Abstract
Epstein–Barr virus-associated gastric cancer (EBVaGC) is a subtype of gastric cancer morphologically characterized by massive lymphocyte infiltration. We herein report the short-term outcomes of three rare cases of intramucosal EBVaGC treated with endoscopic submucosal dissection (ESD). Histologically, the lesions exhibited poorly to moderately differentiated tubular adenocarcinoma without lymphovascular invasion, and in situ hybridization revealed EBV-encoded small RNA. Helicobacter pylori infection was not found in any of the lesions. During the 3 to 12 months of follow-up following ESD, none of the ESD-treated patients showed evidence of local recurrence or distant metastases. Essential characteristics of intramucosal EBVaGC may include lymphocyte infiltration into the mucosal stroma or cancer nests as well as the presence of a lace pattern. We believe that ESD might be a safe and suitable treatment method for intramucosal EBVaGC that avoids needless surgery, particularly in patients with severe comorbidities or a high operational risk.
Keywords
Introduction
Gastric cancer (GC) is the fourth-leading cause of cancer-related death worldwide, accounting for 770,000 fatalities per year. 1 The Cancer Genome Atlas classifies GC into four subtypes: Epstein–Barr virus (EBV)-positive GC, GC with microsatellite instability, genomically stable GC, and GC with chromosomal instability. 2 EBV is a human herpesvirus and has been associated with a variety of human cancers, including nasopharyngeal carcinoma, Hodgkin’s disease, and Burkitt’s lymphoma. 3 EBV-associated GC (EBVaGC) occurs with varying degrees of incidence in different countries, representing approximately 5.2% to 12.0% of all GC cases worldwide, and has recently attracted greater scientific attention because of its unique clinicopathological features. 4 Various studies have revealed a higher prevalence of EBVaGC in male patients and younger patients.5,6 Regardless of sex or age, EBVaGC is predominantly located in the middle and upper stomach and is characterized by a superficially depressed or submucosal tumor-like appearance and a deeper invasion depth than that of non-EBVaGC. 7 Furthermore, different reports have suggested a better prognosis for EBVaGC than for other common GCs because of the lower risk of lymph node metastasis (LNM) and higher survival rate of EBVaGC.8–10
With the development of endoscopic technology, endoscopic submucosal dissection (ESD) has become the main endoscopic treatment method for early GC. However, the use of ESD is restricted to patients with GC who have a minimal risk of LNM. Poorly differentiated GC with invasion into the submucosal layer is usually associated with a high risk of LNM and should be treated surgically with regional lymph node dissection. 8 Uemura et al. 11 described the “lace pattern” (LP) as a histological characteristic of EBVaGC. In the LP, cancer cells form irregular net-like structures composed of small glands.11,12 Another unique pathological feature is lymphocyte infiltration into the stroma or cancer nests (LCI). 13 Histological features of both the LP and LCI were observed in the herein-described cases of intramucosal EBVaGC.
The endoscopic characteristics of early EBVaGC are rather poorly understood. In the present study, we retrospectively examined the clinicopathologic, endoscopic, and ultrasonographic characteristics of histopathology-proven intramucosal EBVaGC in three patients. The patients were treated with ESD and followed up over time.
Case report
Method
Endoscopy was performed with the GIF-H260J or GIF-HQ290 endoscope (Olympus Medical, Tokyo, Japan). The video processor was constantly adjusted in the following manner: for white light imaging (WLI), the structure enhancement function was set at the A3/B6 level, and for magnifying endoscopy with narrow-band imaging (ME-NBI), the function was adjusted to the B7/8 level with the NBI color mode fixed at level 1. ME-NBI was used to evaluate the demarcation line (DL) around the lesion at the highest magnification. The depth of infiltration of the lesions was measured by endoscopic ultrasonography (EUS). Yao et al. 14 proposed the vessel plus surface classification system, which was adopted in the present study as a diagnostic system for ME-NBI. The presence or absence of a DL, microvascular pattern (MV), and microsurface pattern (MS) were used for diagnosis. The presence of evident and distinct differences between tumorous and non-tumorous tissue around the entire tumor circumference was classified as DL(+), whereas an absent or ambiguous all-around tumor boundary was classified as DL(−). The MV and MS were categorized as absent, regular, or irregular. In DL(+) patients with an abnormal MV or MS, cancer was typically detected. The detailed features of the three patients described in this report are shown in Table 1.
Detailed features of EBV-associated intramucosal gastric cancer treated by endoscopic submucosal dissection.
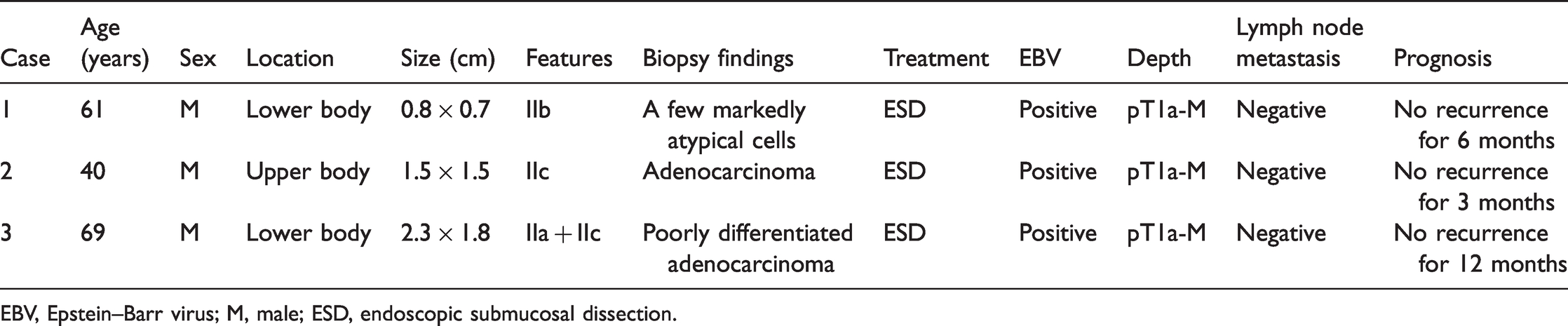
EBV, Epstein–Barr virus; M, male; ESD, endoscopic submucosal dissection.
Case 1
A 61-year-old man was diagnosed with GC despite the absence of obvious symptoms. In our consulting room, physical examination revealed no abnormalities in his abdomen, and he had no family history of GC. He underwent gastroscopy at his routine check-up. WLI revealed a 10-mm reddish lesion with a central slight depression in the lesser curvature of the lower gastric body (Figure 1(a)). Biopsy of the lesion revealed moderately active chronic inflammation, surface erosion and minor intestinal metaplasia, lymphoid tissue hyperplasia in the lamina propria, lymphoid follicle development, and a few atypical cells. ME-NBI revealed DL(+), an irregular MV, and a missing MS in the depressed lesion. However, the presence of mucus on the surface of the lesion slightly affected the observation findings. EUS indicated a hypoechoic mass mostly in the intramucosal layer; the submucosa was normal (Figure 1(b)). Diagnostic ESD was conducted because of high suspicion of early GC (Figure 1(c)). The pathological diagnosis of the ESD-obtained specimen was 8- × 7-mm moderately to poorly differentiated tubular adenocarcinoma with ill-defined tubules and many lymphocytes infiltrating the stroma (Figure 1(d)). The carcinoma was confined in the lamina propria, with a positive horizontal margin and negative vertical margin. No lymphovascular invasion was observed. The nuclei of the carcinoma cells were positive by in situ hybridization targeting EBV-encoded small RNA (EBER) (Figure 1(e)), and the Ki-67 expression level in the cancer cells was 50%. Follow-up gastroscopy 6 months after endoscopic treatment revealed a scar alteration at the ESD site with no signs of recurrence.

Case 1. (a) White light imaging revealed a 10-mm reddish lesion with a central slight depression in the lesser curvature of the lower gastric body. (b) Endoscopic ultrasonography showed a hypoechoic mass located mostly in the intramucosal layer; the submucosa was normal. (c) Diagnostic endoscopic submucosal dissection was conducted because of high suspicion of early gastric cancer. (d) The pathological diagnosis of the specimen was 0.8- × 0.7-cm moderately to partially poorly differentiated tubular adenocarcinoma with lymphocyte infiltration and increased numbers of vessels (hematoxylin and eosin, 100×) and (e) Epstein–Barr virus-encoded RNA in situ hybridization revealed Epstein–Barr virus-positive gastric cancer cells with lymphocyte infiltration into the stroma (100×).
Case 2
Gastroscopy was performed for a 40-year-old man who had no symptoms at his usual check-up. His family members had no history of GC, and his physical examination revealed no evident symptoms of the disease. WLI showed a 20-mm reddish depressed lesion with a surrounding elevation in the lesser curvature of the upper gastric body (Figure 2(a)). Indigo carmine dye clearly revealed the lesion area (Figure 2(b)). ME-NBI revealed DL(+), an uneven MV, and a missing MS. EUS was not performed. Examination of a biopsy specimen led to a pathologic diagnosis of adenocarcinoma. ESD was performed (Figure 2(c)), and the pathological diagnosis of the 15- × 15-mm ESD-obtained tissue specimen was moderately differentiated tubular adenocarcinoma that was confined to the lamina propria without lymphovascular invasion and with negative horizontal and vertical margins (Figure 2(d)). The carcinoma showed an LP with anastomosing and branching glandular structures accompanied by abundant lymphocyte infiltration. EBER in situ hybridization showed that the carcinoma cells were EBV-positive (Figure 2(e)), and the Ki-67 expression level in the cancer cells was 30%. Follow-up gastroscopy at 3 months showed no evidence of recurrence.
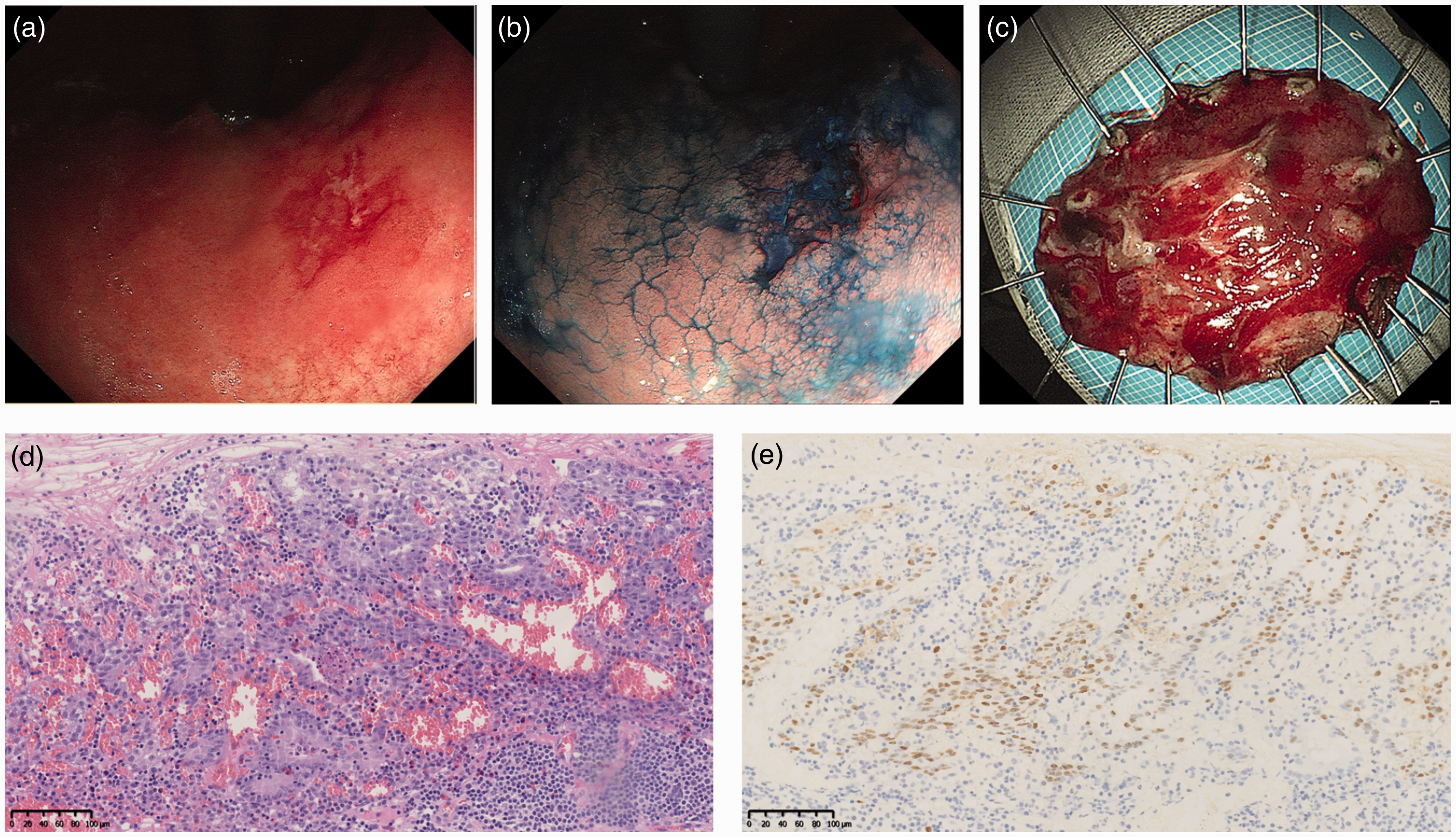
Case 2. (a) White light imaging showed a 20-mm reddish depressed lesion with a surrounding elevation in the lesser curvature of the upper gastric body. (b) The lesion was clearly displayed with indigo carmine dye. (c) Endoscopic submucosal dissection was performed. (d) Histologic examination of the mucosal lesion in the resected tissue showed infiltration of moderately differentiated tubular adenocarcinoma into the mucosal layer without lymphovascular invasion (200×) and (e) Epstein–Barr virus-encoded RNA in situ hybridization revealed Epstein–Barr virus-positive gastric cancer cells with lymphocyte infiltration into the stroma and a lace pattern (200×).
Case 3
A 69-year-old man with a 6-month history of epigastric discomfort underwent upper endoscopy, which revealed a 10-mm raised lesion with a central irregularly depressed region in the greater curvature of the lower gastric body (Figure 3(a)). ME-NBI showed that the lesion was DL(+) and had an irregular MV and no MS (Figure 3(b)). Furthermore, EUS revealed an unevenly moderately hyperechoic lesion situated mostly in the mucosa (Figure 3(c)). Biopsy specimens of the lesion revealed adenocarcinoma with limited differentiation. ESD was performed (Figure 3(d)), and a slightly raised lesion measuring 23 × 18 mm was found on macroscopic inspection of the resected material. The lesion was diagnosed as moderately to poorly differentiated tubular adenocarcinoma with intramucosal invasion (Figure 3(e)) without lymphovascular invasion and with negative horizontal and vertical margins. In situ hybridization revealed that the tumor cells were positive for EBER (Figure 3(f)), and the Ki-67 expression level in the cancer cells was 60%. Follow-up gastroscopy 12 months after endoscopic treatment showed no signs of recurrence.
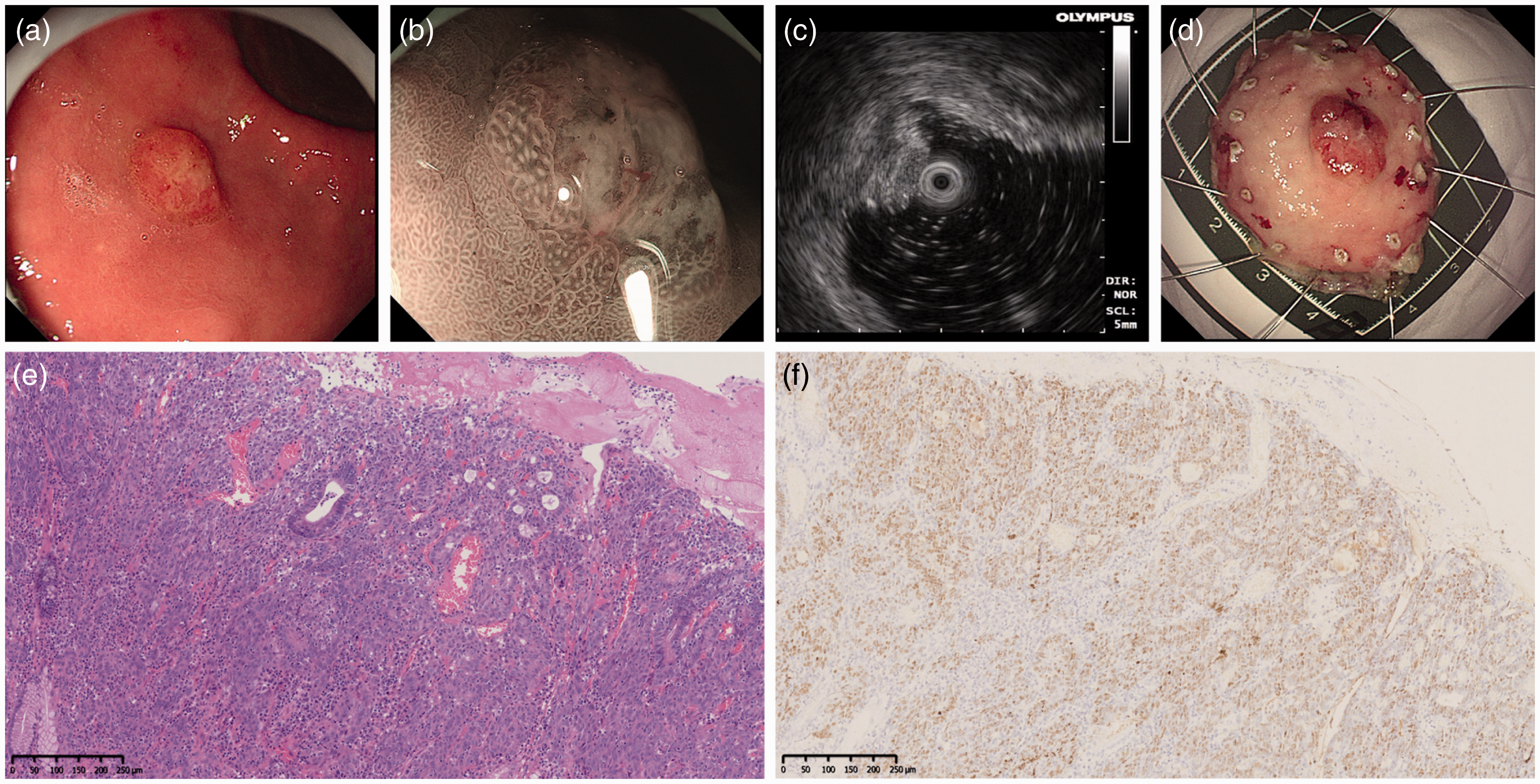
Case 3. (a) Endoscopy revealed a 10-mm raised lesion with a central irregularly depressed region in the greater curvature of the lower gastric body. (b) Magnifying endoscopy with narrow-band imaging revealed a positive demarcation line, irregular microvascular pattern, and no microsurface pattern. (c) Endoscopic ultrasonography revealed an unequal moderately hyperechoic lesion situated mostly in the mucosa. (d) Endoscopic submucosal dissection was performed. (e) A slightly raised lesion measuring 2.3 × 1.8 cm was found on macroscopic inspection of the resected material, comprising moderately to poorly differentiated tubular adenocarcinoma on hematoxylin and eosin staining (100×) and (f) In situ hybridization revealed that the tumor cells were positive for Epstein–Barr virus-encoded RNA and showed lymphocyte infiltration into the stroma (100×).
Discussion
EBVaGC is a form of gastric adenocarcinoma that is generally seen in the upper portion of the stomach, particularly in the gastric body. EBVaGC is a rare disease that mostly affects younger adults. The three patients described in the present report had an average age of 57 years, with the youngest patient being 40 years old. Coinfection with Helicobacter pylori and EBV can hasten the onset of GC. However, H. pylori was negative in all three cases of intramucosal EBVaGC. The patients had either stomach pain or no symptoms at all.
Although the detection of EBER by in situ hybridization ensures a diagnosis of EBVaGC, there are few reports of intramucosal EBVaGC. As a result, intramucosal EBVaGC may be overlooked or undervalued in clinical practice. The characteristic LP shown by ME-NBI, as reported by Kobayashi et al., 12 clearly exhibits an uneven MV, absence of an MS, and a marked DL. Murai et al. 13 found that eight intramucosal early gastric tumors (4.6%) were EBER-positive by in situ hybridization, and none showed LNM. ME-NBI revealed a DL between the non-tumorous mucosa and the carcinoma as well as irregularly dispersed vasculature of varied calibers, supporting the diagnosis of early cancer. The macroscopic types were either depressed (0-IIc) or flat (0-IIb), and the margins were rather obscure. ME-NBI revealed DL(+), an irregular MV, and no MS in all three of our patients. The use of magnification endoscopy can aid in the diagnosis of EBVaGC. The use of EUS to determine the extent of EBVaGC invasion has been reported in only a few previous investigations. Among our patients, EUS showed one hypoechoic and one moderately hyperechoic mass located in the intramucosal layer; EUS was not performed for one patient. The findings in our cases provide preliminary evidence that EUS is useful for determining the depth of EBVaGC lesion invasion.
Radical gastrectomy and lymph node dissection are the most prevalent therapies for advanced EBVaGC. However, some studies have shown a lower frequency of LNM in patients with intramucosal EBVaGC. Murai et al. 13 described eight patients who underwent surgical removal of intramucosal EBVaGC, and none of the patients exhibited LNM. Furthermore, Lim et al. 15 reported 32 cases of intramucosal EBVaGC without LNM. ESD fully removed the intramucosal EBVaGC in all three of our patients. Histological examination of the three mucosal lesions revealed infiltration of poorly to moderately differentiated tubular adenocarcinoma into the mucosal layer without lymphovascular invasion, and EBV positivity in the cancer cells was confirmed by EBER in situ hybridization. A distinctive histological characteristic of EBVaGC, as reported by Uemura et al., 11 is the presence of an LP in which cancer cells form irregular net-like structures fabricated from tiny glands. In the study by Murai et al., 13 only 37.5% (3/8) of the samples had typical LP histology. They also identified another key characteristic of intramucosal EBVaGC: LCI, which might aid in the diagnosis of intramucosal EBVaGC. LCI was found in 87.5 5% (7/8) of their intramucosal EBVaGC samples. 13 In the present study, typical LCI histology was seen in all three cases of intramucosal EBVaGC; only Case 2 showed an obvious LP.
In conclusion, LCI and LP might be essential intramucosal EBVaGC characteristics. Despite the existence of poorly differentiated tubular adenocarcinoma in all three patients, no local recurrence was identified throughout 3 to 12 months of endoscopic follow-up. As a result, we believe that ESD might be a safe and suitable treatment method for intramucosal EBVaGC, thereby avoiding needless surgery, particularly in patients with severe comorbidities or a high operational risk. Endoscopy should be used for extensive follow-up. Further research and evaluation of additional cases are needed to corroborate our findings.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221115162 - Supplemental material for Short-term outcomes of three rare cases of intramucosal Epstein–Barr virus-associated gastric cancer treated with endoscopic submucosal dissection
Supplemental material, sj-pdf-1-imr-10.1177_03000605221115162 for Short-term outcomes of three rare cases of intramucosal Epstein–Barr virus-associated gastric cancer treated with endoscopic submucosal dissection by Rui Cheng, Yao Xu and Bing Yue in Journal of International Medical Research
Footnotes
Acknowledgements
We wish to express our gratitude to the patients for allowing us to use and publish their photographs and for providing us with considerable information about their cases.
Authors’ contributions
Rui Cheng and Yao Xu carried out the studies, participated in the data collection, and drafted the manuscript. Yao Xu and Bing Yue participated in the acquisition, analysis, or interpretation of data and drafted the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available because they contain personally identifiable information; however, they are available from the corresponding author on reasonable request.
Consent for publication
Consent was not applicable because of the retrospective nature of the study and the anonymization of the patients’ data.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Bioethics Committee of Beijing Friendship Hospital, Capital Medical University (Ethics Approval No. 2022-P2-023-01). Written informed consent for participation was obtained from all patients.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Digestive Medical Coordinated Development Center of Beijing Hospitals Authority (Grant No. XXT02).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
