Abstract
Background
Lung cancer is the most common cancer in the world and has become one of the malignancies with the highest incidence and mortality; more than half of patients die within one year of being diagnosed with lung cancer. In recent years, the concept of “patient-centered” service has gained popularity, and patients’ subjective feelings have gradually been used in clinical decision-making. Therefore, this study determined the application of visual patient report outcomes in the field of lung cancer, in order to provide reference for specific clinical practice.
Methods
Using the Web of Science core collection as the main analysis content, Citespace and VOSviewer were used to conduct this scientometric study.
Results
A total of 499 literatures that met the inclusion criteria were retrieved. The most prolific institution was The University of Texas MD Anderson Cancer Center, and the United States dominates this field.
Conclusion
The measurement of patient-reported outcomes is considered the gold standard for effectively evaluating patients' perceptions of clinical interventions or diseases. It is recommended that patient-reported outcomes be integrated into routine care for cancer patients in order to enhance communication between patients and healthcare providers.
Patient or public contribution
Not applicable. All data in this paper are obtained from the web of science database.
Introduction
Lung cancer (LC) is the most common cancer in the world and has become one of the malignancies with the highest incidence and mortality. 1 More than half of patients die within one year of being diagnosed with LC, and the 5-year survival rate is less than 18%. 2 Throughout treatment, patients with LC experience a range of discomfort, which can place physical, emotional, and financial stress on patients and their caregivers, potentially exacerbating the poor prognosis associated with LC .3,4 There is an urgentneed for additional objective tools to help guide clinical practice and improve the lives of those affected by LC.
In recent years, the concept of “patient-centered” service has gained popularity, and patients’ subjective feelings have gradually been used in clinical decision-making. Patient-reported outcome (PRO) refers to the subjective feelings directly from patients, without the intervention of clinicians or anyone else on the patient’s response. 5 PRO can provide unique indicators for clinical research and practice to study disease activity and evaluate treatment effects from the patient’s perspective. 6 Efficacy measures such as pain intensity or pain relief cannot be quantified, leaving patient perception as the only source of data. Therefore, PRO is particularly important in the evaluation of LC patients.
Bibliometrics analysis uses mathematics and statistical measurement to evaluate and predict the current situation of a certain field with literature as the research object. 7 By examining and evaluating the quantity and quality of scientific literature, we can objectively evaluate the development status and level of the field. 8 In recent years, PRO have been widely used in the field of LC. However, there is no literature analysis in this field. This study aims to analyze the research trends, hot spots and research frontiers of PRO in this field, and provide reference for further research.
Methods
We adopt Science citation index expanded Web of Science core collection WosCC (https://webofscience.clarivate.cn/) as the data source. WoSCC includes more than 12,000 high-quality scientific journals, has a relatively reliable database, and has been considered the most ideal database for bibliometrics research in the past.9,10 Our search strategy is as follows: TS=(“lung cancer” OR “carcinoma of lung” OR “pulmonary carcinoma” OR “lung neoplasms” OR “Adenocarcinoma of Lung” OR “Carcinoma, Non-Small-Cell Lung” OR “Small Cell Lung Carcinoma” OR “Squamous cell carcinoma of the lungs” OR “Adenocarcinoma of the lung” OR “large cell carcinoma of the lung” OR “Pulmonary Neoplasms” OR “Cancer of Lung”) AND TS=(“Patient-reported outcomes” OR “Patient-Reported Outcome Measurement Information System”) We chose 2013-2023 as our search dates, at the same time, publication types were limited to original articles and reviews, and only English literature was included. The search flow chart is shown in Figure 1. In addition, we are registered on the INPLASY Registry with the registration number: INPLASY202440061 (INPLASY – International Platform of Registered Systematic Review and Meta-analysis Protocols). This study is a literature review, and all data are obtained from an open database and do not require any approval or permission. This study followed the PRISMA reporting guidelines.
11
The Flowchart.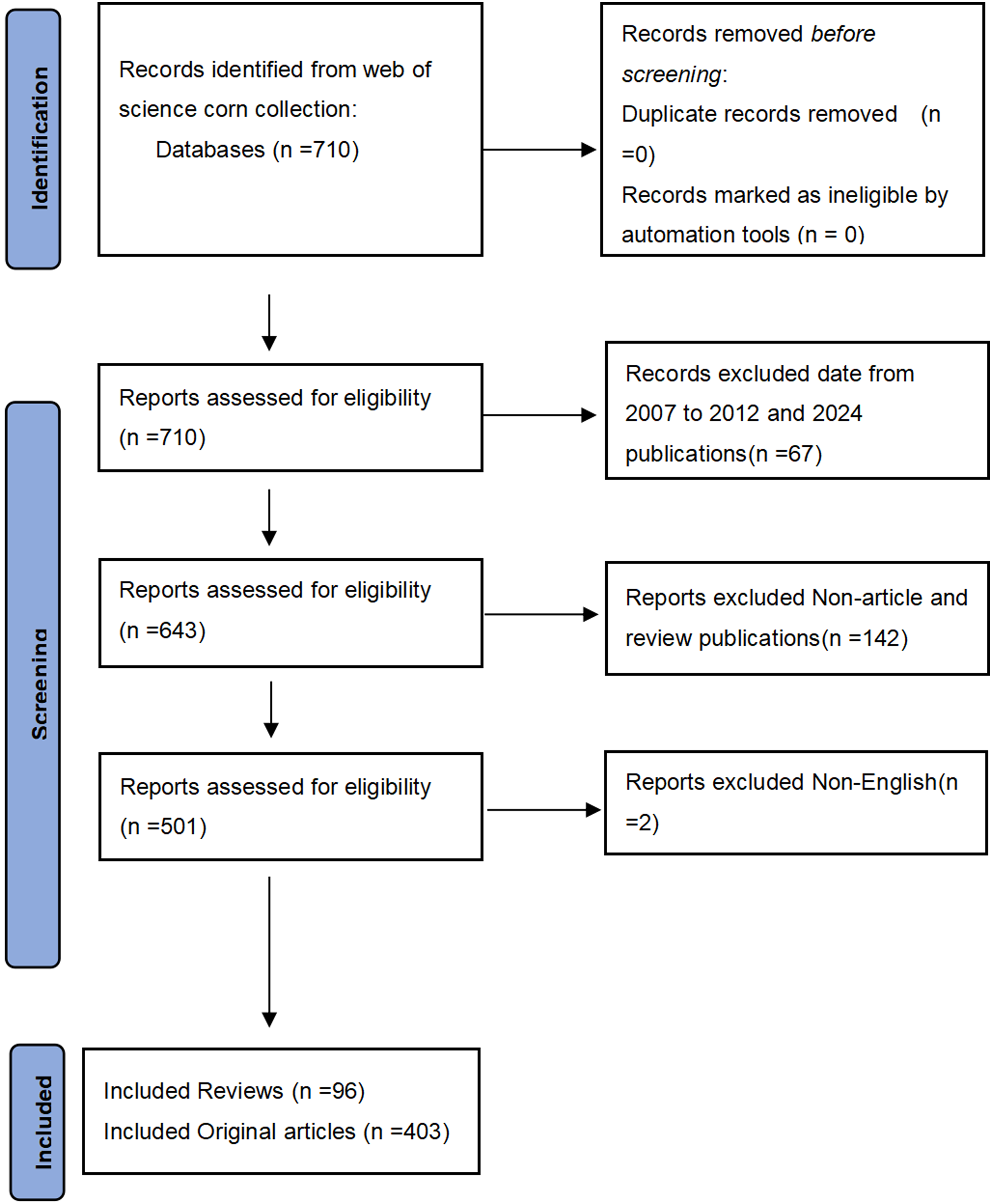
Data Acquisition
Two authors independently conducted the main search. Any differences were discussed with the third investigator before reaching a consensus. The data collected included number of publications, titles, authors, countries, institutions, journals, highly cited articles, and keywords. WOSCC data was converted to text format and imported into VOSviewer 1.6.18 and CiteSpace V. 6.3.R1.
Data Analysis
CiteSpace, as an article that analyzes and identifies hot spots and trends in the visual literature, is mainly used for keywords analysis in our research. 12 VOSviewer was also used to build visual networks, we used VOSviewer to create networks visualization maps. 13 H-index is a hybrid quantitative evaluation method of academic achievements of researchers, which can be used to evaluate the number and level of academic output of researchers. 14 The impact factor (IF) was derived from the 2022 Journal Citation Reports.
Results
Global Publication Results
From 2013 to 2023, WOSC searched 499 literatures, including 403 Original articles (80.76%) and 96 Reviews (19.24%). The total number of citations of the retrieved papers was 11,344 (11,052 without self-citations), and the average number of citations was 27.92. All selected publications have an H-index of 56. Figure 2 shows the number of articles published each year. Figure 3 shows the number of articles published after standardization. Over the past 10 years, the overall trend has increased. The year with the highest number of publications was 2022, with 76. It can be seen that PRO in the field of LC have attracted the attention of many scholars in recent years. Chart of global publication trends. Standardized time distribution map.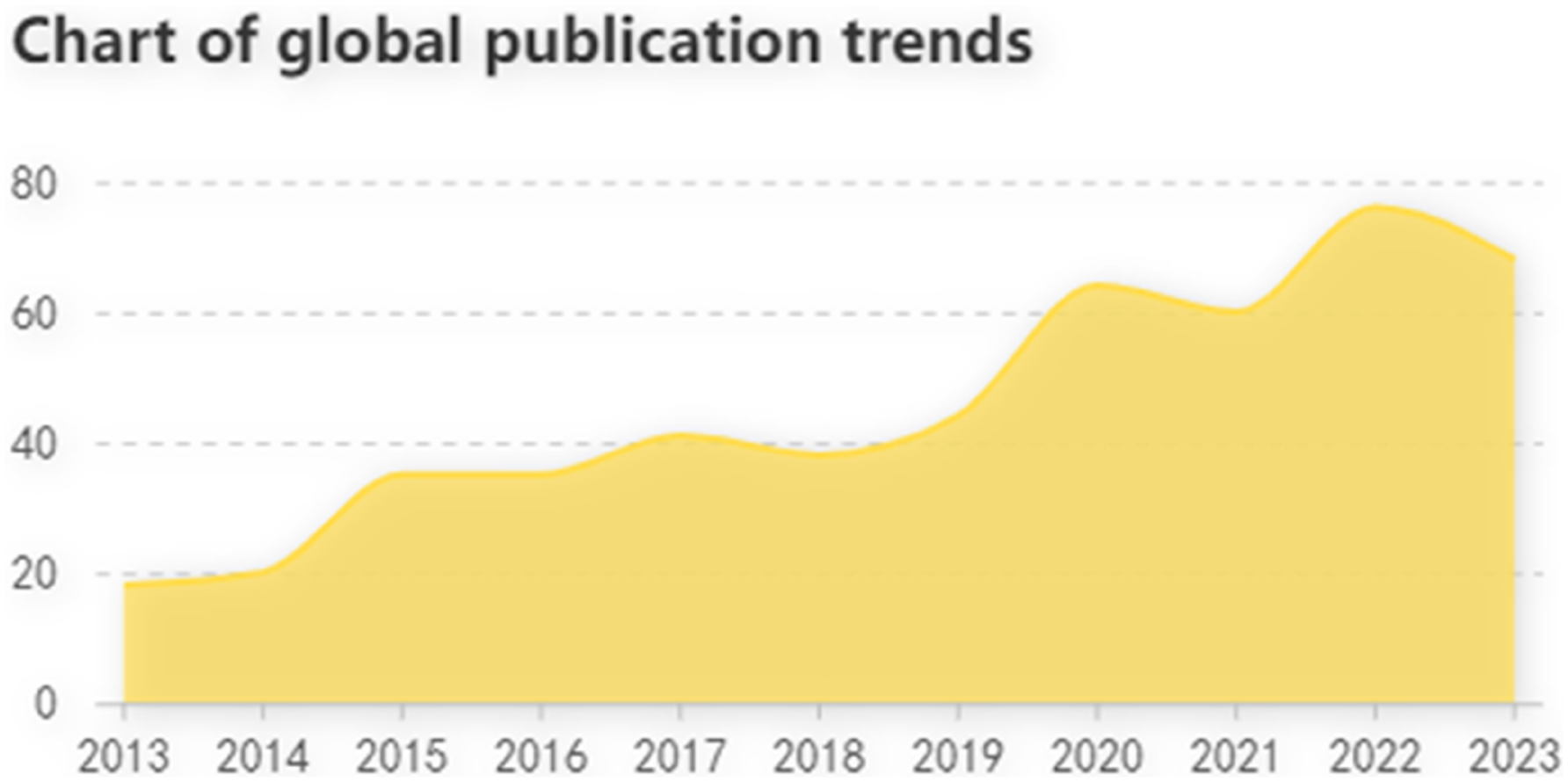

Distribution of Country
The top 10 Countries.
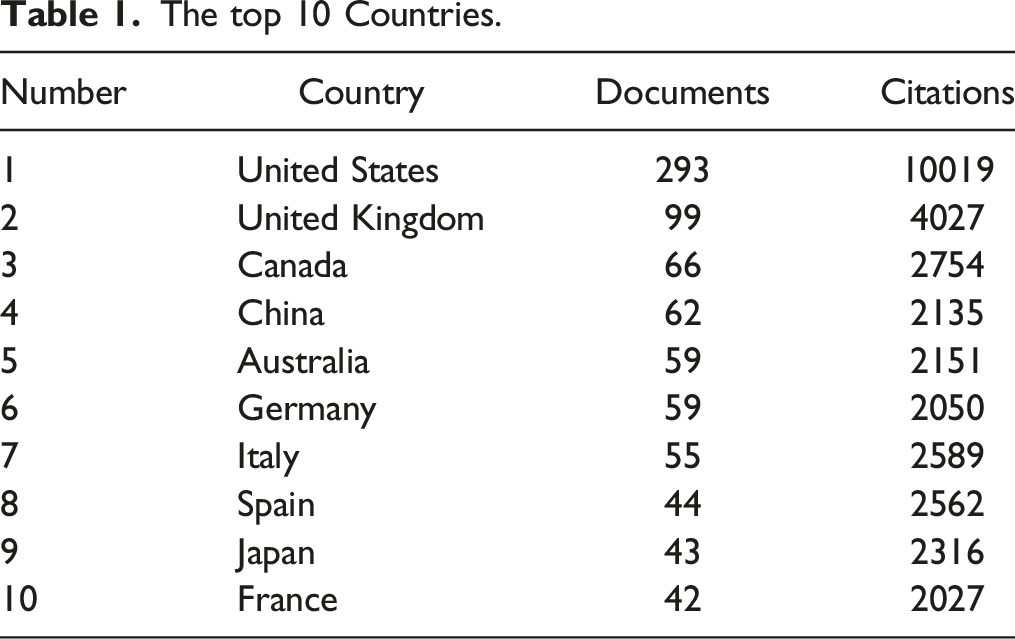

Country cooperation map.
Institution Distribution
The top 10 Institutions.


Institution visual map.
Author Distribution
VOSviewer parameters were set as follows: Method (Linlog/modularity) and a minimum number of documents of an author: 5. The obtained results were retrieved for 3573 authors, and 64 met the thresholds, and the maximum number of authors per document is 25. Each node represents a different author, the circle size represents the number of papers published by the researchers, the lines connecting the circles represent co-occurrence relationships between authors, and the different colors represent collaborative relationships between authors. As shown in Figure 6. Table 3 lists the top 10 authors in terms of number of articles over the past 10 years. Shi Qiuling from China is the most prolific author (n = 20), However, we found that felip, enriqueta, who ranks lower, is the most cited author (n = 1173). The number of papers published by the author can represent the author’s research activities in the field, but we cannot know the contribution of these authors to the article and whether they have the potential to contribute to the future development of the field. Author collaboration map. The top 10 Authors.

Journal Distribution
VOSviewer parameters were set as follows: Method (Linlog/modularity) and a minimum number of documents of a journal: 5. The obtained results were retrieved for 168 journals, and 28 met the thresholds, As shown in Figure 7 and Table 4. The journal with the highest number of publications was lung cancer (IF = 5.3), with 31 articles. However, we found that the most frequently cited Journal (n = 2731) was the journal of clinical oncology (IF = 45.4), which is the most reliable and authoritative resource for disseminating important clinical oncology research. Journal visual map. The top 10 Journals.

The dual map overlay of the journal shows the distribution of topics. The superposition of two periodicals is shown in Figure 8. The citing journal is on the left and the cited journal is on the right. The labels represent the disciplines covered by the journal, and the colored paths represent citation relationships. We can see the two most important paths. Green reference path according to the health, nursing, medicine, journal of molecular studies often been medicine/medical/clinical journal references. Visualization of periodical double map overlay.
Commonly Cited References
The Top 10 Articles cited.


Cited references visualization.
Keywords Analysis
Keywords are generally regarded as important indicators that reflect the research frontiers and hot spots of a subject. Citespace Setting parameters are as follows: Timespan: 2013-2023 (Slice Length=1), Selection Criteria: g-index (k = 25), term source (whole selection), node type (Keyword), as shown in Figure 10A. In this figure, there are 353 network nodes with a network density of 0.0377. Nodes represent keywords, the larger the node, the more keyword occurrences, and different colors represent different clusters. Purple nodes represent high centrality (high importance), such as “cell lung cancer, open label, health-related quality of life, functional assessment”. Through keyword analysis, 9 clusters were obtained (Figure 10B). The cluster ID is the number after the cluster. The number is displayed as # 0, #1, #2, and so on. The larger the size of the cluster, the more members the cluster contains. The most important cluster is “small-cell lung cancer”, including those keywords: “small-cell lung cancer”, “first-line treatment”, “patient-reported outcome”, “first-line nivolumab”, “advanced non-small cell lung cancer”. The timeline viewer can show the dynamic evolution path of the research hotspot represented by the keyword, and explore the time characteristics of the research field and the rise and fall process of the hot keyword research. Based on the keyword co-citation network, keyword emergence words were detected, and the Top 14 keywords with the strongest citation outbreak in LC were reported as PRO, as shown in Figure 10C. The report includes the start year, end year, and duration of the burst, with the blue line representing the timeline and the red segment on the blue timeline representing the burst detection. “Randomized controlled trial” citation outbreak was the strongest (n = 5), followed by “fatigue” (n = 3.6). The most common citation keywords in recent years were “fatigue”, “symptom”, “perbrolizumab” and “efficacy”. (A) Keywords visual map, (B) Keyword clustering, (C) Keyword emergence.
Discussion
With the diversification of LC treatment concepts and treatment modalities, patients are also facing new challenges, including dealing with new side effects and dealing with greater prognostic uncertainty. 15 Compared to patients with other cancers, patients with LC often have a higher burden of comorbidities and overall quality of life (OQL) is affected due to radiation therapy. 16 LC survivors often face a high number of physical symptoms and psychological distress, including breathing disorders, fatigue, depression, anxiety, and fear of cancer recurrence. 17 Consistent use of PRO can better capture the patient experience, improve patient communication with healthcare providers, and improve the OQL.
Capture the Patient Experience
In clinical practice, medical personnel may underestimate the patient’s symptoms or impact on the patient, and this inconsistent assessment may lead to delayed care. 18 In this case, the role of PRO is particularly significant, and its measurement method is still mainly based on paper questionnaires/scales, but different from traditional questionnaires, PRO content is more oriented to patient self-expression and self-analysis, 19 which can better meet the needs of lung cancer patients during clinical treatment and cancer survival.
Promote Patient Expression
Real World studies (RWS) are the collection and analysis of patient-related data in a real world setting to obtain clinical evidence of the use value and potential benefits or risks of medical products. 20 In 2016, the United States passed the 21st Century Cures Act, which clarifies that the Food and Drug Administration (FDA) can use real-world data, where appropriate, as evidence for approval of medical devices and drugs in post market studies and for the development of new indications. 21 PRO is a tool that can collect patients’ data in the real world. Using PRO is conducive to better solving clinical practical problems and predicting the clinical response of LC patients to treatment. 22 In addition, PRO-based symptom management monitoring can provide clinicians with the symptom characteristics of LC patients and provide precise intervention for symptoms, which is conducive to prolonging patient survival, so that patients in the intervention group can tolerate chemotherapy for longer than conventional treatment. 23 However, some studies have pointed out that PRO only reduces the number of visits by recognizing patients’ symptoms, but does not improve the treatment outcome, 24 which indicates that in the process of implementing PRO, strict standard plans and follow-up times need to be formulated, timely early intervention and prevention of disease deterioration.
Curative Effect Evaluation
In the process of drug development/use, patient health preference information is an important influencing factor in the decision-making of drug development and medication practice. 25 This is consistent with the PRO development perspective, which puts the patient perspective at the core, which is conducive to capturing a broader range of patient perceptions of self-symptoms in clinical drug therapy, reflecting the real differences in patient treatment outcomes, providing important evidence for clinical intervention, and enabling multidisciplinary teams to implement early diagnosis and optimal treatment. 26 PRO has been recognized by regulators, and the FDA has successively issued patient-centered drug research and development guidelines, 27 committed to promoting the collection of patient experience data and related information, and its application in drug development and approval, thereby promoting the value evaluation of drugs and medical devices, and maximizing the role of patient-reported data in the patient experience. In a large number of randomized controlled trials of LC drugs, PRO has become an important indicator of clinical comprehensive evaluation of drugs and health benefits for LC patients.28-30 In the era of precision medical services, taking PRO as the primary endpoint can reflect the improvement of life quality and experience of LC patients and its clinical value, providing a new perspective for drug evaluation. However, clinical data provided by the FDA showed that PRO data in cancer trials submitted as part of the drug marketing application lacked a clear sensitivity analysis and was inconsistent across groups.31,32 It also suggests that standardized procedures must be developed to improve PRO reporting and reduce omissions in the process of clinical trials, and multi-disciplinary paths and models should be tried to maximize the function of PRO.
PRO Development
With the continuous development of information technology, PRO measurement methods are also improving, and modern electronic patient report outcomes(ePRO) are not only an electronic version, but based on information systems, real-time data collection, and more as an e-health intervention in LC. 33 The Patient Reported Outcome Measurement Information System (PROMIS) uses item response theory and Computer Adaptive Test (CAT) to collect patients' self-reported physiological, psychological and social information, which is a more advanced electronic application of PRO. It can accurately predict the overall survival of patients and the adverse reactions after treatment.34,35 Several studies have shown that the use of modern electronic and informational PRO in LC is a valuable strategy.36-38 The web-based PRO monitoring not only benefits the overall survival of LC patients, but also reduces the follow-up cost. 39 However, it should be noted that the characteristics of the interviewees will affect the accuracy, female or elderly patients are more inclined to skip the items or take a longer time to fill in the items, resulting in a decline in accuracy. 40 Therefore, we need to invest more patience in the face of this group of lung cancer patients to ensure the effectiveness of PRO results.
Limitation
Although our research has found some results, our selection was based on bibliometric tools, and the study data were only retrieved from the WoSCC database, while data from other relevant search engines (such as PubMed, Embase and Cochrane Library) were ignored. Finally, our study was limited to the last 10 years, and some high-contribution articles or authors were ignored because they were not in our time frame.
Conclusions
In this study, we found that The University of Texas MD Anderson Cancer Center is the number one institution for publication, and the United States is the most productive country. In patient-centered care, PRO is the gold standard for effectively evaluating patients' feelings about clinical intervention or disease. In the future, PRO should be incorporated into routine care for LC treatment, and cloud-based medical records should be combined with electronic applications to maximize medical services for LC patients and improve prognosis.
Footnotes
Author Contributions
(I) Conception and design: Zhirong Gu、Liu Wu; (II) Administrative support: Silin Zheng, Min Huang; (III) Provision of study materials or patients: Jinxing Li; (IV) Collection and assembly of data: Zhirong Gu, Liu Wu; (V) Data analysis and interpretation: Zhirong Gu; (VI) Manuscript writing: All authors; and (VII) Final approval of manuscript: All authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
All data is sourced from an open database, so no additional statement is required.
