, and Li Lyu
(IV) Manuscript writing: Ruofei Du,Jiayu Guo, Jing Li, Jun Lyu, and Li Lyu
(V) Final approval of manuscript: Ruofei Du,Jiayu Guo, Jing Li, Jun Lyu, and Li Lyu
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the This work was supported by Guangdong Provincial Key Laboratory of Traditional Chinese Medicine Informatization (2021B1212040007). The funding agencies had no role in study design, collection and analyses of data, decision to publish, or manuscript preparation.

