Abstract
Introduction
Epithelioid Hemangioendothelioma is a rare vascular neoplasm unresponsive to chemotherapy, radiotherapy, or immunotherapy. Surgical options are considered; however, recurrence rates reach up to 30%. Irreversible electroporation is difficult with large or numerous lesions. This paper reviews treatment options, exploring long-term response after subsegmental bland embolization of hepatic epithelioid hemangioendothelioma (HEHE).
Case Series
This IRB approved retrospective study involved 3 patients after subsegmental bland embolization of 8 total HEHE lesions. Preoperative and postoperative contrast enhanced CT scans were compared up to 4 years post-operatively. Patient 1, a 28-year-old male whose single lesion involved segments 4, 5, and 8, with a mean volume of 458.1 mL that progressed despite treatment with Axitinib and Pembrolizumab. One-month postembolization volume was 439.6 mL and 4-year follow-up volume was 128.9 mL, representing a 72% volume reduction. The second patient, a 54-year-old female could not finish her IRE treatments. Four of the lesions, 2 right-sided and 2 left-sided, were treated with subsegmental bland embolization (mean volume 108.8 mL, SD 86.1 mL). The 1-month postembolization mean volume was 78.1 mL (SD 48.3 mL), and 4-years mean volume was 9.8 mL (SD 8.4 mL), representing a 91% tumor reduction volume. The third patient, a 61-year-old female presented with numerous, bilobar lesions not amenable to IRE. Three lesions were treated with subsegmental bland embolization (mean volume 52.4 mL, SD 55.9 mL). One-month postembolization, the mean volume was 49.9 (SD 49.5 mL). One-and-a-half-year follow-up demonstrated a mean volume of 38.8 mL (SD 48.9 mL), representing a 22% reduction in mean tumor volume. No grade 3 or greater complications after any procedure occurred.
Conclusions
Subsegmental bland embolization is safe and effective, having the potential to stabilize HEHE not otherwise suitable for surgery or IRE. All patients achieved sustained disease control with most lesions demonstrating a partial response. Possible future directions include starting a prospective patient registry to obtain additional long-term data regarding subsegmental bland embolization outcomes.
Keywords
Introduction
Epithelioid Hemangioendothelioma (EHE) is a rare vascular neoplasm that often affects the liver, known more specifically as hepatic epithelioid hemangioendothelioma (HEHE). Characterized by its unique microscopic epithelioid appearance, this tumor exhibits an endothelial origin. Symptomatic HEHE usually presents with right upper quadrant pain (40%-49%), hepatomegaly (6%-24%), and weight loss (9%-16%), but it can also be discovered incidentally.1-3 This disease may manifest in uninodular, multinodular, diffuse patterns, or multifocal form, the most common form. 4 Prognostication is highly dependent on the extent of disease, namely tumor size, evidence of metastasis, liver function, and systemic signs and symptoms.2,5
EHE is rare, with an incidence of roughly 0.038 per 100 000 individuals annually and a prevalence of less than 1 per million people. HEHE is diagnosed primarily in adults between the ages of 30 and 40 years old with a female predominance, showing a female-to-male ratio of 3:2.1,2,5,6 The clinical course of HEHE can range from asymptomatic to severe liver failure in more advanced stages. 4 The rarity and heterogeneity of HEHE presents significant challenges in diagnosis and management. Imaging features often vary; however, common findings on MRI include a slow centripetal enhancement from arterial to portal phase with central hypointesnsity (target sign) or persistent slight rim-like enhancement which when paired with an obstructed portal vein termed the lollipop sign.7,8 Many lesions prefer the subcapsular or peripheral region, often causing capsular retraction.8,9 When greater than 5 cm, lesions often coalesce and exhibit capsular retraction. 8 Surgical resection is often considered for patients with limited disease, curing up to 50% of HEHE cases; however, the distribution pattern and extent of lesions can complicate treatment.2,6 In non-surgical patients with unifocal disease, other options include radiofrequency and microwave ablation. 2 While pazopanib and oral thalidomide are antiangiogenic alternatives, no strong evidence supports first-line use in HEHE management. 10 According to Zhao M et al and 258 HEHE cases pulled from various databases, including PubMed, resection occurs in 29.7% cases, transplantation in 16.1%, palliative treatment in 12.7%, transhepatic arterial chemotherapy and embolization (TACE) in 10.2%, chemotherapy in 11.0%, antiangiogenic therapy in 15.3%, and “other” in 5.1% of cases with surgical treatments providing longer survival times. 5
Given the limited efficacy of conventional therapies in the remaining cases, alternative strategies for HEHE treatment have been investigated. One approach is transplantation but is associated with a recurrence rate of up to 30%. 11 Irreversible electroporation (IRE) has also emerged as a promising modality due to its safety, efficacy, and repeatability,6,10 but it can prove more difficult when dealing with extensive or numerous lesions. The aggressive potential and resistance to many standard treatments necessitates additional treatment options to attain long-term disease control.
Of note, gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumors of the GI tract and, while typically of spindle cell origin, can exhibit pure epithelioid characteristics in 10%-20% of cases and mixed cell type in 10% of cases.12,13 Furthermore, subsegmental bland embolization has been explored as a second-line treatment option to systemic chemotherapy or compared to TACE with promising results.14,15 One study found that subsegmental bland embolization for GIST liver metastases afforded an overall survival (OS) and progression free survival (PFS) of up to 23.8 months and 3.4 months respectively in patients with imatinib and sunitinib refractory treatment. 15 Results of a different study comparing TACE and bland embolization showed longer OS and PFS in the bland embolization group, especially in early-stage GIST liver metastasis defined as Child-Pugh class A or less than 50% liver involvement. 14 Because of their pathologic similarities often being confined to the liver, HEHE may respond well to subsegmental bland embolization similarly to GISTs.
This study aims to investigate tumor response to subsegmental bland embolization in patients with dominant HEHE. This treatment strategy occludes targeted, or subselected, blood vessels supplying the tumor to induce ischemia and necrosis using 100-300-micron embolic agents, namely Embosphere®s (Merit Medical). This size was chosen due to its proved ability to be delivered either inside the tumor or near the tumor margin, thus maximizing tumor selection and minimizing normal liver parenchyma delivery. 16 This study explores the efficacy of subsegmental bland embolization in HEHE to provide valuable insight into alternative therapeutic strategies that could enhance patient outcomes, especially those with tumors unamenable to conventional treatment options.
Materials and Methods
An IRB approved (study number 20070111, approved on 06/17/2024 through the University of Miami) retrospective study of patients who underwent subsegmental bland embolization of liver dominant EHE lesions between 2019 and 2024 at a single institution was performed. Written informed consent was obtained from all patients for the publication of this study and any accompanying images. This revealed 3 patients (1M, 2F) and a total of 8 eligible lesions who were selected for subsegmental bland embolization after Multidisciplinary tumor board (MDTB) review.
All subsegmental bland embolizations were performed under fluoroscopy with the same interventional radiologist with greater than 11 years of experience. All cases used a slurry of 100-300-micron Embosphere® (Merit Medical) composed of 20 mL of pre-packaged sterile saline with 2 mL of Embosphere® and 20 mL of contrast, specifically iodixanol. Two cases were performed under general anesthesia while 1 was performed under moderate sedation.
After subsegmental selection of each lesion, embolization was performed until stasis or near stasis was achieved. Preoperative contrast enhanced CT and post operative CT up to 4 years post-operatively were analyzed by board certified interventional and diagnostic radiologists with over 11 years of experience using MIMS software to compare tumor volumes and monitor disease progression. The reporting of this study conforms to CARE guidelines. 17
Results
Cases
Case 1
A 23-year-old male who presented with right upper quadrant pain was found to have both lung and liver lesions which were HEHE proven on biopsy. Despite being previously controlled with a combination of IRE and Celecoxib for 3 years followed by a clinical trial of Axitinib and Pembrolizumab for an additional 2 years, his disease had progressed.
At this point the dominant liver lesion was found to involve segments IV, V, and VIII while measuring 14.3 cm in maximum diameter and 458.1 mL in volume with smaller satellite lesions treated by IRE. The patient then underwent subsegmental bland embolization of the dominant lesion per MDTB case review. Prior to treatment, liver function tests (LFTs) consisting of total bilirubin (Tbili), aspartate aminotransferase (AST), alanine transaminase (ALT), and alkaline phosphatase (ALP) showed values of 0.6, 10, 16, and 127, respectively. The subsegmental bland embolization was performed by obtaining radial access under general anesthesia and using a 2.4 French Progreat® Microcatheter (Terumo, Somerset, NJ) to select subsegmental branches to segments IV, V, and VIII to deliver a total of 25 mL of slurry composed of 100-300-micron Embosphere®/Saline/Contrast (Figure 1). There were no grade 3-or-above complications per CTCAEv5. Pre-, Intra- and Post-embolization Images From Patient 1. There are Axial and Coronal Images of Pre-embolization and Post-embolization at One Month and Four Years after Treatment as Well as Intraprocedural Images Beneath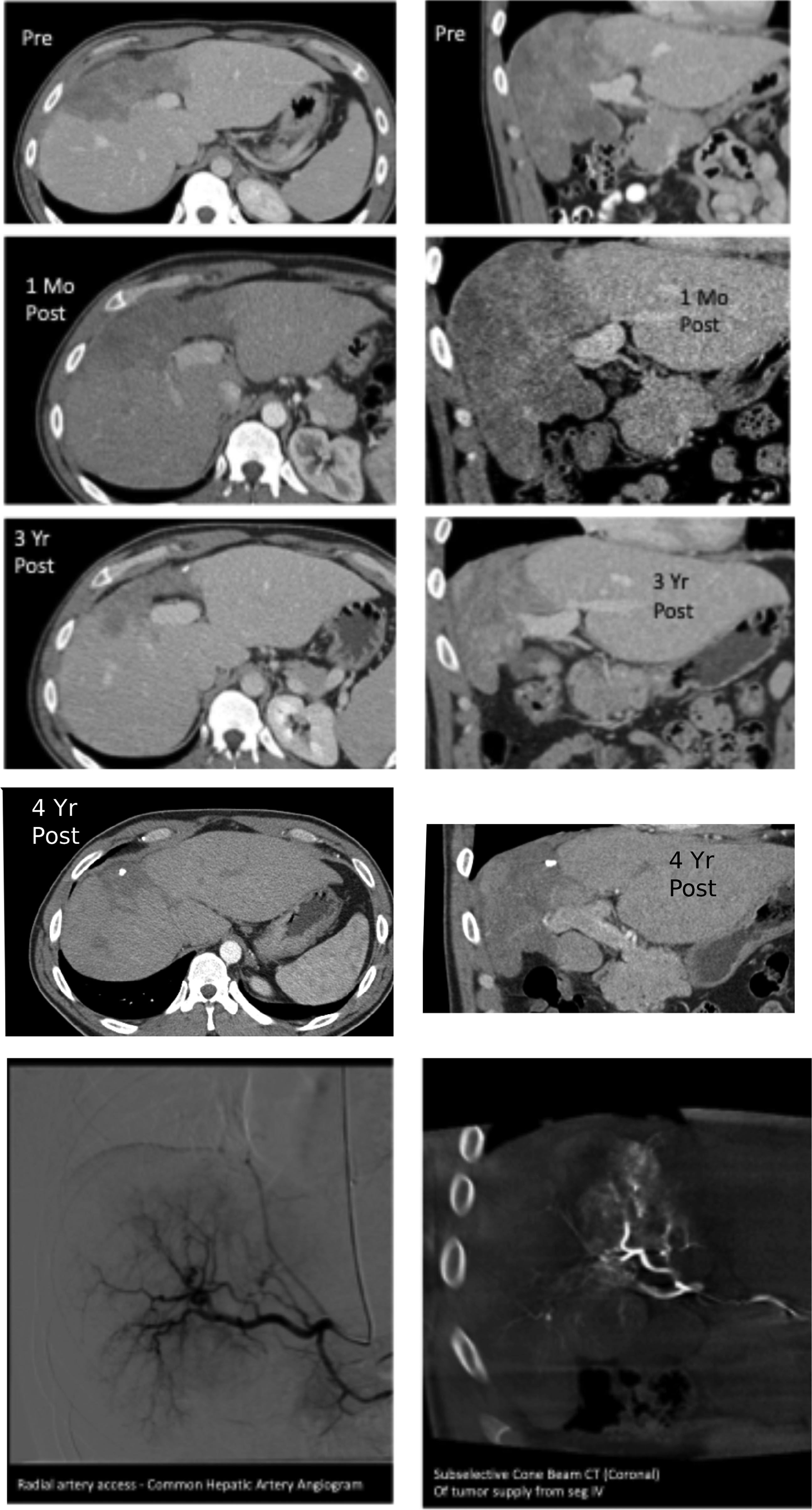
Patient and Tumor Characteristics. This Table Shows the Age of Each Patient Discussed Along With Tumor Distribution, Volume, Percent Volume Reduction, Number of Years Post Treatment, and Averages of Select Values

Case 2
A 54-year-old female who presented with colicky abdominal pain was found to have diffuse liver lesions as well as small, sub-centimeter lung lesions proven to be HEHE on biopsy. One year after diagnosis the patient underwent IRE to segment V. The following year, management targeted segment VIII with IRE; however, intraprocedural tachyarrhythmias limited completion of the treatment.
Pre-embolization CT scan confirmed a total of 4 remaining lesions, 2 in the left lobe, segments II/III and IV, and 2 in the right lobe, segments V and VIII (Figure 2). The lesions in segments II/III and IV measured 29.9 mL and 66.7 mL, respectively, while those in segments V and VIII measured 110.6 mL and 228.1 mL, respectively, with a mean volume of 108.8 mL (SD 86.1 mL). MDTB decided on subsegmental bland embolization for treatment given the tachyarrhythmias during prior IRE. Pre- and Post-embolization Images From Patient 2. There are Photos of Pre-embolization and Post-embolization at One Month and Four Years after Treatment in the Segment II/III and VIII Lesions
Subsegmental bland embolization of the left-lobed lesions in segments II/III and IV were completed first. Pretreatment LFTs showed a Tbili of 0.3, AST 22, ALT of 23, and ALP of 143. The procedure began by gaining femoral access under general anesthesia and using a 2.8 French Progreat® Microcatheter (Terumo, Somerset, NJ) to select and deliver 20 mL of 100-300-micron bead slurry to the appropriate subsegmental branches. Labs obtained the day following the procedure were a Tbili of 0.4, AST of 218, ALT of 120, and ALP of 146 with those same LFTs 1 month after returning to baseline at 0.3, 22, 23, and 123, respectively. The second subsegmental embolization targeted the lesion in segment VIII. Preprocedural labs resulted in Tbili of 0.4, AST of 21, ALT of 21, and ALP of 100. Like the first procedure, femoral access was gained under general anesthesia; however, a 2.7 French Progreat® Microcatheter (Terumo, Somerset, NJ) was used to select and deliver 24 mL of 100-300-micron bead slurry. Demonstrating a transient elevation, labs at 2 days and 1-month post-embolization were a Tbili of 0.7 then 0.4, AST of 178 then 19, ALT of 228 then 18, and ALP of 147 then 103. The third subsegmental embolization treated segment V using a 2.9 French Maestro® Microcatheter (Merit Medical, South Jordan, UT) to select and deliver 10 mL of 100-300-micron bead slurry. Pre- and post-embolization LFTs again showed transient elevation to Tbili of 0.4, AST of 99, ALT of 79, and ALP of 110 before returning to baseline by the next collection of labs approximately 1 month later. The patient tolerated subsegmental bland embolization without grade 3-or-above complications per CTCAEv5.
One-month post-subsegmental-embolization CT revealed that the segment II/III and IV lesions had volumes of 27.4 mL and 61.1 mL, respectively while the segment V and VIII lesions had volumes of 81.9 mL and 142.2 mL. At 4 years post-subsegmental-embolization, their volumes were 5.6 mL (segment II/III), 1.9 mL (segment IV), 21.3 mL (segment V), and 10.8 mL (segment VIII) with a mean volume of 9.8 mL (SD 8.4 mL). These 4-year post-subsegmental-embolization volumes signified a 91% reduction in tumor volume (Table 1).
Case 3
Liver Function Tests. This Table Shows the LFTs of Each Patient Pre- and Peak Postembolization as Well Relative Change From Baseline

Subsegmental bland embolization of all 3 lesions occurred during the same procedure through femoral artery access. A 6 French vascular sheath was placed after gaining access with a 5 French micropuncture set and 0.035-inch guidewire. A series of 5 French catheters were used to select a replacement of the common hepatic artery that arose from the superior mesenteric artery with a common trunk extending from the gastroduodenal artery. Following this, a 2.7 microcatheter and microwire were used to sub-select segment VI and deliver 4 mL of 100-300 µm bead slurry. The patient complained of pain at this point, so repeat angiography was performed without demonstrating nontarget embolization. Segment VII was targeted next with 3 mL of embolic agent delivered, and finally segment VIII received 2 mL off the bead slurry, achieving the desired result without further pain. Aside from the initial pain, the patient did not experience any complications, including any CTCAEv5 grade 3-or-above. Labs obtained the following day demonstrated an elevation of the patient’s Tbili of 0.6, AST and ALT to 35 and 21, respectively, while the ALP was 122. Two weeks later the Tbili, AST, and ALT dropped to 0.5, 19, and 17 while the ALP showed an elevation to 163.
After subsegmental bland embolization, the segment VI, VII, and VIII lesions respective 1-month volumes on CT were 107.1 mL, 23.5 mL, and 19.4 mL with a mean volume of 49.9 mL (SD of 49.5 mL). Four months after the subsegmental embolization, all the patient’s LFT were Tbili of 0.5, AST of 22, ALT of 18, and ALP of 120. At approximately 2 years post-embolization, imaging showed further involution of tumor volumes to 94.9 mL, 17.1 mL, and 4.5 mL in segments VI, VII, and VIII. This represented a mean volume of 38.8 mL (SD 48.9) and a 22% reduction in mean tumor volume (Figure 3). Pre-, Intra- and Post-embolization Images From Patient 1. There are Axial and Coronal Images of Pre-embolization and Post-embolization at Two Years after Treatment for the Third Patient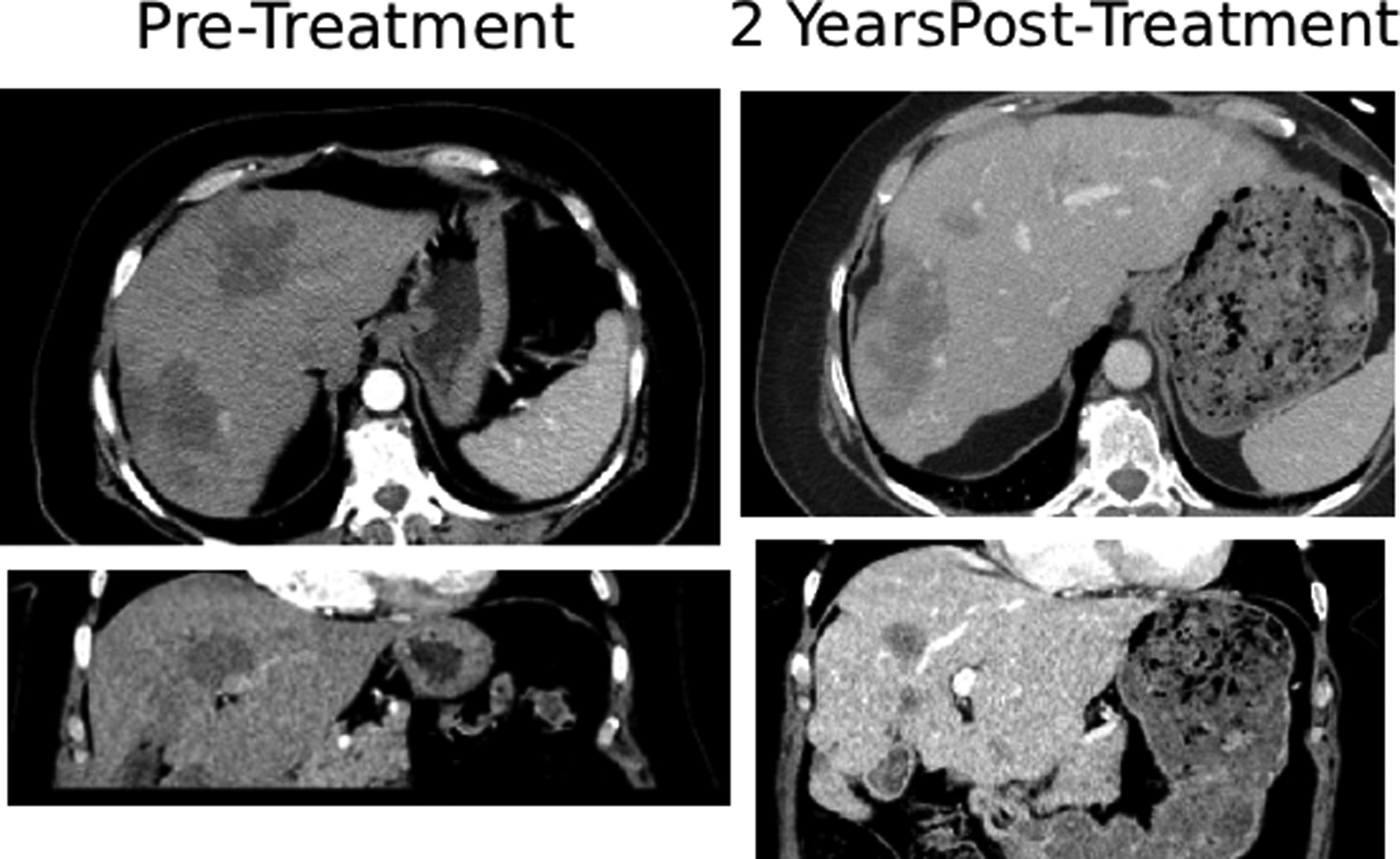
Follow up with the patient 3 years after original diagnosis and 2 years after prior subsegmental bland embolization, patient was noted to have abdominal bloating. Although imaging showed stable liver lesions, it revealed ascites and omental carcinomatosis that developed over 3 months since the previous imaging.
After multidisciplinary discussion, we performed a biopsy of both the segment IV mass, confirmed HEHE positive for WWTR1-CAMTA1 fusion as well as the new omental lesions. During the biopsy the presence of a malignant vascular neoplasm was confirmed on angiogram and favored involvement of the patient’s HEHE, demonstrating active disease in segments previously treated with subsegmental bland embolization, specifically segments V, VII, and VIII. The plan following biopsy was to await the results and transition the patient to systemic therapy.
Unfortunately, her recovery was marked by severe malnutrition and tachycardia with leukocytosis revealing urosepsis compounded by pulmonary embolism and right heart strain. After electing for comfort measures, she passed away secondary to multi-organ failure and metabolic derangements.
Discussion
The unpredictable course and metastatic potential coupled with its rarity pose a challenge to conducting studies into HEHE.1,2,5,6 Agrawal et al. comment in their review that patients not receiving treatment had a mortality rate of greater than 50%. 6 Thus, treatment is a necessity in HEHE patients. While both surgical and nonsurgical options are considered in the treatment of HEHE, there is no standardized regimen, so options continue to be explored.1,2,10 Narayanan et al.’s paper in 2024 describe the lack of feasibility in surgical options due to the multifocal nature of HEHE and that systemic options are chiefly palliative. They discuss the drawbacks of thermal ablative methods and report their experiences with IRE, indicating the lack of consensus on definitive treatment options for HEHE. 10 In 2022, Ajay et al report that 33% of patients in their review underwent surgical treatment, 26% received chemotherapies, and approximately 6.7% received radiation therapy with the remaining unaccounted 1 ; however, others report on other treatment modalities including TACE, IRE, and various ablative techniques such as radio or microwave ablation.5,10,18 This study focused on the efficacy of another nonsurgical option for disease control, subsegmental bland embolization.
A total of 8 HEHE tumors from 3 patients up to 4 years after undergoing subsegmental bland embolization were examined with an average tumor size of 131.3 mL prior to treatment. All patients successfully tolerated their procedures, resulting in a combined 72.8% volume reduction and average of 95.7 mL reduction across all 8 lesions at the time of investigation. Of the 8 tumors, 7 demonstrated partial response while the last tumor showed stable disease per mRECIST criteria
19
(Figure 4). Additionally, this study exhibited the safety of this procedure in HEHE patients. None of the patients experienced CTCAEv5 grade 3 or above adverse events while only 1 patient complained of transient pain intraoperatively and still completed the treatment. Furthermore, the treatment can be repeated in patients to target additional lesions. Duration of Response. Figure 3 Depicts the Duration of Response From Each Patient’s Initial Bland Embolization at Time Zero. Additional Treatments are Marked by Green Diamonds. Follow-Up Imaging is Marked by the Red Triangle, end of Systemic Treatment by the Red Circle, and Patient Death by the Black Square. The Color of Each bar Notes the Patient’s Response to Treatment per mRECIST Criteria
Review of Selected Studies From the Discussion. This Table Presents Data From Various Studies Discussed for Easier Viewing and Comparison of Treatment Modalities Employed in Managing HEHE

Abbreviations: m-PFS (Median Progression Free Survival).
Standard systemic options for vascular tumors have not shown promise in the treatment of EHE. Because of the lack in efficacy, observation in asymptomatic and stable patients is appropriate. However, progressive cases requiring treatment prove difficult with systemic management. Compiling data from 20 sarcoma reference centers in 2021, Frezza et al. found aggressive variants that behave as high-grade sarcomas do not have significant responses. The results showed median (m-) PFS of 5.5 months with anthracycline based regiments, m-PFS of 2.9 months with weekly-paclitaxel, and m-PFS of 2.9 months with pazopanib with the highest m-PFS of 8.9 months with INV-α 2b. 21 Stacchioti et al confirmed activity of sirolimus in patients with progressive EHE demonstrating that pleural and or peritoneal effusion was an indication of poor prognosis with m-PFS of 4.8 months compared with 47.8 months in patients without effusions. 18 Additional work identifying GDF-15 as a biomarker of EHE aggressiveness also favors sirolimus as a systemic option. 22 Trametinib, a MEK inhibitor, showed a m-PFS of 10.4 months and a dramatic improvement in median pain intensity scores. 23 There are reports of response with VEGF inhibitors such as Bevacizumab and Sorafenib.24,25
The ablative techniques and TACE that Stachiotti et al qualitatively describe, note that TACE appeared superior to surgical treatment in more advanced cases of EHE and could potentially be used with palliative intent in the most advanced cases of the disease while ablative measures were best employed with curative intent in limited, small, and single-lesion cases. 2 Previous work by Granito et al. found that AST and ALT served as a clinical marker that predicted treatment response to conventional TACE in hepatocellular carcinoma (HCC). Elevations of 46% and 52% above baseline for AST and ALT were found to be predictive of tumor response. 26 Changes in LFT after IRE reach as high as 20 times the upper limit of normal; however, over 95% of LFT elevations resolve in as early as 7 days and on average within 10 weeks. 27 Subsegmental bland embolization shows transient elevations in LFTs in this patient population; although, the changes are quite variable between each patient ranging from 1.02 of baseline to 10.86 of baseline LFTs (Table 2). Notably, the patient with the largest changes in LFTs demonstrated the greatest reduction in tumor volume, indicating that magnitude of change may predict tumor response. A minimum elevation cannot be established at this time due to the small population. These differences may reflect variation in the biology of each patient or their disease, but the small sample size limits in-depth analysis.
Aside from the variations in lab values, each patient carried varying disease burdens ranging from 1 mass to 4 (Table 1). The total tumor volume also varied from over 400 mL to approximately 150 mL. The disease heterogeneity’s effect on treatment results is unclear; although, the patients with larger total tumor volume demonstrated greater overall responses. Both patients were also younger. Having shorter follow up, the third patient may have needed additional time for a greater treatment response. Furthermore, each patient’s disease pathology or biology likely differed. However, reasons between treatment response variation would be speculative at best. Larger, future, prognostic studies that examine differences in patient characteristics, the pathologic disease differences, and response over time may elucidate exact causes though.
Narayanan et al explored IRE, a nonthermal ablative option, in their retrospective analysis of HEHE patients. They achieved technical success in all cases with complete response in most lesions and at stable disease in the remaining cases over 15 months of follow-up. They evidenced the safety of IRE noting only 1 instance of pneumothorax in 23 treatments. However, they focused on smaller lesions with a median tumor size of 25.8 mm and a max of 60 mm with a median of 3 tumors per patient. 10 Because of this, IRE may prove to be more difficult in patients with larger and numerous lesions.
Subsegmental bland embolization solves this issue, providing a treatment option better suited to patients with larger or numerous lesions. It is safe and efficacious, providing long-term disease control up to 4 years after treatment. However, the small sample size and lack of a control group with only 3 patients from a single institution and relatively short follow-up ranging limit this study. Given its rarity, this disease poses difficulties in performing retrospective studies such as case-control studies or retrospective cohort studies, which provide controls. Despite this, the efficacy of systemic and surgical treatments are reviewed to provide context to subsegmental bland embolization’s efficacy. Performing a larger registry study through the EHE Global Patient Registry, RSNA, or a national cooperative group (Alliance, ECOG/ACRIN) is being proposed. Novel approaches of research recruitment may benefit this unique patient population. For example a global patient-driven Facebook study recruited 115 EHE patients from 20 countries to show the impact of pain, insomnia, and fatigue on daily functioning and psychological distress. 28 Ultimately, this could be a practical treatment option in settings with limited resources. Additional long-term data regarding subsegmental bland embolization outcomes, increasing the sample size, and further investigating the efficacy of repeat treatments would address this study’s limitations.
Conclusion
Subsegmental bland embolization is a safe and effective procedure for stabilizing HEHE not otherwise amenable to surgery or IRE. All patients achieved sustained disease control with majority of lesions (7 out of 8) demonstrating a partial response without major adverse events. Future directions include starting a prospective patient registry and obtaining additional long-term data regarding subsegmental bland embolization outcomes.
Footnotes
Consent for Publication
Written informed consent was obtained from the patients for publication of this case series and any accompanying images. This study was approved 6/17/2024 under IRB 20070111 through the University of Miami.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.
