Abstract
Many aspects of cancer can be explained utilizing well-defined ecological principles. Applying these principles to cancer, cancer cells are an invasive species to a healthy organ ecosystem. In their capacity as ecosystem engineers, cancer cells release cytokines that recruit monocytes to the tumor and polarize them to M2-like protumor macrophages. Macrophages, recruited by the cancer cells, act as a secondary invasive species. The ecosystem engineering functions of M2-macrophages in turn support and stimulate cancer cell survival and proliferation. The cooperative ecosystem engineering of both the primary invasive species of the cancer cell and the secondary invasive species of the M2-macrophage thus creates a vicious cycle of tumor promotion. Targeting a specific aspect of this tumor-promoting ecosystem engineering, such as blocking efferocytosis by M2-like macrophages, may improve the response to standard-of-care anticancer therapies. This strategy has the potential to redirect cooperative protumor ecosystem engineering toward an antitumor ecosystem engineering strategy.
Introduction
In 2019, it is estimated that about 1,700,000 new cases of cancer will be diagnosed, and there will be approximately 600,000 cancer-related deaths in the United States. 1 New paradigms for understanding cancer and cancer lethality are urgently needed. Many aspects of tumor biology are analogous to well-established principles in ecology (Table 1). A tumor itself is not simply an isolated mass of cancer cells but contains many different cell types or “species” that influence each other and the life history of the cancer and therefore the patient. This tumor microenvironment parallels an ecosystem with many interacting cell types and components. As tumors grow, they have substantial impacts on their surrounding local ecosystem and, eventually, the entire human body. Mechanisms including dysregulated cell signaling, cellular proliferation, and recruitment of tumor-promoting host cells lead to ecosystem and ultimately biosphere collapse, resulting in the death of the patient. By applying ecological principles to understand the processes of cancer development and progression, we present a novel strategy to treat cancer: disrupting tumor-promoting ecosystem engineering.
Ecological Definitions in Cancer Biology.

Cancer as an Invasive Ecosystem Engineer
An invasive species is a non-native species that enters an ecosystem. Upon entering the new habitat, these invaders eventually become a destructive species of the ecosystem. These invaders can be recruited to the ecosystem by a number of factors including a need for resources, population growth, or desirable characteristics of the ecosystem to be invaded. 2 -4 Invasive species disrupt homeostasis in the ecosystem, altering the community structure and competing with native species for nutrients and space, ultimately harming the native species. After a lag period, successful invaders have high reproduction and have the ability to survive in a heterogeneous changing environment. 5,6 For example, the European green crab is a native species along the coasts of Europe but is invasive to coasts around the world such as those along the United States and South America. These invasive crabs prey on shellfish and oysters, causing destruction to the ecosystem by competing with native species such as the Dungeness crab and American lobster for resources. 7 -10
Many invasive species are also ecosystem engineers that exert substantial changes to an ecosystem. Ecosystem engineers modify their habitat and by doing so affect other species and the entire adaptive landscape. Allogenic engineers change the ecosystem by mechanically altering the habitat, while autogenic engineers modify their habitat simply by modifying themselves. Beavers are perhaps the most well-known invasive allogenic ecosystem engineer. When beavers build a dam, they exert a substantial and catastrophic influence on the habitat. Both the tree clear-cutting and the resulting redirected water flow have large effects on vegetation and wildlife in the surrounding areas. An autogenic engineer, the kudzu vine is an invasive species in parts of Asia and the Pacific islands as well as the southeastern United States: “the vine that ate the South.” Upon invasion, the kudzu vine spreads and overcomes the native habitat, outcompeting other plants for resources. 11 By growing as a vine on native trees and man-made structures, the kudzu vine blocks sunlight and kills the trees it climbs while also introducing new habitats for small animals, effectively destroying the native ecosystem.
During tumorigenesis, cancer cells are invasive ecosystem engineers. 12,13 Many of the “Hallmarks of Cancer” such as high proliferative rate, evading predation, and resisting death are parallel to the essential characteristics of an invasive species: rapid growth and reproduction, phenotypic plasticity, and high tolerance for different habitats. 14,15 Once the cancer cells are established, long before clinical detection, they engage in ecosystem engineering to modify their environment to favor conditions for their own growth and survival and outcompete native species. As autogenic engineers, the cancer cells proliferate and the tumor grows. For this rapid growth, they consume high amounts of oxygen and nutrients leading to hypoxia and nutrient poverty, generating a “cancer swamp.” 12,16,17 In addition, cancer cells are allogenic engineers that secrete matrix metalloproteinases (MMPs) that enzymatically break down the extracellular matrix and secrete cytokines and chemokines that induce angiogenesis and recruit other species to the invaded and engineered ecosystem: the tumor microenvironment. Through their autogenic and allogenic engineering, cancer cells disrupt the native ecosystem of the healthy organ.
Secondary Invasion of the Tumor Ecosystem
Secondary invasive species follow and depend on invasion of a primary invasive species. These secondary invaders are drawn to the ecosystem due to the changes made by the primary invaders. For example, the

Cancer cells and M2 macrophages as invasive ecosystem engineers. A, Following volcanic eruptions in Hawaii,
In cancer, one of the most common and destructive secondary invaders is protumor M2-like macrophages, recruited to the tumor microenvironment by the cancer cells and the generation of the cancer swamp (Figure 1B). Macrophages are innate immune cells with roles in antigen presentation, phagocytosis, and modifying different types of immune responses. 21,22 In cancer, these cells can play tumor-promoting roles and high macrophage infiltrate correlates with poor prognosis. 23 -25 After differentiation from their monocyte precursor, macrophages polarize toward an M1-like or M2-like phenotype. These phenotypes are highly plastic and can change when the macrophage encounters different stimuli. While M1 and M2 are simplified subtypes used throughout the field, macrophage phenotypes and functions exist on a continuous spectrum and have both M1-like and M2-like characteristics. M1-like macrophages are part of the Th1 immune response and exert an antitumor effect. In contrast, M2-like macrophages are involved in the Th2 immune response and have protumor roles.
Allogenic engineering by cancer cells, specifically secretion of the cytokines macrophage colony stimulating factor and C-C motif chemokine ligand 2 (CCL2), promotes trafficking of monocytes to the tumor. 26 -29 After recruitment and intravasation to the tumor, monocytes differentiate into macrophages. Cytokines such as interleukin (IL)-4 and IL-13 released by cancer cells and other immune cells in the ecosystem polarize these recruited macrophages, as well as tissue-resident macrophages, to the M2 tumor-promoting subtype. These M2-like macrophages further disrupt the healthy organ ecosystem and promote cancer growth.
Cooperative Invasive Ecosystem Engineers
Cancer cells and M2-like macrophages occupy a substantial part of the tumor habitat and have marked influence on the tumor ecosystem as a whole. 30 -32 Cancer cells and macrophages support each other by secreting factors that stimulate the others’ growth, survival, and, importantly, support tumor-promoting ecosystem engineering (Figure 2A). The ecosystem engineering of the cancer cells, the primary invader population, recruits secondary invasion of M2 macrophages by releasing cytokines that recruit and polarize M2 macrophages. In turn, M2 macrophage ecosystem engineering also supports the cancer cell population. For example, M2 macrophages alter resource availability by secreting growth factors such as epidermal growth factor and hepatocyte growth factor that fuel cancer cell proliferation. 33,34 M2 macrophages impact noncancer cell components of the tumor ecosystem by altering the physical habitat and community structure. They also release MMPs that induce extracellular matrix remodeling, physically altering abiotic components of the habitat to permit cancer cell movement.
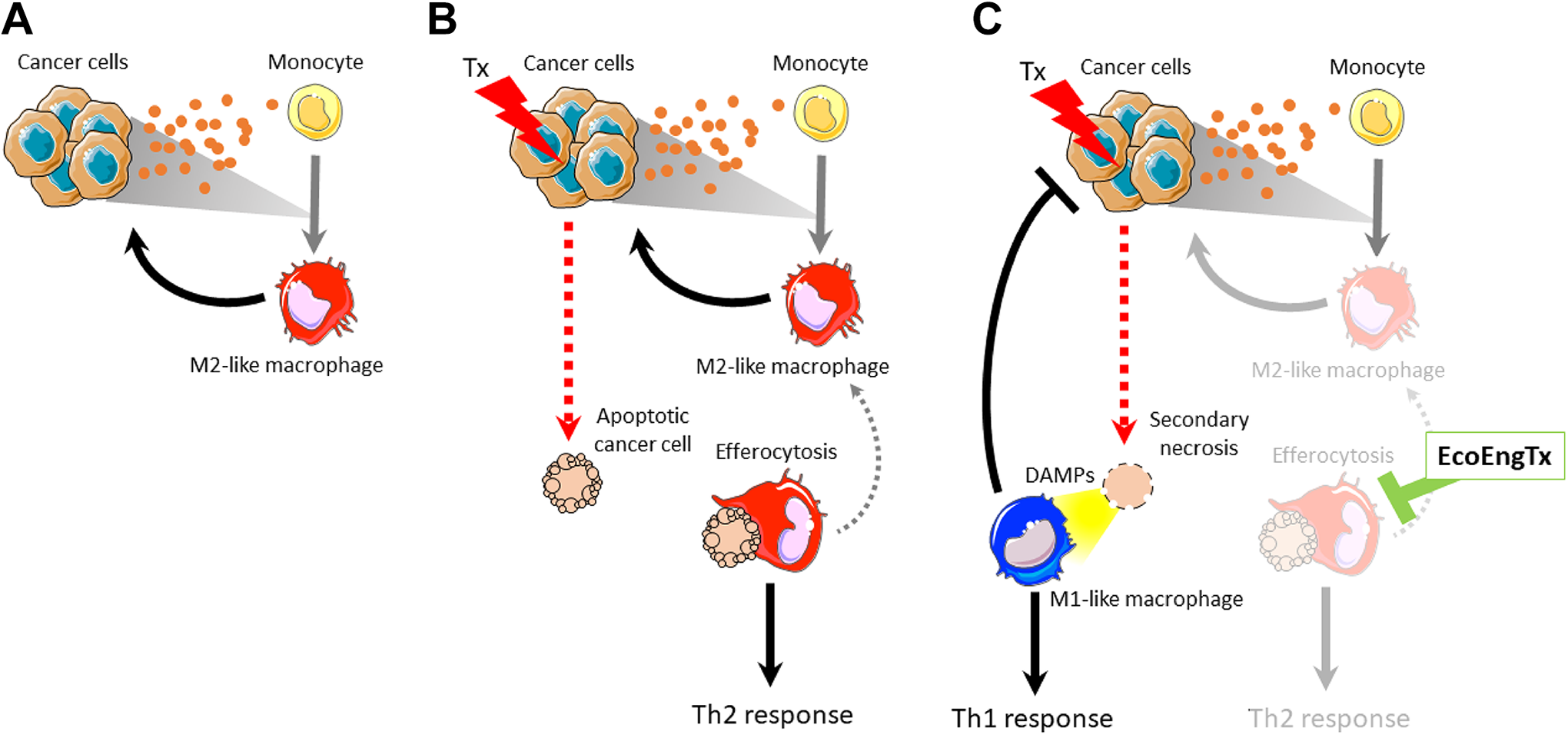
Targeting protumor ecosystem engineering by M2-like macrophages. A, Cooperative protumor ecosystem engineering of cancer cells and M2-like macrophages: Cancer cells release cytokines to the bloodstream through ecosystem engineering. Some of these secreted cytokines recruit monocytes to the tumor and differentiate them to M2-like macrophages. These macrophages support the cancer cells through ecosystem engineering. B, One ecosystem engineering function of M2-macrophages is efferocytosis, the phagocytosis of apoptotic cells. This effect is increased following cancer cell apoptosis due to cytotoxic therapies (Tx). By eliminating apoptotic cells from the ecosystem, M2-like macrophages limit the antitumor Th1 response (allogenic engineering) and support the Th2 response through supporting the M2 macrophage phenotype (autogenic engineering). C, Targeting the ecosystem engineering function (EcoEngTx) of efferocytosis will have an antitumor affect by permitting secondary necrosis of the apoptotic cell and the release of DAMPs. Consequently, this redirects the tumor ecosystem engineering toward an antitumor Th1 immune response and limiting the M2-macrophage protumor influences, weakening the cooperative protumor engineering of cancer cells and M2-macrophages. DAMPs indicates danger-associated molecular patterns.
In addition to interacting with cancer cells and noncellular components, M2-like macrophages also interact with many other cell types in the tumor ecosystem. M2 macrophages release Th2 cytokines that suppress that antitumor immune response. For example, M2 macrophages release IL-10 leading to immune suppression via downregulating antigen presentation on dendritic cells and macrophages. 35 Tumor-associated macrophages also secrete CCL20 and CCL22 to recruit regulatory T cells to the tumor, and M2-like macrophages secrete vascular endothelial growth factor promote angiogenesis. 36 -39 M2-like tumor-associated macrophages have interactions with a myriad of other cell types including cancer-associated fibroblasts, natural killer cells, and neutrophils have been described in the literature and were recently reviewed. 40,41 Through these mechanisms, among others, M2-like macrophages and cancer cells cooperatively engineer the ecosystem to favor tumor growth.
Phagocytosis of apoptotic cells, or efferocytosis, is one major tumor-promoting ecosystem engineering function of M2-like macrophages. Efferocytosis fundamentally alters the tumor environment by clearing apoptotic cells and preventing secondary necrosis, thus further promoting a protumor Th2 response and restricting the antitumor Th1 immune response (Figure 2B). During secondary necrosis, cells lose membrane integrity resulting in their intracellular contents leaking into the extracellular space. Some of these contents, such as adenosine triphosphate, DNA, and high mobility group box-1, are danger-associated molecular patterns that recruit antitumor M1-like macrophages and other Th1 immune cells (Figure 2C). As allogenic engineers themselves, M1 macrophages present tumor antigens on MHC class II and release Th1 cytokines to stimulate immune cells to help generate an antitumor immune response. M1 macrophages can also directly kill viable cancer cells through phagocytosis. Further, released intercellular contents can serve as antigens to generate an antitumor immune response. Therefore, secondary necrosis overall favors an antitumor Th1 immune response. In addition to preventing secondary necrosis, in vitro evidence suggests that efferocytosing M2 macrophages directly promote increased growth of cancer cells. 42 This allogenic engineering function also fuels the M2-macrophage phenotype, thus maintaining the strong autogenic engineering that the tumor ecosystem relies on. After efferocytosis, the macrophage expresses high M2-like characteristics (eg, cytokines IL-4, IL-10, TGF-β), resulting in the maintenance of protumor M2 macrophage effects on the ecosystem. 43 Altogether, ecosystem engineering by efferocytosis promotes tumor growth by restricting the antitumor Th1 immune response and supporting the protumor M2 macrophage phenotype.
Novel Therapeutic Strategy to Treat Cancer by Redirecting Ecosystem Engineering
The cooperation of the invasive ecosystem engineers, cancer cells and M2-like macrophages, creates a vicious cycle of cancer progression supporting protumor macrophage engineering. We propose a novel strategy for therapeutic intervention by specifically targeting the ecosystem engineering functions at play in the tumor ecosystem. By combining this strategy with standard-of-care cancer cell-directed therapy, it may be possible to limit cooperative ecosystem engineering and promote antitumor engineering to reduce tumor burden. MerTK is a promising target to interfere with M2 macrophage protumor ecosystem engineering. MerTK is a receptor tyrosine kinase in the Axl and Tyro3 family (TAM receptors) that plays a critical role in binding apoptotic cells and promoting efferocytosis. 44 -48 Blocking MerTK-mediated efferocytosis may result in altered ecosystem engineering, thus restricting the cancer cell-M2 macrophage cooperative relationship.
Targeting ecosystem engineering to switch the tumor microenvironment from protumor to antitumor by targeting a specific function, rather than a player, of engineering may be a promising treatment strategy. As depicted in Figure 2C, greatest success will likely be found by combination therapy with direct anticancer treatments such as chemotherapy or radiation that engage the targeted engineering function (eg, chemotherapy induces apoptosis that engages M2-macrophages in efferocytosis). Redirecting the ecosystem engineering by targeting MerTK may block efferocytosis and allow apoptotic cells to progress to secondary necrosis. As a result, the dominant immune response will switch from Th2 to Th1. This will fundamentally change the tumor ecosystem and species it contains. In addition to MerTK, other targets on macrophages such as CSF1R, SIRPα, and IL4Rα may similarly target tumor-promoting ecosystem engineering. Overall, this redirection of ecosystem engineering, blocking the vicious cycle of cooperative cancer cell-M2 macrophage engineering, has the potential to improve the antitumor immune response, slow cancer growth, and decrease metastasis.
Footnotes
Acknowledgments
The authors thank Amber de Groot for providing critical feedback to the manuscript. The authors also thank members of the Pienta Lab and the Brady Urological Institute for thoughtful feedback.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kenneth Pienta is a consultant to Cue Biopharma, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by NCI grants U54CA143803, CA163124, CA093900, and CA143055 as well as the Prostate Cancer Foundation, the Patrick C. Walsh Fund and the William and Carolyn Stutt Research Fund.
