Abstract
Background
Natural killer (NK) cell immunotherapy has shown promising therapeutic potential for acute myeloid leukemia (AML), especially with advancements in chimeric antigen receptor-engineered NK cells (CAR-NK) and artificial intelligence (AI). Despite these developments, the field lacks comprehensive bibliometric analyses to identify research hotspots and trends, which could guide future precision treatments.
Methods
A bibliometric analysis of NK cell immunotherapy for AML was conducted using literature from 2000 to 2023 retrieved from the Web of Science Core Collection database. Data visualization tools like CiteSpace, VOSviewer, and RStudio were employed to analyze publication trends, country contributions, institutional collaborations, influential authors, and research themes.
Results
The analysis identified 1513 studies, with the United States and China leading global contributions. Notable institutions include the University of Minnesota and MD Anderson Cancer Center. Hot topics include allogeneic NK therapy, CAR-NK cell therapy, and memory-like NK cells. Emerging trends highlight the integration of intelligent NK cells and combinatory therapies, offering promising avenues for AML treatment. Despite progress, challenges such as NK cell expansion, activation, and resistance mechanisms remain critical areas for research.
Conclusion
This study provides a comprehensive overview of the research landscape, highlighting the transformative potential of NK cell immunotherapy in AML. It underscores the need for international collaboration and continued innovation to overcome existing challenges and advance precision therapies.
Keywords
Introduction
Acute Myeloid Leukemia (AML) is a malignant hematopoietic disorder characterized by the clonal expansion and abnormal differentiation of myeloid progenitor cells. This process results in a significant reduction of healthy blood-forming cells and extensive infiltration of malignant cells into the bone marrow, peripheral blood, and extramedullary tissues. 1 AML is a rapidly progressing malignancy with a high mortality rate. In 2019, approximately 21,450 new cases of AML were reported in the United States, leading to 10,920 deaths. 2 Established risk factors for AML include environmental exposures, genetic predisposition, and previous chemotherapy or radiotherapy. 3 Current therapeutic strategies for AML consist of chemotherapy, radiotherapy, targeted therapy, hematopoietic stem cell transplantation, and supportive care, all aimed at improving survival rates and quality of life. 4
Despite advances in treatment, AML management faces persistent challenges, including high relapse rates, therapy resistance, refractoriness, and complications associated with allogeneic stem cell transplantation.5,6 These limitations not only reduce therapeutic efficacy but also exacerbate patient suffering, underscoring the pressing need for novel, targeted therapies to improve the prognosis of AML.
To address these challenges, ongoing research focuses on unraveling the molecular and cellular mechanisms underlying AML. Key areas of investigation include genetic alterations, the role of the tumor microenvironment, and the dynamic interplay between AML cells and the immune system.7-9 Emerging omics technologies, such as genomics, transcriptomics, and proteomics, have been pivotal in identifying novel biomarkers and therapeutic targets for AML. Furthermore, the development of cancer immunotherapy has opened new avenues for AML treatment, offering promising prospects for overcoming therapeutic barriers.
Natural Killer (NK) cells, a vital component of the innate immune system, represent a subset of lymphocytes distinguished by their innate cytotoxicity against tumor cells and virally infected cells without prior sensitization. 10 Unlike T and B lymphocytes, which rely on antibodies and Major Histocompatibility Complex (MHC) molecules for target cell recognition, NK cells can directly identify and eliminate target cells independently of MHC. 11 Since their discovery in 1973, NK cells have attracted considerable scientific interest. 12 The first NK cell workshop, held in Florida, USA, in 1982, marked the beginning of systematic research in this field. The pace of NK cell research accelerated significantly with the launch of the “Cancer Moonshot” initiative in the United States in 2016. Advances in genetic editing technologies, such as CRISPR-Cas9, and the development of Chimeric Antigen Receptor (CAR) technology, coupled with interdisciplinary integration and artificial intelligence, have further expanded the potential of NK cells in cancer therapy.13,14 In the context of AML, NK cell-based immunotherapy has emerged as a promising area of research, with substantial evidence supporting its therapeutic potential.
Bibliometrics is a quantitative analytical approach that utilizes mathematical and statistical tools to assess scientific publications. It facilitates the evaluation of research dynamics, the identification of hotspots, and the analysis of interdisciplinary integration by studying the distribution, structure, and growth of scientific literature. 15 Bibliometric analyses provide insights into the impact of research activities, identify key journals and influential articles, and trace networks of scientific collaboration. Furthermore, the integration of bibliometric methods with visualization tools enhances the interpretation of complex datasets, enabling readers to discern data patterns and trends more effectively.
This study aims to conduct a systematic bibliometric and visual analysis of the literature on NK cell immunotherapy in AML. By identifying research trends, gaps, and key contributions in this field, the findings are expected to offer valuable insights and references for future scientific exploration, further advancing the development of NK cell-based immunotherapy in AML.
Materials and Methods
Data Source and Literature Search
The literature data were obtained from the Web of Science Core Collection (WoSCC) database, covering the period from January 1, 2000, to February 29, 2024. The search formula used was: (TS = (“AML” or “Acute Myeloid Leukemia”)) AND TS = (“NK” or “Natural Killer Cells”).
Data Screening
Inclusion Criteria
(1) Articles related to Natural Killer Cells and Acute Myeloid Leukemia (AML). (2) Articles published in English. (3) Article types that included clinical trial studies, in vitro experimental studies, in vivo (4)experimental studies, public database analysis studies, and reviews.
Articles with complete bibliographic information, including title, country, author, keywords, and source.
Exclusion Criteria
(1) Conference papers, newspapers, patents, popular science literature, and health-related literature. (2) Duplicate publications. (3) Articles that could not be fully accessed. (4) Data inclusion and exclusion were independently conducted by two reviewers. In cases of discrepancies, a third reviewer participated to resolve conflicts.
Data Standardization
After screening, the literature was exported in RefWorks and plain text formats. Special characters were removed, and keyword names were standardized; for instance, “acute myelogenous leukemia” was unified as “acute myeloid leukemia.” Country and region names were also standardized, grouping terms such as “Northern Ireland,” “Wales,” “England,” and “Scotland” under “United Kingdom.” The CiteSpace software’s Data Import/Export function was used to convert the retrieved literature into the necessary format for analysis.
Data Analysis
Data Extraction
The normalized text data were organized into a structured format designed by two researchers, who subsequently extracted relevant data. Extracted data included publication information such as year of publication, country/region, issuing organization, journal, authors, cited literature, and keywords.
Analysis Methods
Results
Literature Retrieval and Screening Results
The initial search yielded a total of 1800 publications. After performing duplicate checks, selection processing, and a thorough review, 1513 publications were retained based on the inclusion and exclusion criteria. The detailed steps of the literature selection process are presented in Figure 1. Flow chart of data collection in this study.
Trends in Annual Publications and Citations
Figure 2 depicts the trends in publication volume and citation counts related to NK cell immunotherapy research in AML from 2003 to 2023. During this 20-year period, a total of 1513 publications were identified, collectively receiving 65,474 citations, with an average citation rate of 43.27 per publication. As illustrated in Figure 2(A), both the number of publications and citation counts have shown a substantial upward trajectory. Notably, the period between 2014 and 2018 experienced the highest growth in publication volume. Additionally, a significant surge in citation counts was observed from 2018 to 2021, likely reflecting the impact of advancements in tumor immunotherapy following the launch of the 2016 “Cancer Moonshot” initiative. Annual trends of global publications and citations (A) Numbers of publications and citations on NK for AML from 2003 to 2023. (B) Polynomial fitting curve of annual publication in NK for AML.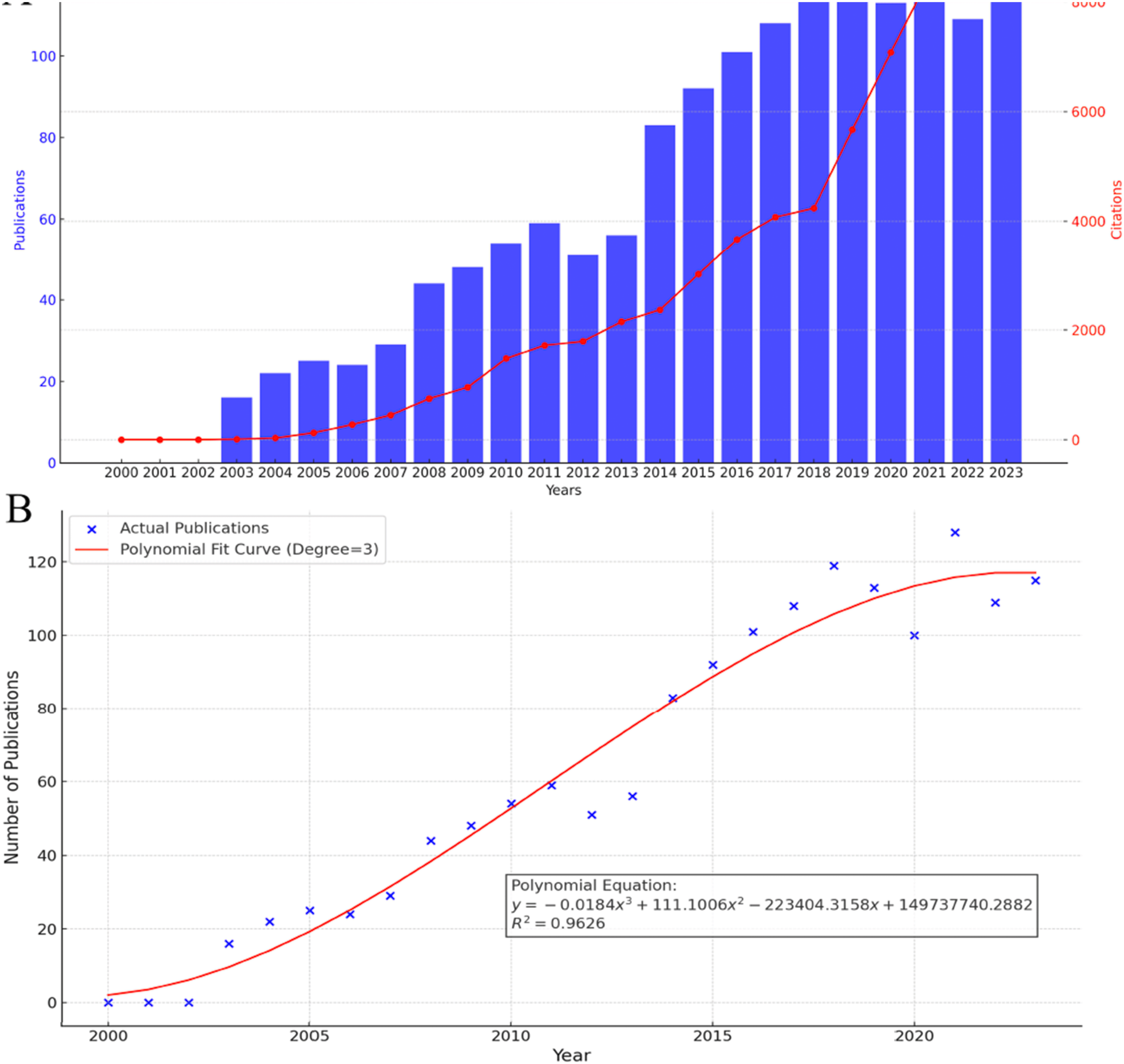
To predict future trends in this field, a polynomial fitting curve was generated and is presented in Figure 2(B). The curve suggests a continued increase in publication output in the coming years. The model’s determination coefficient (R2 = 0.9626) indicates that the curve accounts for 96.26% of the variability in the data, highlighting its strong predictive power.
Overall, the analysis of publication volume and citation counts demonstrates that research on NK cell immunotherapy in AML is a rapidly growing and highly influential area. These findings underscore the substantial focus and promising potential for further advancements in this field.
National and Regional Studies
Over the past two decades, research on NK cell immunotherapy for AML has engaged 61 countries/regions. Figure 3(A) visualizes the collaborative relationships among the top 30 countries based on publication volume. The United States leads in international collaborations with 316 partnerships, followed by Germany with 177 and France with 109. Figure 3(B) illustrates the annual publication trends of the top 10 countries over the last 20 years. The United States has consistently maintained the highest publication output each year, followed by China. Since 2014, these leading countries have demonstrated steady annual contributions, reflecting the establishment of stable research networks within these regions. Analysis of Global Research Trends and Collaborations. (A) Total link strength of publications by country/region, highlighting the intensity of research output and influence across different regions. (B) Annual publication trends, broken down by the top 10 most productive countries/regions, illustrating shifts in research focus and contributions over time. (C) Collaboration network visualization, showing interconnections and partnerships between countries/regions in NK cell research for AML, emphasizing the extent of international cooperation within the field, In the map, darker colors indicate more publications in that country/region.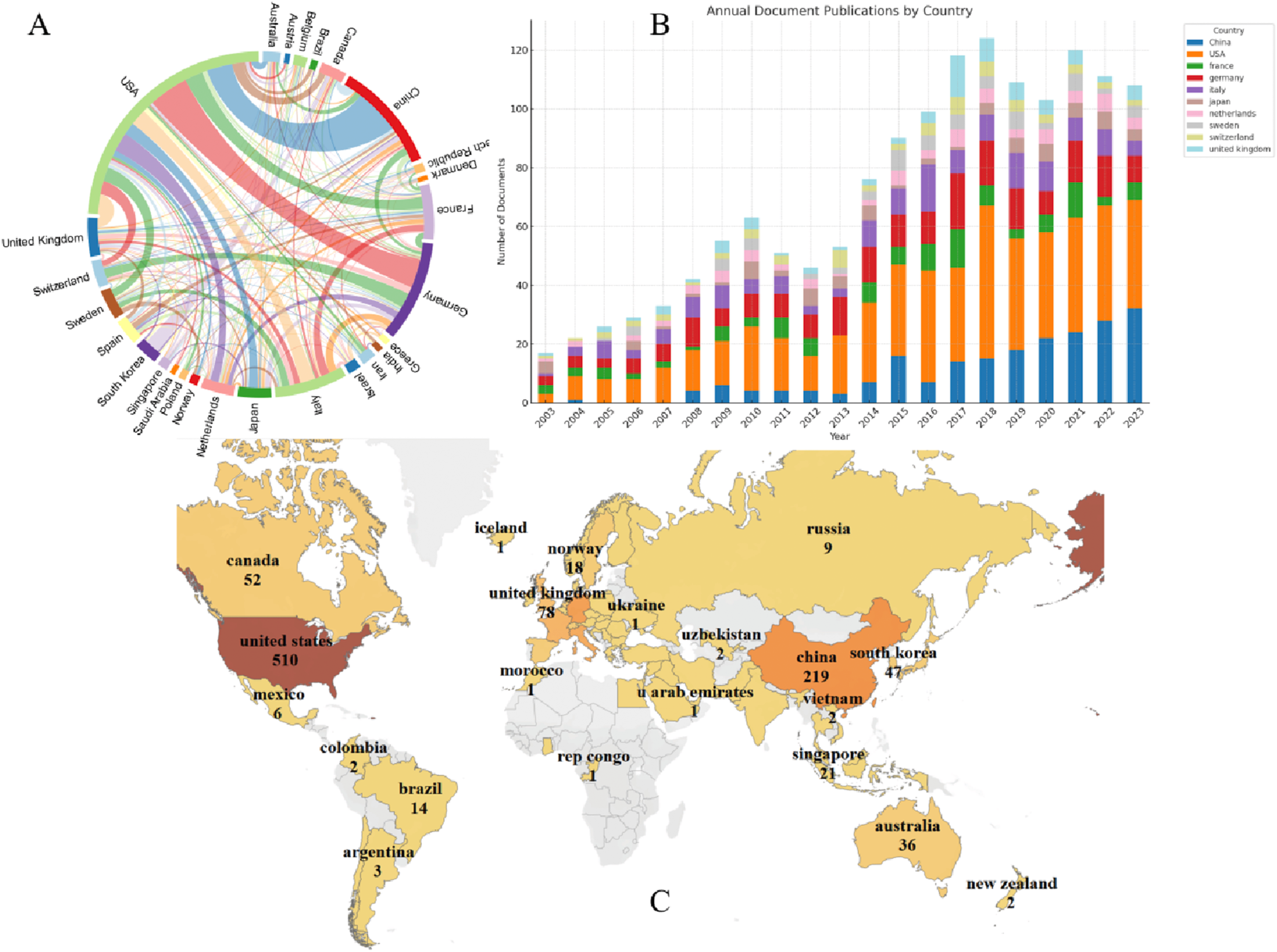
The Top 10 Countries/Regions by Publication Volume in NK Cell Immunotherapy for AML Treatment.
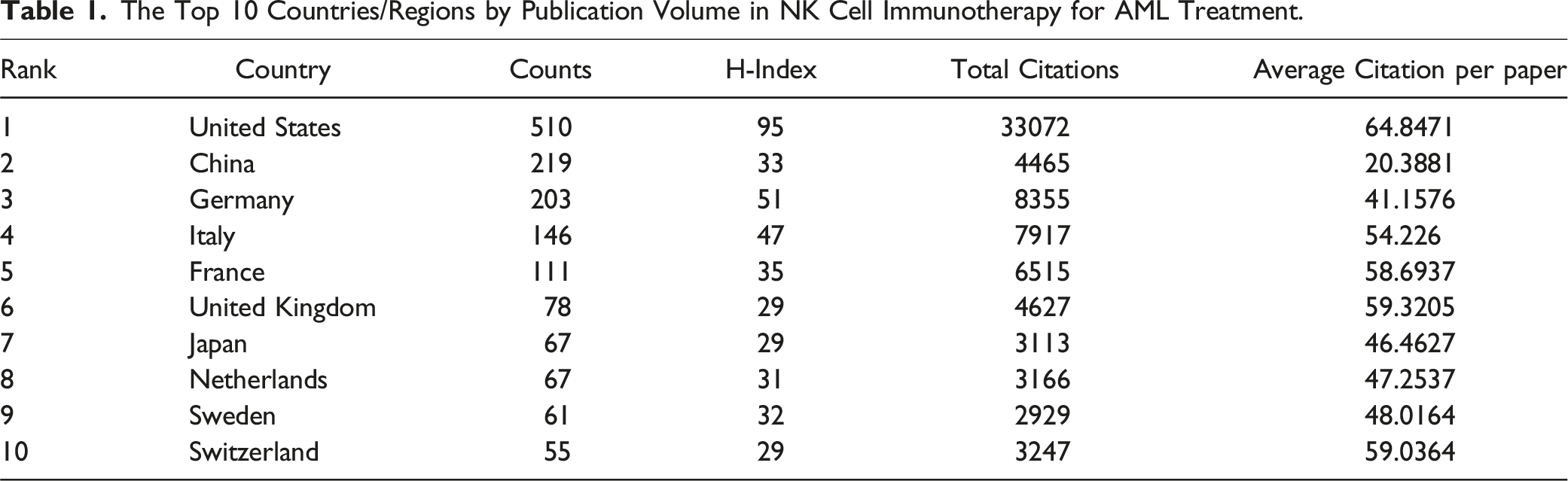
These findings underscore the global engagement and collaborative efforts in NK cell immunotherapy research for AML, highlighting its growing influence and the substantial contributions from researchers worldwide.
Analysis of Research Institutions
A total of 2123 institutions have contributed to research on NK cell immunotherapy for AML. Among these, the University of Minnesota has achieved the highest publication output, with 69 papers, followed by the University of Texas MD Anderson Cancer Center with 50 publications and the University of Genoa with 36. Figure 4(A) highlights the top 30 institutions ranked by publication volume. Over the past two decades, the University of Minnesota has consistently led in publication output, amassing a cumulative citation count of 6,326, as shown in Table 2 and Figure 4(D). Institutional Cooperation and Publication Trends. (A) Visualization of the collaboration network among universities/institutions, illustrating partnerships and collaborative strength within NK cell research for AML. (B) Trend chart displaying the development of bibliographic coupling among universities/institutions, reflecting the growing inter-institutional connections over time. (C) Coupling chart depicting the degree of bibliographic coupling among institutions, indicating shared research focus and citation patterns. (D) Annual publication trends of the top 6 universities/institutions by publication volume, showcasing their research output over the years and highlighting leading contributors in the field. Top 10 Institutions With the Highest Number of Publications.

To explore institutional relationships further, bibliographic coupling analysis was performed. Bibliographic coupling, a concept introduced by M.M. Kessler, suggests that papers sharing a higher number of references are more closely related in subject matter or specialty. Figure 4(C) visualizes the bibliographic coupling among institutions, revealing collaborative relationships where stronger coupling reflects shared citations and often indicates a higher level of collaboration on scientific projects. The University of Minnesota exhibits the strongest degree of coupling, underscoring its pivotal role as a hub for knowledge creation and dissemination in the field of NK cell immunotherapy.
Since 2012, the degree of bibliographic coupling among institutions has shown a marked increase. Recently, institutions such as the University of Texas MD Anderson Cancer Center (USA), Washington University (USA), Harvard Medical School (USA), Johns Hopkins University (USA), Glycostem Therapeutics (Netherlands), University of Science and Technology of China (China), and Sichuan University (China) have demonstrated active participation in international collaborations, as depicted in Figure 4(B). This growing trend of coupling among institutions highlights enhanced cooperation, knowledge exchange, and interdisciplinary integration, suggesting that the field is evolving toward a mature state.
Journals and Co-Cited Journals
Over the past 20 years, research on NK cell immunotherapy for AML has been disseminated across 402 journals. Frontiers in Immunology leads in publication volume with 108 papers, followed by Blood with 80 publications and Cancers with 51, as depicted in Figure 5(A). In terms of impact, Blood has the highest H-index at 54, followed by Frontiers in Immunology at 37 and Cancers at 15, as detailed in Table 3. According to Bradford’s Law, 13 journals have been identified as core journals in this field, as shown in Figure 5(D). Visualization Map of Published Journals. (A) Journal co-occurrence map, illustrating the interrelationships and frequency of co-publication among journals within NK cell research for AML. (B) Double overlay visualization of journals, showing the citation patterns between citing and cited journals, with clusters indicating knowledge dissemination and influence. (C) Annual output trend of the top 5 journals by publication volume, displaying shifts in journal productivity and the growing prominence of key journals over time. (D) Core journals identified based on Bradford’s Law, highlighting the journals that are most central to the field and contribute a substantial volume of research. Top 10 Journals With Published Articles on NK Cell Immunotherapy for AML Treatment.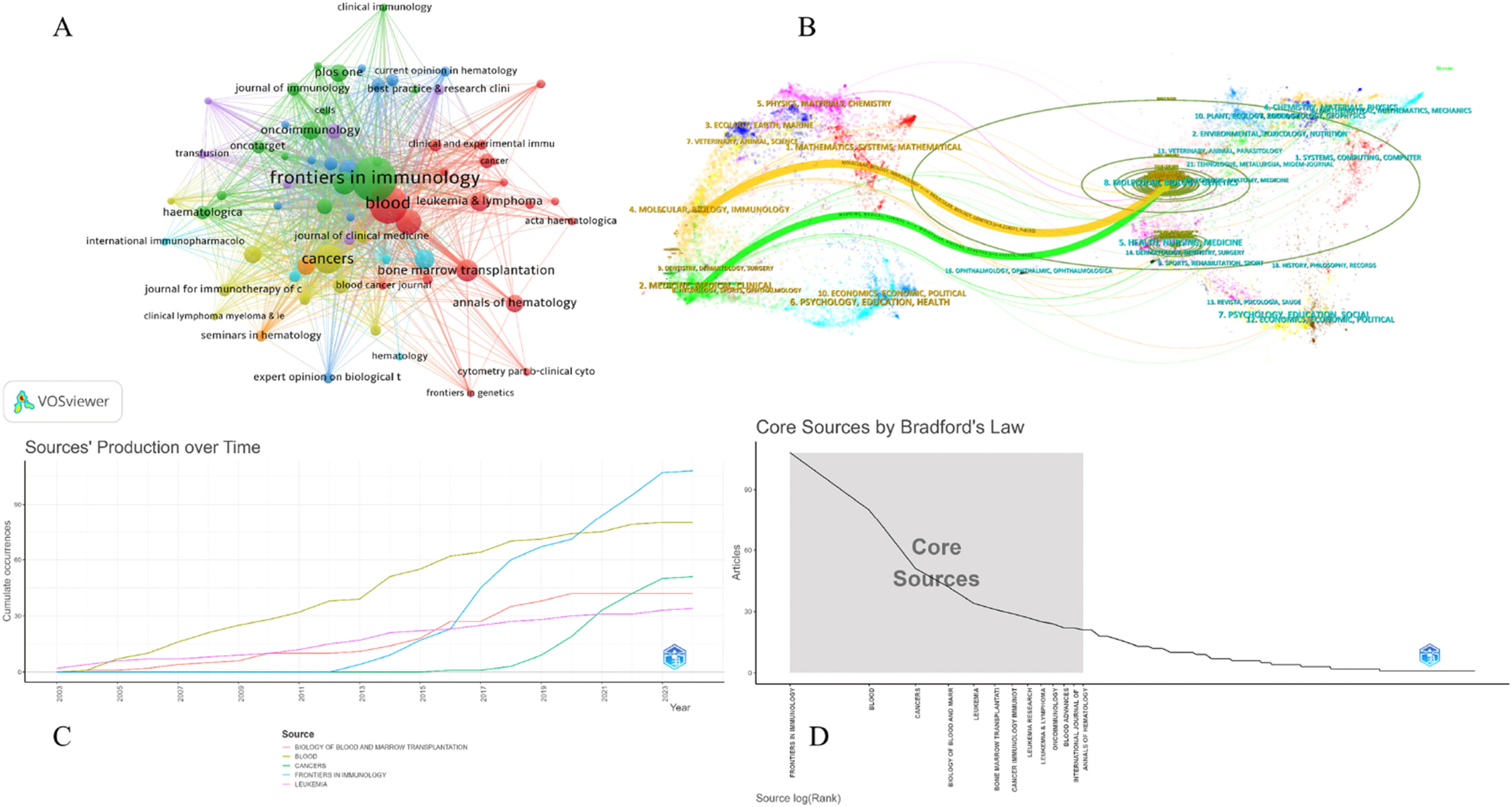

Figure 5(C) illustrates publication trends, showing that Blood dominated publication volume prior to 2021. However, after 2021, Frontiers in Immunology experienced a rapid increase in publications and has since maintained its leading position. The journal co-citation overlay provides insights into citation relationships, where citing journals on the left represent the knowledge frontier, while cited journals on the right reflect the foundational knowledge underpinning the field.
In Figure 5(B), the orange path highlights that research published in journals within the domains of Molecular, Biology, and Genetics is predominantly cited by journals categorized under Molecular, Biology, and Immunology. Additionally, the green path indicates that these journals are frequently cited by those spanning a diverse range of fields, including Medicine, Medical, Clinical, Dentistry, Dermatology, Surgery, Neurology, Sports, and Ophthalmology. These citation patterns underscore the interdisciplinary nature of NK cell immunotherapy research in AML, highlighting its foundational role in advancing knowledge across various clinical and medical specialties.
Authors and Co-Cited Authors
In total, 8711 authors have contributed to research in NK cell immunotherapy for AML, with 33,801 authors being cited. Using Price’s Law, where Mp = 0.749 * √NPmax (core author publication volume > Mp), N = 41, and Mp = 4.79, a publication volume of ≥5 was used to define core authors. This study identified 184 core authors. Eleven relatively fixed research teams have been formed in this field, 11 teams including Miller, Jeffrey S, Dollstra, Harry, Olive, Daniel, etc. have made significant contributions to the research in this field, as shown in Figure 6(A). Author Collaboration Network Graph. (A) Density map of the author collaboration network, illustrating the strength and extent of research partnerships among authors in NK cell research for AML. (B) Co-cited author collaboration network density map, showing the connections and co-citation relationships among influential authors, indicating shared research impact. (C) Jade block diagram of the top 10 authors by publication volume and H-index, highlighting authors with the highest output and academic influence within the field. (D) The top 10 countries ranked by the number of corresponding authors, showcasing the geographic distribution of lead researchers and international research contributions.
The most frequently co-cited author is Ruggeri, L., with 1043 co-citations, followed by Miller, J.S., with 598, and Cooley, S., with 424, as shown in Figure 6(B). The H-index, which indicates a researcher’s academic impact by considering both publication quantity and citation frequency, shows that Velardi, Andrea, has the highest H-index of 21 during this period, as depicted in Figure 6(C).
Authors and Co-Cited Authors (Top 10).

Co-cited References
Citations form the foundation of knowledge in any research field. Figure 7(A) depicts the co-citation network, which is organized into four distinct clusters. The cluster led by Loredana Ruggeri
16
primarily focuses on allogeneic NK cell immunotherapy, while the cluster represented by Régis T. Costello
17
investigates mechanisms of immune escape in AML. The cluster associated with Jeffrey S. Miller
18
emphasizes NK cell expansion techniques, and the cluster led by Eric Vivier
19
explores the regulatory roles of NK cells in immunity and inflammation. Visual Mapping of Cited Literature (A) Thematic coupling analysis of research literature, highlighting the thematic connections between cited works and their influence on NK cell research for AML. (B) Emergent analysis of cited literature, showcasing newly influential studies and identifying recent trends in citations that signal shifts in research focus. (C) Clustering analysis of cited literature, organizing cited works into thematic clusters to reveal key areas of study and foundational research topics within the field. (D) Timeline analysis of cited literature, displaying the temporal evolution of citation clusters, illustrating how research themes have developed and changed over time.
Figure 7(B) highlights the top 25 most-cited documents in this field. The study by Veronika Bachanova et al. 20 holds the highest citation prominence (strength = 38.29). This influential work demonstrates that an IL-2 diphtheria toxin fusion protein can induce NK cell proliferation, thereby enhancing clinical efficacy in refractory AML and underscoring the potential for combined NK cell therapy. Other significant studies include the first human clinical trial of CAR NK-92 cells by Xiaowen Tang et al. 21 and the establishment of AML diagnosis and treatment standards by Hartmut Döhner et al. 22 Additional influential works are by E. Liu, 23 who investigated NK cell engineering; Shannon L. Maude et al, 24 who conducted a clinical trial on Tisagenlecleucel; Enli Liu, 25 who performed clinical research on CAR-NK cell immunotherapy; and Sarah Cooley et al, 26 who led the first human trial of rhIL-15 and haploidentical NK cell therapy for advanced AML.
Citation clustering analysis, as shown in Figure 7(C), identifies key themes within the research. The cluster labeled #0pediatric oncology highlights the growing focus on NK cell therapy in pediatric AML, while #1natural killer cell alloreactivity emphasizes the importance of allogeneic NK cell therapy as a major therapeutic strategy. The timeline clustering analysis in Figure 7(D) indicates emerging research priorities. The cluster labeled #0promising therapeutic target reflects the current focus on identifying novel NK cell immunotherapy targets, while #1car-nk cell therapy highlights the rapid expansion of research on CAR-NK cell therapy in AML.
Keyword Analysis
The keyword cloud in Figure 8(A) highlights that NK cells and immunotherapy are prominent focal points in AML research. A chronological analysis of research topics, depicted in Figures 8(B) and 9(A), reveals distinct phases in the evolution of the field. Between 2003 and 2013, foundational terms emerged, focusing on immune recognition and tumor cell responses. This foundational period advanced the understanding of immune recognition, cellular killing mechanisms, and gene expression. Among the key findings, the NKG2D receptor was identified as a pivotal receptor that enhances NK cell-mediated cytotoxicity against tumor cells. Its role is particularly crucial in addressing immune evasion strategies employed by AML cells, shedding light on the intricate interactions between the immune system and tumor cells. Keyword Visualization Graph (A) Word cloud of Keywords Plus in local clusters, illustrating frequently occurring terms and prominent themes within NK cell and AML research. (B) Trend topics of NK and AML research from 2003 to 2023, showing the progression and emerging areas of interest over the past two decades. Keyword Visualization Graph (A) Thematic evolution in Keywords Plus, mapping changes in keyword focus to reflect the field’s shifting research priorities and developments. (B) Keyword bursts, highlighting terms with sudden increases in usage, indicating emerging trends and shifts in research emphasis.

This period also emphasized the role of apoptosis as a central mechanism of NK cell-mediated tumor destruction. NK cells were found to express activating receptors such as NKG2D and apoptosis-inducing ligands such as Fas ligand and TRAIL, which bind to their counterparts on AML cells, directly triggering apoptosis. Furthermore, CD33 emerged as a significant marker on AML cells, while CD56 was identified as a critical marker on NK cells, aiding in the development of precision immunotherapy strategies by identifying specific therapeutic targets.
Between 2004 and 2017, research shifted towards the activation and functional exploration of NK cells, with stem cell transplantation becoming a cornerstone treatment for AML. From 2018 to 2020, research attention expanded to include residual disease and GVHD (Graft-versus-Host Disease), indicating a broadening scope in AML research. By 2021-2023, the field entered a phase of in-depth exploration, with heightened focus on disease classification and understanding, driving the development of novel treatment strategies targeting both tumors and their microenvironments. NK-92 cells emerged as an exciting area of study, showcasing the potential of harnessing immune cells for innovative cancer therapies.
The term emergence graph in Figure 9(B) highlights notable trends in AML research. Bone marrow transplantation, with a prominence strength of 10.08, remains a cornerstone treatment and shows significant promise when combined with NK cell immunotherapy. Recent emergent terms such as in-vivo expansion, immune checkpoints, and NK-92 cells reflect the dynamic and evolving landscape of AML treatment strategies, underscoring the ongoing innovation and progress in this field.
Discussion
Research Hotspots
Allogeneic NK Cell Therapy and AML
Allogeneic NK cell therapy has emerged as a novel and promising treatment strategy, particularly for hematological malignancies such as AML. This approach involves the transfer of NK cells from a genetically distinct donor to a recipient. 27 Unlike T cells, which rely on Major Histocompatibility Complex (MHC) I molecules for immune recognition, NK cells possess the ability to recognize and eliminate tumor cells with reduced MHC I expression, a common immune evasion mechanism employed by AML cells. This unique capability positions NK cell therapy as a potent tool for targeting AML, fostering growing interest in this field.
In a Phase I non-randomized clinical trial, Mohammad Ahmadvand et al. 28 demonstrated the feasibility and safety of adoptive transfer therapy using allogeneic NK cells from unrelated healthy donors in patients with refractory or recurrent AML. These findings support the advancement of this therapy into Phase II trials. Allogeneic NK cell therapy not only directly activates antibody-dependent cellular cytotoxicity (ADCC) but also enhances the intrinsic cytotoxic effects of NK cells. This dual mechanism effectively targets AML cells while minimizing immune evasion caused by antigenic variation. Importantly, allogeneic NK cell therapy circumvents complications such as graft-versus-host disease (GVHD) and significantly reduces adverse events, including cytokine release syndrome (CRS) and neurotoxicity (NT), making it a safer and more tolerable therapeutic option. 29
In addition, biotechnology company Wugen showcased its memory-like NK cell therapy product, WU-NK-101, at the 2022 European Hematology Association (EHA) hybrid conference. Developed using the proprietary Moneta™ platform, WU-NK-101 targets the CD16 antigen and has demonstrated significant efficacy in treating AML. Notably, this therapy achieves therapeutic benefits without inducing CRS or NT, even in patients with high disease burdens. These advancements highlight allogeneic NK cell therapy as a promising treatment option for refractory or recurrent AML, paving the way for new therapeutic opportunities in hematological malignancies.
CAR-NK Cell Therapy and AML
Chimeric Antigen Receptor (CAR) technology has emerged as a cornerstone in cellular immunotherapy. Compared to CAR-T cell therapy, CAR-NK cell therapy offers several advantages, including enhanced tumoricidal activity, greater target specificity, and a significantly reduced risk of adverse effects such as graft-versus-host disease (GVHD) and cytokine release syndrome (CRS).14,30 A critical challenge in the development of CAR-NK therapy lies in identifying tumor-specific antigens that are highly expressed on AML cells but minimally or not expressed on normal hematopoietic tissues. This ensures that CAR-NK cells effectively target tumor cells while minimizing off-target toxicity. 31
Several CAR-NK targets are currently under investigation. CD33 and NKG2D have advanced to clinical trials, while other targets, such as CD123, CD25, CD96, and CD371 (CLL-1), are demonstrating significant anti-AML activity in preclinical studies.32,33 Among these, NKG2D has garnered considerable attention due to its ability to recognize tumor antigens and initiate robust anti-tumor responses. NKG2D ligands are typically overexpressed on malignant cells or upregulated in response to DNA damage or infection, making NKG2D an attractive target for AML therapy.34,35
Recent advances in CAR-NK cell technology include the work of Zhicheng Du et al, 36 who developed CAR-NK cells targeting NKG2D ligands using a non-viral piggyBac transposon system. Their in vitro and in vivo experiments confirmed that these CAR-NK cells could effectively lyse NKG2D ligand-expressing AML cells, suppress tumor growth, and improve survival rates in a xenograft KG-1 AML mouse model.
Another promising target is CD371 (CLL-1), which is expressed in approximately 68% of AML CD34+/CD38- cells. This population is strongly associated with AML persistence and recurrence. CAR-NK cells targeting CD371/CLL-1 hold the potential to eliminate leukemia at its source by specifically targeting these resistant cell populations. 37
While CAR-NK therapy has shown significant promise in preclinical studies, its clinical application is still in the early stages and faces substantial challenges. These include optimizing cell persistence, improving antigen selection, and overcoming potential immunosuppressive effects within the tumor microenvironment. Addressing these obstacles is critical for realizing the full therapeutic potential of CAR-NK cells in AML.
Research Trends
Intelligent NK cells
Intelligent cells are genetically engineered to incorporate logical genetic circuits, enabling them to interact with the body’s internal environment while being externally controlled. These cells can be equipped with various sensors—such as electrical, thermal, magnetic, and acoustic—that allow for remote manipulation outside the body. With the continuous evolution of artificial intelligence (AI) and gene-editing technologies, the development of intelligent NK cells has encountered remarkable opportunities. AI’s application in precision medicine has already demonstrated significant potential. 38 By analyzing genomic, transcriptomic, and proteomic data, AI can identify novel targets associated with acute myeloid leukemia (AML) and predict which patients are most likely to respond favorably to NK cell therapy.
AI not only enhances the specificity of NK cell immunotherapy for targeting specific cancer biomarkers but also facilitates dynamic adjustments to address the heterogeneity of tumor cell characteristics, thereby improving both efficacy and safety. For instance, AI can guide CRISPR-Cas9 gene-editing technology to optimize the design of chimeric antigen receptor (CAR) NK cells. By predicting the functional outcomes of different CAR constructs, AI can enhance the specific cytotoxicity of NK cells against AML cells. 39
The biological activity of NK cells is governed by the interplay of multiple activating and inhibitory receptors on their surface, which are critical to their function. In engineered NK cells, the selection of appropriate logic gates is essential for achieving intelligent function. For example, a study designed NK cells equipped with an OR-NOT logic gate to precisely target AML cells. In this system, the OR gate simultaneously targets CD33 and FLT3, while the NOT gate employs an inhibitory CAR directed at intrinsic mucin, thereby sparing healthy hematopoietic stem cells. 40 This selective approach represents a promising strategy for intelligent NK cell design, significantly enhancing the precision and safety of AML treatments.
NK cell combination therapy
NK cells combined with chemotherapy
Chemotherapy for AML typically comprises two phases: induction remission and consolidation therapy. The induction phase aims to eliminate the majority of leukemia cells through intensive chemotherapy to achieve clinical remission, while consolidation therapy focuses on eradicating residual leukemia cells and preventing relapse. Despite its efficacy, chemotherapy often involves cytotoxic drug combinations that can lead to severe adverse effects, including myelosuppression, infections, and bleeding tendencies.
In a clinical trial, Uday Kulkarni et al. 41 evaluated the safety and efficacy of haploidentical donor CD56-positive NK cell transfusions in 14 AML patients who were refractory to initial chemotherapy. The study reported a 2-year overall survival rate of 28.6% ± 12.1% and a treatment-related mortality (TRM) of 38.5% ± 13.5%. These results suggest that haploidentical NK cell therapy, used as an adjunct to transplantation, may be a safe and feasible option for AML patients, warranting further investigation in larger cohorts.
Furthermore, Jérôme Rey et al. 42 examined the impact of chemotherapy on the expression of activating and inhibitory receptors on NK cells, CD8+ T cells, and γδ T cells in 29 AML patients. The study revealed that NK cells were the most significantly affected by chemotherapy, with a partial recovery of activating receptor expression observed 6 weeks post-treatment. This finding underscores the importance of monitoring NK cell levels and receptor expression in AML patients undergoing chemotherapy to better understand immune recovery and potential therapeutic interventions.
NK cells combined with targeted Drugs
Programmed cell death protein 1 (PD-1) is a critical immune checkpoint that regulates NK cell activation. Within the tumor microenvironment, tumor cells often exhibit high PD-L1 expression, which binds to PD-1 on NK cells, suppressing their activity and limiting their ability to eliminate tumor cells. Recent studies suggest that combining interleukin-15 (IL-15) with PD-1 inhibitors can significantly enhance the cytotoxic potential of NK cells against AML.43,44
Additionally, transforming growth factor-beta 1 (TGF-β1) has been identified as a key immunosuppressive factor that negatively impacts NK cell function. 45 Targeting TGF-β1 pathways offers a promising strategy to reverse NK cell suppression in AML therapy. Furthermore, high expression and activation of NKp46, an activating receptor on NK cells, have been shown to correlate with improved overall survival in AML patients undergoing allogeneic stem cell transplantation (allo-SCT). 46
These findings underscore the therapeutic potential of modulating NK cell activity through multiple pathways. Combining TGF-β1 inhibition or NKp46 activation with existing NK cell-based therapies may produce synergistic effects, providing novel strategies to enhance treatment outcomes in AML.
NK Cells Combined with Hematopoietic Stem Cell Transplantation (HSCT)
Natural killer (NK) cells play a pivotal role in both graft-versus-host disease (GVHD) and graft-versus-tumor (GVT) effects following hematopoietic stem cell transplantation (HSCT) in AML patients. Optimizing killer immunoglobulin-like receptor (KIR) and human leukocyte antigen (HLA) pairings has been shown to enhance the clearance of residual leukemia cells while minimizing the risk of GVHD. 47
In a clinical trial (NCT02782546), 15 patients with relapsed or refractory AML were treated with memory-like (ML) NK cells induced by N-803, an IL-15 superagonist, in combination with HSCT. The study demonstrated good tolerance to donor-derived ML NK cells, with 87% of patients achieving composite complete remission by day 28. This included the eradication of high-risk mutations such as TP53 variants. Additionally, ML NK cells exhibited prolonged persistence, remaining active for over two months. 48
These findings underscore the potential of combining NK cell therapies with HSCT to reduce GVHD occurrence and improve therapeutic outcomes for relapsed and refractory AML. The ability of ML NK cells to target high-risk mutations and maintain activity over extended periods highlights their promise as a key component of advanced AML treatment strategies.
Limitations
This study has some limitations that should be considered. First, the analysis relied exclusively on the Web of Science Core Collection (WoSCC) database, which, although comprehensive, may not fully capture the global research landscape on NK cell immunotherapy for AML. Databases such as PubMed, Scopus, and Embase, which include additional relevant studies, were not incorporated. This limitation might lead to a less complete representation of the field, as important publications indexed in other databases could be omitted.
Second, the study only included English-language publications. As a result, significant research published in other languages, especially from countries where English is not the primary language, may not have been captured. This language restriction could introduce a bias in the analysis, potentially underrepresenting important contributions from non-English speaking regions and impacting the observed global trends and research hotspots.
Future research should aim to incorporate a broader range of databases and include non-English publications to provide a more comprehensive overview. Such an approach would allow for a more accurate representation of global research activities, reflecting diverse contributions to the field of NK cell immunotherapy for AML.
Challenges and Prospects
In recent years, NK cell immunotherapy has demonstrated immense potential in the treatment of AML. However, several significant challenges remain. One key obstacle is the production of sufficient quantities of highly active NK cells and resolving issues related to donor matching and the long-term maintenance of activity in allogeneic NK cells. Although CAR-NK cells significantly enhance tumor cytotoxicity through specific targeting, tumor cells may evade NK cell-mediated attacks by altering surface antigen expression or leveraging immunomodulatory factors, leading to immune escape. These limitations highlight the pressing need for further research into AML-specific antigens to improve therapeutic specificity and efficacy.
Moreover, the immunoregulatory roles of NK cells and their potential in combination therapies with T cells remain underexplored. These interactions could offer synergistic effects in AML treatment and warrant deeper investigation. The sensitive anti-tumor responses of memory-like NK cells, alongside strategies to sustain their memory characteristics within the tumor microenvironment, represent critical directions for future research. Additionally, the development of NK cell vaccines designed to boost anti-tumor immunity holds significant promise.
Another major challenge involves addressing NK cell senescence, exhaustion, and functional suppression, which are pivotal mechanisms underlying AML immune evasion. Overcoming these barriers is essential for achieving more precise and durable NK cell-based therapies for AML. Advancing our understanding of these aspects will be crucial for fully unlocking the therapeutic potential of NK cells in the fight against AML.
Conclusion
Over the past 20 years, researchers from 2123 institutions across 61 countries/regions have contributed to this field, with 8711 authors publishing their studies in 402 journals. The volume of publications in this domain is expected to continue growing. Currently, the United States, China, Germany, Italy, and France lead in this area of research. Prominent institutions such as the University of Minnesota, the University of Texas MD Anderson Cancer Center, and the University of Genoa have emerged as influential contributors. The most prolific journals include Frontiers in Immunology, Blood, and Cancers. Leading teams, including those led by Jeffrey S. Miller, Harry Dolstra, and Daniel Olive, have made significant contributions to this field.
To address emerging opportunities and challenges, greater collaboration and exchange between countries, institutions, and authors are essential. Current research hotspots focus primarily on allogeneic NK cell therapy, CAR-NK cell therapy, and memory-like NK cell therapy. Looking forward, intelligent NK cell engineering and combination therapies involving NK cells are expected to be key trends in future research.
Footnotes
Acknowledgments
We would like to thank Dr Nana Tang for her guidance and assistance in literature screening and the use of graphic tools.
Author Contribution
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (82260914); General Project of Jiangxi Provincial Natural Science Foundation (20192BAB205100); Traditional Chinese Medicine Advantageous Disease Cultivation Project of Jiangxi Provincial Administration of Traditional Chinese Medicine (Gan Cai She Zhi [2023] No. 70); Innovation and Entrepreneurship Training Program for College Students at Jiangxi University of Traditional Chinese Medicine (202410412256).
