Abstract
Background
Macrophages are a critical component of the innate immune system, derived from monocytes, with significant roles in anti-inflammatory and anti-tumour activities. In the tumour microenvironment, however, macrophages are often reprogrammed into tumour-associated macrophages (TAMs), which promote tumour growth, metastasis, and therapeutic resistance.
Purpose
To review recent advancements in the understanding of macrophage polarisation and reprogramming, highlighting their role in tumour progression and potential as therapeutic targets.
Research Design
This is a review article synthesising findings from recent studies on macrophage polarisation and reprogramming in tumour biology.
Study Sample
Not applicable (review of existing literature).
Data Collection and/or Analysis
Key studies were identified and summarised to explore mechanisms of macrophage polarisation and reprogramming, focusing on M1/M2 polarisation, metabolic and epigenetic changes, and pathway regulation.
Results
Macrophage reprogramming in the tumour microenvironment involves complex mechanisms, including phenotypic and functional alterations. These processes are influenced by M1/M2 polarisation, metabolic and epigenetic reprogramming, and various signalling pathways. TAMs play a pivotal role in tumour progression, metastasis, and therapy resistance, making them prime targets for combination therapies.
Conclusions
Understanding the mechanisms underlying macrophage polarisation and reprogramming offers promising avenues for developing therapies to counteract tumour progression. Future research should focus on translating these insights into clinical applications for effective cancer treatment.
Plain language summary
Macrophages are an important part of the innate immune system that originate from monocytes in the blood and help the host fight inflammation and tumour development. Macrophages are often transformed into tumour-associated macrophages (TAMs) in the tumour microenvironment, which not only promotes tumour growth and metastasis, but also leads to resistance to chemotherapy and immunotherapy, making macrophages attractive targets for combination therapy in oncology. Macrophage reprogramming refers to the modulation of their role in the immune response and the tumour microenvironment by altering their function and phenotype and involves multiple mechanisms, including classical M1/M2 polarisation, metabolic reprogramming, epigenetic regulation, pathway modulation and regulation in the tumour microenvironment. Here, we review recent studies on macrophage polarisation and therapy in tumours, the different mechanisms of macrophage reprogramming and look ahead to the future of macrophage reprogramming.
Introduction
Tumours are complex multicellular ecosystems in which there are multiple interactions between cancer cells, immune cells and the extracellular matrix (ECM), which consists mainly of noncellular components such as laminin, collagen and fibronectin. In contrast, the cellular components surrounding tumour cells include immune cells (eg, lymphocytes, NK cells, macrophages and dendritic cells) and non-immune cells (eg, fibroblasts and vascular endothelial cells). In the early stages of a tumour, cells in the tumour microenvironment, such as infiltrating inflammatory cells, endothelial progenitor cells and cancer-associated fibroblasts, form the basic structure of the tumour ecosystem. This environment promotes the malignant potential of tumour development. Within this ecosystem, the number of innate immune cells is very high, with macrophages being the dominant immune cell population.1,2 Macrophages play a critical role in the tumour microenvironment. Not only are they an important component of the immune system, but they also play a key role in tumour development, metastasis and immune escape. Depending on their origin and activation status, macrophages can be classified into ‘classically activated' macrophages (M1 type) and ‘alternatively activated' macrophages (M2 type), and in tumours the main functions of M1 and M2 types are anti-tumour pro-inflammatory and anti-tumour anti-inflammatory, respectively.1-5 Given the dual role of macrophages in inflammation and tumour biology, targeting macrophages has emerged as a promising strategy for cancer therapy. Current therapeutic approaches focus on depleting TAM or reprogramming its function to enhance anti-tumour immunity.6,7
In the tumour microenvironment, macrophage reprogramming is a key process that influences tumour progression and the immune response. This reprogramming can be achieved through classical gene signalling pathways, metabolic pathways and epigenetic mechanisms. Reprogramming of classical signalling pathways allows macrophages to adapt to different tumour microenvironments and take on different biological functions by altering their gene expression and signalling pathways. For example, signalling pathways such as Notch, mTOR and STAT play a critical role in macrophage polarisation and modulation of these pathways can drive macrophages towards pro-inflammatory or anti-tumour phenotypes.8-13
Metabolic reprogramming of macrophages is also closely linked to their function, for example M1 and M2 macrophages rely on aerobic glycolysis and fatty acid oxidation respectively, and these metabolic properties directly influence their role in the tumour microenvironment.14-17 In the metabolic regulation of macrophages, M1-type macrophages show increased glycolytic activity and pentose phosphate pathway (PPP), accompanied by decreased tricarboxylic acid (TCA) cycle function and reduced mitochondrial oxidative phosphorylation (OXPHOS). In contrast, M2-type macrophages are more dependent on OXPHOS and reduce PPP activity, and both M1- and M2-type macrophages promote fatty acid synthesis. For amino acid metabolism, M1-type macrophages convert
In addition, epigenetic reprogramming further affects macrophage gene expression and function by regulating reversible modifications of RNA. Epigenetic reprogramming of macrophages affects the immune response by altering their functional state through epigenetic mechanisms that regulate gene expression (eg, DNA methylation, histone modification and non-coding RNA regulation). Unlike gene mutations, epigenetic reprogramming does not alter the DNA sequence but regulates macrophage immune function by reversibly altering gene expression patterns. 22 Drug- and nanotechnology-mediated macrophage reprogramming is emerging as a revolutionary strategy for cancer immunotherapy. Through the action of drugs or nanocarriers, macrophage function can be precisely modulated to influence their role in the immune response. Drugs can alter the immune activity of macrophages by modulating their receptors, cytokine secretion and metabolic pathways. For example, certain small molecule immunomodulatory drugs can induce the conversion of macrophages from the immunosuppressive M2 type to the anti-tumour M1 type, while immune checkpoint inhibitors can block immune escape mechanisms in the tumour microenvironment. Nanotechnology, on the other hand, enhances immune clearance by providing an efficient drug delivery system capable of targeting macrophages and precisely modulating their polarisation state. Combination therapies combining nanoparticles with immunomodulatory molecules have shown significant potential, particularly in the treatment of tumours. Although this area is still in the early stages of research, immune reprogramming optimised by nanotechnology offers new possibilities for cancer immunotherapy.23-26 Therefore, an indepth understanding of macrophage reprogramming mechanisms in tumours will provide an important theoretical basis and practical guidance for the development of new anti-tumour therapies.
Macrophages in Tumours
Introduction to Macrophages
Macrophages have a variety of classifications and are divided into 2 main subtypes based on the tissue classification of their origin: 1 belongs to the mononuclear phagocytosis-type system (MPS), where some macrophages differentiate from monocytes released from the bone marrow. Secondly, other tissue macrophages are derived from embryonic progenitor cells and are maintained by in situ self-renewal without replenishment from the bone marrow.3-5 Classification by activation pathway: Macrophages are classified as ‘classically activated' macrophages involved in the Th1 response and ‘alternatively activated' macrophages involved in the Th2 response. 27 Based on their homeostatic activities (host defence, wound healing and immune regulation), macrophages are further classified as classically activated macrophages, wound healing macrophages and regulatory macrophages. 19 Macrophages are phenotypically and functionally heterogeneous, and based on their phenotypic characteristics and local microenvironment, macrophages can also be divided into 2 groups: pro-inflammatory ‘classically activated macrophages (CAM)', ie, M1, and anti-inflammatory ‘alternatively activated macrophages (AAM)', ie, M2.28,29
TAM (tumour-associated macrophages) are the most abundant immune cells in the TME (tumour microenvironment) and are characterised by their polarisation towards AAM-type macrophages, which are not only closely associated with the inflammatory response in the tumour, but also contribute to tumour progression and metastasis.30,31 Macrophages contribute to the survival of tumour cells when they become tumour-associated macrophages (TAMs) in the tumour microenvironment. For example, TAMs will interact with tumour cells through paracrine EGF/CSF1 signalling to promote tumour progression. Cancer cells will secrete CCL2 and CSF1 to recruit macrophages and simultaneously secrete IL-10 and PGE2 to enable immune escape. In the hypoxic tumour microenvironment, TAMs secrete pro-angiogenic factors such as VEGFA, VEGFC and PDGF, and secrete proteases to help cancer cells invade new blood vessels. In addition, TAMs express the Tie2 receptor, which further enhances angiogenesis and inhibits the anti-tumour function of effector T cells through the PD-L1/PD-1 pathway. As the tumour grows, MDSCs and Treg cells interfere with immune function and promote tumour angiogenesis through activation of the TGF-B and CXCL5-CXCR2 pathways. MDSCs also reduce resistance to tumour metastasis through stromal degradation. 2
Macrophage Polarization
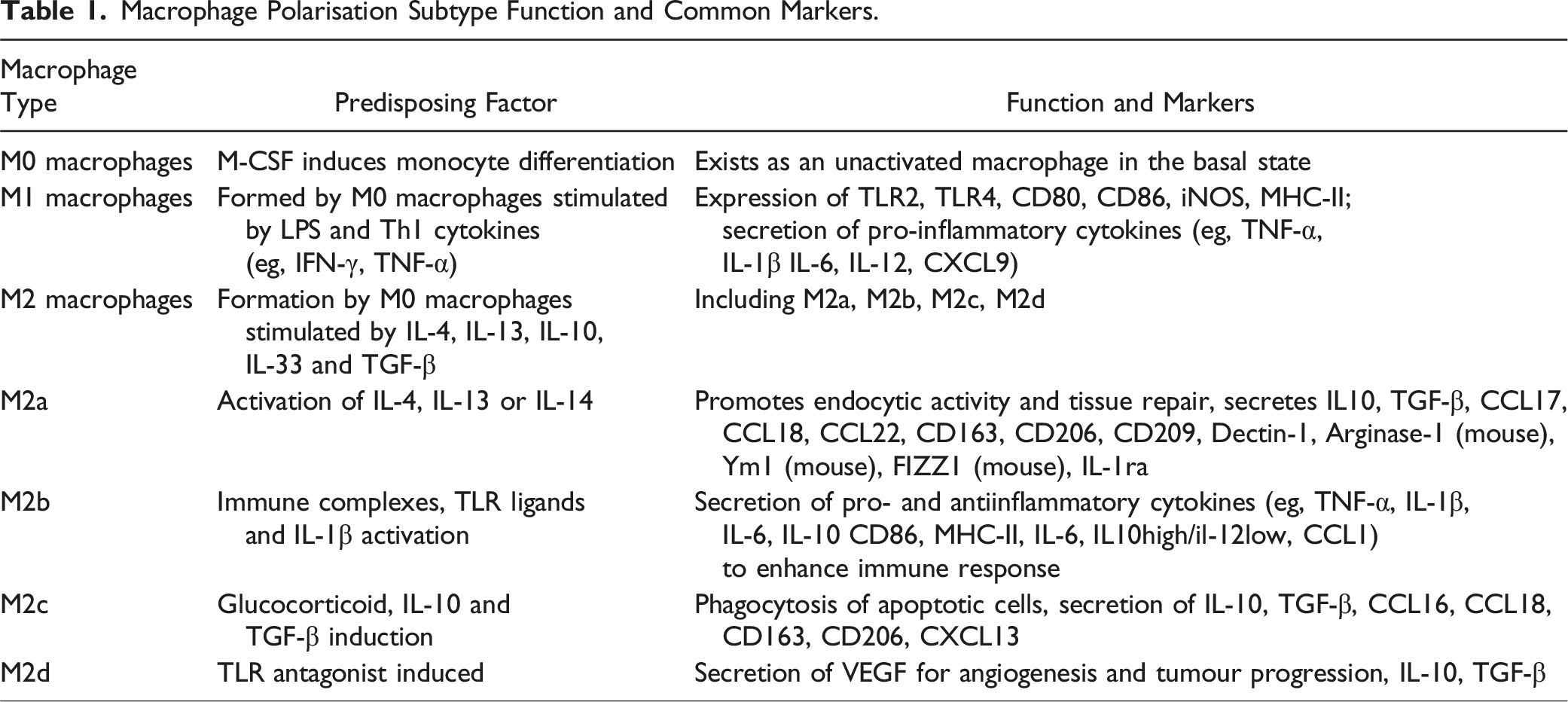
M0 macrophages are induced to differentiate from monocytes by M-CSF (macrophage colonystimulating factor), while DC cells (dendritic cells), which are also monocytes, are induced to differentiate from monocytes by a combination of GM-CSF (granulocyte/macrophage colonystimulating factor) and IL-4.32-35 M0 macrophages form M1 macrophages upon stimulation with LPS and Th1 cytokines (such as IFN-γ and TNF-a).36,37 Whereas M0 macrophages give rise to M2 macrophages upon stimulation with IL-4, IL-13, IL-10, IL-33 and TGF-β.
38
M1 macrophages express TLR2, TLR4, CD80, CD86, iNOS and MHC-II on their surface, which are markers of M1 macrophages. In the nucleus of M1 macrophages, these key transcription factors, such as NF-KB, STAT1, STAT5, IRF3, regulate gene expression in M1 macrophages, which in turn promotes the release of various cytokines and chemokines (eg, TNF-a, IL-1β, IL-6, IL-12, CXCL9, etc.) from M1 macrophages.39-41 M2 cells are classified into M2a, M2b, M2c and M2d macrophages based on the different factors that activate them. IL-4, IL-13 or IL-14 activate M2a macrophages.42-44 M2a macrophages promote enhanced endocytosis activity through increased expression of IL10, TGF-B, CCL17, CCL18 and CCL22 to promote macrophages and promote cell growth and tissue repair.45-47 Immune complexes, TLR ligands and IL-1β activate M2b macrophages,48,49 causing them to secrete pro- and anti-inflammatory cytokines (eg, TNF-a, IL-1B, IL-6 and IL-10), which enhance immune and inflammatory responses. Glucocorticoids, IL-10 and TGF-β induce M2c macrophages, which secrete IL-10, TGF-B, CCL16 and CCL18, and their main function is to phagocytose apoptotic cells.50,51 M2d macrophages are induced by TLR antagonists and secrete VEGF, which promotes angiogenesis and tumour progression. M2 macrophages will express proteins such as mannitol receptor, CD206, CD163, CD209, FIZZ1 and Ym1/2. In M2 macrophages they are regulated by transcription factors such as STAT3, STAT6, IRF4, JMJD3, PPARδ and PPARγ, which in turn express cytokines and chemokines such as IL-10, TGF- β, CCL1, CCL17, etc52-56 (Figure 1). Another marker of M2 macrophages is Arg1, also known as arginase. Unlike M1 macrophages, where The mechanism of action of macrophage polarization. CCL: C-C motif chemokine ligand; cMaf: c-tendon membrane sarcoma; CXCL: C-X-C motif chemokine ligand; FIZZ1: anti-resistin-like protein 1; HIF: hypoxia-inducible factor; iNOS: inducible nitric oxide synthase; IFN-γ: interferon gamma; IL: interleukin; IRF: interferon regulatory factor; JMJD: Jumonji structural domain protein; NF-κB: nuclear factor κB; LPS: lipopolysaccharide; MHC: major histocompatibility complex; PPAR: peroxisome proliferator-activated receptor; STAT: signalling and transcriptional activator; TLR: Toll-like receptor; TNF-α: tumour necrosis factor α; TGF-β : transforming growth factor β; VEGF: vascular endothelial growth factor; Ym1: chitosanase 3-like protein. Created with BioRender.com. Macrophage Polarisation Subtype Function and Common Markers.
Targeting Macrophages in Tumours
Due to the key role of tumour-associated macrophages (TAM) in tumour immunity and therapy, current therapeutic strategies focus on targeting macrophages, which can usually be divided into 2 categories: Depletion of monocytes or TAMs; Reversal of TAM tumour-promoting activity.6,7
Depletion of monocytes or TAMs
Monocyte depletion has become an effective strategy to reduce TAMs. Currently, commonly used methods include bisphosphonates, trabectedin and peptide molecules. Bisphosphonates reduce TAMs by inducing apoptosis in monocytes and macrophages, while inhibiting angiogenesis and tumour cell invasion; Trabectedin not only kills tumour cells by alkylation, but also induces caspase-8-mediated apoptosis in TAMs by stimulating the TNF-related apoptosis-inducing ligand receptor (TRAILR), with specific effects only on monocytes and TAMs, without affecting normal cells; Peptide molecules, on the other hand, optimise therapeutic efficacy by specifically targeting M2-type macrophages, delivering therapeutic agents to TAMs or using them as imaging agents, and can further reprogramme M2-type macrophages into M1-type macrophages with anti-tumour functions.58-61
The depletion of macrophages in the tumour microenvironment is used to treat cancer, either alone or in combination with chemotherapy. The main approach is to inhibit the signalling pathway of colony stimulating factor-1 (CSF-1) and its receptor (CSF-1R), which in turn leads to massive TAM apoptosis. In addition, CSF-1R inhibition in combination with radiotherapy or chemotherapy enhances the T-cell response. In a murine breast cancer model, CSF-1R blockade effectively depleted immunosuppressive TAM while stimulating CD8+ T-cell responses and enhancing the efficacy of paclitaxel, and clinical trials are ongoing. 62 CSF-1 R inhibition also enhances the efficacy of a variety of immunotherapies, including PD-1, CTLA4 antagonists and CD40 agonists.63-67 Early studies have shown that the monoclonal antibody RG7155 significantly reduces tumour size by blocking CSF-1 or IL-34-induced receptor dimerisation, which in turn selectively inhibits CSF-1R signalling, reduces TAM levels and increases CD8+ T cell infiltration. 68 Another way to reduce TAM is to block the recruitment of circulating inflammatory monocytes to the tumour site, thereby cutting off their supply. Recruitment is largely dependent on the CCL2CCR2 pathway, and inhibition of CCR2 retention of monocytes in the bone marrow leads to a reduction in circulating monocytes, impairing their migration to primary and metastatic tumour sites. This process reduces the amount of TAM, which in turn inhibits tumour growth and improves patient survival.69-73
In addition to the above pathways, macrophage recruitment also involves the CXCL12-CXCR4 and Angiopoietin 2 (ANG2)-TIE2 axes. TIE2 + macrophages (TEMs) are bone marrow-derived myeloid cells that are closely associated with the vasculature and significantly influence tumour angiogenesis, playing a key role in tumour metastasis and angiogenesis. It plays a key role in tumour metastasis and angiogenesis, and CXCL12 functions mainly by promoting TEM recruitment. 74 Depletion of TEM promotes vascular destruction and neutralisation of ANG2 enhances the therapeutic effect of VEGFA blockade.75,76
Reversal of TAM tumour-promoting activity
Reversing the pro-tumourigenic activity of TAM is a more desirable strategy, as direct depletion of TAM attenuates its tumour-supporting effects, but also loses its phagocytosis and antigen presenting potential in the tumour microenvironment. To preserve these immune advantages, the current therapeutic focus is on functional reprogramming or repolarisation of TAM to confer antitumour properties and inhibit pro-tumour effects. 77
Tumour-associated macrophages (TAMs) are highly plastic, a property that can be exploited to restore their anti-tumour function. Blocking the CD47/SIRPα axis is a commonly used strategy to enhance macrophage phagocytosis, and anti-CD47 antibodies such as SIRPα-Fc not only enhanced the anti-tumour activity of CAR-T cells, but also inhibited the negative regulatory signals of CD47/SIRPα on CAR-T cell function, providing a new direction for combination therapy. 78 In addition, increased antigen presentation through enhanced CD40/CD40 L interactions can inhibit tumour growth by inducing T-cell activation and promoting the secretion of factors such as NO and TNF-α. 79 Studies have shown that the antitumour effect of platelets is mediated by P2Y12-dependent CD40 L release, which can activate CD8⁺ T cells via CD40 receptors, thereby enhancing antitumour immunity and informing antiplatelet therapy in specific patients. 80 On the other hand, chimeric antigen receptor (CAR) macrophages create a pro-inflammatory milieu through specific phagocytosis or recruitment of other immune cells (eg, dendritic cells, T cells and NK cells) to significantly suppress tumours. 81 CAR macrophages targeting HER2 and CD47 showed antigen-specific phagocytosis in ovarian cancer models and altered the phenotype of TAMs by activating CD8⁺ T cells to secrete anti-tumour factors, ultimately promoting tumour regression. 82
Immune checkpoint inhibitors (CPIs), such as those targeting programmed cell death protein-1 (PD1) and its ligand (PD-L1), have become the standard first-line treatment for metastatic non-small cell lung cancer (NSCLC). The key mechanism of PD-1 resistance is the suppression of immune function in the tumour microenvironment, and therefore remodelling immunity in the tumour microenvironment has become a key strategy to overcome drug resistance. 83 NSCLC has been shown to be resistant to CPI treatment through activation of the TAM receptor family (Tyro3, Axl, MerTK), and TAM receptor inhibitors (TAM RIs) are emerging as a potential means to improve the tumour immune microenvironment. In the treatment of NSCLC, the combination of TAM RI and CPI has shown potential in overcoming drug resistance and improving long-term efficacy. 84 Therefore, targeting TAM receptors and reprogramming their phenotype and function is not only important in tumour therapy, but also provides new ideas for the clinical application of immune responses through modulation of the tumour microenvironment.
Macrophage Reprogramming in Tumours
Reprogramming Classic Gene Signalling Pathways
Classical pathway reprogramming involves cells responding to changes in their internal and external environment by altering gene expression patterns and the activation status of signalling pathways. Classical gene signalling reprogramming of macrophages in tumours occurs mainly through gene silencing, which in turn affects their signalling pathways in cancer, and is mainly represented by signalling pathways such as Notch, mTOR and STAT, which play a key role in macrophage polarisation.8-13 Regulation of specific signalling pathways can promote the shift of macrophages towards pro-inflammatory or anti-tumour phenotypes.
In non-small cell lung cancer (NSCLC), Jagged2 has been found to be highly expressed in NSCLC and correlates with poorer survival and immune escape, while Jagged2 knockdown promotes the expansion of immunostimulatory macrophages and activates T-cell-mediated anti-tumour immune responses. Following Jagged2 deletion, the Notch ligand DLL1/4 is upregulated in tumour cells, which induces the expression of the transcription factor IRF4 through the Notch1/2 signalling pathway, which in turn reprograms macrophages towards an immunostimulatory phenotype. Among these, IRF4 expression is critical for macrophage-mediated anti-tumour effects following Jagged2 deletion. Analysis of clinical samples showed that high Jagged2 expression in tumours inhibits CD3+ T cell infiltration, suggesting that Jagged2 may inhibit anti-tumour immunity in the tumour microenvironment
8
(Figure 2). YTHDF2 is highly expressed in a variety of tumour types, including breast cancer, colon adenocarcinoma and melanoma. The research team established a mouse model by generating TAMs deficient in the YTHDF2 gene, and the results of the study showed a significant reduction in tumour volume and weight, as well as prolonged survival in YTHDF2-deficient mice. It was shown that YTHDF2 expression in TAMs promotes tumour development and that YTHDF2 inhibition can effectively suppress tumour growth. In addition, TAMs deficient in YTHDF2 exhibited enhanced antigen presentation, which could effectively activate CD8+ T cells and enhance their recognition and killing of tumour antigens. YTHDF2 limits the anti-tumour function of tumour associated macrophages (TAMs) by inhibiting the IFNγ-STAT1 signalling pathway. YTHDF2 deletion increased the expression and phosphorylation of STAT1 in macrophages, which enhanced IFNγ-induced expression of downstream anti-tumour genes, and activation of this pathway was the central mechanism by which YTHDF2 deletion led to reprogramming of TAMs to an anti-tumour phenotype. Furthermore, YTHDF2 expression is regulated by IL-10 in the tumour microenvironment, which promotes YTHDF2 transcription through activation of STAT3, suggesting that YTHDF2 expression correlates with pro-tumour polarisation of TAMs
9
(Figure 2). In addition, MTHFD2 has been reported to modulate macrophage polarisation by inhibiting PTEN PIP3 phosphatase activity and enhancing Akt activation. It has been shown that MTHFD2 deficiency impairs the function of M(IL-4) macrophages and reduces their pro-functional effects in vivo.
10
Another study found that TAMs maintain their pro-tumour function in the tumour microenvironment through low activity of the mTORC1 pathway. TSC1 is a repressor of mTORC1, and when TSC1 is repressed in TAMs, the mTORC1 pathway is activated, which then induces TAMs to migrate to the perivascular area of the tumour to inhibit the growth of endothelial progenitor cells, which ultimately leads to a reduction in tumour angiogenesis and hypoxia of the tumour tissue, which in turn triggers tumour cell death. This ultimately leads to reduced tumour angiogenesis and hypoxia of the tumour tissue, which in turn triggers tumour cell death. TSC1deficient TAMs have anti-inflammatory properties and studies have shown that these TAMs exhibit features associated with metabolic reprogramming at the gene expression level, particularly in lysosomal function, lipid metabolism and antioxidant responses, enabling them to adapt to the nutrient- and oxygen-rich conditions of the tumour microenvironment. TAMs lacking TSC1 are metabolically reprogrammed in response to increased mitochondrial respiration and exhibit not only higher metabolic activity but also enhanced phagocytosis, enabling them to engulf dead endothelial cells more efficiently, further enhancing their dominance in the tumour microenvironment. This phagocytosis may also reduce neoangiogenesis in tumours by removing endothelial progenitor cells, ultimately leading to hypoxia in tumour tissue and further inhibiting tumour growth.
11
The mTOR signalling pathway, which regulates cell metabolism, proliferation and survival, plays a key role in metabolic reprogramming of macrophages, and studies in breast cancer have shown that overexpression of MYC leads to an mtorc1-dependent ‘winner' cancer cell state. Low protein diets inhibit tumour growth by suppressing mTORC1 signalling in cancer cells, which in turn inhibits tumour growth and is associated with activation of the transcription factors TFEB, TFE3 and mTORC1 in TAM.
12
Reprogramming of classical signalling pathways in macrophages induced by Jagged2 and YTHDF2 representatives. Created with BioRender.com.
Qiu Li et al extracted the polysaccharide IAPS-2 from hollyhock leaves, which increased the expression of genes associated with M1-type macrophages (eg, NOS2 and MHC II) and downregulated the expression of genes usually considered as M2-type markers (eg, Arg1 and Ym1). IAPS-2 promotes IL-12 secretion and reduces IL-10 levels, and significantly increases INF-γ expression in tumour tissue, which in turn activates Th1-mediated immune responses against tumours. In addition, IAPS-2 enhances immune surveillance in the tumour microenvironment by activating the NF-κB and STAT1 pathways and inhibiting the STAT3 pathway. 13
Metabolic Reprogramming
Glucose
Macrophage function and polarisation are closely linked to altered metabolism. In particular, M1 macrophages and M2 macrophages are mainly dependent on aerobic glycolysis and oxidative metabolism, respectively.
In terms of glucose metabolism, tumour cells in desperate need of more glucose make greater use of aerobic glycolysis as opposed to oxidative phosphorylation. Glycolysis is the metabolism of glucose to pyruvate under anaerobic conditions and the subsequent conversion of pyruvate to lactate catalysed by lactate dehydrogenase. It has been shown that lactate in malignant pleural effusions affects macrophage function by regulating norepinephrine synthesis, which may alter the epigenetic profile of TAM, giving it an M2-type macrophage profile.14-16
In the hypoxic tumour microenvironment (TME), the expression of hypoxia-inducible factor-1a (HIF-1a) is activated as a key transcription factor that regulates the transcription of several genes related to the macrophage glycolysis pathway or glucose transport. 85 HIF-1a increases lactate levels by upregulating the expression of lactate dehydrogenase (LDH, which catalyses the conversion of pyruvate to lactate) and pyruvate dehydrogenase kinase, which promotes the conversion of pyruvate to lactate. Elevated lactate levels in turn promote the expression of vascular endothelial growth factor (VEGF) and induce macrophage polarisation towards the M2 type via
HIF-1a. 86 Lactate produced by tumour cells is converted to pyruvate via the monocarboxylic acid transporter (MCT1) upon entry into macrophages, which inhibits proline hydroxylase (PH), thereby stabilising HIF-1a and triggering the glycolytic pathway. Lactate also promotes tumour growth by activating ERK1/2, STAT3 and HIF-1a, inducing Arg-1 expression in macrophages and inhibiting T-cell function. 87 With increased glycolysis, glucose in the TME decreases, further suppressing T-cell function and exacerbating immunosuppression.88,89 Finally, macrophage energy acquisition shifts from glycolysis to oxidative phosphorylation as lactate accumulates and oxygen decreases. In addition, catalase can reprogramme macrophages through a hypoxic mechanism. 90 TAM differentiates into M2-like TAM by increasing glycolysis and lactate secretion to exert immunosuppressive effects. In nutrient competition, M2-like TAM preferentially uses oxidative phosphorylation to avoid competing with other cells for glucose.91,92 Despite increased oxidative phosphorylation activity, TAM retains a glycolysis-dependent phenotype and is independent of the oxidative phosphorylation and pentose phosphate pathways. 93 Although glycolysis produces less ATP, it is more important for TAM function.
Lipids
TAM accumulates lipids and relies on fatty acid oxidation (FAO) as a major energy source through high expression of the scavenger receptor CD36. High levels of FAO promote mitochondrial oxidative phosphorylation (OXPHOS), which generates reactive oxygen species (ROS), which in turn activates phosphorylation of Janus kinase (JAK) 1 and dephosphorylation of Src homology 2 structural domain phosphatase 1 (SHP1), ultimately activating the STAT6 signalling pathway. This process is a key step in TAM re-education. 17
Receptor-interacting protein kinase 3 deficiency increases fatty acid oxidation (FAO) via the ROS/caspase-1/PPAR pathway, which promotes M2-type polarisation of TAM and enhances immunosuppression. Re-education of TAM and restoration of anti-tumour activity can be achieved by up-regulation of RIPK3 or inhibition of FAO. 94 Activation of RIPK3 balances lipid storage and degradation in tumour cells through a time-dependent mechanism. In addition, the degradation of PPAR-g is dependent on caspase-1, and dysfunctional PPAR-g can be translocated to the mitochondria, which in turn negatively regulates fatty acid oxidation (FAO), induces lipid droplet accumulation and promotes TAM differentiation. 95
Myeloid neutral ceramidase is required for lipid droplet formation and induction of lipolysis, which generates fatty acids for fatty acid oxidation and coordinates macrophage metabolism. The metabolite ceramide leads to reprogramming of macrophages towards immunosuppressive TREM2 + tumour-associated macrophages, which promotes CD8 T cell depletion. 96
Exocytosis of membrane cholesterol in tumour-associated macrophages (TAM) enhances the IL-4 pathway and inhibits interferon (IFN)-γ-induced gene expression, thereby promoting tumour development. In addition, the reduction of intracellular cholesterol contributes to the transformation of macrophages into M2-type TAMs, and thus cholesterol transporters play a key role in macrophage polarisation. 97 The absence of ABC transporter proteins (eg, ABCA1 and ABCG1), which mediate cholesterol efflux, reverses the pro-tumour function of TAMs and reduces tumour progression. 98
In tumour-associated macrophages (TAM), lipid deposition activates genes associated with fatty acid β-oxidation, including CPT1A (carnitine palmitoyltransferase 1a). 99 Fatty acids are mainly derived from the catabolism of triglycerides (TG), which are first converted to diacylglycerol (DG) by adipose triglyceride lipase, then to monoacylglycerol (MG) by hormone-sensitive lipase (HSL), and finally to free fatty acids and glycerol by monoacylglycerol lipase (MAGL/MGLL). 100 Among these, MGLL deficiency is a key factor in lipid accumulation in TAM, leading to activation of M2-type macrophages. In contrast, overexpression of MGLL results in complete inhibition of lipid accumulation in TAM. 101
Phospholipids are a source of lipid second messengers that activate the PI3K/AKT/mTOR pathway, which is associated with tumour progression and poor prognosis. 102 Arachidonic acid (AA) is an important phospholipid involved in inflammation and cancer regulation and is converted to metabolites such as prostaglandins through various pathways. In the tumour microenvironment, changes in AA metabolism are associated with different stages of macrophage development. Prostaglandin E2 (PGE2) is mediated by COX2 production and stimulates the production of tumour promoting chemokines in TAM, conferring a pro-tumour phenotype to TAM.103,104 Relevant studies have shown that SLC3A2 knockdown not only promotes the expression of M1 markers (IL6, IL-12) and inhibits the expression of M2 markers (ARG1, IL-10, CD163) in the tissues of LUAD patients, but also alters the metabolite levels of tumour cells, reducing AA and PGE2 concentrations. In addition, experiments showed that SLC3A2 deficiency inhibited tumour growth and reduced the number of M2 macrophages. Injection of AA reversed this effect. Thus, SLC3A2 may promote tumour progression by promoting M2-type polarisation via AA. 105
The liver is a major organ for ketogenesis, and the key enzyme OXCT1 (3-oxoacyl coenzyme A transferase 1) is responsible for ketone body metabolism in peripheral tissues. In hepatocellular carcinoma (HCC), OXCT1 expression is reactivated to promote tumour progression, and Chu-Xu Zhu et al showed that OXCT1 is highly expressed in liver macrophages at steady state and further increased in tumour-associated macrophages (TAMs). Macrophages lacking OXCT1 reprogrammed TAMs to an anti-tumour phenotype, reducing CD8+ T cell depletion and increasing their toxicity. High OXCT1 expression induces the accumulation of succinate (a by-product of ketolysis) in TAMs, which promotes Arg1 transcription by increasing H3K4me3 levels in the Arg1 promoter. In addition, the OXCT1 inhibitor pimozide inhibits Arg1 expression as well as TAM polarisation towards a pro-tumourigenic phenotype, resulting in reduced CD8+ T cell depletion and slowed tumour growth. 106
Proteins
TAM upregulates the expression of Arg-1 and IDO and promotes glutamine synthesis and catabolism, leading to the accumulation of related metabolites. These changes contribute to the polarisation of TAM into M2-type macrophages. TAM activates the transcription of Arg- 1, which stimulates the catabolism of
TAM depletes tryptophan in the local microenvironment through uptake and metabolism in malignant tumours, leading to immunosuppression. In addition, both TAM and tumour cells can activate IDO (indoleamine 2,3-dioxygenase), which depletes tryptophan and accumulates its metabolites (eg, kynurenine), creating an immunosuppressive environment. 111 Activation of IDO can be induced by tumour necrosis factor-a, IFN-g or prostaglandins, and TAM tends to have an M2-like phenotype when IDO is overexpressed, whereas silencing of IDO reveals anti-tumour macrophage characteristics. 112
The role of glutamate and glutamine metabolism in TAM in their functional phenotypes is less well understood, but they provide energy for TAM and tumour cells and are involved in the polarisation of TAM towards an immunosuppressive phenotype. Glutamine synthetase (GS) maintains the M2like TAM phenotype by converting glutamate to glutamine, and GS inhibition reverses TAM to a pro-inflammatory M1-like phenotype.113-115 In glioblastoma studies, GS upregulation was found to contribute to the polarisation of TAM towards a pro-tumourigenic phenotype. 116 The a-KG produced by glutamine catabolism is essential for M2 activation, whereas a low a-KG/succinate ratio promotes a pro-inflammatory phenotype in M1-like macrophages. 117
Rag GTPase regulates mTORC1 signalling in TAM by sensing dietary cytoplasmic amino acids. Deficiency of GATOR1 in TAM accelerates tumour growth under low protein dietary conditions, whereas deletion of FLCN or Rag GTPase slows tumour growth under a normal protein diet. 118
Iron and nucleotides
Iron accumulation in tumour cells promotes aggressiveness, and in the tumour microenvironment, TAM is the major source of iron. Iron metabolism influences TAM polarisation.119,120 In particular, intracellular iron deficiency activates the hypoxia-inducible factor (HIF), high intracellular iron concentrations promote an M1-like phenotype, and M2-like macrophages tend to export iron.121-124 In addition, adenosine inhibits TAM function in the tumour microenvironment, affecting its phagocytosis and cytokine production, and the adenosine receptor A2A affects tumour progression by upregulating CXCL5 secretion via the NF-kB pathway. 125
Epigenetic Reprogramming
Epigenetic reprogramming of macrophages regulates their gene expression and function through reversible RNA modifications, which are particularly important in the tumour microenvironment (TME). n6-methyladenosine (m6A) is a reversible mRNA modification that is widely distributed in eukaryotic cells and plays an important role in regulating a variety of biological processes. Aberrant m6A modifications are closely associated with a variety of cancers, and in different tumours, m6A modifications can either promote or inhibit cancer progression. 126 METTL3 is an RNA methyltransferase responsible for adding N6-methyladenosine (m6A) modifications to RNA molecules. It normally forms a complex with METTL14 and other cofactors (eg, WTAP) to complete the modification near the 3′UTR and termination codon of the RNA. This m6A modification has an important impact on RNA stability, translation efficiency and splicing, thereby regulating cellular function and the development of several diseases, and its role in cancer is particularly critical. 127 In the tumour microenvironment (TME), METTL3 deficiency remodels the tumour immune environment, which in turn drives immunosuppression by increasing the infiltration of M1 vs M2-like tumour-associated macrophages (TAMs) and regulatory T cells (Tregs). METTL3 deficiency also activates the NF-κB and STAT3 pathways, further promoting macrophage M1 or M2 polarisation. In addition, METTL3 deficiency leads to downregulation of Spred2 (a target gene of m6A) expression and reduces its translational efficiency via YTHDF1, which in turn increases the phosphorylation levels of ERK, NF-κB and STAT3. 128 In addition, YTHDF2 has been shown to regulate the stability of STAT1 mRNA by binding to m6A modification sites, and deletion of YTHDF2 increases the stability of STAT1 mRNA, which in turn increases the anti-tumour capacity of TAMs. 129 Epigenetic reprogramming of macrophages significantly affects their function in the tumour microenvironment through the regulation of m6A modifications and associated signalling pathways. Changes in key molecules such as METTL3, YTHDF1 and YTHDF2 can drive macrophage polarisation and enhance immunosuppressive or anti-tumour responses.
miR-1322 is a key miRNA that regulates the expression of CCL20. In colorectal cancer (CRC), the oral pathogenic bacterium
Other studies have reported that inhibition of histone deacetylase (HDAC) epigenetic reprogramming of macrophages induces T cell immune responses. Studies have shown that selective class IIa HDAC inhibitors induce an anti-tumour macrophage phenotype, promote T-cell immune responses and increase responses to chemotherapy and immune checkpoint blockade in breast cancer models. 131
Drug- and Nanotherapeutically-Mediated Reprogramming
Macrophage reprogramming is emerging as an innovative strategy for cancer immunotherapy. Precise modulation of macrophage polarisation status in the tumour microenvironment by drugs and nanomaterials can effectively enhance anti-tumour immune responses and reduce immunosuppression. Most of these therapeutic approaches focus on remodelling the tumour microenvironment (TME) by switching tumour-associated macrophages (TAMs) from an immunosuppressive M2 phenotype to a pro-inflammatory M1 phenotype.
Dickkopf-1 (DKK1) is a secreted protein that promotes tumour progression by interacting with the CKAP4 protein on the surface of macrophages, activating the PI3K-AKT pathway, inducing macrophage polarisation towards an immunosuppressive M2 phenotype and decreasing CD8+ T-cell and NK-cell activity. Tumour-associated macrophages (TAMs) in a murine gastric cancer model were reprogrammed to the M1 phenotype with enhanced CD8+ T-cell and NK-cell activity after treatment with a DKK1 monoclonal antibody (mDKN-01), and combination treatment with DKK1 blockade and a PD-1 antibody showed a stronger tumour-suppressing effect than monotherapy. 132
Triple-negative breast cancer (TNBC) is an aggressive breast cancer with a poor prognosis for which treatment is based on immune checkpoint inhibitors (ICIs) and chemotherapy with limited efficacy. Eganelisib, a selective PI3K-γ inhibitor, has been shown in preclinical studies to reduce tumour recruitment of myeloid cells by switching tumour-associated macrophages (TAMs) from an immunosuppressive to an immunoactivated phenotype, thereby enhancing ICI activity and improving the tumour microenvironment (TME). A phase II clinical trial of eganelisib in combination with the anti-PD-L1 drug alizumab and albumin-bound paclitaxel for the treatment of metastatic TNBC (MARIO-3, NCT03961698) is currently underway to evaluate its efficacy in first-line therapy. 133 In bladder cancer studies, type III interferon (IFN-λ) was found to promote the infiltration of CD8+ T cells, Th1 cells, natural killer (NK) cells and pro-inflammatory macrophages, while reducing the accumulation of neutrophils. IFN-λ3 induced a shift of tumour-associated macrophages (TAMs) towards M1-type macrophages by upregulating phagocytosis and pro-inflammatory gene expression, enhancing their phagocytic activity and reducing the proportion of M2-type macrophages. In addition, MB49-IFN-λ3-expressing cells significantly inhibited tumour growth and increased apoptosis (increased caspase-3 expression). IFN-λ3 increased the sensitivity of bladder cancer to PD-L1 antibody treatment by activating T-cell immune responses, demonstrating the potential for synergistic enhancement of anti-tumour immunity. 134 Gold nanoclusters (GNA) effectively reprogrammed macrophage polarisation via the Nrf2 pathway, and GNA can be used as a low-toxicity nanomedicine for the treatment of macrophage-driven inflammatory diseases and related cancers, showing promising therapeutic effects in IBD (inflammatory bowel disease) and CRC (colitis-associated colorectal cancer). 135
An innovative single agent therapy, exoASO-STAT6, reprogrammed tumour-associated macrophages (TAMs) into M1-type macrophages by exosomally delivered ASO targeting and silencing the STAT6 gene, significantly enhancing the anti-tumour immune response. ExoASOSTAT6 acts as a key transcription factor for the polarisation of TAMs to the M2-type and its silencing reduces the expression of M2-type markers (eg, Arg1, CD206) and upregulates M1-type markers (eg, NOS2, IL-12), thereby altering the tumour microenvironment. Its silencing reduces the expression of M2-type markers (eg, Arg1, CD206) and upregulates M1-type markers (eg, NOS2, IL-12), thereby altering the tumour microenvironment. ExoASO-STAT6 inhibits the M2 phenotype and activates M1-type macrophages through STAT6 gene silencing, demonstrating broad anti-cancer potential. In addition, exoASO-STAT6 monotherapy was more effective than PD1 antibody and CSF1R inhibitor, while free ASO showed almost no significant efficacy. And in combination with PD-1 antibody, tumour burden was further reduced by 85%, demonstrating a strong synergistic anti-tumour effect. 136
In addition to the drug aspect, several research teams have developed innovative nanomedicine systems to enhance anti-tumour effects by reprogramming tumour-associated macrophages (TAMs) and improving the tumour microenvironment (TME). These include: (1) Hb-DOXM@Cel achieves reprogramming of tumour associated macrophages (TAMs) and remodelling of the tumour microenvironment (TME) through targeted delivery of oxygen, chemotherapeutic agents and immunomodulators. Hb-DOXM@Cel uses haemoglobin (Hb) as an oxygen carrier to alleviate tumour hypoxia, reduce recruitment of M2type TAMs and dramatically inhibit 4T1 COX2 and PGE2 expression in cells. Its high accumulation and prolonged retention at the tumour site can effectively promote T-cell infiltration, reduce the proportion of M2-type TAMs and enhance the anti-tumour immune response. Compared with chemotherapeutic agents alone, Hb-DOXM@Cel showed more significant anti-tumour effects, promoted apoptosis or necrosis of tumour cells, and enhanced the effect of DOX-based chemotherapy by synergistically transforming immunosuppressive TME into an immunostimulatory environment through synergistic TAM reprogramming
137
; (2) NPPDT@Reg was used to deliver the vascular normalising agent Reglanis (Reg). Under 808 nm laser irradiation, the system achieves efficient release of Reg, improving tumour hypoxia and facilitating more oxygen and drug entry into the tumour through vascular normalisation. NP-PDT@Reg not only generates more reactive oxygen species (ROS) to kill tumour cells, but also induces immunogenic cell death (ICD), which activates anti-tumour immune responses. In addition, Reg further reverses the immunosuppressive tumour microenvironment (TME) by reprogramming tumour-associated macrophages (TAMs) from the pro-tumour M2 type to the anti-tumour M1 type, thereby enhancing overall anti-tumour efficacy
138
; (3) HMMDN-Met@PM specifically targets M2-type tumour-associated macrophages (TAMs) for repolarisation. The nanoparticles are based on hollow porous manganese dioxide nanoparticles (HMMDN) extensively loaded with metformin and modified with M2-type macrophage targeting peptide (M2pep) on their surface to ensure precise drug delivery to M2-type TAMs. Ex vivo and in vivo experiments showed that HMMDN-Met@PM reduced the expression of M2-type macrophage markers and inflammatory factors (eg, CD206, Arg-1 and IL-10) while increasing the expression of M1-type markers and pro-inflammatory factors (eg, CD80, TNF-α and iNOS), successfully repolarising M2-type macrophages to M1-type, which in turn effectively inhibited tumour growth
139
; (4) HBPdC This material is capable of generating reactive oxygen species (ROS) in the tumour microenvironment, triggering mitochondrial dysfunction and promoting tumour cell apoptosis. HBPdC-induced ROS accumulation not only induces tumour-associated macrophages (TAMs) to polarise towards a pro-inflammatory M1 phenotype and enhance their phagocytosis, but also exhibits excellent anti-tumour effects in 4T1 tumour models, including inhibition of tumour growth, reduction of tumour burden, reduction of inflammatory factor levels and prolongation of survival. Compared to transient antibody blockade, HBPdC provides a durable gene editing approach. In addition, the team found that knockdown of CD47 and APMAP using CRISPR technology further enhanced phagocytosis of TAMs, improved therapeutic efficacy and reduced tumour metastasis; (5) R848@M2pep-MPAFP is able to target immunosuppressive M2-type tumour-associated macrophages (TAMs) and reprogram them into M1-type macrophages. These reprogrammed TAMs not only act as antigen-presenting cells, delivering AFP antigens to activate anti-tumour immunity in CD8+ T cells, but also maintain and promote the differentiation and maintenance of stem cell-like CD8+ T cells by providing ecological niche support within the tumour (Figure 3). Nanomaterial-mediated reprogramming of macrophages. Created with BioRender.com.
Future Perspectives: The Potential of Macrophage Reprogramming in Tumour Therapy
Macrophage reprogramming in the tumour microenvironment has been shown to be a key factor in regulating tumour progression and immune response. Future research can focus on the following directions to advance the application of macrophage reprogramming in tumour therapy: (1) Continue to investigate in depth how signalling pathways such as Notch, mTOR, STAT, etc. affect macrophage polarisation and function. Using gene-editing technologies such as CRISPR-Cas9 to systematically analyse the role of these signalling pathways in different tumour types will help to identify new therapeutic targets; (2) Alterations in macrophage metabolism have important implications for macrophage function and polarisation. Future research should continue to focus on how to regulate macrophage function by interfering with pathways such as glucose metabolism, lipid metabolism and amino acid metabolism; (3) Continue to expand research in epigenetics and develop drugs or therapeutic strategies that target epigenetic regulation to open new avenues for immunotherapy; (4) As nanotechnology advances, strategies for using nanocarriers to deliver specific genes or drugs to reprogramme macrophages will become more sophisticated. Future clinical trials will test the safety and efficacy of these new therapies; (5) Through the combined use of gene editing, metabolic intervention, epigenetic modulation and nanotechnology, future research will hopefully lead to effective remodelling of the tumour microenvironment, thereby improving the success rate of cancer treatment.
However, macrophage reprogramming therapies still face many challenges in clinical translation. (1) Drug tolerance is the main challenge, as macrophages are highly plastic and their phenotype is regulated by the tumour microenvironment, and they are prone to regain pro-tumour functions through other pathways or trigger negative feedback loops due to prolonged use of a single drug. In this context, combination therapies (eg, immune checkpoint inhibitors, chemotherapy or multi-pathway targeting strategies) have emerged as a solution to slow down the development of tolerance by inhibiting pro-tumour signals at multiple levels; (2) Uncertainty in efficacy arises from significant individual differences in patient TAM phenotypes, as well as interference from other cells in the complex tumour microenvironment. Precision medicine combines multi-omics data to analyse patient TAM characteristics and develop personalised therapies, while optimising treatment by monitoring phenotypic changes in macrophages in real time. In vitro models that mimic the tumour microenvironment can also be used to screen potential drugs and study cellular interactions in detail; (3) Lack of reliable biomarkers makes predicting and monitoring efficacy challenging. By combining single-cell histology and liquid biopsy techniques, new functional markers, such as secreted factors or metabolites, can be used to dynamically monitor efficacy and optimise treatment strategies.
Although macrophage reprogramming has significant potential in cancer immunotherapy, it may face a number of safety issues in practical clinical applications: (1) excessive or dysregulated immune responses may occur, in particular excessive release of pro-inflammatory cytokines during the reprogramming process, which may lead to cytokine storms or immune-related organ damage. The intensity of the immune response can be regulated by precisely controlling the macrophage reprogramming process, monitoring the immune response in real time and using targeted inhibitors (eg, anti-IL-6 antibodies or immune checkpoint inhibitors); (2) Tumours can resist treatment through immune escape mechanisms, and immunosuppressive factors in the tumour microenvironment, such as TGF-β and IL-10, can limit macrophage function or direct their immunosuppressive polarisation, thereby attenuating the therapeutic effect. Therefore, strategies combining immune checkpoint inhibitors, anti-tumour vaccines or modulation of the tumour microenvironment may enhance the immunotherapeutic effect and prevent immune escape; (3) The small-molecule drugs or techniques used may induce local or systemic toxicity affecting non-tumour tissues or normal immune cells, so the use of targeted drug delivery systems (eg, nanocarriers or antibody-coupled drugs) and rigorous drug safety assessments may reduce toxic responses. Macrophage dysfunction or instability is also a potential problem, and excessive or insufficient reprogramming may impair their immune function or even trigger immune-related diseases. Adaptable reprogramming methods and long-term immune monitoring could be developed to maintain stable macrophage function; (4) Due to differences in patients’ immune systems, treatment effects may be heterogeneous due to differences in immune tolerance, and individualised treatment and immune preconditioning measures (eg, vaccination) may enhance patients' immune response; (5) Although macrophage reprogramming can provide significant anti-tumour effects in the short term, tumours can develop resistance over time through mechanisms such as immune escape or epigenetic changes, and the use of dynamic treatment regimens and combinations of therapies can reduce the risk of resistance developing.
Overall, future research should focus on the following directions to promote clinical translation and application in this field. First, the role of key signalling pathways in regulating macrophage polarisation and function needs to be explored in depth, and the mechanisms of these pathways need to be systematically analysed in conjunction with gene editing technologies (eg, CRISPR-Cas9) to provide new targets for precision therapy. In addition, it is a key area to investigate the effects of other metabolic changes in macrophages on their polarisation and function, and to enhance their anti-tumour effects through metabolic intervention. Meanwhile, epigenetic research needs to be expanded to develop specific drugs that target epigenetic modifications to precisely regulate macrophage function. The application of nanotechnology will also be a focus of research to efficiently reprogramme macrophages through targeted delivery of genes or drugs and to explore their clinical safety and efficacy in combination with immunotherapy. In the future, multi-dimensional strategies such as gene editing, metabolic intervention, epigenetic regulation and nanotechnology will need to be integrated to reshape the tumour microenvironment to improve therapeutic efficacy and reduce immune escape. Meanwhile, the development of personalised precision medicine will also contribute to individualised treatment through single-cell sequencing and multi-omics analysis to map the characteristics of patients’ macrophages and optimise treatment options in real time. Finally, efficacy and safety will be further improved by designing specific drug delivery systems and implementing controlled reprogramming methods to control immune imbalances and side effects during treatment, and by designing dynamically adapted combination therapies that target immune escape and resistance mechanisms. By overcoming the challenges outlined above, macrophage reprogramming is expected to become an important tool for tumour therapy in the clinic, providing patients with precise and efficient treatment options.
Limitations of the Review
Although this review summarises the functions of macrophages in the tumour microenvironment and the potential of their reprogramming in anti-tumour therapy, the following limitations remain. First, the mechanisms of macrophage polarisation and reprogramming are very complex, involving multiple aspects such as gene signalling pathways, metabolic regulation and epigenetic inheritance, etc. Current studies have mostly focused on the analysis of a single mechanism, while systematic studies of the synergistic effects of multiple mechanisms have been less frequent, which has limited the comprehensive understanding of the dynamic changes in macrophage function. Secondly, the vast majority of studies have been conducted in vitro or in animal models, which to some extent may not fully reflect the complexity of the human tumour microenvironment and may lead to limitations of experimental results in clinical applications. In addition, drug- and nanotechnology-mediated macrophage reprogramming is theoretically promising, but its long-term safety, target specificity and individual differences in therapeutic effects need to be further validated and optimised. Finally, with the development of tumour therapies, the role of macrophages in combination therapy strategies needs to be further explored to clarify the optimal timing and modalities of their application in different treatment modalities. These limitations suggest that future studies need to integrate multidisciplinary resources to develop more precise and effective therapeutic strategies to better promote the clinical translation of macrophages in tumour therapy.
Conclusion
In this review, we discuss the role of macrophages in the tumour microenvironment and their potential therapeutic strategies. Macrophages, as important immune cells, play a dual role in tumour initiation, progression and metastasis, both promoting tumour progression and participating in antitumour immune responses. By studying the mechanisms of macrophage polarisation, we have found that macrophages can be converted into different subtypes under different microenvironments to form tumour-associated macrophages (TAMs), which play a key role in tumour growth and metastasis. Therapeutic strategies against TAMs include depletion and reversal of their tumour-promoting activity. Depletion of TAMs has been shown to be effective in several studies, particularly by inhibiting signalling pathways such as CSF-1 and CCR2, which can significantly reduce the number of TAMs and thus inhibit tumour growth. However, reversing the tumour-promoting properties of TAMs is considered a more desirable therapeutic direction, by reprogramming TAMs into cells with anti-tumour properties, thereby enhancing the immune response. In addition, studies of classical gene signalling pathways, metabolic reprogramming and epigenetic reprogramming have provided new perspectives on macrophage reprogramming in the tumour microenvironment, and drug- and nanotherapeutics-mediated reprogramming provide important theoretical foundations and practical guidance for the development of new immunotherapies. Future studies should further explore the mechanism of interaction between macrophages and tumour cells and, based on the current macrophage reprogramming, explore in depth and develop new areas of reprogramming, which in turn will provide more effective strategies for tumour diagnosis and treatment.
Footnotes
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nantong University’s 2023 Academic Level Research Project (Special Project of Yancheng Third Institute) (YXY-Z2023008), 2021 Yancheng Medical Science and Technology Development Plan Project (YK2021060), The Special Funds for Science Development of the Clinical Teaching Hospitals of Jiangsu Vocational College of Medicine (20209113), The Special Funds for Science Development of the Clinical Teaching Hospitals of Jiangsu Vocational College of Medicine (20219105), 2021 Jiangsu Provincial Health and Health Commission Medical Research Guidance Project (Z2021087).
Ethical Statement
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
