Abstract
Background
Docetaxel alone or in combination with ramucirumab or nintedanib is recommended as second-line treatment in metastatic non-small cell lung cancer (NSCLC) patients after progression on first-line platinum-base chemotherapy in combination with immune-checkpoint inhibitors (ICI). The purpose of our study was to compare the toxicity profile and efficacy of docetaxel after paclitaxel- or pemetrexed-platinum chemotherapy alone or in combination with ICI.
Patients und Methods
Metastatic NSCLC patients treated with docetaxel after pemetrexed or paclitaxel-platinum with chemotherapy with or without ICI were included in our retrospective analysis.
Results
We included in our analyses a total of 53 patients (median age 60.3 years (SD 9.35) in the paclitaxel-group, and 62 years (SD 12) in the pemetrexed-group) treated at our Institution with docetaxel following either paclitaxel or pemetrexed chemotherapy. No treatment-related deaths and no new safety signals were observed.
Conclusion
Docetaxel alone or in combination with ramucirumab or nintedanib demonstrated encouraging antitumor activity with a manageable safety profile in patients who have progressed on previous paclitaxel-or pemetrexed platinum-based chemotherapy. The results of this analysis can be a helpful reference in conducting further trials of new second-line treatment options.
Introduction
Lung cancer continues to be the most common cause of cancer-related morbidity and mortality worldwide. 1 The approval of immune checkpoint inhibitors (ICI) has led to remarkable survival improvements in first-line non-small cell lung cancer (NSCLC) without driver alterations. In addition to the evidence of the efficacy of first-line pembrolizumab, atezolizumab or cemiplimab for Programmed death ligand-1 (PD-L1) high expression NSCLC, several phase III studies have shown the survival superiority of platinum-based chemotherapy combined with ICI to platinum-based chemotherapy alone.2-8
Despite the improvement in the first-line therapy most patients develop primary or secondary resistance to ICIs resulting in disease progress. The actual standard of care is docetaxel either alone or in combination with ramucirumab or nintedanib based on two phase 3 studies, the REVEL study and the LUME-Lung 1, that demonstrated a significant prolonged overall survival (OS) with the addition of these drugs compared to docetaxel alone.9,10
However, the REVEL and the LUME-Lung 1 studies have been conducted before the regulatory approval of first- line ICIs; thus, the efficacy and safety of docetaxel plus either ramucirumab or nintedanib following ICI plus platinum-based chemotherapy have not been adequately studied previously, and we need to consider the residual effect of ICIs.
Furthermore, there are no prospective clinical trial evaluating the safety profile and efficacy of docetaxel following previous chemotherapy with either platinum-paclitaxel combination or platinum-pemetrexed alone or with ICI, which represents the standard of care in first-line NSCLC without driver alterations and with a PD-L1 expression level below 50%.11,12 The aim of our retrospective study was to evaluate the safety of docetaxel following a chemotherapy with either paclitaxel or pemetrexed with or without ICIs.
Methods
Study Design and Study Population
Patients with metastatic NSCLC treated at Ludwig-Maximilians-University (LMU) Hospital in Munich from September 2013 to December 2023 with second line Docetaxel after first-line platinum plus paclitaxel or pemetrexed alone or in combination with ICI were included in our retrospective analysis. The main eligibility criteria were: (I) age older than 18 years, (II) histologically confirmed diagnosis of stage IV NSCLC according to the VIII Edition of the American Joint Committee Cancer Staging System, (III) undergone at least 1 course of antitumor treatment, (IV), receiving previously to docetaxel either paclitaxel-or pemetrexed platinum chemotherapy alone or in combination with ICI (V), relatively complete baseline data in electronic medical records (VI).
The exclusion criteria for patients were the following: (I) lack of antitumor therapy, (II) histologically not non-small cell lung cancer (IV), patients with suspected thoracic malignancy without histological confirmation, and patients with a cancer of unknown primary (CUP) or patients with lung metastases from other tumors.
Clinical data including sex, age, tumor histology, tumor stage, performance status measured in Eastern Cooperative Oncology Group (ECOG) score, toxicity under docetaxel according to Common Terminology Criteria for adverse events (CTCAE) version 5.0, best response to treatment, progression-free survival (PFS) and overall survival (OS).
The study was conducted in accordance with the Declaration of Helsinki, and was approved by the Institutional Ethics Committee of the University Hospital of Munich (LMU) (Approval-code: 476-16 UE, approval date 5 August 2016). Personal identifiable information was not involved, therefore informed consent was not required. The reporting of this study conforms to STROBE guidelines. 13
Statistical Analysis
Patient and tumor characteristics were presented as absolute and relative frequencies and means with standard deviation and compared using Student’s t-test and Chi2-test, respectively. We compared distributions of therapy, response to treatment, and toxicities between patients receiving first-line pemetrexed and first-line paclitaxel using Chi2-test. To analyze factors influencing the occurrence of grade 3 or 4 toxicities we used logistic regression models including the variable pemetrexed/paclitaxel, alone or in combination with ICIs as first-line treatment and docetaxel was used as second line or a therapy line. Kaplan-Meier curves displayed differences in univariate overall and progression-free survival depending first-line therapy of paclitaxel or pemetrexed. Multivariate Cox regression analysis was used to analyze differences in overall and progression-free survival according first-line therapy, age, sex, histological subtype, TTF1 status, type of platinum therapy, whether docetaxel was used as second line or a therapy line beyond second line, and which add on to docetaxel was used. A significance threshold of alpha <0.05 was applied for all analyses.
All analyses were performed using R Studio with R version 4.0; tables and figures were created in Microsoft Excel and R Studio.
Results
Patient Characteristics
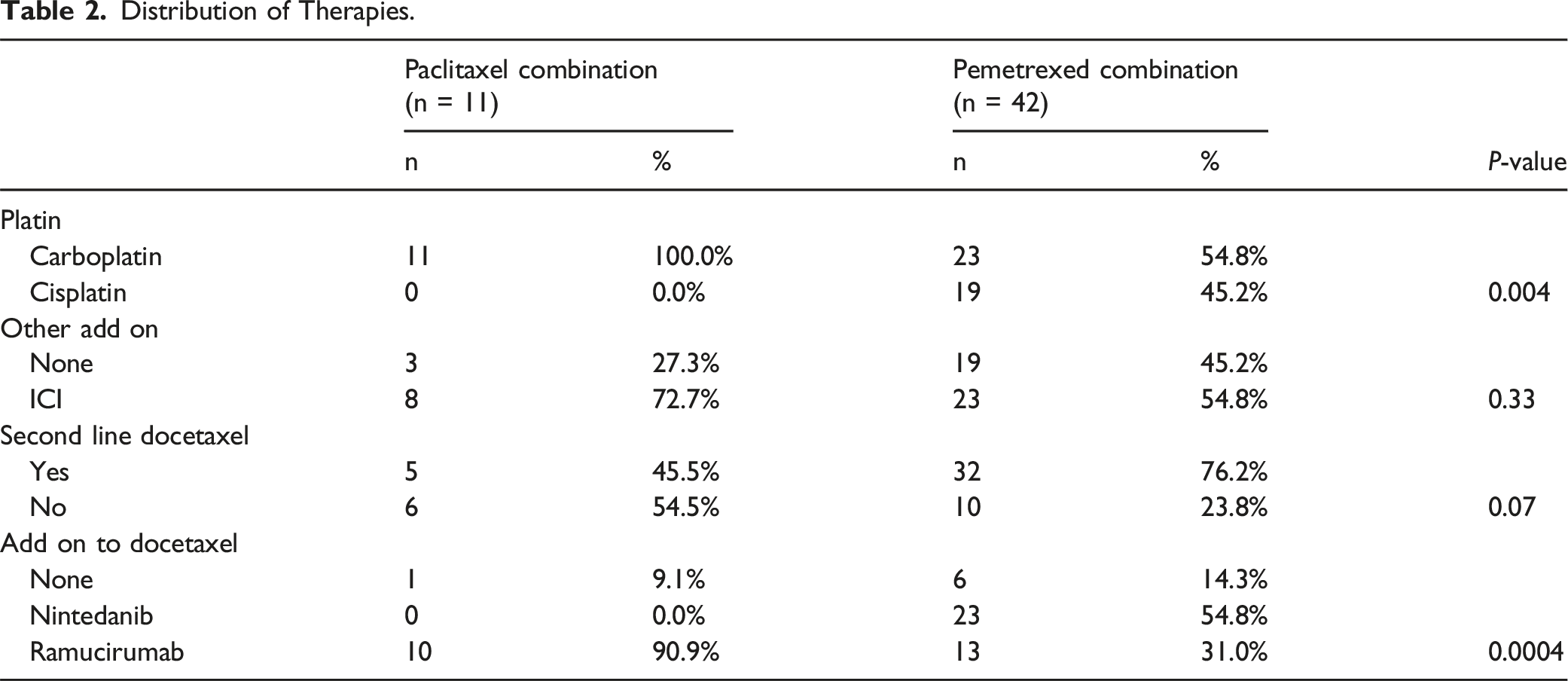
We included in our study a total of 53 patients after excluding all patients who did not meet the inclusion criteria. A total of 11 (20.7%) patients received docetaxel after paclitaxel and 42 (79.3%) patients received docetaxel after pemetrexed.
The median age in the paclitaxel group was 60.3 years (SD 9.35) and most patients were male (54.5%). In the group of patients receiving docetaxel after pemetrexed combination chemotherapy the median age was 62 years (SD 12), and most patients were male (64.3%).
6 patients (54.5%) in the paclitaxel group had ECOG 0, and 27 patients (64.3%) in the pemetrexed group presented with ECOG 0.
41 (77%) were histologically adenocarcinoma, 8 (15%) had squamous carcinoma, 1 (2%) patient had adenosquamous carcinoma, 2 (4%) patients large-cell lung carcinoma and 1(2%) presented with sarcomatoid histology. 72.8% of the patients in the paclitaxel group had a history of smoking (27.3% current-smoker and 45.5% past-smoker). 73.8% of the patients in the pemetrexed group had a history of smoking (35.7% current-smoker and 38.1% past-smoker). 9.1% of the patients in the paclitaxel combination were non-smokers and 11.9% of the patients in the pemetrexed group had never smoked.
57.1% of patients receiving docetaxel after pemetrexed had metastasis in one organ. In the Paclitaxel group 27.3 % of the patients presented with metastases in one organ. 54.6% of the population in the Paclitaxel group presented with metastases at diagnosis. The most frequent site of metastases was liver (36.4%), brain (27.3%) and adrenal (27.3%) in the paclitaxel group. In the pemetrexed combination the most frequent site of metastases were bone (42.9%), brain (26.2%) and liver (21.4%).
Patient Characteristics.

Distribution of Therapy
Distribution of Therapies.
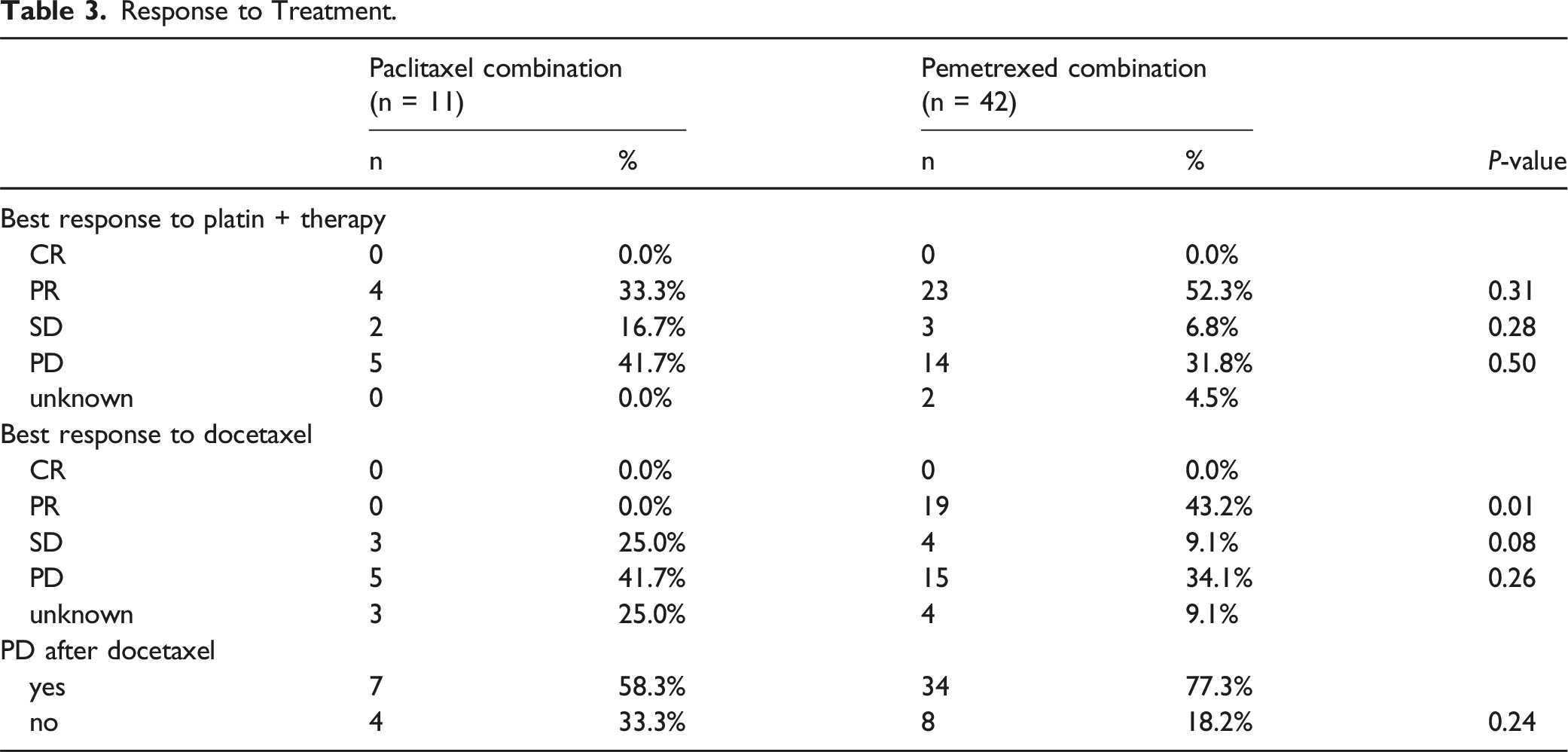
Response to Treatment
Best response to treatment with paclitaxel combination or pemetrexed was not significantly different. The proportion of patients with partial response was higher in patients with pemetrexed compared to paclitaxel (52.3% vs 33.3%, P = 0.31). The proportion stable disease (16.7% vs 6.8%, P = 0.28) and progressive disease (41.7% vs 31.8%, P = 0.50) was higher in paclitaxel patients than in pemetrexed.
Response to Treatment.
Toxicities Under Docetaxel
Overall, we found that all patients with docetaxel following pemetrexed in first line experienced a toxicity under docetaxel therapy. This was significantly different from patients with paclitaxel as first-line, where only 81.8% had a toxicity (P = 0.04).
Overall increased gamma-glutamyl transferase (GGT) was the most common adverse event, followed by anemia being the next common and hyponatremia and alopecia in the group of patients who received docetaxel after a paclitaxel combination therapy.
In the group of patients who received docetaxel after pemetrexed the most frequent adverse event was anemia and neutropenia followed by increased GGT, glutamic-oxaloacetic transaminase (GOT) and glutamic-pyruvic transaminase (GPT).
We did not find significant differences regarding toxicity grades between the two groups in our analysis. There were no grade 5 toxicities observed among patients analyzed.
In the univariate analysis ECOG emerged in our analysis as not statistically significant for the toxicities under docetaxel.
Figure 1 displays all types of toxicities measured in this study. We did not find significant differences between the groups regarding toxicity grades, this is displayed in Figure 2. Toxicities Under Docetaxel Stratified by Pemetrexed and Paclitaxel. Toxicity Grades Under Docetaxel Stratified by Pemetrexed and Paclitaxel.

Multivariate Logistic Regression
The multivariate analysis of having a grade 3 or 4 toxicity revealed no significant association with pemetrexed or paclitaxel. However, if patients received pemetrexed or paclitaxel in combination with platinum therapy alone vs ICI therapy as add-on this was associated with a lower risk for a grade 3 or 4 toxicity (OR = 0.10, P = 0.01). Additionally, if docetaxel was given as second-line vs beyond second line this was associated with a lower risk as well (OR = 0.15, P = 0.03).
Association of Overall and Progression-Free Survival and First-Line Therapy
Overall survival since the start of first-line therapy was significantly better with pemetrexed compared to paclitaxel (P = 0.0039), as showed in Figure 3. In the pemetrexed group we observed a longer median OS of 6.8 months (95% CI, 3.1-10.3) compared to 2.1 months in the paclitaxel group (95% CI, 0.1-15.0). Patients with an ECOG performance status of 2, as opposed to ECOG 0, experienced poorer survival (HR = 4.04, P < 0.0001) in our analysis. Kaplan-Meier Curves of Overall Survival Stratified by Pemetrexed and Paclitaxel.
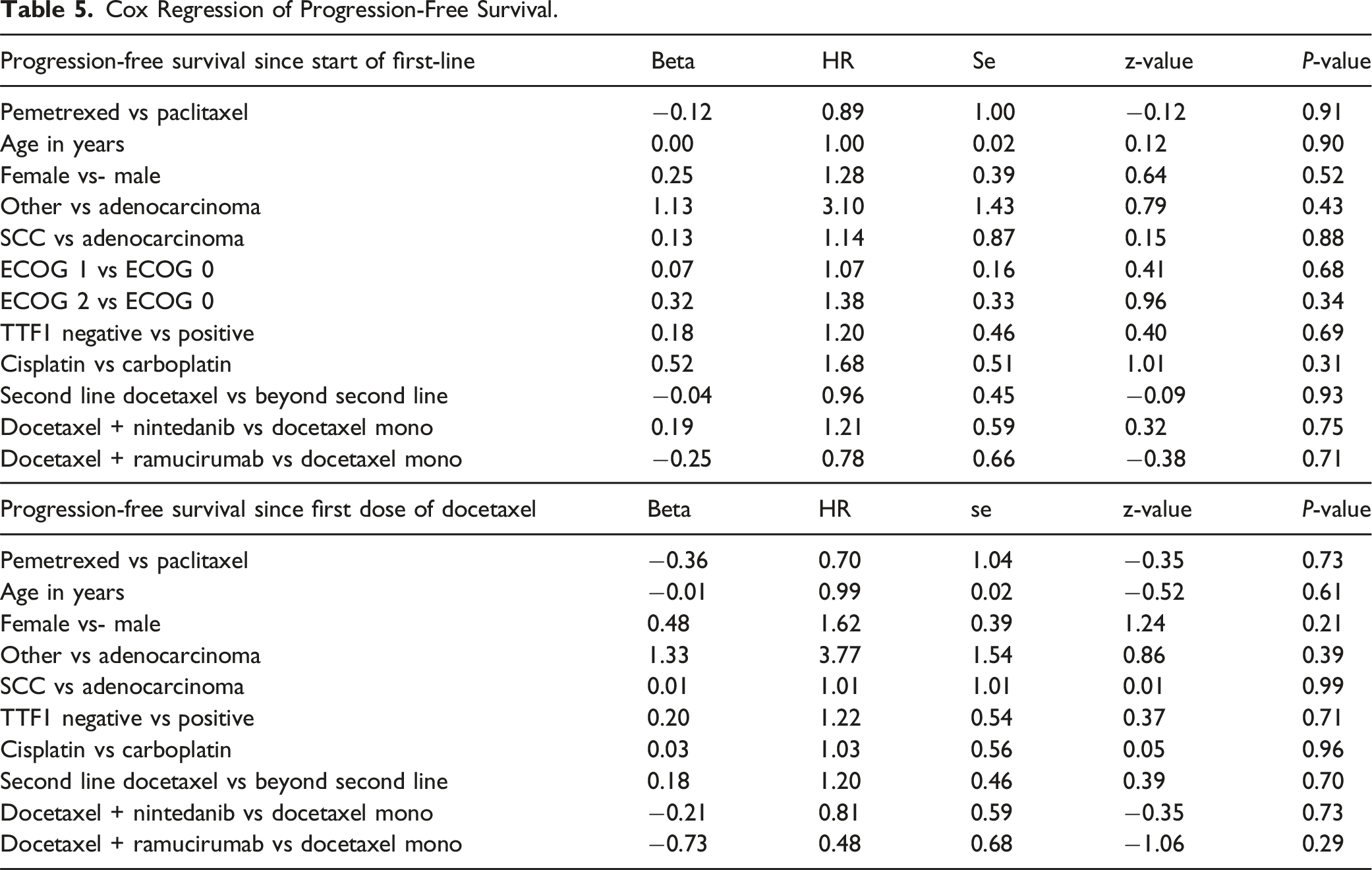
Progression-free survival (PFS) since first line therapy, as well as since first dose of docetaxel, did not differ significantly between the two groups. In patients who received pemetrexed we observed a longer median PFS (4.1 months; 95% CI, 2.1-5.2) compared to the patients who received paclitaxel (1.9 months; 95% CI, 1.0-14.9). Kaplan-Meier curves of progression-free survival are displayed in Figure 4. Kaplan-Meier Curves of Progression-Free Survival Stratified by Pemetrexed and Paclitaxel.
Cox Regression of Overall Survival.

Cox Regression of Progression-Free Survival.
Discussion
In this retrospective single center-analysis, we sought to analyze the efficacy and safety of docetaxel after a previous treatment with either paclitaxel or pemetrexed -combination in advanced NSCLC patients. Despite the use of novel agents in the first-line therapeutic setting, such as PD-1/PDL1 inhibitors for non-oncogene addicted NSCLC. Unfortunately, survival outcomes remain poor and patients often experience substantial toxicity, underscoring a high unmet need. 14 Additionally, several novel immunotherapeutic agents and new combination of ICIs as well as antibody drug conjugates (ADCs) are being investigated in second-line NSCLC patients with promising data.15-19 Based on available data, these novel therapies and particularly ADCs will soon replace chemotherapy as second-line standard of care for NSCLC.20-22
Currently, the standard second-line treatment consists primarily of systemic cytotoxic therapies, which typically yield poor outcomes. Recently, several novel therapeutic strategies have emerged that may improve patient outcomes. Docetaxel is presently considered the standard of care, alone or in combination with anti- angiogenic agents such as nintedanib or ramucirumab.9,10 In the REVEL study, a phase III study comparing docetaxel plus ramucirumab vs docetaxel in patients with stage IV NSCLC who had disease progression during or after prior first-line platinum- based chemotherapy, docetaxel plus ramucirumab significantly prolonged overall survival (OS) compared to docetaxel alone. 9
The phase 3 LUME-Lung 1 study assessed the efficacy and safety of docetaxel plus nintedanib as second-line therapy for non-small-cell lung cancer (NSCLC). THE data from the LUME- Lung 1 study evaluating docetaxel plus nintedanib in the adenocarcinoma histology subgroup demonstrated an ORR of 4.7% and a disease control rate (DCR) of 60.2%, a median progression-free survival (PFS) of 4.4 months, and a median OS of 12.6 months. 10
These studies were carried out before approval of immunotherapy or chemoimmunotherapy in first line. In the clinical practice we usually use docetaxel alone or in combination with either ramucirumab or nintedanib following a previous combination of chemotherapy with either platinum-paclitaxel or platinum-pemetrexed based on histology in addition to ICIs. Most of the patients do not benefit long from the therapy, and even more patients experience side effects which impact their quality of life.
To the best of our knowledge there has been no other study to investigate the efficacy or the safety of docetaxel following a previous pemetrexed or paclitaxel-based combination either alone or with ICIs. Therefore, we conducted this retrospective analysis in order to investigate the safety and efficacy of docetaxel after previous pemetrexed compared to paclitaxel-based chemotherapy alone or in combination with ICI.
We found that all the patients receiving docetaxel after pemetrexed experience toxicity under docetaxel, with myelosuppression (anemia and neutropenia) being the most common adverse effect. All the patients included in our analysis received paclitaxel in combination with carboplatin before docetaxel. In the pemetrexed group half of the patients received pemetrexed in combination with cisplatin and half with carboplatin.
We did not observe in our analysis a statistical different best response between the two groups, even though the rates of partial response to treatment with docetaxel following pemetrexed compared to paclitaxel were higher. In the paclitaxel group we observed higher proportion of stable or progressive disease compared to the pemetrexed group.
In the group of patients receiving docetaxel after paclitaxel we observed toxicities under docetaxel in 81.8% of the patients. We expected more toxicities in this group considering the fact that both, paclitaxel and docetaxel, belong to the taxane class of cytotoxic agents. They share major parts of their structures and mechanism of action, but differ in several other aspects. We found out that increased GGT, followed by anemia and hyponatremia were the more frequent side effects of docetaxel following a previuos therapy with paclitaxel.
Our findings indicated that patients who received docetaxel after a regime with pemetrexed compared to paclitaxel experienced a longer overall survival since the start of the first line treatment as well as survival since the start of first dose docetaxel.
Progression free survival did not statistically differ between the two groups, however a longer mPFS was observed in the pemetrexed group compared to the docetaxel group.
The observed differences in overall survival and progression-free survival between the Pemetrexed and Paclitaxel groups may primarily be attributed to the underlying histological subtypes of non-small cell lung cancer such as squamous vs non-squamous, rather than being solely related to the effect of Docetaxel.
Our study harbors some limitations. Firstly, the small sample size of the cohort and the retrospective nature of the study. Secondly, we only included patients who received docetaxel after either pemetrexed or paclitaxel and we did not include in our study comorbidities that could have influenced the toxicity reported on docetaxel. However, as we included all patients treated with docetaxel after a previous treatment with either a paclitaxel or a pemetrexed-combination, we analyzed a true real-world cohort and our results may help to guide treatment decisions in second-line in order to identify patients who are more likely to benefit from docetaxel. Furthermore, given the low efficacy of the docetaxel in combination with high toxicity, patients should be considered to be enrolled in clinical trial.
Conclusion
Docetaxel has been the foundation of second-line treatment for metastatic NSCLC for over two decades, however it is associated with modest clinical benefit at the cost of substantial toxicity.
The analysis of our cohort revealed a manageable safety profile in patients who have progressed on previous paclitaxel-or pemetrexed-combination treatment. We consider that personalized combination strategies that are developed according to the pathways or hallmarks that specifically drive each patient’s tumor biology will remain the main challenge. Consequently, we believe that reconsidering tumor mutational status with tissue re-biopsy or liquid biopsy after immunotherapy failure could represent an intriguing approach.
ORCID iDs
Blerina Resuli https://orcid.org/0000-0003-2024-2771
Maria Arredondo Lasso https://orcid.org/0000-0003-0191-8887
Statements and Declarations
Footnotes
Author Contributions
Conceptualization, B.R. and A.T.; methodology, B.R. J.W. and M.A.; investigation, B.R. J.W.; data curation; B.R. and J.W.; writing-original draft preparation B.R. and J.W.; writing-review and editing; B.R., J.W., D.K.G., P.M. and A.T.; visualization, B.R., J.W., D.K.G., P.M., M.A., J.B., P.A., Ch.S. and A.T., supervision, A.T. All authors have read and agreed to the published version of the manuscript.
Ethical Statement
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors state to share the original data on reasonable request.
