Abstract
Pancreatic cancer has traditionally been associated with a dismal prognosis, even in early stages of the disease. In recent years, the introduction of newer generation chemotherapy regimens in the adjuvant setting has improved the survival of patients treated with upfront resection. However, there are multiple theoretical advantages to deliver early systemic therapy in patients with localized pancreatic cancer. So far, the evidence supports the use of neoadjuvant therapy for patients with borderline resectable pancreatic cancer. The benefit of this treatment sequence for patients with resectable disease remains elusive. In this review, we summarize the data on adjuvant therapy for pancreatic cancer and describe which evidence backs the use of neoadjuvant therapy. Additionally, we address important issues faced in clinical practice when treating patients with localized pancreatic cancer.
Introduction
Pancreatic cancer currently represents the seventh most common cause of cancer-related death worldwide. 1 This disease is associated with a dismal prognosis, with only 12% of patients alive 5 years after the diagnosis, and these figures have minimally changed over the past 2 decades. 2 Importantly, recent studies foresee a significant rise in the incidence and mortality of pancreatic cancer in the next decade both in developed and developing countries, emphasizing the growing epidemiological burden of the disease.3-5
Pancreatic ductal adenocarcinomas (PDACs) represent 90% of all pancreatic malignancies. 6 Traditionally, 15–20% of patients with PDAC present disease amenable to upfront resection, 7 and for these patients, surgery remains the cornerstone of curative-intent treatment. 8 By the 1980s, developments in patient selection, surgical techniques, and perioperative management had enabled pancreatoduodenectomy to be performed with less than 5% mortality rates. 7 However, despite these betterments in surgical outcomes, overall survival for patients with resected PDAC remained poor, with less than 20% of patients alive 5 years after surgery. 9 Therefore, it became clear that further improvements in survival should be attained with the use of chemotherapy and radiotherapy. With that goal in mind, many groups designed and carried out studies evaluating the role of adjuvant and neoadjuvant therapies in resectable PDAC.
Adjuvant Therapy
Adjuvant Chemotherapy
Studies Evaluating the Role of Adjuvant Chemotherapy (vs Surveillance) After Pancreatectomy.

GEM, gemcitabine; 5-FU, 5-fluorouracil; LV, leucovorin; DOXO, doxorubicin; MMC, mitomycin; CDDP, cisplatin.
aTime to recurrence.
Studies Comparing Multiagent Regimens to Gemcitabine in the Adjuvant Setting.

GEM, gemcitabine; CAP, capecitabine; NAB, nab-paclitaxel.
cRelapse-free survival.
dInvestigator-assessed disease-free survival.
aAfter R0 resection.
bAfter R1 resection.
While recent data have ratified the role of gemcitabine plus capecitabine in the adjuvant setting,30,31 its use in clinical practice faces some challenges. Real-world data from Western countries suggest this regimen to be relatively toxic. 32 Indeed, it has the lower overall treatment completion rate among all multi-agent regimens used in the adjuvant setting. Also, in some countries like Brazil, there is no reimbursement for capecitabine in the treatment of PDAC. Furthermore, the vast majority of the patients included in the ESPAC-4 had stage III disease with positive surgical margins, only one-third of the patients underwent salvage therapy at relapse, 33 and the study failed to show improvements in relapse-free survival (a secondary outcome).
In the case of gemcitabine plus nab-paclitaxel, its use is not currently endorsed by the guidelines due to the fact that the APACT trial failed to meet the primary end-point of independently reviewed disease-free survival.34,35 However, the limitations of this outcome have been recently highlighted,24,36 and the study met the secondary outcomes of overall survival and investigator-assessed disease-free survival. 23 Also, as seen with gemcitabine plus capecitabine, retrospective data also suggest that the combination of gemcitabine plus nab-paclitaxel is associated with improved outcomes in the adjuvant setting. 37 Moreover, this regimen is frequently used in the neoadjuvant setting, with efficacy comparable to that of FOLFIRINOX. 38 For that, the authors believe that gemcitabine plus nab-paclitaxel could be an alternative to gemcitabine plus capecitabine in patients not eligible to modified FOLFIRINOX. However, one should point out that patients in the adjuvant polychemotherapy trials, especially in PRODIGE 24, were highly selected by biological (absence of disease progression until adjuvant treatment initiation and complete macroscopic resection) and clinical (relatively younger age, good performance status, and limited comorbidities) factors when compared to the general population of patients with PDAC, potentially explaining these survival figures. Therefore, while real-life data support the efficacy of adjuvant polychemotherapy, especially of FOLFIRINOX in this setting, 39 many patients in real world will not be candidates to adjuvant polychemotherapy. For such patients, single-agent gemcitabine or 5-FU/LV are still reasonable options.
It is important to highlight that overall treatment completion seems to be an important prognostic factor. Landmark analyses from the ESPAC-3 and PRODIGE 24 trials have demonstrated that patients who were able to complete the 24 weeks of adjuvant treatment fared much better that those who discontinued treatment.40,41 Moreover, in the PRODIGE 24, a dose intensity higher than 80% for all drugs was not associated with overall survival. 41 Currently, it is not known how much the doses of the individual drugs in the polychemotherapy regimens can be reduced before the efficacy of adjuvant polychemotherapy is significantly compromised. That said, we believe that adhering to the 24-week treatment duration should be the primary goal, even if dose reductions are deemed necessary. One aspect that seems to be overrated in clinical practice is time to initiate adjuvant chemotherapy. Although some data suggest that the early initiation of adjuvant chemotherapy might be associated with longer survival,42,43 most data support that there is no to minimum difference in outcomes based on the time to initiate chemotherapy. 44 Data from the ESPAC-3 trial have not shown improved survival for patients starting adjuvant chemotherapy within 8 weeks of surgery (vs 8–12 weeks). 40 Additionally, multiple analyses suggest that the benefit of adjuvant chemotherapy stands even for patients who start chemotherapy after 12 weeks.45-47 Therefore, given the impact of adjuvant chemotherapy in survival, for those who develop significant postoperative complications but have a full recovery, delayed adjuvant chemotherapy should still be considered. However, for patients who are fit after surgery, adjuvant chemotherapy should be administered as soon as possible, based on the concept of higher chances of cure with early treatment of micrometastases. 48
Currently, the only molecular biomarker used to tailor decisions regarding adjuvant chemotherapy for PDAC is the presence of a pathogenic germline mutation in core genes of the homologous recombination repair mechanism, namely, BRCA-1/2 and PALB2. Patients with these alterations frequently have tumors that are more sensitive to platinum-based chemotherapy,49-51 with some evidence suggesting improved disease-free and even overall survival when such regimens are employed in the adjuvant setting.52,53 However, it should be said that very often this information is not available by the time decisions regarding adjuvant chemotherapy must be taken. Other biomarkers have been evaluated, such as TP53 mutations for gemcitabine-based chemotherapy 54 and GATA6 expression for 5-fluorouracil-based chemotherapy. 55 However, so far these markers have not been validated and their use is not recommended in clinical practice.
Adjuvant Radiotherapy
While the role of adjuvant chemotherapy in resected PDAC remains undisputed, the benefit of post-operative radiotherapy is still a matter of debate. So far, only 1 study has shown that adjuvant chemoradiation was associated with improved overall survival. In the GITSG 9173, 43 patients with resected PDAC were randomized to undergo radiotherapy (split course, 2 20-Gy segments 2 weeks apart) concomitant to 5-FU (days 1–3 of the radiotherapy segments) followed by weekly 5-FU for up to 2 years or surveillance. 56 The study showed that adjuvant chemoradiation was associated with improvements in median overall survival (21.0 vs 10.9 months). However, the small sample size, the suboptimal radiation technique, and the fact that patients in the radiotherapy arm received adjuvant chemotherapy for up to 2 years cast doubt on the true benefit of adjuvant radiotherapy for patients with resected PDAC.
Studies Evaluating the Role of Adjuvant Radiotherapy for Resected PDAC.

GEM, gemcitabine; 5-FU, 5-fluorouracil; LV, leucovorin; CDDP, cisplatin; INF, interferon alpha-2b; CI, continuous infusion.
aTime to recurrence.
bRelapse-free survival.
Patients with positive surgical margins are at a higher risk for local recurrence after surgery.62,63 In this scenario, retrospective data suggest that adjuvant radiotherapy can be associated with improved local control. 64 Indeed, data from individual patient meta-analyses of randomized trials suggest that chemoradiation might be associated with improved survival in the subgroup of patients with positive surgical margins.65,66 On the other hand, retrospective multi-institutional series and population-based studies conducted in the US suggest benefit of adjuvant chemoradiation for patients with positive lymph nodes67,68 and even for patients without high-risk features. 69 Therefore, despite the overall negative results observed in randomized trials, the use of adjuvant radiotherapy in the settings of positive surgical margins or (to a lesser extent) positive lymph nodes is supported by low-quality evidence.
These controversies are highlighted in the contrasting recommendations from different guidelines. According to the NCCN guidelines, adjuvant chemoradiation is an option for patients with features that portend high risk for local recurrence, such as positive surgical margins. 34 The ASTRO guidelines also suggest that chemoradiotherapy can be conditionally recommended to patients with high-risk clinical features such as positive surgical margins or positive lymph nodes. 70 On the other hand, the ESMO guidelines recommend that patients should not receive adjuvant radiotherapy outside the setting of randomized clinical trials. 71 To settle this controversy, the results of the phase III part of the RTOG 0848 trial, in which patients were treated with state-of-the-art radiation techniques, are eagerly waited, and it is hoped that they will finally establish the role of adjuvant radiotherapy in resected PDAC.
To date, no molecular marker of improved efficacy of radiotherapy in pancreatic cancer has been established. Some,72,73 but not all,74,75 studies have found an association between intact SMAD4 expression and locally aggressive PDAC, while mutant SMAD4/SMAD4 loss has been associated with resistance to radiotherapy mediated by autophagy. 76 That would possibly help selecting patients with a higher chance of benefiting from adjuvant chemoradiation. Indeed, limited evidence suggests that radiotherapy might improve locoregional control and survival outcomes for patients with preserved SMAD4 expression.77,78 However, given the controversial role of SMAD4 in locoregional recurrence and the retrospective nature of these studies, the use of SMAD4 status to guide treatment decisions regarding the use of adjuvant radiotherapy is not currently recommended.
Neoadjuvant Therapy
Rationale
Potential Advantages of Neoadjuvant Therapy in Potentially Resectable PDAC. 87

Evidence Supporting the Use of Neoadjuvant Therapy
Despite the strong rational behind neoadjuvant therapy for localized PDAC, stronger evidence is needed to make it standard-of-care, especially in cases of clearly resectable disease. The first evidence of improved outcomes with neoadjuvant therapy came from studies carried out in population databases. Mokdak et al showed that neoadjuvant therapy was associated with improved survival when compared to upfront surgery, even when the comparison was restricted to those submitted to upfront surgery followed by adjuvant chemotherapy. 106 Other studies suggest the survival increment secondary to neoadjuvant therapy might be more pronounced for patients with more advanced disease 107 and for those undergoing multi-agent neoadjuvant chemotherapy regimens. 108 However, one must highlight that not all population-based studies suggest neoadjuvant therapy is associated with improved outcomes. Two recent European studies, 1 from Germany and another from Sweden, failed to show an association between neoadjuvant therapy and improved overall survival.109,110 Additionally, many of the studies evaluating the role of neoadjuvant therapy suffer from immortal time bias, as they compared patients submitted to surgery who received either neoadjuvant or adjuvant therapy. Additionally, limited evidence suggests that for patients who are able to receive all planned chemotherapy cycles, neoadjuvant therapy offers no benefit in overall survival. 111
Randomized Clinical Trials Evaluating the Role of Neoadjuvant Therapy for Localized PDAC.

GEM, gemcitabine; CAP, capecitabine; NAB, nab-paclitaxel; PEXG, cisplatin + epirubicin + capecitabine + gemcitabine; CT, chemotherapy; RT, radiotherapy.
a3-year overall survival.
b1-year overall survival.
cAvailable only as meeting abstracts.
Neoadjuvant Chemotherapy
So far, 7 randomized controlled trials have evaluated the role of neoadjuvant chemotherapy for patients with potentially resectable pancreatic cancer. For the NEONAX, a non-comparative trial including patients with resectable disease, both disease-free survival (11.5 vs 5.9 months) and overall survival (25.2 vs 16.7 months) results favor the arm of neoadjuvant gemcitabine plus nab-paclitaxel for 2 cycles.104,119 Two other studies have been presented with short follow-up times. The ESPAC-5F compared the pathological and survival outcomes of patients with borderline resectable PDAC after treatment with neoadjuvant chemoradiation, capecitabine plus gemcitabine, FOLFIRINOX, or upfront surgery.100,118 At 12 months, survival numerically favored the use of neoadjuvant therapy, especially FOLFIRINOX and capecitabine plus gemcitabine. In the PANACHE-01/PRODIGE 48, patients with resectable disease were randomized to perioperative FOLFIRINOX, perioperative FOLFOX, or upfront surgery followed by adjuvant chemotherapy. 115 At 12 months, more patients were alive in the FOLFIRINOX and upfront surgery arms. This led to the discontinuation of the neoadjuvant FOLFOX arm.
More mature results are available for other studies. In a small trial by the Swiss group, 4 cycles of neoadjuvant GEMOX (oxaliplatin plus gemcitabine) failed to improve survival for patients with resectable PDAC. 94 In the PACT-15, patients with resectable disease were randomized to upfront surgery with adjuvant gemcitabine, upfront surgery with adjuvant PEXG (cisplatin, epirubicin, capecitabine, and gemcitabine), or perioperative PEXG. 98 Event-free survival and overall survival times were numerically longer in the perioperative arm. Perhaps the most important study presented so far in this setting is the Prep-02/JSAP-05, a Japanese phase II/III randomized trial evaluating the role of 2 cycles of neoadjuvant gemcitabine plus S-1 for patients with potentially resectable PDAC. 99 The authors identified a significant improvement in overall survival for patients undergoing neoadjuvant chemotherapy (median overall survival: 36.7 vs 26.7 months; HR = .72; P = .015). However, an alert sign came with the results of the NEPAFOX trial, a small phase II trial comparing upfront surgery followed by adjuvant gemcitabine to perioperative FOLFIRINOX for patients with resectable (roughly 80%) or borderline resectable PDAC. 114 The median overall survival for patients treated with FOLFIRINOX was only 10 months (vs 25.7 months in the upfront surgery arm). Importantly, less than 50% of patients in the FOLFIRINOX arm eventually underwent tumor resection. These conflicting results are potentially explained by heterogenous treatment regimens, populations, and methods of staging (discussed ahead) across trials, along with differences in how survival data were analyzed. Additionally, some of the studies (NEPAFOX, PANACHE-01/PRODIGE 48, and Prep-05/JSAP-02) have only been presented as meeting abstracts, and further data from these trials are awaited.
There have been few studies comparing different chemotherapy regimens in the neoadjuvant scenario. One initial study showed that single-agent gemcitabine was associated with inferior survival when compared to cisplatin plus gemcitabine in the neoadjuvant setting. 120 More recently, the SWOG S1505 compared the outcomes of patients with resectable PDAC treated with perioperative FOLFIRINOX or gemcitabine plus nab-paclitaxel. 38 There was no difference in the primary outcome of 2-year overall survival rate between the 2 arms (47% for FOLFIRINOX arm and 48% for gemcitabine plus nab-paclitaxel arm). Also, secondary outcomes of R0 resection, node-negative resection, pathological response, and disease-free survival were similar between the 2 arms. However, this trial has been criticized for its relatively small sample size and the need to remove almost one-third of the participants after central radiological review detected they did not fulfill the study’s inclusion criteria. Additionally, multiple retrospective analyses and 1 meta-analysis suggest FOLFIRINOX might be associated with better outcomes in the neoadjuvant setting.121-124 Therefore, FOLFIRINOX is still considered to be the standard chemotherapy regimen in the neoadjuvant setting, and gemcitabine plus nab-paclitaxel should be reserved for those not able to tolerate FOLFIRINOX.
There is very little evidence to support the use of molecular data to tailor the choice of neoadjuvant chemotherapy. In a recent study, patients with BRCA-1/2 mutation carriers with borderline resectable PDAC treated with platinum-based neoadjuvant chemotherapy achieved a complete pathological response rate of 44% (vs 10% for non-carriers). 125 Therefore, even though this information is not usually available at the start of neoadjuvant therapy, patients with known germline BRCA-1/2 (and PALB2) mutations should receive platinum-based neoadjuvant chemotherapy regimens. Also, limited data also suggest that higher intratumoral expression of carboxylesterase 2, the most efficient enzyme activating the pro-drug irinotecan into its active form SN-38, is associated with improved survival for patients undergoing neoadjuvant FOLFIRINOX in the neoadjuvant setting. 126 However, this marker is currently not used in clinical practice.
Neoadjuvant Radiotherapy
Four randomized trials evaluated the role of neoadjuvant gemcitabine-based chemoradiation for patients with potentially resectable PDAC. Two small initial trials conducted in Germany and Italy included patients with potentially resectable (resectable or borderline resectable) PDAC and randomized them between upfront surgery and neoadjuvant chemoradiation.97,112 Both arms received adjuvant single-agent gemcitabine. There were no significant differences in time to progression or overall survival. A Korean trial included only patients with borderline resectable PDAC and again randomized them between upfront surgery and neoadjuvant chemoradiation. 92 The study’s primary outcome, 2-year overall survival rate, favored patients treated with neoadjuvant chemoradiation (40.7 vs 26.0%; P = .028). Secondary outcomes, such as pathological staging and resection margins, were also improved in the neoadjuvant chemoradiation arm. Lastly, so far, the most important trial evaluating the role of neoadjuvant chemoradiation for PDAC is the PREOPANC trial. 93 In this study, 246 patients with resectable or borderline resectable PDAC were randomized to gemcitabine-based chemoradiation or upfront surgery. Patients received 4 to 6 months of adjuvant gemcitabine after surgery. In an updated analysis, there was a significant improvement in the primary outcome of overall survival (15.7 vs 14.3 months; HR = .73; P = .025). 113 Secondary outcomes of disease-free survival, locoregional failure-free survival, and distant metastasis-free survival also favored the neoadjuvant treatment arm. In subgroup analysis, the benefit of neoadjuvant chemoradiation was more robust among patients with borderline resectable disease, although this interaction was not statistically significant.
As a whole, these studies suggest improvements in overall survival, especially for the subgroup of patients with borderline resectable PDAC. However, systemic therapy is of utmost importance in these patients, and none of these trials assessed the role of radiation with systemic doses of chemotherapy in the neoadjuvant setting. In the recently published ALLIANCE 021501 trial, 126 patients with anatomical borderline PDAC were treated with either 8 cycles of FOLFIRINOX followed by surgery, and 4 additional cycles of FOLFOX or with 7 cycles of FOLFIRINOX, followed by radiotherapy, surgery, and 4 cycles of FOLFOX. 127 Radiotherapy consisted of either stereotactic body RT (SBRT) or hypofractionated image-guided RT (HIGRT). The primary outcome was 18-month overall survival rate. Patients treated with radiotherapy had a 20% decrease in the chances of being alive at 18 months (67.9 vs 47.3%) and were less likely to undergo pancreatectomy (48 vs 35%) and R0 resection (42 vs 25%). Although the relatively small sample size and the use of non-conventional radiation techniques hamper the extrapolation of these data to all sorts of preoperative radiotherapy, most experts believe that given the small chance of tumor downstaging associated with neoadjuvant chemoradiation, 128 its use after neoadjuvant chemotherapy should be reserved for those at the highest risk of positive surgical margins, such as patients with arterial involvement. 129
Immunotherapy for Localized PDAC
The use of immunotherapy in pancreatic cancer is sound due to the pivotal role played by tumor microenvironment and its immune cells in the physiopathology of PDAC. 130 Also, its use is expected to be more effective in the neoadjuvant setting (before or concomitant to chemotherapy), due to improved immune response priming in the presence of the tumor and the stronger presence of exhaustion markers before chemotherapy. 131 However, in line with data from the metastatic setting, non-selected patients (without microsatellite instable tumors) with potentially resectable PDAC do not benefit from immunotherapy. In the IMPRESS trial, the use of the whole-tumor vaccine algenpantucel-L failed to improve the survival of patients with resected PDAC. 132 Moreover, in a randomized phase Ib/II trial, the addition of the anti-PD-1 antibody pembrolizumab to neoadjuvant capecitabine-based chemoradiation did not lead to improved outcomes for patients with resectable or borderline resectable PDAC. 133 Indeed, recent data suggest other immune checkpoints, such as TIGIT, 134 are important drivers of immune surveillance evasion in PDAC, and studies are ongoing to evaluate the efficacy of inhibitors of other immune checkpoints in the advanced disease setting.
Practical Aspects in the Management of Potentially Resectable PDAC
Initial Workup
Upon suspicion of the diagnosis of pancreatic cancer, patients should undergo triphasic abdominal computed tomography (CT) scan with pancreatic protocol and standardized reporting template.135-137 Magnetic resonance imaging (MRI) should only be used in cases of indeterminate (small or isoattenuating) pancreatic lesions, cystic pancreatic lesions, indeterminate small hepatic lesions, allergy to iodinated contrasts, or impaired renal function. 137 Also, MRI with diffusion-weighted imaging (DWI) has been shown to detect liver metastases in 14–24% of patients with non-metastatic PDAC on CT scans, suggesting that abdominal MRI could become a new standard in terms of PDAC staging.138,139 It is important to look for indirect signs of pancreatic cancer, as they may aid in the detection of pancreatic lesions. 140 Importantly, scans performed more than 25 days before treatment start should be updated, as patients with older scans are at a higher risk of unexpected progression.141,142
Patients with a high probability of resectable PDAC based on clinical, imaging, and laboratory grounds can undergo upfront surgery without the need for pathological confirmation. However, it should be highlighted that 5–13% of patients submitted to pancreatectomy for presumed malignancy are found to have benign diseases (mostly chronic or auto-immune pancreatitis). 143 For patients with equivocal findings or candidates to neoadjuvant therapy, biopsy is mandatory to establish the diagnosis. Whenever biopsy is needed, endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) and CT-guided fine-needle aspiration (or core biopsy) have similar diagnostic yield, with very low complication rates.144,145 Given its lower sensitivity and higher rates of complication, endoscopic retrograde cholangiopancreatography (ERCP) should be used for diagnostic purposes only in patients with suspected pancreatic malignancy that could not be confirmed using the abovementioned methods. 146
In patients presenting with jaundice, routine preoperative biliary drainage (PBD) before upfront surgery has been associated with increased rates of postoperative complications.147,148 Therefore, PBD should be indicated only for patients with cholangitis, expected delayed surgery, or planned neoadjuvant therapy. 149 Whenever PBD is needed for patients with PDAC, ERCP is considered to be the standard approach, while endoscopic ultrasound biliary drainage (EUS-BD) and percutaneous transhepatic biliary drainage (PTBD) are considered valuable alternatives. 149 As modern neoadjuvant treatment schedules usually span several months, PBD should employ fully or partially covered self-expandable metal stents due to their longer patency compared to plastic stents. 150 In the neoadjuvant scenario, these types of metal stents should be preferred to uncovered metal ones as the latter are more difficult and riskier to remove during surgery. 151
Patients also should undergo chest CT scan and have blood drawn for complete blood counts, renal and hepatic function tests (including bilirubin levels), and baseline CA 19-9 measurement (ideally when bilirubin levels are within the normal range). Some authors 152 recommend routinely performing positron emission tomography/computed tomography (PET/CT) based on 1 randomized controlled trial (PET-PANC) that showed that PET/CT led to changes in staging and planned management in 14 and 45% of the patients, respectively. 153 However, while many specialists agree PET/CT might have a role in PDAC staging, 154 most suggest it should be used selectively in patients with high-risk features (National Comprehensive Cancer Network [NCCN] and Brazilian Society of Clinical Oncology [SBOC] guidelines).34,155 Another controversial issue is the role of staging laparoscopy, especially for patients’ candidates to neoadjuvant therapy. Studies have shown that 15–25% of patients with potentially resectable disease are found to have occult intra-abdominal disease in staging laparoscopy. 156 For that, guidelines suggest that staging laparoscopy could be selectively indicated for patients with high-risk features.34,155,157 Lastly, in clinical scenarios where it is feasible, we suggest routinely searching for dihydropyrimidine dehydrogenase (DPD) deficiency in patients who are eligible to treatment with a fluoropyrimidine, as this strategy is also likely to be cost-effective also in the management of localized PDAC. 158 Recent data also suggest a potential role for testing patients undergoing treatment with FOLFIRINOX for uridine diphosphate glucuronosyltransferase 1A1 (UGT 1A1) deficiency, and patients treated with gemcitabine plus nab-paclitaxel for cytidine deaminase (CDA) and ATP Binding Cassette Subfamily B Member 1 (ABCB1) polymorphisms in search for variants associated with increased toxicity. 159 However, apart from DPD deficiency test, these tests are not routinely used in our clinical practice.
Then, patients should be clinically staged according to the eighth edition of the AJCC TNM staging criteria (supplementary table 1). 160 However, 1 caveat of this staging system is the lack of an immediate translation into patient management, given the fact that it does take into consideration the probabilistic nature of vessel invasion according to imaging features and the low accuracy of pre-operative lymph node staging in PDAC. Then, many groups devised classifications aiming at separating patients who have disease resectable with probable R0 resection (resectable disease), probable R1 resection (anatomical borderline resectable disease), or probable R2 resection (locally advannced unresectable disease). 161 As a result, there were slight differences in these classifications that prevented cross-study comparisons and hampered proper communication in the field. With that in mind, during the 2017 meeting from the International Association of Pancreatology, specialists developed a consensus classification (supplementary table 2). 162 One great advantage of this classification is that it also incorporates prognostic features associated with high risk of disease progression (eg, baseline CA 19-9 levels ≥500 UI/ml, clinical finding suspicious for metastatic disease, or regional lymph node metastasis diagnosed by biopsy or PET/CT) and clinical features associated with higher risk of post-operative complications (eg, Eastern Cooperative Oncology Group [ECOG] performance status ≥2).
Systemic Treatment for Potentially Resectable PDAC
Given the high rates of R1 resection when upfront surgery is undertaken for patients with anatomical borderline resectable PDAC, all such patients should receive neoadjuvant chemotherapy (Figure 1). For patients with resectable disease, there is still a great deal of controversy on the optimal strategy. Pending further data from randomized trials, many experts recommend neoadjuvant chemotherapy for patients with high-risk features, as defined by the NCCN or the IAP consensus. For patients treated with upfront surgery, elevated pre-operative CA 19-9 levels (≥500 UI/ml) are associated with lower R0 resection rates and inferior survival outcomes, and neoadjuvant therapy should be considered for these patients.
163
Patients with compromised performance status (ECOG 2) or decompensated comorbidities have a higher risk of major post-operative complications, and a period of neoadjuvant chemotherapy along with a structured re-habilitation program might optimize treatment delivery and reduce post-operative complications.
164
In the experience of the MD Anderson Cancer Center, 37% of patients with compromised performance status or decompensated comorbidities achieved resection.
105
With this goal, optimal clinical management of such patients is paramount. Indeed, recent data from the advanced disease setting suggest even a short-course integrative care program can lead to clinical benefit in as much as 50% of the patients, allowing then to undergo polychemotherapy.
165
However, patients who have ECOG performance status 3 or 4 despite adequate clinical management are not candidates to chemotherapy or surgery and should be considered for best supportive care.162,166 Staging and treatment algorithm for patients with localized pancreatic cancer.@ High-risk features: large (>2–3 cm) tumors, positive lymph nodes (biopsy-proven or based on PET-CT), elevated CA 19-9 levels (≥500 UI/ml), celiac-type pain, significant weight loss (≥10% of body weight), or suspicious hepatic or pulmonary lesions.# Unfit for upfront surgery: ECOG 2.$ During neoadjuvant therapy, consider physical and nutritional rehabilitation.
Selected Ongoing Randomized Trials in Neoadjuvant Treatment for PDAC.
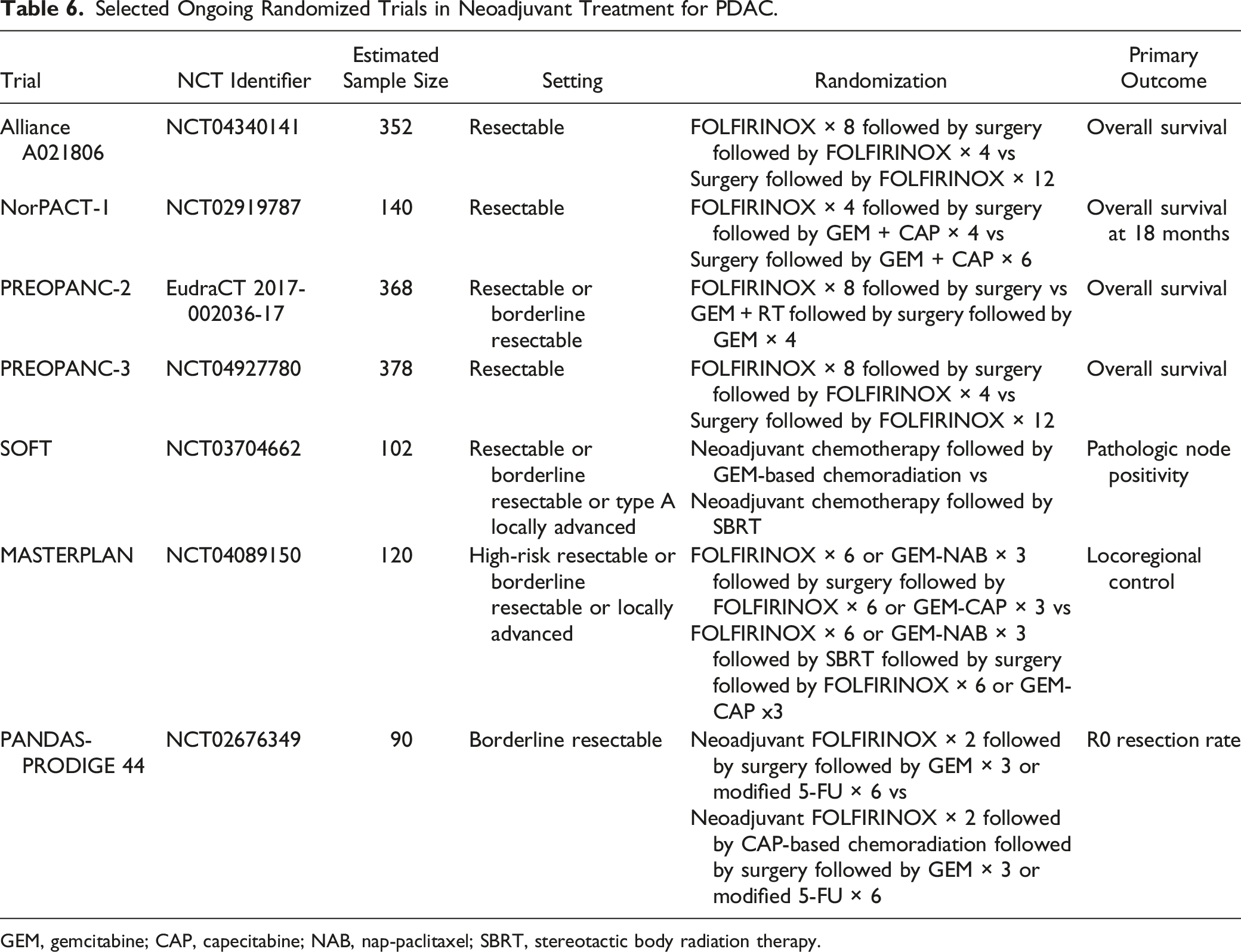
GEM, gemcitabine; CAP, capecitabine; NAB, nap-paclitaxel; SBRT, stereotactic body radiation therapy.
Another unsettled question is the role of adjuvant chemotherapy after neoadjuvant chemotherapy. Retrospective and population-based data suggest patients with poor prognostic factors, such as failure to normalize CA 19-9 levels, 170 positive surgical margins, 171 or positive lymph nodes, 172 might benefit from adjuvant chemotherapy. However, other studies suggest adjuvant chemotherapy might play a role after neoadjuvant therapy even for patients without poor prognostic features.173-175 Therefore, given these controversial findings and in accordance with current guidelines, 34 we suggest whenever patients have adequately recovered from surgery, they should receive adjuvant chemotherapy to complete a total of 6 months of systemic therapy. In our opinion, consideration should be given to switch treatment regimen in case of poor pathological response to neoadjuvant chemotherapy. 176
During neoadjuvant chemotherapy, patients should undergo response assessment scans every 2 months. 87 Given the limitations of response assessment of neoadjuvant therapy, 177 patients with resectable or borderline resectable PDAC and stable disease at the end of neoadjuvant therapy should undergo surgical exploration. However, patients with exclusive local progression during neoadjuvant chemotherapy pose a challenging clinical scenario. While some would consider salvage chemoradiation, the probability of tumor downstaging with chemoradiation is considered to be low (<10%). 128 On the other hand, recent data support that many patients can be adequately salvaged when shifted to a different chemotherapy regimen.95,96 Thus, despite the lack of randomized data, whenever exclusive local disease progression takes place during neoadjuvant chemotherapy, strong consideration should be given to switch neoadjuvant chemotherapy to an alternative regimen. Likewise, lack of a significant reduction in CA 19-9 levels (at least ≥50% drop) despite neoadjuvant chemotherapy is a poor prognostic factor.178–180 In such cases, only half of the patients will have their CA 19-9 levels normalized post-operatively. Therefore, instead of proceeding with tumor resection, one should consider changing the chemotherapy regimen of these patients. 96
Survivorship
Currently, there is no randomized data to guide the optimal follow-up strategy for patients with resected PDAC. However, a structured follow-up protocol has many advantages, including nutritional and metabolic surveillance, psychological reassurance, and early initiation of systemic therapy in case of disease relapse. 181 In fact, retrospective data suggest an intense follow-up schedule allows patients to have their disease relapse diagnosed when they are asymptomatic or while they have adequate performance status, and to receive standard-of-care therapies. 181 After surgery, patients should undergo CT scans and have blood drawn for CA 19-9 levels. These studies should be repeated every 3 months during the first 2 years and every 6 months thereafter up to 5 years after treatment completion.22,34 Also, during treatment or follow-up periods, patients are strongly encouraged to undergo genetic consultation. 34
Conclusion
In the past 2 decades, we witnessed considerable advances in the management of potentially resectable PDAC. However, even when more active polychemotherapy regimens are used in the adjuvant setting, most patients still experience disease relapse. With that in mind, the neoadjuvant treatment approach has been brought to the treatment of PADC with the goal of improving treatment exposure and disease-related outcomes. Currently, neoadjuvant therapy is considered to be the standard-of care for patients with borderline resectable pancreatic cancer. While randomized trials are ongoing to settle the role of neoadjuvant chemotherapy in resectable PDAC, we think neoadjuvant chemotherapy with either FOLFIRINOX (preferred) or gemcitabine and nab-paclitaxel should be considered for patients with PDAC and high-risk features. Future trials of neoadjuvant treatments for patients with PDAC should implement strong internal validity to reduce heterogeneity observed in previous randomized trials—which have focused more on external validity. That said, we suggest these trials include high-quality image tests, consider laparoscopy and FDG-PET for proper staging, and ideally identify (and stratify) BRCA-1/2 (and PALB2/ATM) mutation carriers.
Supplemental Material
Supplemental Material - Current Treatment of Potentially Resectable Pancreatic Ductal Adenocarcinoma: A Medical Oncologist’s Perspective
Supplemental Material for Current Treatment of Potentially Resectable Pancreatic Ductal Adenocarcinoma: A Medical Oncologist’s Perspective by Victor Hugo Fonseca de Jesus, MD, MSc, and Rachel P. Riechelmann, MD, PhD in Cancer Control.
Footnotes
Author Contributions
Victor Hugo Fonseca de Jesus: conceptualization, writing, and visualization; Rachel P Riechelmann: conceptualization, writing, and visualization.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
As a review of the current medical literature, approval by Institutional Ethic Board Review is not needed by local standards.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
