Abstract
Among the three primary gynecological malignancies, ovarian cancer has the lowest incidence but the worst prognosis. Because of the poor prognosis of ovarian cancer patients treated with existing treatments, immunotherapy is emerging as a potentially ideal alternative to surgery, chemotherapy, and targeted therapy. Among immunotherapies, immune checkpoint inhibitors have been the most thoroughly studied, and many drugs have been successfully used in the clinic. CD47, a novel immune checkpoint, provides insights into ovarian cancer immunotherapy. This review highlights the mechanisms of tumor immune evasion via CD47-mediated inhibition of phagocytosis and provides a comprehensive insight into the progress of the relevant targeted agents in ovarian cancer.
Introduction
Ovarian cancer (OC) is one of the deadliest female malignancies, with the lowest incidence but the worst prognosis among all gynecological malignancies. According to clinical guidelines and expert consensus, cytoreductive surgery followed by platinum-based chemotherapy is the standard treatment for most patients with OC. 1 Related clinical symptoms are relieved under this standard treatment; however, OC is prone to recurrence and drug resistance, resulting in a five-year survival rate of less than 50%. 2 Despite significant progress in surgical techniques and drug therapies, the survival of patients with advanced OC has not improved. Therefore, there is an urgent need to get OC out of therapeutic dilemmas. 3
Growing evidence has demonstrated that malignancy is a heterogeneous disease with immunogenicity, and its occurrence, development, and metastasis rely on immune suppression. 4 Immunotherapy targeting immunogenicity is a hotspot of anti-tumor therapies and has been successfully used in clinics as a novel anti-tumor option after traditional treatments (e.g., surgery, radiotherapy, chemotherapy, and endocrine therapy). 5 Along with the discovery of immune checkpoints (ICs), immune checkpoint inhibitors (ICIs) have become the focus of research in tumor immunotherapy.6–8 ICIs restore self-clearing and monitor the function of the immune system by blocking inhibitory signals. Various ICIs have shown anti-tumor activity in preclinical models, and some have been successfully used in clinics.6–11 Programmed cell death protein-1 (PD-1) and its ligand (PD-L1) inhibitors have been the most successful and widely used ICIs. 12 Nevertheless, new ICIs are continuously being identified and developed.
The known ICIs primarily act on the adaptive immune system. However, only 10%-30% of patients with OC show long-term and durable responses, followed by acquired resistance, which remains a substantial dilemma.7,12–14 CD47 has been identified as the first innate IC, restoring macrophages phagocytosis by blocking the “don’t eat me” signal.15,16 CD47 is a promising therapeutic target to provide insights into new treatment options for patients with OC.
The Development of ICIs
Adaptive ICs
T cells mediated adaptive immune dominates in anti-tumor reaction.5,14 Both the primary signals produced by the interaction between the major histocompatibility complex and T cells receptor (TCR) and the secondary signals offered by the co-stimulatory molecules are necessary to elicit intact T cell responses. 14 Functioning as the assistant of T cell activation and proliferation, co-stimulatory molecules are also essential regulators of humoral immunity and cytokine production. 17 However, increasing evidence has shown that some members, such as cytotoxic T-lymphocyte-associated protein-4 (CTLA-4), PD-L1, PD-L2, PD-1 homolog, and B7-H3, also provide critical inhibitory secondary signals. 18 These inhibitory signals act as a “brake” to protect normal cells from the excessive T cells’ attack by attenuating T cell response.18,19 Conversely, multiple tumors suppress anti-tumor immune responses and evade immune attacks by overexpressing these molecules. 7
These brake-like molecules and related inhibitors have been dubbed as ICs and ICIs in cancer immunotherapy because of the satisfactory anti-tumor effects of targeting CTLA-4. 14,20,21 Mechanically, the increased anti-tumor effects appear to be the result of the simultaneous enhancement of effector T cell function and concurrent inhibition of Treg activity. In 2011, the FDA (the Food and Drug Administration) authorized the first ICI, Ipilimumab, a CTLA-4 monoclonal antibody (mAb), for the treatment of advanced melanoma. Notably, subsequent approvals of Ipilimumab for the treatment of other malignancies have involved its use with the PD-1/PD-L1 ICIs.22–24 PD-1/PD-L1 inhibitors are the most successful ICIs, which primarily act in the adaptive immune system to trigger tumor-specific T cells responses. 25 PD-L1 overexpresses on various human tumors and induces the phosphorylation ordephosphorylation process of intracellular signaling pathways when it binds to PD-1.26–29 These signaling cascades suppress T cell proliferation, activation, and cytokine production, inhibiting the anti-tumor immune responses and promoting tumor growth.9,13 Various anti-PD-1/PD-L1 mAbs have been shown to have effective anti-tumor activities in, for example, melanoma, lung cancer, and Hodgkin’s lymphoma (HL).29–32 Multifarious PD-1/PD-L1 inhibitors, for example, Pembrolizumab, Atezolizumab, and Nivolumab, have been approved worldwide for various malignancies.33,34
Despite their proven efficacy, only 10%-30% of patients with solid tumors receiving PD-1/PD-L1 or CTLA-4 ICIs show long-term, durable responses, and the remainder mostly do not respond. This result is true in OC.35–37 How to overcome acquired resistance in the development of ICIs is critical, prompting us to explore new targets in tumor immunotherapy to solve this predicament.
Innate ICs
The immune system consists of the innate and adaptive immune systems, both of which must be activated simultaneously to obtain sufficient anti-tumor effects. 38 CTLA-4 and PD-L1 has garnered considerable attention, which mainly acts on adaptive immunity, but the impact of innate immunity is ignored. In innate immune responses, antigen-presenting cells (APCs) present antigens after phagocytes take up tumor-specific antigens; NK cells directly kill tumor cells, or APCs trigger an adaptive immune response to participate in anti-tumor responses by presenting antigens to T cells. 39 Brake-like molecules are also found during the innate immune response.
CD47 is the first identified innate IC, 40 and Oldenborg described its anti-phagocytic effect by observing the rapid clearance of erythrocytes from CD47−/− mice injected into wild-type mice, but macrophage depletion removed this effect. 41 Van Buerger found that macrophages rapidly cleared senescent erythrocytes with reduced CD47 expression. 42 Notably, CD47 has become a research hotspot since the revelation of its innate IC identity.
Biological Characteristics and Function of CD47
CD47 is a 50-kDa highly glycosylated-transmembrane immunoglobulin 43 possessing an extracellular IgV-like domain at the N-terminal, five highly hydrophobic membrane-spanning regions, and a short intracellular tail at the C-terminal, ubiquitously expressing on various normal cells.44,45 The known ligands of CD47 include integrin, 40 thrombospondin-1 (TSP-1), signal regulatory protein α (SIRPα), SIRPγ.16,45 Integrin or TSP-1 binding to CD47 is involved in cell adhesion, migration, aging, angiogenesis, platelet activation, and other biological functions.45–51 Additionally, the connection of TSP-1 and CD47 inhibits anti-tumor immunity by regulating the immune system.49,52 On the one hand, TSP-1/CD47-dependent signaling inhibits T cell function via limiting TCR-mediated first signal transduction and blocking H2S-mediated T cell activation.50,53 On the other hand, TSP-1 reduces macrophage activation by limiting lipopolysaccharide-induced IL-1β expression and total protein production in macrophages. 51 Notably, mice myeloid macrophages without TSP1 or CD47 showed the reduced induction of mature IL-1β of lipopolysaccharide.49,54 Simultaneously, decreased macrophage infiltration was observed in the absence of TSP-1. 55 Moreover, TSP-1 was initially identified as an angiogenesis inhibitor to suppress tumor growth by limiting angiogenesis and perfusion in the tumor vasculature.56,57 Perhaps the bi-phasic effects of TSP-1 in cancers that therapies targeting TSP-1/CD47 have not progressed from preclinical studies into human trials.
SIRPα is the most researched ligand of CD47, which is mainly restricted to its expression on myeloid cells.46,47,58,59 SIRPα consists of three immunoglobulin-like extracellular domains, a transmembrane region, and a cytoplasmic domain containing four immunoreceptor tyrosine inhibitory motifs (ITIMs).
59
The interactions between CD47 and SIRPα on macrophages recruit Src homology-2 (SH2)-containing tyrosine phosphatase 1 (SHP-1) and SHP-2, leading to the phosphorylation of ITIMs. These upstream signals prevent myosin-IIA accumulation to form phagocytic synapses, leading to phagocytosis inhibitions.45,47,59–61 (Figure 1) This phenomenon could further induce APCs cross-presentation and initiate adaptive immunity. In a sense, the CD47-SIRPα axis bridges innate and adaptive immunity.11,62,63 Overall, CD47 sends a “don’t eat me” suppressant signal to maintain immune tolerance and avoid excessive phagocytosis. The CD47-SIRPα signal axis. The interaction between CD47 and SIRPα recruits SHP-1/2, leading to the phosphorylation of two tyrosine residues of ITIMs in the intracellular structural domain of SIRPα. Phosphorylation of SHP-1/2 prevents the accumulation of myosin-IIA, which forms phagocytic synapses, resulting in sending a “don’t eat me” signal. Blocking the CD47-SIRPa axis could restore phagocytosis. Abbreviations: SHP-1, Src homology 2 containing tyrosine phosphatase 1; SHP-2, Src homology 2 containing tyrosine phosphatase 2; ITIMs, immunoreceptor tyrosine inhibitory motifs.
CD47 Expression in OC and Its Possible Regulatory Mechanism
CD47 is overexpressed on OC cells and was identified as an OC tumor-specific marker in 1986. 63 Subsequently, diverse insights into its expression and regulation in OC have been proposed.
Regulatory factors, including Myc, NF-κB, and HIF-1, induce CD47 expression at the transcriptional level.64–66 Myc oncogene has been confirmed to regulate CD47 and PD-L1 expression through direct joining to their promoters. CD47 and PD-L1 expression was decreased in a mouse xenograft tumor model with Myc suppression, and the anti-tumor immune response was enhanced. Conversely, the opposite phenomenon was observed with Myc activation. 64 HIF-1 binding to the promoter initiates transcription and increases CD47 expression, protecting breast cancer cells from phagocytosis. 65 Moreover, NF-κB directly connects with a super-enhancer near the CD47 gene to promote its expression upon stimulation by TNF-α in breast cancer. 67 Notably, the aforementioned transcriptional regulatory pathways must be further verified in OC models.
At the cellular level, the CD47 expression levels of exosomes in OC cells were observed to correlate with macrophage phagocytosis. Inhibition of exosome release and uptake can downregulate CD47 expression, increasing macrophage phagocytosis. 68 In general, the multifaceted regulatory mechanisms of CD47 expression imply diverse perspectives on CD47-targeted therapies.
Clinical Significance of Abnormal CD47 Expression in Patients With OC
Similar to various malignancies, CD47 high expression is associated with disease development and adverse clinical outcomes in OC.38,46,69
In 2012, Weissman et al first observed that the expression level of CD47 mRNA is associated with prognosis in OC and other solid tumors. They revealed that CD47 expression in tumor tissues was more than three times that in the surrounding normal tissues. Further analysis demonstrated that high CD47 mRNA expression correlated with poorer progression-free survival and overall survival (OS) in OC. 46 Another team found that CD47 overexpression enhanced growth and motility in TOV OC cell lines. 70 Wang et al not only observed high CD47 expression in 86 ovarian clear cell carcinomas by IHC but also confirmed that CD47 expression was correlated with surgical stage, drug resistance, and prognosis. Furthermore, it was an independent risk factor for prognosis. 71 Subsequently, the same researchers proposed that CD47 comprised Lewis-y antigen, implicated in tumor cells adhesion, metastasis, and resistance. CD47 and Lewis-y were confirmed to be significantly highly expressed in OC, and their expressions were linearly correlated. High expression of CD47 and Lewis-y was statistically correlated with the FIGO stage, lymph node metastasis, and differentiation degree. 72 The effective component of CD47 was clarified in this study.
Opinions on the association between CD47 expression and prognosis differ. A retrospective exploratory analysis of 316 serous patients with OC in the Cancer Genome Atlas database showed no difference in disease-free survival and OS between patients with different CD47 expression levels. IHC was used on another 265 patient tumor samples to validate this conclusion. Upregulation of CD47 expression was observed in 48.7% of the patients, and the low-expression group showed better treatment responses. However, this result did not improve their prognosis, which might partially be explained by the presence of multiple immune evasion mechanisms in the tumor immune microenvironment (TME) of OC. 73
A recent bioinformatics study offers several references to this confusion. The OC TME with CD47 overexpression in the Tumor Immune Estimation Resource database contained more M2 and Treg cell infiltration, promoting tumor cell immune escape. Additionally, the positive correlation between CD47 overexpression and T cell exhaustion further confirmed that CD47 might influence the infiltration abundance of immune cells in the TME to regulate OC biological behavior. 74
Mechanism of CD47-SIRPα Axis Targeted Therapy in OC
In 2009, Weissman et al observed the disappearance of AML cells in vitro and in xenografted mice with B6H12.2 (anti-human CD47 mAb). Surprisingly, the depletion of macrophages eliminated this phenomenon. It suggested that targeting the CD47-SIRPα axis eliminates tumors by restoring macrophage phagocytosis.
75
Anti-CD47/SIRPα mAbs and recombinant SIRPα-fusion proteins are thus considered promising immunotherapies based on these inspiring theories. These attractive immunotherapies may facilitate tumor clearance through the following pathways: (Figure 2). Mechanisms of blocking CD47-SIRPα axis therapies in OC. First, blocking the CD47-SIRPα axis could clear tumor cells by restoring macrophage phagocytosis and promoting tumor-associated macrophages to differentiate toward the M1 subtype (upper right). Second, anti-CD47/SIRPα-blocking mAbs or SIRPα-fusion proteins could clear tumor cells through classical Fc-dependent mechanisms, including NK cell-mediated ADCC (lower left) and macrophage-mediated ADCP (upper right). Third, blocking the CD47-SIRPα axis might indirectly stimulate adaptive immunity by promoting DCs uptake by tumor cells and cross-presentation to CD8+ T cells (upper left). Fourth, blocking the CD47-SIRPα axis could directly clear tumor cells by caspase-independent apoptosis (lower right). Abbreviations: ADCC, antibody-dependent cytotoxicity; ADCP, antibody-dependent phagocytosis; DC, dendritic cells; M1, M1-like macrophages; M2, M2-like macrophages; NK cells, natural killer cells; OC, ovarian cancer.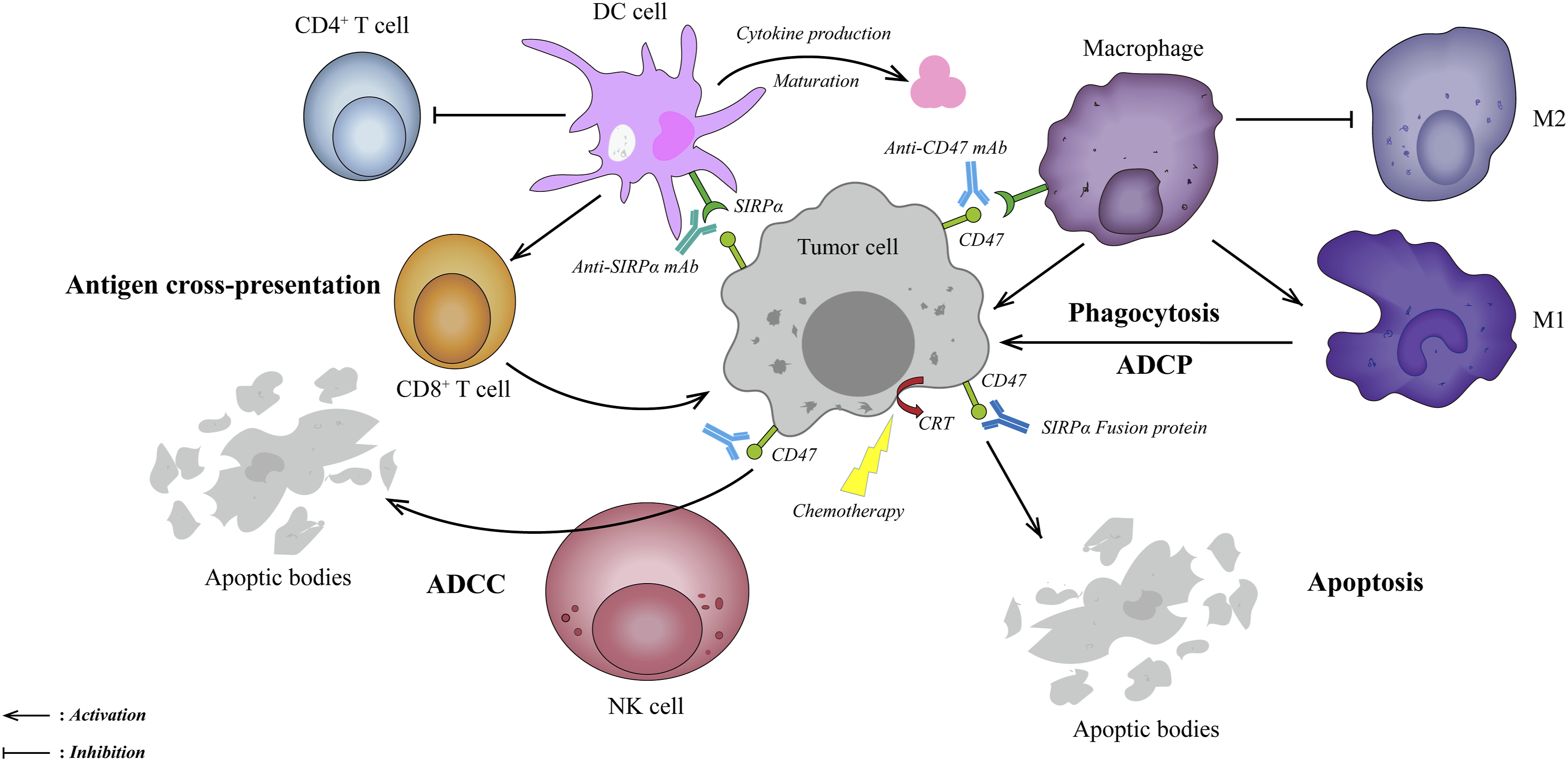
First, anti-CD47/SIRPα-blocking mAbs restore macrophage-mediated phagocytosis by blocking the “don’t eat me” signal and non-blocking antibodies without such function. In xenotransplantation models, the exhaustion of clodronate-mediated phagocytes eliminated such mAbs-dependent anti-tumor effects, confirming that phagocytes as the primary effectors.15,46,76 In this pathway, tumor-associated macrophages differentiate toward the M1-like phenotype, shifting immune-tolerant TME to attacking tumor cells.46,77
Second, the balance between pro-phagocytic and anti-phagocytic signals determines phagocytosis. In other words, blocking the “don’t eat me” signal alone may be insufficient to gain effective anti-tumor effects.62,78 Calreticulin (CRT), a dominant pro-phagocytic signal, highly expresses on various cancer cells’ surfaces, whereas barely on normal cells.62,79 The Fc receptor is another pro-phagocytic molecule, which eliminates tumor cells through NK cells’ mediated antibody-dependent cytotoxicity (ADCC) and macrophage-mediated antibody-dependent phagocytosis (ADCP). Importantly, most anti-CD47/SIRPα mAbs or SIRPα fusion proteins contain an IgG (Fcγ) skeleton. The SIRPα-Fc fusion protein constructed by Huang et al. significantly enhanced ADCC and highlighted a potent anti-tumor effect in SK-OV3 and HO8910 OC cells. 80 Moreover, further research is necessary to clarify the roles of other pro-phagocytic molecules, including SLAMF and PtdSer, during the CD47-SIRPα axis blockade.62,79
Third, inhibition of the CD47-SIRPα axis bridges innate and adaptive immunity. Weissman et al first observed that anti-CD47 antibody-mediated phagocytosis of tumor cells promoted CD8+ T cell activation but inhibited CD4+ T cell initiation in vitro and in vivo. 81 Furthermore, the interactions of CD47 with SIRPα suppress the biological behaviors of dendritic cells (DCs), such as maturation and cytokine production, resulting in the inhibition of T cell antigen presentation and adaptive immunity initiation. 82 Blocking CD47-SIRPα can remove these inhibitory effects, enabling the cross-presentation of tumor antigens and activating adaptive immunity.83,84
Fourth, direct induction of apoptosis is another mechanism, which has been observed in breast cancer, chronic myeloid leukemia (CML), and multiple myeloma (MM).15,76,85,86 Notably, anti-CD47 antibodies mediated apoptosis via a caspase-independent pathway. 86
Clinical Drug Development Targeting CD47-SIRPα Axis in OC
Registered Clinical Trials of Drugs Blocking CD47-SIRPα Axis in the NCT and CDT System.
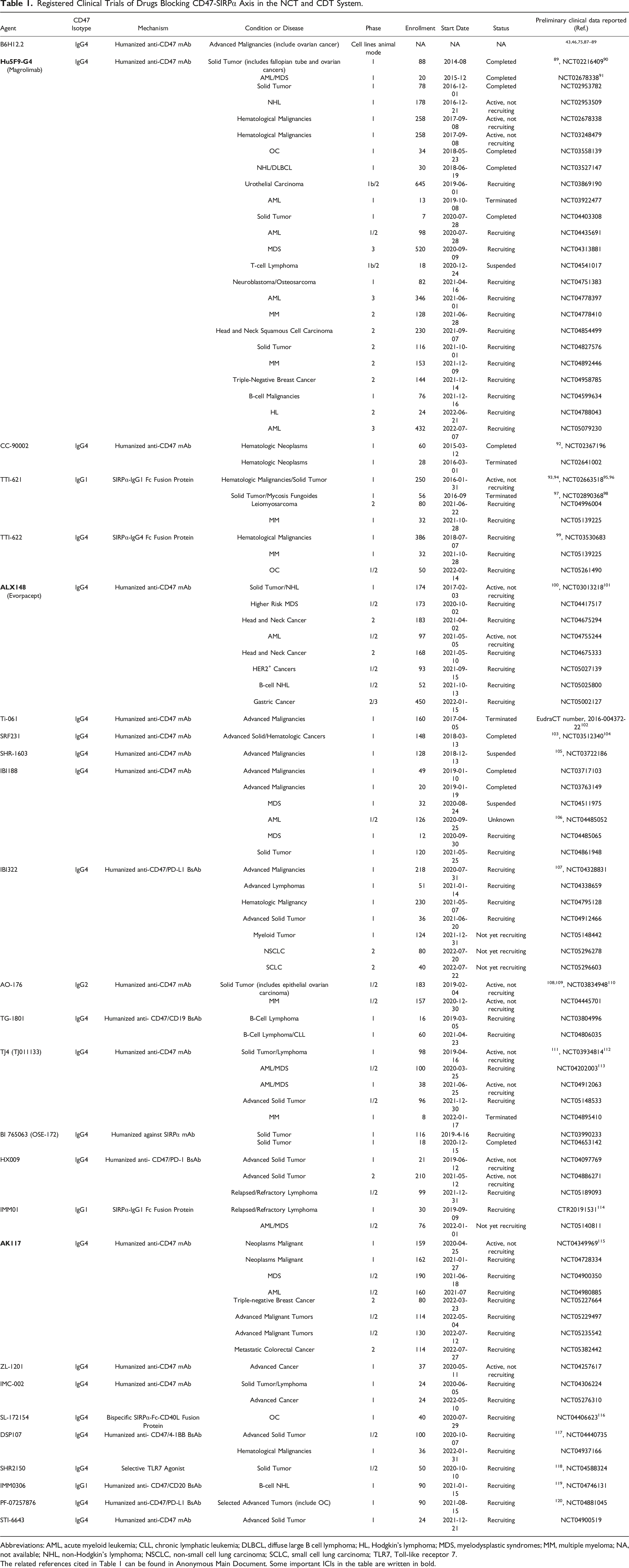
Abbreviations: AML, acute myeloid leukemia; CLL, chronic lymphatic leukemia; DLBCL, diffuse large B cell lymphoma; HL, Hodgkin's lymphoma; MDS, myelodysplastic syndromes; MM, multiple myeloma; NA, not available; NHL, non-Hodgkin’s lymphoma; NSCLC, non-small cell lung carcinoma; SCLC, small cell lung carcinoma; TLR7, Toll-like receptor 7.
The related references cited in Table 1 can be found in Anonymous Main Document. Some important ICIs in the table are written in bold.
Blocking CD47-SIRPα axis Applied in OC.

Abbreviations: CAR-T, chimeric antigen receptor T; OV, oncolytic virus; PLD, PEGylated liposomaldoxorubicin; TAG-72, tumor-associated glycoprotein 72.
The related references cited in Table 2 can be found in Anonymous Main Document.
Blocking CD47-SIRPα Axis Monotherapy
The research onanti-CD47 mAbs in OC started later than the research on AML, ALL, NHL, breast cancer, and bladder cancer, which may be related to its complex histological types and heterogeneities. In 2012, Weissman et al found that B6H12.2 and Bric126 effectively promoted human and mouse macrophage phagocytosis of SK-OV-3 OC cells in vitro, respectively. Furthermore, tumor growth suppression and significant survival improvement were demonstrated in xenotransplantation mice. 46 In 2017, Liu et al further observed increased macrophage infiltration abundance with B6H12.2 in xenograft OCs. In this study, OC stem cells (CSCs) were also found to highly express CD47, and robust phagocytosis of CSCs was induced with anti-CD47 mAbs. CSCs are known for their proliferation, recurrence, and resistance, suggesting that anti-CD47 mAbs may prevent and treat metastatic and recurrent OC.87,126 Significant inhibition of tumor cell growth, migration, and invasion in TOV OC cell lines has been observed by applying CD47 shRNA and anti-CD47 mAbs. 70
BI 765063, a selective humanized-IgG4 mAb, blocks the CD47-SIRPα axis by antagonizing SIRPα. During a phase I study including nine advanced patients with OC, BI 765063 was well tolerated without reported dose-limited toxicities or hemotoxic adverse drug reactions and showed well-pharmacokinetics and efficacy (NCT04653142).
121
Details are available on
Blocking CD47-SIRPα Axis Therapy Combined With Tumor-Targeting Antibody
Blocking the CD47-SIRPα axis monotherapy has shown promising utilization, but the combination strategies of CD47-SIRPα inhibition may offer more therapeutic potential than that treatment.
The pro-phagocytic effects of CD47-SIRPα axis mAbs combined with the Fc-dependent effects (ADCC and ADCP) of tumor-targeting antibodies exert synergistic roles for maximum efficacy.63,83 In 2010, Cao et al combined B6H12.2 with anti-CD20 mAb (rituximab) to eliminate lymphoma in mice engrafted with primary human NHL. 88 Hu5F9-G4 (Magrolimab), a humanized-IgG4 antibody, is the first CD47-targeting mAb with good clinical data to be tested in humans. 90 In a subsequent phase Ib trial, Magrolimab combined with rituximab showed promising activity and well-tolerated safety events in aggressive and indolent patients with NHL (NCT02953509). 127 Similarly, trastuzumab-mediated ADCP can significantly suppress ADCC-tolerant HER2+ breast cancer growth with the combination of anti-CD47 mAb. 128
In the absence of specific targets for OC, the combination of targeting PD1/PD-L1 therapies is the mainstream research in this field. Blocking the CD47-SIRPα axis has been shown to simultaneously restore macrophage phagocytosis and activate T cells, indicating that combinations of innate and adaptive ICIs may yield promising results.64,81,129 In 2016, Weissman combined anti-CD47 mAb with anti-PD-L1 mAb to synergistically promote tumor cell phagocytosis in vitro, and tumor growth was suppressed in vivo, demonstrating the therapeutic synergy between innate and adaptive ICIs. 130 Subsequently, a complete phase 1b trial of Hu5F9-G4 combined with avelumab (PD-L1) showed a 56% stable disease rate in 18 platinum-resistant or refractory patients with OC, which facilitated further phase II trial evaluation (NCT03558139). 122 AO-176 (NCT03834948), 108 TJ011133 (NCT03934814), 113 and TTI-621 (NCT02663518) 96 in combination with various PD-1/PD-L1 ICIs are now in different trial stages.
Blocking CD47-SIRPα Axis With Bispecific Antibody
Bispecific antibodies (BsAbs) have been developed based on combination therapy with targeting-tumor antibodies, which are expected to be more effective and precise. The BsAbs skeleton contains two binding arms, one arm blocks the CD47-SIRPα axis, and the other arm binds tumor-specific antigens, which ensures that BsAbs specifically kill cancer cells.
Majeti et al constructed a BsAb targeting CD47 and CD20 arms with less affinity for CD47 and more affinity for CD20 than the parental antibodies. Decreased tumor burden and prolonged survival in human NHL-engrafted mice were observed with BsAb; more importantly, less blood cell destruction occurred in the BsAb group than in the combination therapy group. 119 IMM0306, the related agent, is in the recruitment stage (NCT04746131).
Similar to combination therapy, BsAbs were designed to co-target CD47/SIRPα and PD-1/PD-L1 in OC. PF-07257876, a dual checkpoint inhibiting BsAb, simultaneously blocks CD47 and PD-L1 to maximize anti-tumor immunity. The preferential targeting of tumor cells in the TME is attributed to its high affinity for PD-L1 and weak affinity for CD47. It can additionally activate DCs and macrophages and increase CD8+ T cells in the TME. 119 In a phase I dose escalation and expansion study of PF-07257876, patients with OC tolerated the priming and subsequent dose (NCT04881045).
Other Combination Therapy
The strategy of anti-CD47/SIRPα monotherapy combined with chemotherapy relies on a pro-phagocytosis effect rather than an Fc-dependent effect. Almost all chemotherapies can cause inflammatory reactions and further increase macrophage infiltration in the TME. 131 As aforementioned, CRT is an essential pro-phagocytic signal, and some chemotherapy drugs, such as anthracyclines, can induce additional CRT translocation to the cell surface, which might enhance the activity of anti-CD47 mAbs. Notably, normal cells also highly express CRT with these chemotherapies, suggesting that this strategy may increase toxicity.60,78 Hongrapipat et al. found that the combination of chemotherapy and photodynamic therapy drugs with the Fab’ segment of anti-CD47 antibody might improve the reactivity of OC OVCAR-3 cells to drugs. 123
CAR-T cells have revolutionized anti-tumor therapy for hematological malignancies but have shown disappointing activity in most solid tumors. 132 Shu et al generated dual CAR-T cells co-targeting CD47 and TAG-72. TAG-72 is an aberrantly glycosylated glycoprotein overexpressed in adenocarcinomas, including ovarian, colorectal, and gastric adenocarcinomas. These dual CAR-T cells effectively eliminated OVCAR-3 cells in vitro and suppressed tumor growth in OC xenografted mice. Furthermore, the damage to normal tissues was reduced owing to the specific targeting of TAG-72. 124 The ability of oncolytic virus (OV) to infiltrate drugs into the TME makes it an attractive alternative for anti-tumor therapy. 133 Researchers constructed OV-αCD47, which delivered anti-CD47 mAb. OV-αCD47, especially those with the IgG1 skeleton, prolonged survival in OC-engrafted mice by enhancing innate immunity (NK cell cytotoxicity and macrophage phagocytosis) and performing an oncolytic function. 125
Most of these studies are in the preclinical stage, and additional efforts are warranted to promote the early application of research results to clinics.
Limitations and Future Perspectives
Although preclinical data on CD47-SIRPα axis blockade has highlighted the promising therapeutic potential of blocking the “don’t eat me” signal to recover phagocytosis, the development of related drugs is not all plain sailing. The most common AE of anti-CD47 mAbs is the off-target effect caused by widespread expression of CD47 in normal cells, among which hematotoxicity is the most intractable.
As early as 2017, the phase 1 clinical trial of Ti-061(CD47 mAb) was prematurely terminated owing to unexpected case deaths. 134 Subsequently, CD47 mAb CC-90002 was declared to have failed the phase I clinical trial (NCT02641002) owing to severe hemagglutination. 92 The repetitive failures have resulted in the uncertain future of such drug development. The superior phase 1b results of Magrolimab in combination with azacitidine in patients with MDS and AML promoted the enthusiasm for CD47-targeted drug development to a high priority until 2019. A low priming dose (1 mg/kg) was used to remove the aged erythrocytes and induce compensatory hematopoiesis to overcome anemia. Moreover, newborn erythrocytes can tolerate the maintenance (30 mg/kg) dose.127,135 Unfortunately, the FDA suspended some clinical trials (i.e., NCT04313881) of the Magrolimab in combination with azacitidine owing to apparent imbalances of suspected unexpected serious adverse reactions across study arms in January 2022. However, most experts have posited that these SUSARs may be more related to the hematotoxicity of Magrolimab or the additive toxicity of azacitidine than to affecting the overall CD47 mAbs. As expected, the FDA quickly lifted the restrictions after reviewing the combined safety data from each trial in April. Pharmaceutical companies have different attitudes toward CD47-targeted drugs. The recent discontinuation of several CD47-targeting programs by AbbVie and Zai Lab has been attributed to strategic reasons rather than safety concerns. By contrast, Innovent and Akesobio are actively advancing CD47-related projects.
In summary, within controlled AEs, CD47-targeted drugs remain the focus of tumor immunotherapy. Notably, strategies have been developed to overcome these limitations, such as anti-SIRPα mAbs, SIRPα-fusion proteins, BsAbs, dual ICIs BsAbs, dual CAR-T cells, and OC-CD47. Alternatively, researchers have engineered TAX2, the first-ever antagonist of the TSP-1/CD47 axis. TAX2 selectively targets tumor-overexpressed TSP-1 and inhibits tumor angiogenesis while activating the anti-tumor immune system without destroying blood cells. 136
Although blocking the CD47-SIRPα axis has shown promising activities in OC preclinical models, based on the experience with PD-1/PD-L1, 137 this novel therapy might have use as an adjunctive therapy or in combination with other therapies to maximize efficacy. In the future, with the disclosure of the complete upstream and downstream pathways of the CD47-SIRPα axis, more potent blocking CD47-SIRPα axis therapies with fewer side effects will be certain to the clinical application of OC treatment.
Conclusion
In conclusion,CD47-SIRPα axis plays a notable role in inhibiting macrophage phagocytosis, promoting tumors to evade immune surveillance through sending “don’t eat me” anti-phagocytic signals. Therapeutic approaches that inhibit the CD47-SIRPα axis are novel, promising treatments for OC. However, the limitations of these therapies cannot be ignored, and further studies and clinical trials are necessary for continuous improvement.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Obstetrics and Gynecology Hospital of Fudan University.
Ethical Approval
All authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
