Abstract
Background
“Crawling-type” early gastric carcinoma (EGC) is a rare subtype of gastric cancer (GC) that is challenging to diagnose at an early stage due to its low-grade nuclear heterogeneity and morphology that mimics intestinal metaplasia. This study aimed to explore the clinical characteristics and pathological features of patients with crawling-type EGC.
Methods
This case series study retrospectively included patients with crawling-type EGC who underwent endoscopic submucosal dissection (ESD) or gastrectomy at the East Hospital Affiliated to Tongji University between January 2019 and March 2022.
Results
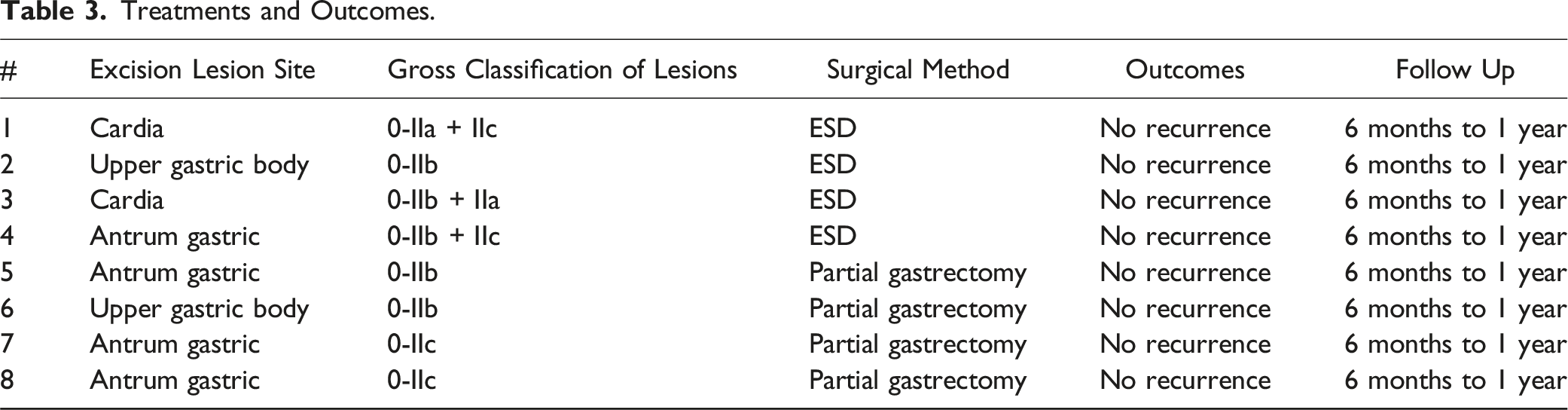
8 patients (mean age 63.5 ± 7.8 years) were included: 4 underwent ESD, and 4 underwent partial gastrectomy. In 4 patients, the tumors were primarily located in the gastric cardia and the basal gland area of the upper stomach, while the other 4 patients had tumors in the antral region. Preoperative gastroscopy revealed atrophic gastritis and intestinal metaplasia in all patients. The lesions were generally flat in morphology. Submucosal infiltration was found in only one case. Signet ring cells were present in the tumors of 5 patients. The mucinous type was observed in 7 patients. Seven tumors were of the gastrointestinal mixed type. Curative resection was achieved in all patients. No recurrence events were observed in any patient at 1 year after surgery.
Conclusions
The crawling-type EGC may exhibit distinct clinical characteristics and pathological features compared with classical GC. Curative resection was achieved in all patients. The short-term prognosis of surgical treatment may be favorable.
Keywords
Introduction
Gastric cancer (GC) is a malignant neoplasm originating from the epithelium of the gastric mucosa. Although its incidence has declined in recent years, it remains one of the most common malignant tumors globally. 1 In China, GC was the third most important cancer in terms of incidence and mortality in 2022. 2 GC is a heterogeneous disease, and clinicians may occasionally overlook rare variants of GC due to their rarity.
“Crawling-type” early gastric carcinoma (EGC) is one such rare variant of GC and is characterized by irregular glandular fusion, low-grade cellular atypia, and a tendency for lateral diffusion within the mucosa.3,4 The microscopic examination of crawling-type EGC typically reveals intestinal metaplasia cells, irregular glandular structures, occasional signet ring cells, “handshake structures,” or “Whyx-type” structures.5,6 Despite minimal atypia and extension into the area of epithelial hyperplasia, the mucosal surface of crawling-type EGC generally appears normal, posing a diagnostic challenge, particularly in small, limited biopsy specimens. Cytological features lacking distinct characteristics are often misdiagnosed as ambiguous tumor formation or reactive intestinal metaplasia, leading to an underestimation of their malignant potential.7,8 Moreover, the boundaries of crawling-type EGC within the mucosa are frequently indistinct due to the lack of comparison with surrounding non-neoplastic mucosa, 5 making complete removal through endoscopic submucosal dissection (ESD) difficult.
Studies on the clinicopathological and molecular characteristics of crawling-type EGC are limited, complicating the accurate diagnosis of this condition.9-11 In addition, there has been insufficient focus on the prognosis after surgical intervention. Therefore, this study aimed to report the clinical characteristics, diagnostic methods, and short-term prognosis of patients with crawling-type EGC after ESD or gastrectomy.
Material and Methods
This case series study retrospectively included consecutive patients diagnosed with crawling-type EGC who underwent ESD or gastrectomy at the East Hospital Affiliated to Tongji University between January 2019 and March 2022. The inclusion criteria were (1) patients diagnosed with crawling-type EGC for the first time and (2) who underwent ESD or gastrectomy. The exclusion criteria were (1) received cancer treatments before surgery for crawling-type EGC or (2) incomplete clinical data. This study was approved by the Ethics Committee of the East Hospital Affiliated to Tongji University (2024YS-270), and the requirement for individual informed consent was exempted due to the retrospective nature of the study. The reporting of this study conforms to STROBE guidelines. 12
Data Collection
Data were collected from the original patient records. All surgical specimens were examined by experienced pathologists. Variables such as preoperative and postoperative pathological results, postoperative lesion/tumor size and shape, depth of invasion, atypia, 52-week prognosis, TNM staging, and type of surgery were collected. All patients were routinely followed up at 6 and 12 months after surgery using gastroscopy to observe eventual changes in gastric and scar mucosa and using computed tomography (CT) to observe lymph nodes and nearby organs. All patient details were de-identified.
Statistical Analysis
Only descriptive analysis was performed. Categorical data were expressed as n (%).
Results
Clinical Information.

Pathology.

Treatments and Outcomes.
Typical Cases
Patient 4: A 64-year-old man presented with abdominal distension and belching for more than 1 month. Gastroscopic examination revealed a light red lesion classified as 0-IIb + IIc under white light and Indigo blush dyeing endoscopy (Figure 1(A) and (B)). Magnifying narrow-band imaging showed irregular microsurface and microvascular patterns (Figure 1(C)). The preoperative pathological examination suggested chronic inflammation of gastric mucosa in the minor curvature of the gastric antrum and high-grade intraepithelial neoplasia of focal glands. The patient underwent ESD (Figure 1(D)). The postoperative pathological examination revealed highly to moderately differentiated tubular adenocarcinoma/tub2 > tub1 (in part, crawling type morphological changes as the major differentiation). Patient #4. Endoscopic images. (A) Under white light. (B) Under Indigo blush dyeing endoscopy. (C) Magnifying narrow-band imaging showed irregular microsurface and microvascular patterns. (D) Endoscopic submucosal dissection.
The mucinous type was classified as the gastrointestinal mixed type, and the visual classification was superficial uplift and concave type (0-IIb + IIc). The tumor size was approximately 20 mm × 11 mm under the microscope. The carcinoma tissue infiltrated into mucous lamina propria (LPM/pT1a). There was no ulcer formation (UL0). The postoperative diagnosis was crawling-type EGC (Figure 2). There was no recurrence after 1 year of follow-up. Patient #4. Postoperative histopathological findings of the primary lesion. There was a handshake structure in the glands of the lesion. Leica 3000 type light microscope (Leica, Wetzlar, Germany), eyepiece 10 × 22, objective lens ×10, ×20, ×40, ×60, and ×100 (oil lens). The magnification of the images is ×100. The section was stained with hematoxylin & eosin.
Discussion
This study suggests that crawling-type EGC may exhibit distinct clinical characteristics and pathological features compared with classical GC. The rate of curative resection post-surgery appears satisfactory, and the short-term prognosis following surgical treatment may be favorable. These findings may provide valuable insights for diagnosing and treating this GC subtype.
In this case series, the mean age of the patients was 63.5 ± 13.45, slightly higher than previously reported by Woo et al. 10 The present study found that crawling-type EGC was mainly located in the upper part of the stomach, from the cardia to the fundus gland region and the gastric antrum. This finding differs from Haruta et al 5 earlier, who demonstrated that the middle third of the stomach was the preferential site for crawling-type EGC. The EGCs observed in the present study were mainly characterized by a flat morphology, whereas previous reports showed that more than 70% of crawling-type EGCs exhibited a depressed structure.5,8,9 According to the Lauren classification, 13 most of these lesions were of the gastrointestinal mixed type. This result aligns with the classification of crawling-type EGC as a very well-differentiated GC of the intestinal type.7,14,15 Notably, most of the lesions were limited to the mucosa, with only one tumor penetrating the submucosal layer, a finding supported by previous research. 14
As the literature describes,5,9-11 crawling-type EGC is characterized by unique histological features. In agreement with these reports,5,9-11 the cases reported here displayed heterotypic structures, crawling-type cells, and irregular glandular formations. These features are the most significant pathological characteristics of crawling-type EGC.5,9-11 Among the 5 tumors containing signet ring cells, 4 had a small amount of signet ring cell carcinoma, and one case had a large area of signet ring cell carcinoma. Nevertheless, postoperative pathology still suggested curative resection. It is worth considering whether there is an association with older patient age, as most previous reports of signet ring cell carcinoma were in younger patients with poorer prognosis. This point warrants further exploration. The finding indicates that the presence of signet ring cells could be a helpful indicator for diagnosing crawling-type EGC, but it will have to be validated in future studies.
In addition, the present case series showed large tumors, with 6 being larger than 2 cm. The borders of the lesions were frequently poorly defined due to a lack of contrast with the surrounding non-neoplastic mucosa. These features may pose difficulties for the complete resection of lesions by ESD, as the absolute indications for ESD include highly or moderately differentiated intramucosal adenocarcinoma less than 2.0 cm in diameter without ulcerative changes. 16 Fortunately, no recurrence events were found in any patient. In this case series, 4 patients underwent ESD, and 4 underwent partial gastrectomy. Although the long-term outcomes remain to be determined, treatment outcomes for all patients were optimal after 12 months of follow-up. ESD for crawling-type EGC can be difficult because of the similar appearance of the lesion to the adjacent tissue, 5 but it can still be possible in selected cases. Nevertheless, according to the “Guidelines for endoscopic submucosal dissection and endoscopic mucosal resection for early gastric cancer,” 17 ESD treatment for crawling-type EGC should preferably meet the following indications: (1) UL0 cT1a differentiated-type carcinomas with a long diameter greater than 2 cm; (2) UL1 cT1a differentiated-type carcinomas with a long diameter measuring 3 cm or less; (3) UL0 cT1a undifferentiated-type carcinomas with a long diameter of 2 cm or less. Of course, the resection margins must be carefully evaluated, and the patient and surgeons must be prepared for an eventual gastrectomy if ESD has positive margins.
The present study had several limitations. Firstly, the sample size was small, precluding the observation of certain features. Secondly, the postoperative follow-up was short, impeding an assessment of the long-term efficacy of surgical resection. In addition, the study did not investigate the expression profiles of crawling-type EGC, a factor that could offer supplementary information for diagnosing this condition.
Conclusions
Crawling-type EGC may exhibit distinct clinical characteristics and pathological features from classical GC. Curative resection was achieved in all patients, and the short-term prognosis of surgical treatment may be favorable. Preoperative gastroscopy may potentially misdiagnose crawling-type EGC. The exact prognosis of crawling-type EGC remains unknown, and the selection of adjuvant treatments should be made based on the final pathological results.
Footnotes
Author Contributions
(I) Conception and design: Kehan Li. (II) Administrative support: Jia Cao. (III) Provision of study materials or patients: Xiaofeng Zhuang, Bingyue Yao. (IV) Collection and assembly of data: Kehan Li, Qinwei Xu, Li Zhang. (V) Data analysis and interpretation: Tao Chen. (VI) Manuscript writing: All authors. (VII) Final approval of manuscript: All authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Academic Leaders Training Program of Pudong Health Bureau of Shanghai (Grant No. PWRd2021-02).
