Abstract
Introduction
Nanoliposomal irinotecan (nal-IRI) plus 5-fluorouracil and leucovorin (5-FU/LV) is an established second-line therapy for metastatic pancreatic ductal adenocarcinoma (PDAC). We previously developed a prognostic model (CGMH nomogram) to predict overall survival (OS) in patients receiving second-line chemotherapy before the nal-IRI + 5-FU/LV era. Herein, we aimed to validate the CGMH nomogram in a real-world cohort treated with nal-IRI plus 5-FU/LV, the current standard second-line treatment for metastatic PDAC.
Methods
A retrospective cohort of 148 patients with metastatic PDAC treated with second-line nal-IRI + 5-FU/LV was analyzed. Prognostic scores were assigned using the CGMH nomogram, with patients stratified into tertiles as good, intermediate, and poor prognostic groups. Predictive performance was assessed using the concordance index (c-index) and calibration plots.
Results
Our cohort had a median OS of 6.1 months. Patients in the good, intermediate, and poor prognostic groups had median OS of 8.7 (95% confidence interval [CI], 6.7-10.7), 5.7 (95% CI, 5.3-6.3), and 4.0 (95% CI, 2.8-5.2) months, respectively. Compared with the good group, intermediate and poor groups had hazard ratios of 1.99 (95% CI, 1.29-3.07, P = .002) and 3.18 (95% CI, 1.87-5.40, P < .001), respectively. The nomogram demonstrated strong predictive ability, with c-indices of 0.73 and 0.70 for 6- and 12-month OS predictions, respectively. Calibration plots displayed excellent agreement between predicted and observed survival.
Conclusion
The CGMH nomogram reliably predicted survival outcomes in nal-IRI + 5-FU/LV-treated patients with metastatic PDAC, and validation supported its use in clinical decision-making and personalized treatment planning.
Plain Language Summary
Why was the study done? For patients with metastatic pancreatic cancer, the combination of nal-IRI (nanoliposomal irinotecan) with 5-fluorouracil and leucovorin (5-FU/LV) is the standard second-line treatment. Predicting how long patients might survive with this treatment can help doctors personalize care. A tool called the CGMH nomogram was developed earlier to predict survival before this treatment became standard. This study tested whether the CGMH nomogram works well for patients receiving nal-IRI + 5-FU/LV. What did the researchers do? The researchers studied the medical records of 148 patients with metastatic pancreatic cancer treated with nal-IRI + 5-FU/LV. They used the CGMH nomogram to predict patients’ survival and divided them into three groups based on their predicted outcomes: good, intermediate, and poor prognosis. They then checked how accurate these predictions were by comparing them to the actual survival times. What did the researchers find? The average survival for all patients was about 6.1 months. Patients in the good prognosis group lived longer (8.7 months) than those in the intermediate (5.7 months) or poor (4.0 months) groups. The predictions made by the CGMH nomogram were very accurate, with strong agreement between predicted and actual survival outcomes. Statistical analyses confirmed that the nomogram is reliable for making predictions. What do the findings mean? The CGMH nomogram is a helpful tool for predicting survival in patients with advanced pancreatic cancer treated with nal-IRI + 5-FU/LV. It can guide doctors in making personalized treatment plans and support better decision-making for patients based on their expected outcomes.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the most aggressive types of cancer and was ranked as the seventh leading cause of cancer-related deaths worldwide in 2022. 1 Palliative chemotherapy is the standard treatment for patients with metastatic disease, eliciting a median overall survival (OS) of 8.5 to 11.1 months during first-line treatment.2-4 Despite the modest efficacy of first-line therapy, approximately 60% of patients undergo second-line treatment owing to disease progression.5,6
The NAPOLI-1 trial was a crucial milestone in the management of metastatic PDAC in 2016. 7 This pivotal study demonstrated that combining nanoliposomal irinotecan (nal-IRI) with 5-fluorouracil and leucovorin (5-FU/LV) could substantially improve the survival outcomes of patients who progressed to gemcitabine-based therapy. Specifically, nal-IRI + 5-FU/LV extended the median OS to 6.1 months compared with 4.2 months with 5-FU/LV alone (P = 0.012). Based on these results, nal-IRI plus 5-FU/LV was approved and included for reimbursement by the Taiwan National Health Insurance Program in November 2018, providing a standardized second-line treatment option for metastatic PDAC. 8
Prior to reimbursement of nal-IRI in Taiwan, the second-line management of metastatic PDAC was inconsistent and lacked a unified standard of care.5,6 Treatment strategies were highly heterogeneous and driven by local practices and expert opinions. Patients who progressed to gemcitabine-based therapy predominantly transitioned to 5-FU-based regimens, whereas others received additional cytotoxic agents incorporated into the gemcitabine-based regimens during second-line therapy. 6
In a previous study, we developed a prognostic nomogram to predict survival outcomes in patients with metastatic PDAC undergoing second-line palliative chemotherapy, using data from 183 patients treated at Chang Gung Memorial Hospital (CGMH). 9 The CGMH nomogram was constructed using eight independent factors: sex, duration of first-line treatment, reason for discontinuation of first-line therapy, Eastern Cooperative Oncology Group (ECOG) performance status, metastatic status, neutrophil-to-lymphocyte ratio (NLR), carbohydrate antigen (CA) 19-9 level, and body mass index (BMI) (supplemental Figure 1). Each patient was assigned a score ranging from 0 to 500 based on the nomogram. The patients were subsequently divided into tertiles classified as good, intermediate, and poor prognostic groups. The median OS rates of these groups were 11.0, 4.6, and 2.2 months, respectively.
Although the CGMH nomogram has undergone external validation for predicting survival outcomes, it is yet to be validated for use in patients treated with nal-IRI + 5-FU/LV, which is the current standard second-line therapy for metastatic PDAC that progressed despite treatment with a gemcitabine-based regimen. In the current study, we aimed to assess the applicability and predictive accuracy of the CGMH nomogram in a real-world cohort of patients with metastatic PDAC receiving nal-IRI plus 5-FU/LV as second-line treatment.
Patients and Methods
This retrospective study included 148 consecutive patients who received nal-IRI plus 5-FU/LV as second-line palliative chemotherapy for metastatic PDAC between November 2018 and December 2019 at two affiliated branches of the Chang Gung Memorial Hospital. In this study, we applied eligibility criteria consistent with those used to derive the CGMH nomogram. 9 Specifically, patients were required to have a histologically or radiographically confirmed diagnosis of locally advanced or metastatic PDAC. All patients received nal-IRI + 5-FU/LV according to the NAPOLI-1 protocol 7 for second-line treatment of metastatic PDAC. Exclusion criteria were as follows: patients who had not received gemcitabine-based therapy as first-line treatment; had participated in clinical trials; had insufficient follow-up data; had received concurrent radiotherapy during second-line treatment; or had incomplete medical records. Additionally, patients with recurrent tumors within 6 months of completing adjuvant chemotherapy were classified as having undergone unsuccessful first-line therapy and were included in the analysis.
Demographic and clinicopathological variables, as well as chemotherapy regimens for PDAC, were retrospectively collected through a chart review. Primary care physicians selected first-line chemotherapeutic regimens and treatment schedules. OS was measured from the start of second-line chemotherapy to the date of death from any cause. Patients were monitored until death or until the data cutoff date (December 31, 2020). The study was approved by the Institutional Review Board (IRB) of Chang Gung Memorial Hospital (IRB Approval Number: 202100783B0), located in Taiwan. The approval date was May, 31, 2021. Since this study was a retrospective analysis of anonymized data, the requirement for obtaining informed consent was waived by the IRB. The reporting of this study conforms to the TRIPOD guidelines for transparent reporting of a multivariable prediction model. 10
Statistical Analysis
Baseline demographic data are presented as frequencies and percentages for categorical variables and as medians with interquartile ranges (IQRs) for continuous variables. Descriptive statistics were used to summarize and compare variable distributions between our cohort and the original CGMH cohort. Continuous and ordinal variables were analyzed using the Kruskal–Wallis test, while categorical variables were compared using the chi-square or Fisher’s exact test when the cell count was <5.
The endpoint of the CGMH model is survival time, 9 which is consistent with the endpoint used in this study. All clinicopathological variables identified in the original CGMH nomogram study 9 were assessed using univariate Cox regression analysis to determine their impact on OS. All variables in the univariate analysis with P-values <0.10 were further analyzed using multivariate analysis. Prognostic scores for each patient were calculated based on the CGMH nomogram by assigning points corresponding to the eight independent factors, aligning each variable’s value with the “points” scale. The total score for each patient was determined by summing the individual points and mapping the total to the “total points” scale, which provided predictions for OS. Subsequently, patients were stratified into tertiles based on the CGMH nomogram-predicted risk groups, and survival outcomes were compared using Kaplan-Meier curves. Differences between the survival curves were analyzed using the log-rank test.
The CGMH nomogram was validated using the concordance index (c-index) and calibration plots. The c-index, a generalization of the area under the receiver operating characteristic curve, was used to assess the discriminative capability of the nomogram. Calibration plots were generated to compare the observed survival probabilities with those predicted by the nomograms. All statistical analyses were performed using SPSS (version 17.0, SPSS Inc., Chicago, IL, USA) and R version 2.9.1 (The R Foundation for Statistical Computing, Vanderbilt University, TN, USA) with the Hmisc and Design libraries. A P-value of <0.05 was considered statistically significant for all tests.
Results
Patient Demographics and Clinical Characteristics
Comparisons of Patient Characteristics Between the Current Cohort and the Original CGMH Model Cohort.
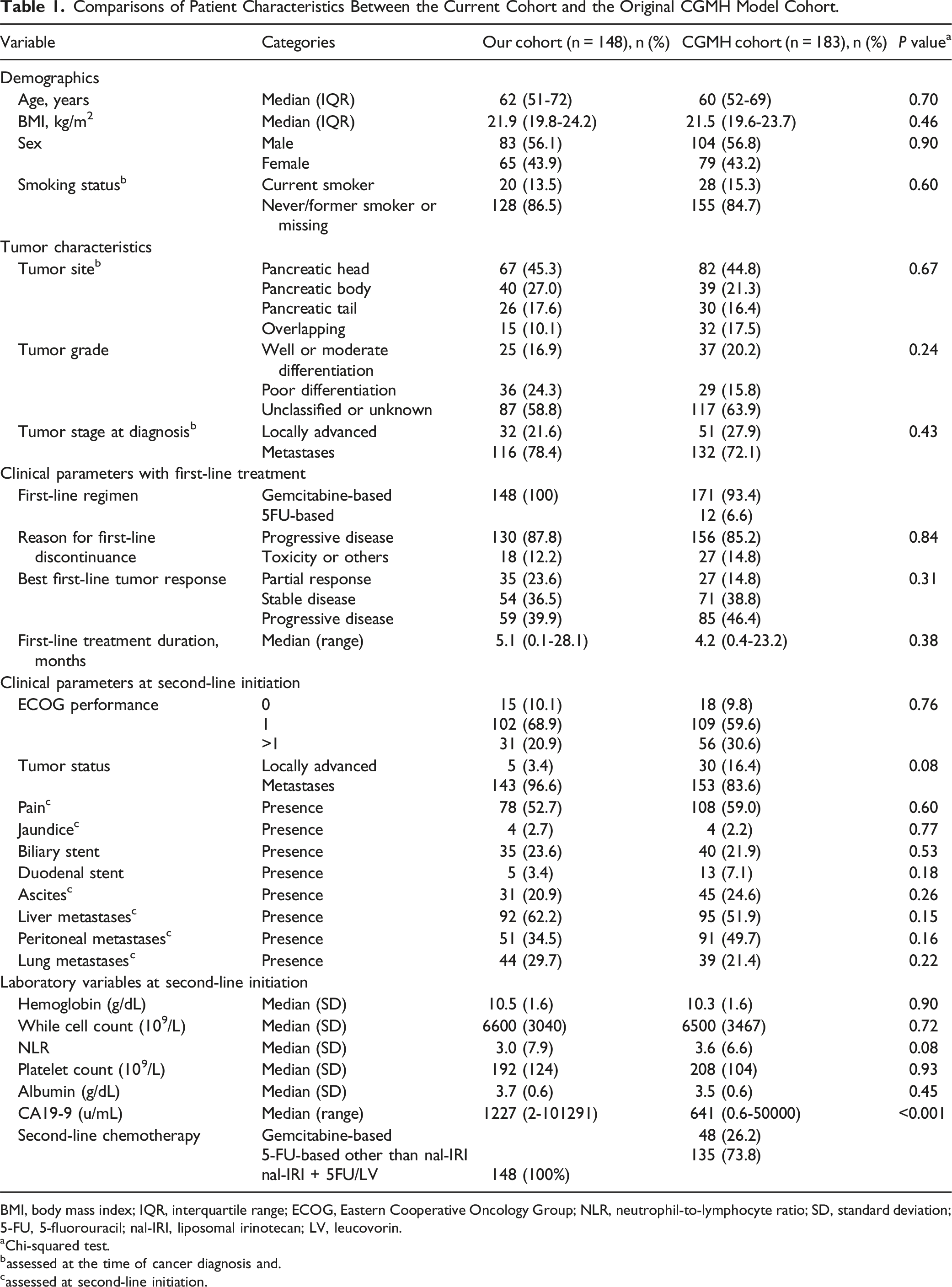
BMI, body mass index; IQR, interquartile range; ECOG, Eastern Cooperative Oncology Group; NLR, neutrophil-to-lymphocyte ratio; SD, standard deviation; 5-FU, 5-fluorouracil; nal-IRI, liposomal irinotecan; LV, leucovorin.
aChi-squared test.
bassessed at the time of cancer diagnosis and.
cassessed at second-line initiation.
In both groups, the reason for discontinuing first-line treatment was predominantly progressive disease (87.8% vs 85.2%, P = .84). The median duration of the first-line treatment was slightly longer in our cohort than in the CGMH cohort (5.1 vs 4.2 months, P = .38). At the start of second-line treatment, our cohort had a lower prevalence of ECOG performance status >1 (20.9% vs 30.6%) and locally advanced disease (3.4% vs 16.4%). Key laboratory values, such as hemoglobin, white blood cell count, and albumin levels, were comparable between the groups. However, the median CA19-9 level was significantly higher in our cohort (1227 vs 641 U/mL, P < .001).
Univariate and Multivariate Analysis of OS
Univariate and Multivariate Analyses of Overall Survival in Our Patient Cohort.

BMI, body mass index; CI, confidence interval; HR, hazard ratio; ECOG, Eastern Cooperative Oncology Group.
In multivariate analysis for OS, ECOG performance status, presence of pain, lung metastases, and NLR remained the independent prognostic factors in our cohort.
OS Curves Categorized by CGMH Nomogram
Patients were stratified into good, intermediate, and poor prognostic groups based on CGMH nomogram predictions (Figure 1). The median OS times for these groups were 8.7 months (95% CI, 6.7-10.7), 5.7 months (95% CI, 5.3-6.3), and 4.0 months (95% CI, 2.8-5.2), respectively. Compared with the good prognostic group, the intermediate prognostic group had an HR for OS of 1.99 (95% CI, 1.29-3.07, P = .002), while that of the poor prognostic group was 3.18 (95% CI, 1.87-5.40, P < .001). Kaplan‐Meier Overall Survival Curve of Patients With Metastatic Pancreatic Ductal Adenocarcinoma Stratified by Tertiles Using the CGMH Nomogram. CI, Confidence Interval; CGMH, Chang Gung Memorial Hospital; HR, Hazard Ratio.
Validation and Performance of the CGMH Nomogram
The c-indices for the CGMH nomogram in predicting 6- and 12-month survival probabilities were 0.73 (95% CI, 0.67-0.78) and 0.70 (95% CI, 0.62-0.79), respectively (supplemental Figure 2). Figure 2 presents the calibration plot for the CGMH nomogram, with the observed 6-month (green line) and 12-month (red line) survival probabilities plotted against the predicted probabilities. The green line aligned closely with the diagonal, reflecting good calibration and strong predictive accuracy at 6 months. Conversely, the red line deviated significantly from the diagonal, indicating a discrepancy between the nomogram predictions and the actual observed survival probabilities at 12 months in this cohort. This deviation suggests a reduced calibration accuracy for longer-term survival predictions. Calibration Plot of the Observed 6-Month (Green) and 12-Month (Red) Survival Probabilities Against the Predicted Survival Probability of the CGMH Nomogram. The Dotted Line Represents the Ideal Line where the Actual Probability of Survival Matches the Predicted Probability. CGMH, Chang Gung Memorial Hospital.
Association of CGMH Prognostic Group and Tumor Response
The tumor response rates to nal-IRI plus 5-FU/LV treatment were as follows: partial response, 7.6%; stable disease, 26.0%; and progressive disease, 66.4%. Patients in the good prognostic group had a higher rate of partial response (16.9%) and stable disease (25.4%) than those in the intermediate (6.3% and 12.7%, respectively) and poor prognostic groups (3.8% and 23.1%, respectively) (Figure 3). Differences in tumor response rates among the three prognostic groups were statistically significant (P = .045). Best Tumor Responses to Nal-IRI + 5-FU/LV in Patients From Different CGMH Prognostic Groups. CGMH, Chang Gung Memorial Hospital; 5-FU, 5-Fluorouracil; LV, Leucovorin; Nal-IRI, Nanoliposomal Irinotecan.
Discussion
The CGMH nomogram was originally developed to predict the OS of patients with metastatic PDAC undergoing second-line chemotherapy before initiating the nal-IRI + 5-FU/LV regimen. 9 In our cohort of 148 patients treated with nal-IRI + 5-FU/LV as the second-line therapy for metastatic PDAC, the median OS was 6.1 months, consistent with the findings of the NAPOLI-1 trial, 7 thereby demonstrating the effectiveness of nal-IRI in a real-world clinical setting. Our study also highlights the utility of the CGMH nomogram in stratifying patients according to expected survival outcomes, supporting its applicability in clinical practice for guiding treatment decisions and patient counseling.
The original CGMH nomogram development cohort analyzed patients with metastatic PDAC who had received various second-line treatment regimens between 2010 and 2016, yielding a median OS of 4.2 months. 9 In contrast, our current cohort included patients treated with a standardized nal-IRI + 5-FU/LV regimen between 2018 and 2019, which elicited a higher median OS of 6.1 months. This improvement in survival aligned with advancements in treatment protocols, as demonstrated by the NAPOLI-1 trial, 7 which established nal-IRI + 5-FU/LV as a superior option.
Patients treated with nal-IRI + 5-FU/LV in the NAPOLI-1 trial achieved a median OS of 6.1 months 7 and an objective response rate of 16%. In the subgroup analysis of Asian patients in NAPOLI-1, the median OS was 8.9 months, with objective response and disease control rates of 8.4% and 36.9%, respectively. 11 In real-world settings, the median OS of patients with metastatic PDAC who received nal-IRI ranged between 4.2 and 9.4 months.12-18 In our study, the median OS was 6.1 months, with an objective response rate of 7.6% and a disease control rate of 33.6%, which are comparable to the outcomes reported in the NAPOLI-1 trial and real-world practice, supporting the efficacy of nal-IRI to treat metastatic PDAC.
The CGMH nomogram incorporates eight prognostic factors, 9 but only four factors —ECOG performance status, albumin levels, CA19-9 levels, and metastatic sites—were associated with significant differences in OS in our cohort. This discrepancy may be due to differences in baseline characteristics between the cohorts. Notably, the median CA19-9 levels are significantly higher in our cohort (1227 U/mL) compared to the CGMH cohort (641 U/mL, P < .001). Since CA19-9 is a tumor marker associated with pancreatic cancer burden, this difference suggests that our cohort may have had a more aggressive disease at baseline, potentially contributing to poorer survival outcomes. Additionally, while the proportion of patients with an ECOG performance status >1 is lower in our cohort (20.9%) than in the CGMH cohort (30.6%), the difference is not statistically significant (P = .08). ECOG status is a key factor in assessing patient functionality and prognosis, and a lower proportion of patients with a poor performance status in our cohort may slightly mitigate the impact of the higher CA19-9 levels on survival outcomes. Furthermore, differences in protocols for second-line therapy may have influenced treatment outcomes. These findings underscore the need to refine nomograms to accommodate the evolving therapeutic landscape.
Comparison of Different Nomograms or Prognostic Models in Predicting Overall Survival in Patients With Metastatic Pancreatic Adenocarcinoma Undergoing Second-Line Chemotherapy.

CGMH, Chang Gung Memorial Hospital; 5-FU, 5-fluorouracil; LV, leucovorin; nal-IRI, nanoliposomal irinotecan; NLR, neutrophil-to-lymphocyte ratio; CA19-9, cancer antigen 19-9; BMI, body mass index; LDH, lactate dehydrogenase.
Our previous study retrospectively analyzed 645 patients with newly diagnosed metastatic PDAC in Taiwan who received palliative chemotherapy between 2010 and 2016, highlighting the evolution of treatment patterns and their impact on OS. 6 In contrast, the current study focuses on a contemporary cohort of 148 patients treated with nal-IRI plus 5-FU/LV as second-line therapy following gemcitabine-based treatment. While the earlier study demonstrated an improvement in OS over time with the increased adoption of multi-line chemotherapy strategies, 6 our findings further refine prognostic stratification by validating the CGMH nomogram in the era of nal-IRI treatment. Additionally, whereas the earlier study documented the increased utilization of S-1 following its reimbursement, 6 our study illustrates the transition to a standardized second-line treatment with nal-IRI + 5-FU/LV. This shift in therapeutic strategies underscores the evolving treatment landscape for metastatic PDAC and the necessity of incorporating validated prognostic models, such as the CGMH nomogram, to personalize patient care and optimize survival outcomes.
Our analysis indicated that the presence of pain and jaundice were markedly associated with reduced OS, consistent with the existing literature on their prognostic value.19,22,23 Pain in PDAC often results from nerve involvement, reflecting advanced tumor burden and poor prognosis. 24 Similarly, jaundice typically signifies biliary obstruction due to tumor invasion or compression, indicating extensive disease involvement. 18 Both conditions severely affect the patient’s quality of life, and their management can be challenging.24,25 Recognizing their impact highlights the importance of prioritizing symptom control. Effective management strategies, such as nerve blocks for pain and biliary stenting for jaundice, can enhance the patient’s quality of life, even if they do not substantially extend survival.
Our current validation was performed within a single institutional dataset, and although the results are promising, external validation using larger, multi-center cohorts is necessary to confirm the model’s reliability across different clinical settings. Furthermore, the reduced calibration accuracy for 12-month survival probabilities in our cohort suggests that survival predictions may become less precise over extended time frames owing to variations in disease biology, therapeutic responses, and progression patterns that are not fully captured by the prognostic factors in the CGMH model. Additionally, differences in patient responses to second-line or subsequent treatments could contribute to greater variability in survival outcomes at the 12-month follow-up. Future studies should focus on applying this nomogram to independent datasets, assessing its predictive accuracy across varying demographic, clinical, and molecular profiles to further strengthen its clinical utility in guiding treatment decisions for patients with metastatic pancreatic cancer.
Conclusion
The CGMH nomogram provides a valuable tool for predicting survival outcomes in patients with metastatic PDAC undergoing second-line treatment with nal-IRI + 5-FU/LV. Our validation study demonstrated the effectiveness of the CGMH nomogram in accurately stratifying patients into prognostic groups, thereby guiding treatment decisions and optimizing patient care. Although second-line chemotherapy remains the standard treatment option, tailoring care based on prognostic assessments can improve outcomes and enhance the quality of life in patients with advanced pancreatic cancer.
Supplemental Material
Supplemental Material - Validation of a Prognostic Nomogram for Patients with Metastatic Pancreatic Cancer Treated with Nanoliposomal Irinotecan as Second-Line Therapy
Supplemental Material for Validation of a Prognostic Nomogram for Patients with Metastatic Pancreatic Cancer Treated with Nanoliposomal Irinotecan as Second-Line Therapy by Yu-Shin Hung, MD, Tai-Jan Chiu, MD, PhD, Yen-Yang Chen, MD, and Wen-Chi Chou, MD, PhD in Cancer Control
Supplemental Material
Supplemental Material - Validation of a Prognostic Nomogram for Patients with Metastatic Pancreatic Cancer Treated with Nanoliposomal Irinotecan as Second-Line Therapy
Supplemental Material for Validation of a Prognostic Nomogram for Patients with Metastatic Pancreatic Cancer Treated with Nanoliposomal Irinotecan as Second-Line Therapy by Yu-Shin Hung, MD, Tai-Jan Chiu, MD, PhD, Yen-Yang Chen, MD, and Wen-Chi Chou, MD, PhD in Cancer Control
Footnotes
Acknowledgments
The authors gratefully acknowledge the assistance of the patients who participated in this study.
Statements and Declarations
Author Contributions
Conception and design of study: YSH, TJC, YYC, WCC; Acquisition of data: YSH, TJC, YYC, WCC; Analysis and interpretation of data: YSH, TJC, YYC, WCC; Drafting of the manuscript: YSH, TJC, YYC, WCC.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
