Abstract
Purpose
Hepatic metastasis frequently occurs in patients who have undergone radical pancreatic resection for pancreatic cancer. Besides chemotherapy, various local treatment approaches targeting hepatic lesions have been explored. However, research on radiofrequency ablation (RFA) as a localized therapy for hepatic metastasis is limited. Therefore, we conducted this retrospective study to provide clinical evidence.
Methods
This is a single-center, retrospective, cohort study. After radical pancreaticoduodenectomy, 32 patients developed metachronous hepatic metastasis with fewer than 3 lesions, the largest of which was less than 3 cm in diameter. These patients underwent combined treatment with chemotherapy and RFA. After 8 weeks of chemotherapy, patients received RFA for hepatic lesions. Additional chemotherapy was administered, and the patients’ tumor status and survival were monitored. The primary endpoint of this study was overall survival (OS). Factors affecting OS were analyzed using the Cox risk model.
Results
Among the 32 patients, the mean OS was 28.4 months. Univariate and multivariate Cox regression analysis revealed that the time (in months) of liver metastasis (HR = 0.04, 95% CI: 0.01 to 0.19; P < 0.001), the number of liver metastases (HR = 7.08, 95% CI: 1.85 to 27.08, P = 0.004), and PD (progressive disease) response to the second round of chemotherapy (HR = 29.50, 95% CI: 1.46 to 597.27; P = 0.027) were independent predictors of poorer survival.
Conclusion
Combined therapy with RFA and chemotherapy is safe in patients with hepatic metastasis after radical pancreaticoduodenectomy. Early recurrence (≤12 months), three liver metastatic lesions, and a poor response to the second round of chemotherapy were associated with poor survival.
Keywords
Introduction
Pancreatic cancer is a devastating disease with an extremely poor prognosis. It was estimated that in 2023, the number of new cases in the USA would be 64,050 and that 50,550 would die from the disease. 1 Despite advancements in basic and clinical research into pancreatic cancer, little improvement in prognosis has been achieved over the past decades. Most patients have advanced, unresectable lesions at the time of diagnosis. The median overall 5-year survival rate of pancreatic cancer does not exceed 11%, even in countries with well-developed medical systems. 1 Surgical resection remains the only curative option, applicable only to patients with local lesions confined to the pancreas. The 5-year survival rate for patients with pancreatic cancer has improved after radical resection in large centers. 2 However, even for patients with resectable carcinomas, the median overall survival time is only 14-38 months after radical resection and systemic chemotherapy.3,4 Hepatic metastasis, the most common sign of tumor recurrence, occurs in approximately 60%-80% of patients after radical resection,5,6 typically signifying a poor prognosis and limited life expectancy. Recommended treatments for patients with metastatic hepatic lesions include various chemotherapy regimens. 2 New regimens and pharmacological developments, including FOLFIRINOX, 7 gemcitabine/nab-paclitaxel, 8 and PARP inhibition as maintenance therapy in patients with germline BRCA1 or BRACA2 mutations have extended life expectancy for these patients. 9 In the treatment of pancreatic tumors and synchronous liver metastases, several clinical trials have attempted local treatment of metastatic hepatic lesions, including transcatheter arterial chemoembolization (TACE), percutaneous ethanol injection (PEI), and surgical resection.10-13 Significantly, these local treatments have proven helpful only in select patients. For patients who develop metastasis after radical resection, systemic treatment is the only recommended therapy 2 known to improve prognosis. Most attempts at local treatment targeting metastatic lesions have proven to be largely ineffective. However, combined therapy involving local treatment and chemotherapy has shown improved outcomes in some trials compared to patients who only received chemotherapy. 14
Radiofrequency ablation (RFA) is an efficient local treatment for both primary and metastatic hepatic lesions.15-18 RFA has been shown to have similar effects to surgical resection for hepatic lesions no larger than 3-5 cm.18,19 The application of RFA for metastatic hepatic tumors of pancreatic carcinoma of an appropriate size has seldom been reported. In this study, we report 32 cases of patients with metastatic hepatic tumors following radical pancreaticoduodenectomy. These patients, with tumors suitable for RFA in size and number after a chemotherapy cycle of 8-10 weeks, underwent combined therapy of chemotherapy and RFA.
Material and Methods
Study Population
This single-center, retrospective, cohort study included 32 patients who developed metastatic hepatic lesions after radical pancreaticoduodenectomy and underwent combined therapy between January 2012 and January 2018 at the Second Affiliated Hospital of Zhejiang University School of Medicine (Figure 1). Patient demographics and baseline characteristics are summarized in Table 1. At the time of diagnosis of hepatic metastasis after radical pancreaticoduodenectomy, there was no evidence of peritoneal, retroperitoneal, or other metastasis sites beyond the hepatic lesions. All patients had 3 or fewer hepatic lesions, with the largest being less than 3 cm in diameter. Patients underwent chemotherapy for 8-10 weeks before reassessment of the tumors. No new lesions were found, and the largest hepatic lesion was less than 5 cm in diameter. Percutaneous RFA of the hepatic lesion was performed, followed by subsequent chemotherapy. Serum glucoprotein antigen 199 (CA19-9) levels, metastatic hepatic lesions, and patient outcomes were monitored and evaluated, according to relevant researches.3,20 Overall survival (OS) was defined as the time from surgery to either the date of death or last follow-up. The exclusion criteria were as follows: non pancreaticoduodenectomy surgery histo; an intraductal papillary mucinous carcinoma or a pancreatic intraepithelial neoplasia; incomplete records owing to follow-up at other institutions or <12 months of follow-up. The present study was approved by the Institutional Review Board of the Second Affiliated Hospital of Zhejiang University School of Medicine (Approval No. 2023LSYD0235). The analysis used anonymous clinical data obtained after all the participants agreed to treatment by written consent, so patients were not required to give informed consent for the study. All methods were carried out in accordance with relevant guidelines and regulations. The reporting of this study conforms to STROBE guidelines.
21
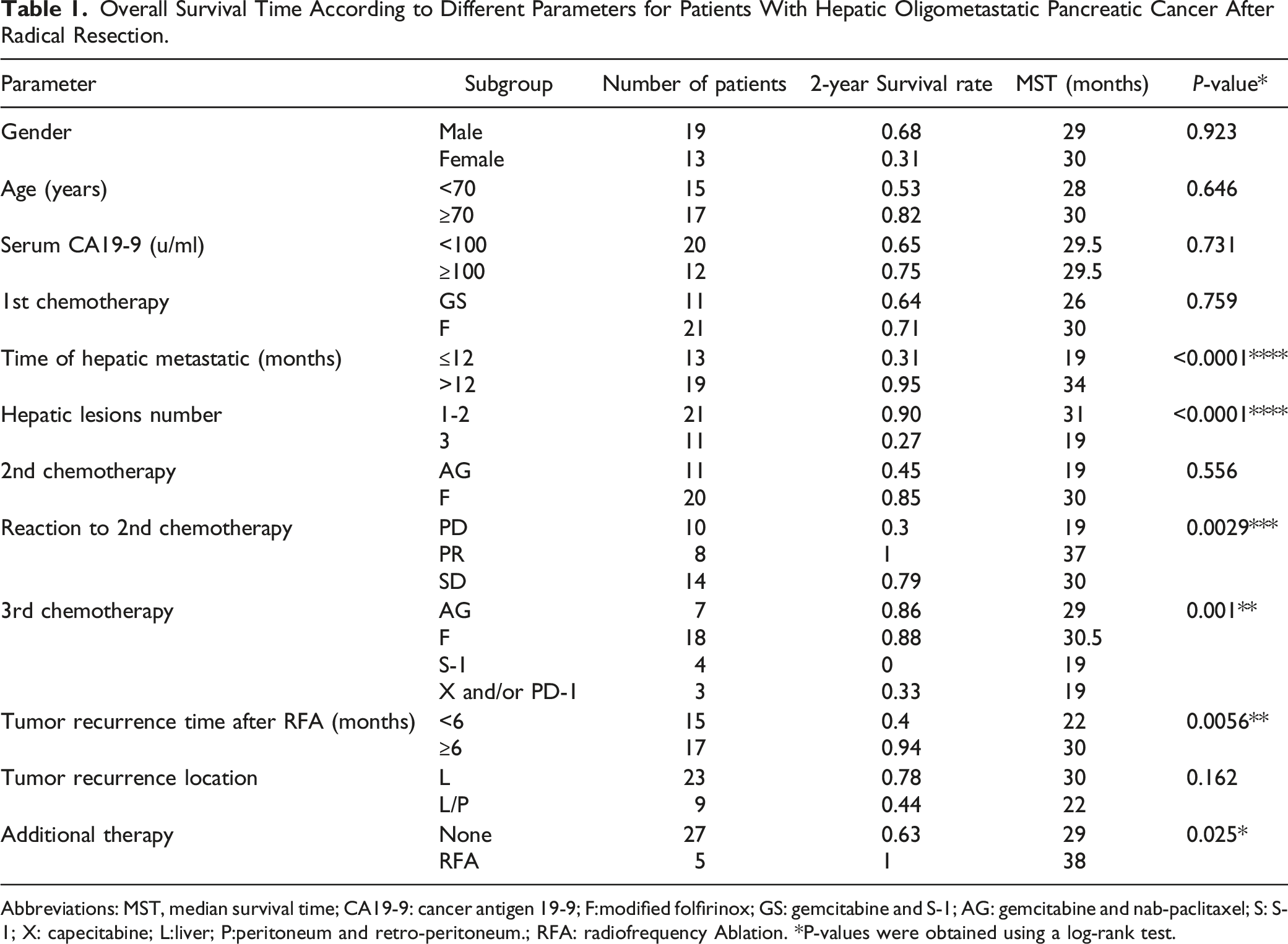
Patient selection flow chart. Overall Survival Time According to Different Parameters for Patients With Hepatic Oligometastatic Pancreatic Cancer After Radical Resection. Abbreviations: MST, median survival time; CA19-9: cancer antigen 19-9; F:modified folfirinox; GS: gemcitabine and S-1; AG: gemcitabine and nab-paclitaxel; S: S-1; X: capecitabine; L:liver; P:peritoneum and retro-peritoneum.; RFA: radiofrequency Ablation. *P-values were obtained using a log-rank test.
Owing to the distinctive inclusion criteria adopted in this study, we encountered limitations in searching for additional similar research data, which could facilitate sample size calculation. Therefore, we resorted to utilizing the data generated within the current study for the estimation of the requisite sample size. We defined 12 months as a threshold for differentiating between early and late recurrence. The 2-year OS in the late recurrence group was 95% and 31% of the early recurrence group. Calculation formula:
Initial Management of Patients
All patients underwent enhanced computer tomography (CT) or magnetic resonance (MR) scanning before surgery and showed no signs of hepatic metastatic tumors. No palpable liver masses were found during surgical exploration. All patients received adjuvant chemotherapy starting 6-8 weeks post-surgery and lasting for 4 months. Regimens included modified FOLFIRINOX (21 of 32 patients) and Gemcitabine plus S-1 (11 of 32 patients). Patients were monitored every 3 months in the first year and every 6 months thereafter. Blood tumor marker testing and contrast-enhanced CT scans were performed at each surveillance visit.
Management After the Discovery of Hepatic Metastasis
All patients underwent chemotherapy after the discovery of hepatic lesions. For the 19 patients who developed metastasis more than one year after surgery, the regimen was identical to the adjuvant therapy. A second-line regimen, including gemcitabine plus nab-paclitaxel and modified FOLFIRINOX, was given to the remaining 13 patients, according to the previous regimen. Serum CA19-9 levels were tested monthly during chemotherapy. The response of the hepatic lesion to chemotherapy was assessed by enhanced CT or MR after 2 months of chemotherapy, following the Response Evaluation Criteria in Solid Tumors (RECIST) standard version 1.1.
After confirming that the size and number of hepatic lesions met the previous criteria, RFA for the hepatic lesions was performed. The RFA procedure was performed percutaneously under the guidance of ultrasonography. The patients underwent general anesthesia and intubation. After the cool-tip needle was punctured into the core of the lesion under ultrasonographic guidance, the ablation lasted for 6-8 minutes per lesion. The ablation areas were confirmed to fully cover the lesion using an ultrasonography scan after ablation.
For those assessed as having a partial response (PR) or stable disease (SD) after the second round of chemotherapy, the same drugs were administered in the third round of chemotherapy, which started 2-4 weeks after ablation. For patients with progressive disease (PD), a different regimen was applied. Serum tumor markers were tested monthly during therapy, and CT/MR scans were performed every 2 months. Repeated RFA was applied to patients with newly developed hepatic lesions still suitable for ablation. The chemotherapy regimen was adjusted according to patient status and the tumor’s reaction to chemotherapy. The detailed scheme is shown in Figure 2. Treatment scheme diagram.
Statistical Analysis
Overall survival, from the date of radical resection, was estimated using the Kaplan-Meier method and survival curves were compared using the log-rank test. Continuous variables were categorized, and the cutoff values referred to previously published data. Multivariate analysis was performed using the Cox proportional hazards model, which tested all clinical and tumor-related factors significantly associated with OS in the univariate analysis. P < 0.05 was considered statistically significant. Statistical analyses were performed using MedCalc 20.0.3, R programming language 4.3.0, and SPSS 19.0.
Results
Patient Characteristics and Clinical Background
All patients, with primary tumors located in the pancreatic head, underwent radical pancreaticoduodenectomy. The pathological diagnoses for these patients were all pancreatic duct adenocarcinoma. The demographic characteristics are shown in Table 1.
Four patients developed hepatic lesions on computed tomography (CT) scan during adjuvant chemotherapy, within 6 months after surgery. Nineteen patients had hepatic metastasis over one year after surgery. The remaining 9 patients experienced liver metastasis between 6 and 12 months after surgery (also within 6 months after completing adjuvant chemotherapy). Thirteen patients developed a single hepatic lesion, 9 had two lesions, and 10 had three lesions at the time of diagnosis of metastasis in the liver. Twenty-five patients had elevated serum CA19-9 levels before treatment. Ten patients experienced hepatic lesion progression after 2 months of chemotherapy. Eight cases showed partial response (PR) and 14 had stable hepatic tumors (SD) after 2 months of chemotherapy. The original data are shown in Additional File 1.
RFA was performed before the third round of chemotherapy. All detectable lesions were ablated, confirmed by ultrasonography immediately after ablation. Of the 28 patients with elevated serum CA19-9, all experienced a decrease in CA19-9 2 weeks post-RFA. Among these, 14 patients’ CA19-9 levels decreased to the normal range.
Hepatic abscesses developed in 2 patients 8-10 days post-first RFA and one patient post-second RFA. All three patients had a primary pancreatic tumor located in the head of the pancreas and had undergone a pancreaticoduodenectomy procedure. They experienced abdominal pain and fever, and an abscess was detected via ultrasonography, with a percutaneous drainage tube placed simultaneously. Intravenous antibiotics were administered to the patients for 3-5 days, based on their temperature and blood test results. High fever and upper abdominal pain subsided after percutaneous abscess drainage and intravenous antibiotics. Two patients kept the drainage tube for 7-10 days. One patient kept the drainage for over 4 weeks due to bile leakage. Chemotherapy was initiated more than a week after their temperatures returned to normal. None of the 32 patients experienced bleeding or other complications after RFA.
Patients started the third round of chemotherapy 2-4 weeks post-ablation. Those assessed as PR and stable disease (SD) to previous chemotherapy continued with the same regimen. The remaining 10 patients, assessed as progressive disease (PD), changed their regimen. Among these, 4 switched from AG to S-1, 2 from FOLFIRINOX to AG, 1 from GS to FOLFIRINOX, 1 from AG to capecitabine, 1 from AG to capecitabine plus PD-1, and 1 from FOLFIRINOX to PD-1. Five other patients underwent additional RFA when new lesions suitable for ablation emerged.
Survival Outcomes
For the entire cohort,the median follow-up period was 29.4 months (range, 12-46 months). At the end of the follow-up period, all 32 patients had died. The mean OS was 28.4 months. The 2-year survival rate was 68.8%. Patient characteristics significantly associated with OS are shown in Table 1, labeled by P value*.
For patients with early recurrence (≤12 months), the median OS was shorter than patients with recurrence time >12 months (median, 19 vs 34 months; P < 0.0001) (Figure 3A), and 2-year survival rates were 31% and 95%, respectively. For liver metastasis number, having 3 lesions was significantly associated with poorer survival compared to those with 1-2 metastatic lesions (median, 19 vs 31 months; P < 0.0001) (Figure 3B), and 2-year survival rates were 27% and 90%, respectively. For the response to the second round of chemotherapy, PD was associated with poorer survival compared to PR and SD (median, 19 vs 36 vs 30 months; P = 0.0029) (Figure 3C), with 2-year survival rates of 30%, 100%, and 79%, respectively. For the third round of chemotherapy, AG (gemcitabine & nab-paclitaxel) and modified FOLFIRINOX chemotherapy regimens showed better survival compared to S-1/capecitabine/PD-1 (P = 0.0037) (Figure 3D). For tumor recurrence time after RFA, patients with early recurrence (<6 months) had shorter median OS than patients with recurrence time >12 months (median, 22 vs 30 months; P = 0.0056) (Figure 3E), with 2-year survival rates of 40% and 94%, respectively. For additional therapy, patients with repetitive RFA treatment had better median OS than patients with no further treatment (median, 38 vs 29 months; P = 0.0251) (Figure 3F), with 2-year survival rates of 100% and 63%, respectively. Overall survival curves for patients with hepatic metastatic pancreatic cancer after radical resection according to risk factors: (A) Occurrence time of hepatic metastasis after surgery; (B) Number of metastatic liver tumors; (C) Reaction to the second round of chemotherapy; (D) Third chemotherapy regimen; (E) Tumor recurrence time after RFA; (F) Additional therapy.
Univariate and Multivariate Cox Proportional Hazards Regression Analysis for Prediction of Overall Survival in Patients With Hepatic Oligometastatic Pancreatic Cancer After Radical Resection.

Abbreviations: RFA: radio-frequency ablation; F:modified folfirinox; GS: gemcitabine and S-1; AG: gemcitabine and nab-paclitaxel; S: S-1; X: capecitabine; L:liver; P:peritoneum and retro-peritoneum.
Discussion
Hepatic metastasis is a common cause of unresectable disease in pancreatic cancer. Treatments commonly accepted for these patients are restricted to chemotherapy. FOLFIRINOX and gemcitabine/nab-paclitaxel are the most recommended regimens for patients with hepatic metastasis. The median survival time for patients with hepatic metastasis is about 11.1 months with FOLFIRINOX, which provides a disease control rate of approximately 70.2% and a response duration of 5.9 months. 7 In some cases, a complete response to FOLFIRINOX has been observed, and patients with hepatic metastasis have achieved R0 resection of the primary tumor after several chemotherapy courses.22,23 Simultaneous resection of the primary pancreatic tumor and metastatic hepatic lesion has proven beneficial only to patients with one or a few metastases in some small-sample reports.24-28
In patients who have undergone radical resection of a pancreatic tumor, hepatic metastasis is the most common sign of tumor relapse and the cause of death. Chemotherapy is almost the only accepted treatment for these patients. There is limited data for surgical treatment of hepatic metastasis that occurs after radical resection. In some small-sample reports, TACE, PEI, and even surgical resection have been attempted for treating metastatic hepatic lesions.29-32 Since neither TACE nor PEI is a complete tumor cell killing method for local hepatic tumors, it’s reasonable that TACE and PEI yielded poor benefits for metastatic hepatic lesions. Surgical resection of metastatic lesions for pancreatic cancer may cause significant injury to patients and often leads to chemotherapy interruption, the only proven beneficial treatment for metastatic pancreatic cancer prognosis. Therefore, such a treatment procedure is not widely accepted by most oncologists.
RFA provides a considerably high temperature around the needle. This heat causes necrosis in a certain area of the liver, usually 3-5 cm in diameter depending on the duration and power of ablation. RFA has proven as effective as surgical resection for hepatic cell carcinoma (HCC) lesions smaller than 3 cm and is recommended as a curative option as an alternate to surgical resection for HCC patients,33,34 and patients with colorectal cancer with hepatic metastasis. 16 For most patients, a percutaneous RFA procedure is safe and will not interrupt the chemotherapy course. 35 In our study, RFA caused complete necrosis, as confirmed by imaging scans, suggesting its effectiveness for hepatic lesions.
RFA proved safe for all patients, despite the presence of three cases of hepatic abscess in the necrosis area. Reflux through cholangiojejunostomy may explain why all abscesses occurred in patients who underwent pancreaticoduodenectomy. The typical time for abscess occurrence was 8-10 days after ablation, corresponding to the time of coagulation necrosis turning into liquefactive necrosis after RFA. One patient had bile leakage into the abscess cavity after drainage, likely caused by ablation involving small bile ducts and the coagulation necrosis decomposing in the abscess. Bleeding is another common complication of RFA, usually occurring in patients with cirrhosis and when the tumor is near the liver’s surface. In our study, no patients experienced bleeding, fistula of the gastrointestinal duct, or other complications of RFA. Most patients started subsequent chemotherapy 2 weeks after RFA. Even for those patients with abscess and bile leakage, chemotherapy was started after the patient had a normal temperature for over one week; the postponement period was less than 2 weeks.
The combined therapy provided a median OS of 28.4 months. For patients who responded well to chemotherapy, the OS data were significantly better. These results demonstrated that chemotherapy played a crucial role in the overall therapy. Residual living tumor cells resistant to the chemicals after certain rounds of chemotherapy will thrive and develop new metastatic lesions, leading to treatment failure. An effective local treatment, including surgical resection and RFA, can eliminate the residual tumor cells. Theoretically, tumor relapse can be prevented. But the new metastasis may also be due to circulating tumor cells in the bloodstream36-38 and undetectable micro-lesions already present in the liver,39,40 which also lead to the failure of treatments, including surgical resection in metastatic diseases.12,13,41 In our study, patients detected with 3 liver metastatic lesions and metastasis occurrence time within a year, as well as those having poor reactions to the second round of chemotherapy, had poor outcomes. These can be explained as these patients had a higher risk of the presence of circulating tumor cells and micro-metastasis. Compared with reported data, our patients in this study seemingly showed a better outcome than chemotherapy alone.7,8
In addition, the time point (in months) of metastasis after pancreatectomy was an important predictor of poorer survival. Groot et al 42 involved 957 resected PDAC patients in the study, and first established a classification criteria, which set a recurrence free interval of 12 months as a threshold, according to the post-recurrence survival (PRS). In the study of Groot et al, patients with resectable PDAC had a Median OS was 34.6 months for patients with late recurrence (>12 months), compared to 13 months for patients with early recurrence (≤12 months) (P < 0.001). In our work, we used the same grouping method, patients with early recurrence (≤12 months) had a 2-year OS rates of 31% compared with 95% for the late recurrence group (both P < 0.0001). The median OS was shorter than patients with recurrence time >12 months (median, 19 vs 34 months; P < 0.0001), which is similar to the previous results.3,42,43 It was worth mentioning that, in these reports recurrence locations included several categories: “local only”, “liver only”, “lung only”, “multiple-site” and “other”. However, our study only focused on cases of liver metastasis.
To avoid possible confounding factors, we limited case selection to pancreatic head carcinoma suitable for radical pancreaticoduodenectomy. In addition, all the patients had no more than three metastatic lesions only in the liver, and the largest lesion was no more than 3 cm in diameter. These conditions limit the number of patients enrolled. Whether such combined therapy is more beneficial needs further data support and analysis. Finally, adjuvant therapy comprises a diverse array of treatment regimens tailored to patients’ individual conditions at the time, and this variability may introduce a degree of error stemming from discrepancies in analysis.
Conclusion
The combined therapy of RFA and chemotherapy is safe and effective in patients with hepatic metastasis after radical pancreatic resection. Early liver recurrence (≤12 months), a higher number of liver metastasis (≥3 lesions), and a poor reaction to the second round of chemotherapy were associated with poor survival.
Footnotes
Author Contributions
Lantian Wang wrote the final manuscript. Lan Wang and Jia Yang performed the statistical analysis. Jian Wu assisted in data collation and performed the literature search. Wenjie Lu designed this research and supervised the writing of the manuscript. All authors participated in the care of the patients. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Zhejiang Province of China (no. LY19H160049).
Ethical Statement
Data Availability Statement
All data generated or analysed during this study are included in this published article.
