Abstract
Tumor immune escape is a major challenge in cancer treatment, and targeted immune escape therapy has become a key strategy for cancer treatment. As an important deubiquitinating enzyme, ubiquitin-specific protease 10 (USP10) participates in the process of tumor development by adjusting the balance between ubiquitination and deubiquitination of substrate proteins. Recently, USP10 has been shown to be closely related to tumor immune escape, where it serves to reduce the immunogenicity of tumor cells by stabilizing immune checkpoints and promotes tumor immune escape. In this review, we focus on the structural and functional characteristics of USP10 and elaborate on the biological function of USP10 in the occurrence and development of tumors, as well as its role in immune escape, including the regulation of immune checkpoints and the effect on immune cells in the immune microenvironment. It is possible to improve the efficacy of traditional cancer therapies by appropriately regulating the expression of USP10. The aim of this review is to provide a reference for further understanding the mechanism of tumor immune escape and the development of new tumor treatment methods.
Background
Ubiquitination is a reversible process in which deubiquitinating enzymes (DUBs) specifically hydrolyze small ubiquitin molecules from target proteins or precursor proteins by hydrolyzing peptide bonds, isopeptide bonds, or ester bonds at the carboxy-terminal of ubiquitin to protect target proteins from ubiquitin–proteasome degradation.1,2 Ubiquitin-specific proteases are the largest subfamily of DUBs, comprising approximately 56 members; these enzymes are characterized by a catalytic core that is associated with ubiquitin binding (Figure 1).3,4 USPs are involved in various intracellular life activities and are also closely related to human diseases, such as tumors and inflammation.5,6 USP10, a member of the USP family, is a cysteine protease that is widely distributed in human tissues and has enormous significance in diverse cellular processes and various human diseases.
7
Studies have shown that USP10 acts as an anti-stress molecule in oxidative stress, viral infection, heat shock, and other stressful environments.8,9 In terms of metabolism, USP10 regulates lipid formation, lactic acid production, and glycolysis gene expression.
10
Schematic representation of the domain architectures of human USPs. The red arrow points to the specific structural composition of USP10
Tumor immune escape is a process by which tumor cells evade the recognition and attack of the immune system through a variety of mechanisms, which enables tumors to continue to grow and metastasize in the body. The mechanism of tumor immune escape is complex, involving changes in the characteristics of tumor cells, immunosuppression of the tumor microenvironment, and dysfunction of the host immune system. 11 Among the many molecular mechanisms that affect tumor immune escape, the role of DUBs has gradually attracted attention.12,13 As an important member of the DUB family, USP10 plays a key role in the process of tumor immune escape by deubiquitinating substrate proteins to regulate their stability, localization, and function.14,15 An in-depth study of the mechanism of USP10 in tumor immune escape is crucial to elucidate the pathogenesis of tumors and develop new treatment strategies.
Biological Function of USP10
Role and Biological Function of USP10 in Various Types of Cancer

The Role of USP10 in Cell Proliferation and Apoptosis
As an important tumor suppressor protein, p53 inhibits cell proliferation and tumor formation by promoting cell apoptosis.58,59 In normal cells, p53 is ubiquitinated by interaction with the E3 ligase murine double minute 2 in the nucleus, then exported from the nucleus to the cytoplasm, and finally degraded by the proteasome, thereby promoting apoptosis.56,60–62 Continuous degradation keeps the p53 protein at a low level.
Studies have shown that USP10 deubiquitinates and stabilizes p53 in the cytoplasm, enhances the function of p53, and enables p53 to re-enter the nucleus.
63
In the case of DNA damage and ataxia-telangiectasia mutant (ATM) activation, USP10 is phosphorylated and accumulates in the nucleus.
56
Moreover, USP10 knockout has been shown to inhibit colon cancer cell apoptosis by promoting wild-type p53 degradation.
56
In addition, previous research has shown that USP10 knockout in HCT116 cells with wild-type p53 increased their proliferation, but USP10 knockout in HCT116 cells with p53 double deletion had no effect on their proliferation.
56
USP10 has been found to promote cell proliferation in p53 mutant cells, but the specific molecular mechanism remains unclear.
64
Another study revealed that USP10 regulated Musashi-2 (MSI2) stability via deubiquitination and promoted tumor proliferation in colon cancer.
32
These results demonstrate that USP10 stabilized the protein level of p53 in cells by deubiquitination of p53, thus enhancing the anticancer effect of wild-type p53 on cells and achieving the effect of inhibiting cell proliferation and apoptosis (Figure 2A).
56
Currently, studies on the effect of USP10 on tumor cell proliferation and apoptosis mostly focus on the p53 protein pathway, which may also be related to the tumor suppressor effect of the p53 protein itself. The biological function of USP10. Some mechanistic pathways of USP10 in proliferation and apoptosis (A), cell cycle (B), autophagy (C), and DNA damage repair (D) pathways
The Role of USP10 in Cell Cycle
As a member of the histone deacetylase family, sirtuin-6 (SIRT6) is a tumor suppressor protein encoded by genes localized to telomeres in human cells. SIRT6 controls cellular senescence and telomere structure by deacetylating histone H3 lysine 9 (H3K9). 65 Through proteomic analysis of SIRT6, some scholars found that USP10 deubiquitinated and stabilized SIRT6. 66 In addition, the overexpression of USP10 or SIRT6 significantly reduced the G2/M and S phases and increased the G0/G1 phase of HCT116 colon cancer cells; the overexpression of USP10 and SIRT6 had a more significant effect on the cell cycle. 67 The knockout of USP10 or SIRT6 promoted cell cycle progression by increasing S and G2/M phases, and the effect of the knockout of USP10 and SIRT6 was more obvious. 67 The aforementioned information suggested that USP10 and SIRT6 synergistically regulated the cell cycle of colon cancer cells. 67 Because SIRT6 performed its anticancer function by inhibiting the transcriptional activity of c-Myc, it regulated tumor cell cycle progression by regulating the expression levels of some key genes.34,68 Subsequent experiments found that the overexpression of SIRT6 or p53 partially restored the interference of USP10 on the cell cycle, while the co-expression of p53 and SIRT6 completely restored the interference. Therefore, the researchers concluded that USP10 inhibited the cell cycle process by enhancing the SIRT6-and p53-mediated inhibition of c-Myc. 37
A recent study showed that USP10 interacted with CCND1 and prevented the polymerization of its K48- but not K63-connections in glioblastoma U251 and HS683 cells, thereby increasing the stability of CCND1. 40 In line with the effect of USP10 on CCND1, the USP10 knockdown of CCND1 resulted in glioblastoma multiforme (GBM) cell cycle arrest in the G1 phase and induced GBM cell apoptosis (Figure 2B). 40 Despite its role as a double-edged sword in tumors, USP10 is widely recognized as an oncogene in several settings, driving the malignant progression of tumors by driving cell cycle progression.
The Role of USP10 in Autophagy
Autophagy is a basic metabolic mechanism that degrades unnecessary or abnormal cellular components through lysosomes, before recycling through autophagy. 69 Vacuolar sorting protein 34 (Vps34) is an essential component of the PI3 kinase (PI3K) family and the only known PI3K class. Vps34 exists in mammalian cells in the form of complexes, including Vps complex 1 and Vps complex, and plays a central role in autophagosome formation and maturation.70,71 Both complexes share a core component, Beclin1, which is an essential molecule in the formation of autophagosomes and can mediate the localization of other autophagic proteins to phagophores.72–74 USP10 and USP13 regulate Vps34 complex–mediated autophagy through Beclin1 deubiquitination. 75 However, some experiments have proven that Beclin1 interacts directly with USP13 and only indirectly or instantaneously with USP10. 75 Moreover, downregulation of the Vps34 complex or Beclin1 reduced endogenous levels of USP10 and USP13. 75 Further experiments revealed that USP13 deubiquitinated USP10, suggesting that Beclin1 and Vps34 also regulate USP10 activity by interacting with USP13, thus affecting autophagy. 75 Furthermore, researchers have found that USP10 alleviated hepatic steatosis in patients with nonalcoholic steatohepatitis in an autophagy-dependent manner (Figure 2C). 49 In general, USP10 interferes with the autophagy process of tumor cells mainly by regulating the autophagy initiation complex, stabilizing key proteins of autophagy nucleation, and interfering with autophagosome–lysosome fusion, thereby affecting the progression of tumor cells.
The Role of USP10 in DNA Damage Repair
The external environment and internal factors often lead to DNA molecular damage. After damage, certain mechanisms can restore DNA structure and function or improve the survival rate of damaged cells. 76 In normal cells, USP10 is mostly located in the cytoplasm, where it deubiquitinates p53, which allows p53 to re-enter the nucleus. 77 However, after DNA damage, USP10 is phosphorylated by activated ATM and transferred from the cytoplasm to accumulate in the nucleus, thereby stabilizing p53 in the nucleus. 56 Therefore, USP10 is a necessary factor for p53 to participate in DNA damage repair. 56 Moreover, USP10 interacts with MutS and deubiquitinates and stabilizes MutS homologous 2 to regulate cell sensitivity to DNA damage (Figure 2D). 54 The genomic instability caused by DNA damage repair defects promotes the development of cancer and causes adverse outcomes such as tumor drug resistance. USP10 mediates tumor DNA damage repair through ubiquitination regulation and promotes tumor chemotherapy resistance, which provides a new idea for targeted tumor therapy.
USP10 and Tumor Immune Escape Mechanism
USP10 in Cancer Types
As a specific deubiquitination enzyme, USP10 plays a vital role in cell life activities, with its role in cancer attracting increasing attention in recent years. USP10 is involved in the occurrence and progression of various cancer types and is a potential molecular marker for cancer diagnosis and prognosis prediction (Figure 3). Next, we summarize the specific role and mechanistic pathway of USP10 in some cancers. Role and function of USP10 in various cancers
In addition, USP10 inhibits hepatic steatosis, insulin resistance, and inflammation through SIRT6.
66
In the context of DNA damage, USP10 inhibits the activation of nuclear factor kappa B (NF-κB) and the production of inflammatory cytokines.
78
In addition, as a DUB, USP10 has a crucial role in the development and progression of tumors. For example, USP10 inhibits the growth of cancer cells and tumor development by inhibiting the ubiquitination and protein degradation of p53 and SIRT6.37,56 USP10 regulates the stability of KLF transcription factor 4 (KLF4) and suppresses lung tumorigenesis.
79
The association of USP10 with GTPase-activating protein (SH3 domain)-binding protein 2 inhibits p53 signaling and contributes to poor outcome in prostate cancer.
75
USP10 regulates MSI2 stability via deubiquitination and promotes tumor proliferation in colon cancer.
32
Herein, we evaluated USP10 expression in patients with different types of cancer according to the UALCAN Cancer Database. As shown in Figure 4A, esophageal carcinoma (ESCA) and liver hepatocellular carcinoma showed the highest and lowest USP10 expression, respectively. Furthermore, most cancerous tissues exhibit higher expression levels of USP10 than normal tissues. Among them, cholangiocarcinoma, ESCA, GBM, and pancreatic adenocarcinoma differ significantly from normal tissues (P < .05), suggesting that USP10 may be involved in the development of these tumors and that its inhibition may show better efficacy. The expression level of USP10 in DLBC, LAML, and PCPG cells is lower than that in normal tissues, which might be associated with the tumor-suppressor function of USP10 in these tumor types. For these tumors, up-regulating the expression of USP10 may lead to a better prognosis. (Figure 4B). Based on the GEPIA database Survival Analysis, USP10 expression in breast cancer (BRCA) and head and neck squamous cell carcinoma (HNSC) shows an inverse proportional relationship with the overall survival rate. In contrast, in kidney renal clear cell carcinoma (KIRC), USP10 expression is directly proportional to the overall survival rate, and these differences are statistically significant (P < .05) (Figure 4C). Whether USP10 plays a pro- or anti-tumor role is related to its expression level and downstream substrates in different tumors, which needs to be personalized designed in the treatment of tumors. The relationship of USP10 with tumors by bioinformatics. (A) The expression level of USP10 in different cancer types. (B) Comparison of USP10 expression between tumor and normal tissues. Blue columns stand for normal tissues. Red columns stand for cancerous tissues. (C) The relationship between USP10 and the overall survival rate of several types of tumors; BRCA, breast cancer; HNSC, head and neck squamous cell carcinoma; KIRC, kidney renal clear cell carcinoma; The data is sourced from the Gene Expression Profiling Interactive Analysis (GEPIA) database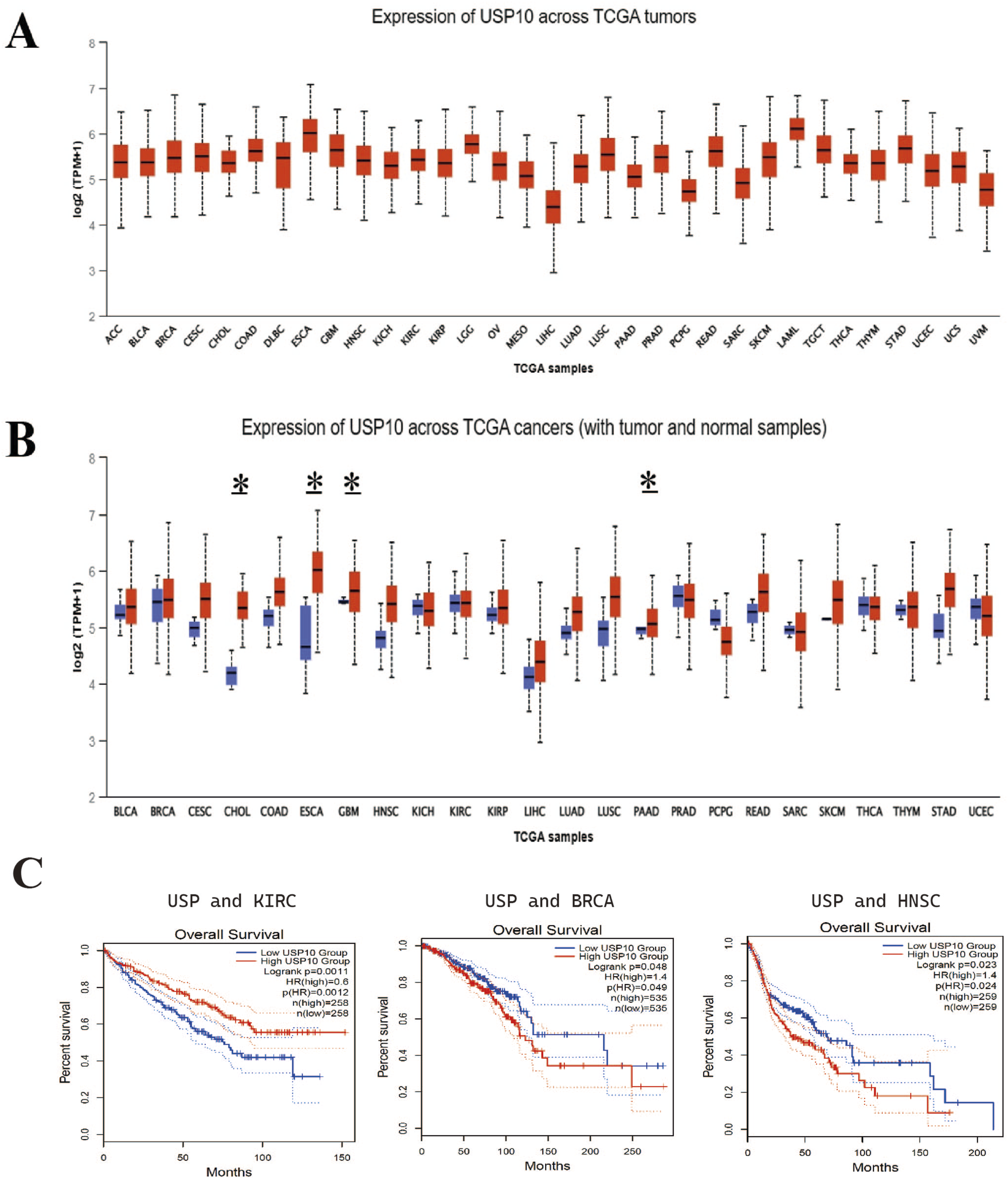
Immune Checkpoint Proteins and USP10
Immune checkpoint proteins play a critical role in tumor immune escape, and USP10 has been found to regulate the expression and stability of multiple immune checkpoint proteins.
Regulation of PD-L1
In tumor cells, USP10 can interact with programmed death ligand 1 (PD-L1) to inhibit its degradation through deubiquitination, thereby upregulating its expression level. In prostate cancer studies, NDR1 hinders the ubiquitination and degradation of PD-L1 by increasing the interaction between USP10 and PD-L1, resulting in the abundant expression of PD-L1 on the surface of tumor cells (Figure 5). After binding to the programmed death receptor 1 (PD-1) on the surface of T cells, PD-L1 inhibits the activation and proliferation of T cells and promotes immune escape of prostate cancer cells.
14
In a study on esophageal cancer (EC), FOXP4-AS1 was found to stabilize PD-L1 through USP10, reduce PD-L1 ubiquitination, and promote immune escape of EC cells by hindering the function of CD8 + cytotoxic T lymphocytes (CTLs).
15
USP10 mainly maintains the protein stability of PD-L1 through deubiquitination, ultimately promoting tumor immune escape and influencing the response to immunotherapy. However, the specific molecular mechanism still needs to be further clarified, providing a theoretical basis for improving the efficacy of immunotherapy. The mutual regulatory relationship between USP10 and PD-1/PD-L1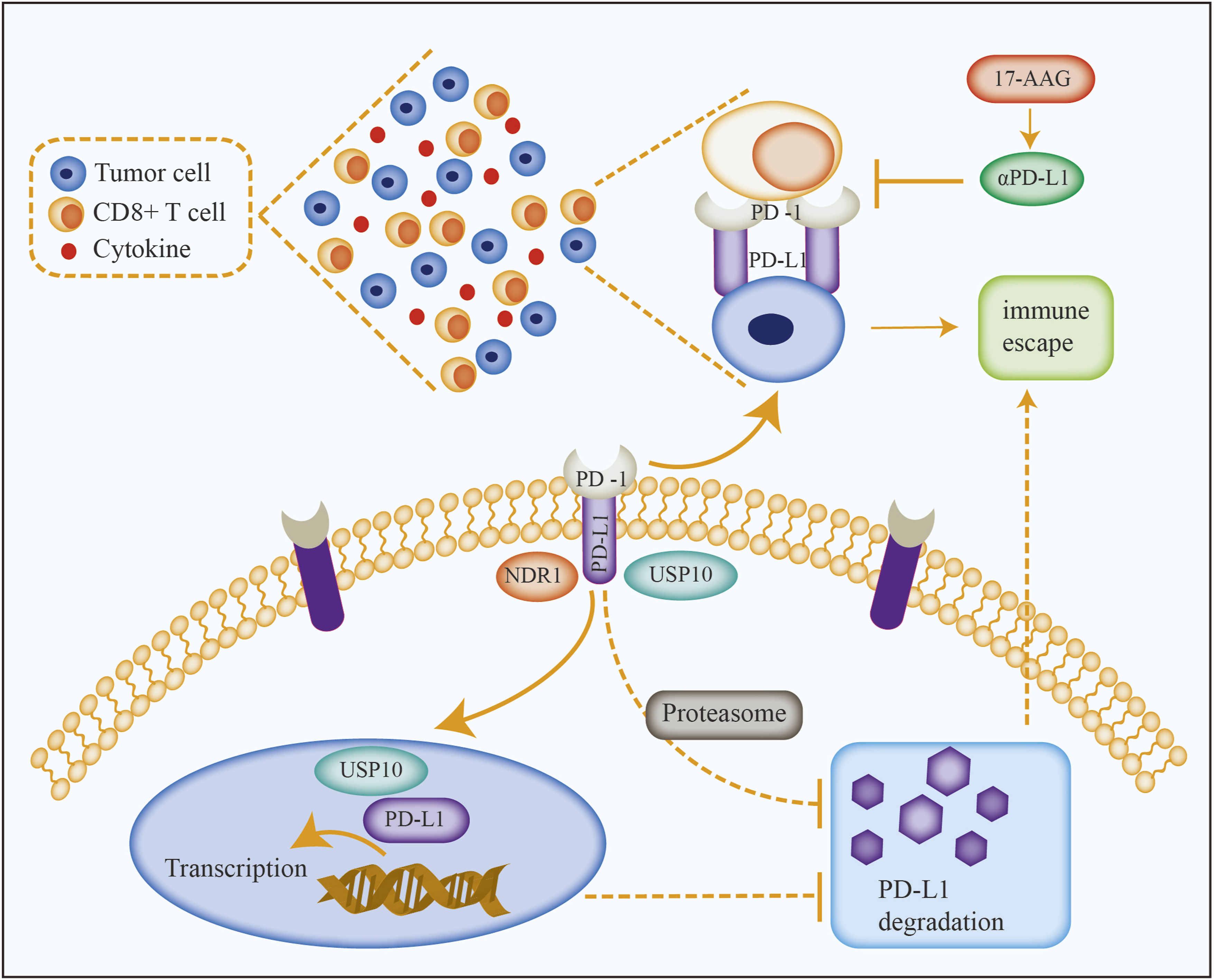
Regulation of B7-H4
B7-H4, a coinhibitory B7 family ligand, is a key immune checkpoint that inhibits CD8 T cell activity. B7-H4 mRNA can be detected in multiple normal tissues, but protein levels are barely detectable in normal tissues; the inconsistency between B7-H4 mRNA and protein levels suggests that B7-H4 undergoes post-transcriptional modification. Studies have shown that B7-H4 can be glycosylated or ubiquitinated. 80 Zeng et al discovered that in triple-negative breast cancer, USP10 acts as a critical DUB to suppress tumor immunogenicity by inhibiting autocrine motor factor receptor-mediated B7-H4 stabilization, which in turn mediates tumor immune escape by stabilizing protein expression. Inhibition of USP10 expression can reduce the stability of B7-H4, increase tumor immunogenicity, and enhance the ability of immune cells to recognize and kill tumor cells. 81 Currently, there are relatively few studies on USP10 and B7-H4. The immunotherapy for cold tumors based on these two factors has only been verified in breast cancer research. Further studies are needed to explore how they can overcome immune suppression and treat drug resistance.
Effect of USP10 on Immune Cell Function in the Tumor Microenvironment
The tumor microenvironment is a complex ecosystem of non-malignant components surrounding a tumor cell, including immune cells, stromal cells, and the factors produced and released by them. 82 Their functional status is crucial for tumor immune escape, and USP10 plays a regulatory role in it.
Effects on T Cell Function
T cells are involved in coordinating multiple aspects of adaptive immunity throughout life, including immune responses, homeostasis, and the establishment and maintenance of memory. 83 USP10 indirectly affects the function of T cells through the expression of immune checkpoint proteins. USP10 can stabilize immune checkpoint proteins such as PD-L1, prevent their degradation, and promote the exhaustion of CD8 + CTLs while reducing their killing activity against tumor cells. 15 USP10 may also directly affect T cell differentiation and function through other pathways. Indeed, in some tumor models, aberrant expression of USP10 has been found to lead to an imbalance in T cell subsets, an increase in the proportion of regulatory T cells, and a decrease in the proportion of effector T cells with antitumor activity, thereby promoting tumor immune escape.
Effect on Macrophage Function
Macrophages are the main type of immune cells and play a crucial role in the tumor microenvironment. Macrophages mainly polarize into either the classically activated “M1” type, which promotes antitumor immune responses, or the selectively activated “M2” phenotype, which leads to immunosuppression and tumor development. 84 The expression level of USP10 has been shown to correlate with the immunosuppressive M2 polarization of macrophages. Studies in colorectal cancer (CRC) have revealed that USP10 can interact with NLRP7 and catalyze its deubiquitination in CRC cells, which stabilizes NLRP7 and increases its protein level. NLRP3 promotes the secretion of CCL2 by activating the NF-κB signaling pathway, which further promotes the polarization of M2-like tumor macrophages. 26 In addition, several studies have shown that USP10 inhibits YAP1 ubiquitination and degradation, enhances Cyr61 expression, and enhances M2 macrophage polarization in the tumor microenvironment, leading to immune escape and malignant development of pancreatic cancer cells. Knockdown of USP10 has been shown to increase the ubiquitination and degradation of YAP1 in pancreatic cancer cells, increase the proportion of M1-polarized macrophages, and decrease the proportion of M2-polarized macrophages. 84 USP10, in different tumors or diseases, exerts its deubiquitination-stabilizing protein function by acting on different protein substrates. USP10 promotes the transformation of macrophages into M2-like cells and plays various roles, including facilitating tumor progression and alleviating disease.
Effect on Other Immune Cell Function
USP10 has also been discovered to exert an influence on other immune cells. For example, USP10 transcriptionally upregulates PD-L1 and galectin-9 (Gal-9) by deubiquitinating YAP1, desensitizing pancreatic ductal adenocarcinoma (PDAC) cells to NK cell-mediated cytotoxicity. 85 The high expression of USP10 is also positively associated with neutrophils, dendritic cells, macrophages, etc. 86 Currently, research on the impact of USP10 the functions of immune cells primarily focus on T cells and macrophages. Further in-depth studies on the effects of USP10 on other immune cells are needed to enhance the theoretical credibility.
The Current Status of Tumor Treatment Targeting USP10
In view of the key role of USP10 in tumor immune escape, targeting USP10 has become a popular research direction in tumor treatment. USP10 is overexpressed in various tumors and is closely related to a poor prognosis. Functionally, USP10, as an oncogene, can increase the proliferation and metastasis of tumor cells both in vivo and in vitro. 87 Some small-molecule inhibitors targeting USP10 have been developed. These inhibitors can specifically inhibit the deubiquitination enzyme activity of USP10 and block its regulatory effect on substrate proteins. USP10 is overexpressed in osteosarcoma and significantly induces autophagy, cell proliferation, and invasion by enhancing ULK1 expression. Treatment with the USP10 inhibitor Spautin-1 inhibits the proliferation of osteosarcoma cells and increases their sensitivity to the chemotherapy drug cisplatin. Studies in an animal xenograft tumor model also revealed that using Spautin-1 to inhibit USP10 can slow tumor growth. 88 However, many USP10 inhibitors are still in the research stage and need further optimization and improvement in terms of drug specificity, efficacy, and safety.
Therapeutic Potential of USP10 Inhibitors
Summary of Inhibitors Targeting USP10

Spautin-1
Specific and potent autophagy interactivator-1 (spautin-1) is the most commonly used small-molecule inhibitor of USP10, which has a strong inhibitory effect on autophagy. Previous research has shown that spautin-1 regulates osteoclast formation by inhibiting autophagy induced by receptor activator for NF-kB and 1α,25-(OH)2D3. 96 Furthermore, spautin-1 has been shown to alleviate acute pancreatitis in animal models established using the combination of frog skin peptide and lipopolysaccharide by inhibiting pancreatic cell autophagy. 97 In tumors, spautin-1 enhances the sensitivity of osteosarcoma and chronic myeloid leukemia to chemotherapeutic agents by inhibiting autophagy. Spautin-1 also decreases cell survival and colony formation in osteosarcoma cells.98,99 Most importantly, spautin-1 inhibits autophagy by interacting with USP10 and USP13 through the inhibition of Beclin-1 protein expression levels. Spautin-1 suppresses melanoma growth via ROS-mediated DNA damage and exhibits synergy with cisplatin.100,101 Currently, there have been no reports on the efficacy of spautin-1 in vivo animal studies; thus, spautin-1 is still in preclinical development.
HBX19818
HBX19818 is another USP10 inhibitor. Originally thought to be an inhibitor of USP7, HBX19818 targets USP7 and reduces UV-induced phosphorylation of Chk1 and the levels of the checkpoint mediator Claspin. 102 Later, Yang et al found that HBX19818 leads to the dual degradation of spleen tyrosine kinase and FMS-like tyrosine kinase 3 (FLT3), resulting in the death of acute myeloid leukemia cells. 103
Wu-5
Wu-5 is a little-known USP10 inhibitor. The kinase FLT3 internal tandem duplication (FLT3-ITD) is known to be associated with adverse clinical outcomes in AML. 104 Wu-5 has been found to induce synergistic cell death in FLT3-ITD-positive cells by reducing FLT3 and AMP-activated protein kinase proteins. 45 However, further in-depth research is still needed to explore the specific efficacy and clinical outcomes of Wu-5 in the treatment of human cancers.
LY-2
Dong et al developed the small molecular inhibitor LY-2 via structural optimization of the previously identified USP10 inhibitor D2. By substituting the 1,2,4-oxadiazole moiety with an amide bond linkage, LY-2 exhibited substantially enhanced binding affinity for USP10 compared to its lead compound D2. Comprehensive structure-activity relationship (SAR) analyses further validated its potential as a promising therapeutic candidate. Additionally, LY-2 is the first USP10 inhibitor to achieve nanomolar binding affinity. In liver cancer, LY-2 effectively inhibits the proteasomal degradation of downstream proteins YAP and p53 mediated by USP10, thereby leading to the subsequent downregulation of CDK4 in the p53 signaling pathway. 92 However, LY-2 has currently only been validated at the cellular level, and its activity in vivo animal models as well as clinical settings remains to be further confirmed.
F806
F806 is a natural macrolide compound isolated from the Chinese actinomycete strain FIM-04-806. It selectively targets the deubiquitinase USP10 and suppresses its catalytic activity, which disrupts the homeostasis of the USP10-Cdh1-ANLN complex, thereby exerting a potent inhibitory effect on cell cycle progression in esophageal squamous cell carcinoma (ESCC) cells. 93 In addition, F806 has been shown to induce ESCC cell apoptosis through blocking the activation of integrin β1. 105 Furthermore, it exerts anti-ESCC activity by facilitating the internalization of glucose transporter 1 and its subsequent degradation within autolysosomes. 93
Other Inhibitors
Notably, several natural products and their derivatives also exert regulatory effects on USP10 activity. For example, curcumin induces mitochondrial expansion in breast cancer cells through targeting USP10. Specifically, silencing or inhibiting USP10 expression effectively abrogates curcumin-induced upregulation of polyubiquitinated proteins and CHOP, attenuates curcumin-mediated activation of ERK and JNK signaling pathways, and diminishes mitochondrial peroxide production, ultimately mitigating curcumin induced breast cancer cell death in a marked manner. 94 Quercetin also exerts a distinct inhibitory effect on USP10. In the context of asthma, USP10 interacts with the transcription factor T-bet and sustains its structural stability via deubiquitination, thereby protecting T-bet from proteasomal degradation. As a key regulator of adaptive immunity, T-bet orchestrates Th1 immune responses by upregulating the expression of Th1-signature cytokines. Importantly, quercetin suppresses USP10 activity to abrogate T-bet upregulation, consequently mitigating asthmatic inflammation. Collectively, quercetin has been proven to be a potential anti-inflammatory drug in autoimmune diseases. 95 However, currently the inhibitors of USP10 are only used in preclinical experiments, and there is still a long way to go before they can make a substantive contribution to human diseases.
To date, no new USP10 inhibitors have been reported. With the development of high-throughput proteomics technology, it is possible to screen specific substrates and specific deubiquitination sites of USP10 through various research methods and develop new specific inhibitors, providing new drugs for targeted therapy of tumors.
Conclusion and Prospect
As a member of the ubiquitin-specific protease family, USP10 affects the degradation and stability of substrate proteins by removing the substrate ubiquitin and participates in the whole process of tumor development, 93 including regulating immune checkpoint proteins, regulating the infiltration of specific immune cell subsets, and affecting cell migration and invasion. Thus, USP10 can be used as a potential marker for tumor prognosis. Currently, research on USP10 in tumor immune escape has made some progress, but there remain many problems to be solved. In terms of basic research, it is still necessary to further clarify whether USP10 affects tumor immunity and progression through other pathways, and whether there are other unknown substrate proteins. In terms of clinical application, how to safely and effectively transfer USP10 targeted therapy strategies to cancer treatment remains a major challenge. In the future, it will be important to elucidate the mechanism of USP10 in tumors and conduct more clinical trials to evaluate its efficacy and safety in various tumors and patient populations. Furthermore, future research should explore the potential of a combined treatment approach involving USP10 modulation alongside chemotherapy, radiotherapy, targeted therapy, and immunotherapy. The ultimate goal is to create personalized treatment plans that consider the patient’s specific gene mutations and the unique characteristics of their immune microenvironment.
The Future Direction of Combination Therapy With USP10 Inhibitors
USP10 participates in crucial processes by regulating the stability of substrate proteins, including tumor proliferation, apoptosis, and microenvironment remodeling. Nevertheless, owing to its tumor-type-dependent bidirectionality, the monotherapy of USP10 inhibitors is restricted. Consequently, combination therapy has emerged as the core approach to overcome the bottleneck of USP10 monotherapy and enhance the anti-tumor efficacy. The following focuses on exploring the mechanism and development prospects of the combined application of USP10 inhibitors with immune checkpoint inhibitors and chemotherapy drugs. Research on the relationship between USP10 and immune checkpoint inhibitors has mainly focused on PD-L1 and B7-H4, and the specific mechanism have been comprehensively elaborated in the preceding content.14,81 The mechanisms underlying the combined application of USP10 inhibitors and chemotherapeutic drugs primarily encompass the following aspects. USP10 promotes tumor resistance to chemotherapy by facilitating DNA damage repair. Inhibiting USP10 can enhance the efficacy of oxaliplatin in the chemotherapy of colorectal cancer.100,101 USP10 can promote the migration and cisplatin resistance of esophageal squamous cell carcinoma (ESCC) through deubiquitination and stabilization of integrin β1/YAP. Deficiency of USP10 can significantly suppress the migratory ability and chemoresistance of ESCC cells. 106 In the treatment of thyroid cancer, USP10 plays a unique role. Overexpression of USP10 suppresses the invasion, migration, and epithelial-mesenchymal transition (EMT) characteristics of doxorubicin-resistant (FTC133-DOX) cells, promotes cell apoptosis, and enhances the chemosensitivity of FTC133 cells to DOX treatment. 107 Targeting USP10 can also generate a synergistic effect with anti-tumor drugs by amplifying the cytotoxicity of chemotherapy and optimizing the response to chemotherapy, thus overcoming drug resistance in tumors. In the future, more efforts should be made to focus on issues such as precise tumor classification and in-depth exploration of mechanisms, so as to promote the combination of USP10 inhibitors in clinical tumor treatment.
Potential Immunomodulatory Function of USP10: Potential Perspectives
Effective immunotherapy currently includes immune checkpoint blocking, chimeric antigen receptor-T cell, and natural killer cell therapy; however, patients with tumors still have low survival rates because most are diagnosed at an advanced stage due to non-specific symptoms and a lack of biomarkers, especially in the early stage of the disease. Thus, finding a valid target for tumors remains an urgent issue. 108 USP10 affects oncogenic and tumor-suppressor signals, tumor immunogenicity, and immune cells and is involved in pro-tumor responses. Thus, USP10 inhibitors have important implications for the development and application of both epigenetic therapies and cancer immunotherapies, and combinations thereof.
Challenges in USP10 Inhibitors Development for Tumors Therapy
There are currently only a few available USP10 inhibitors, and none of them have entered clinical studies. In addition, USP10 inhibitors in combination with other compounds may be an effective way to treat tumors. However, there is a lack of compound combination therapy and exploration of combination mechanisms. Of course, it is also critical to improve the subtype selectivity of compounds.
In this paper, we have discussed the biological function of USP10 and its key role in tumor progression. Many detailed molecular mechanisms of USP10 in the context of tumorigenesis and development remain unclear. We have also summarized the current knowledge of USP10 inhibitors in treating tumors. USP10 has attracted attention in research on epigenetic regulation of tumor development because of its involvement in the whole progression of many tumors. With the advances in methods such as structural biology and computer-aided drug design, we hope that this review will encourage and accelerate advances in the continuing search for novel USP10 inhibitors for tumor therapy. Additionally, the application of USP10 as a target for tumor diagnosis and targeted therapy in clinical practice is expected in the future.
Footnotes
Acknowledgments
Ethical Considerations
This manuscript is a review article and does not involve a research protocol requiring approval by the relevant institutional review board or ethics committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Henan Provincial Medical Science and Technology, Joint Construction Project (No LHGJ20220423).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
