Abstract
Objective
Our study aimed to update demographic profiles of sinonasal adenocarcinoma (SNAC) between 2000 and 2020, identify independent prognostic risk factors, and devise a predictive nomogram for overall survival (OS).
Methods
Utilizing the Surveillance, Epidemiology, and End Results (SEER) database, cases of SNAC from 2000 to 2020 were analyzed for incidence trends. Univariate and multivariate Cox regression models helped pinpoint factors impacting patient survival. A prognostic nomogram was then created based on these factors and assessed via concordance index, ROC analysis, and calibration curves.
Results
Analysis of 488 SNAC patients indicated a rising incidence, peaking among 60-69-year-olds, with a male predominance and the highest occurrence in White populations. Common sites of occurrence included the nasal cavity, and the maxillary and ethmoid sinuses. The nomogram, developed with 184 patients, highlighted older age (≥70), male sex, advanced T stages (T4b, T4a), distant metastasis, larger tumors (≥5 cm), and lack of surgery as poor prognostic indicators. Nomogram performance demonstrated strong predictive capabilities in both training and validation cohorts.
Conclusion
This investigation, leveraging the SEER database, elucidates the epidemiology of SNAC and pinpoints key adverse prognostic determinants: age ≥70, male gender, T stages T4b and T4a, presence of distant metastasis, tumor size exceeding 5 cm, and absence of surgical intervention. Moreover, it introduces a novel nomogram capable of accurately forecasting 1-, 3-, and 5-year OS for SNAC patients, thereby enhancing disease comprehension and facilitating the formulation of tailored therapeutic strategies by clinicians.
Background
Sinonasal adenocarcinoma (SNAC) is a rare malignancy with an annual incidence of only 0.44 cases per million in the US population. 1 In the 5th edition of the World Health Organization (WHO) classification in 2022, SNAC was divided into two subtypes: intestinal adenocarcinoma (ITAC) and non-intestinal adenocarcinoma (non-ITAC). ITAC is the predominant type, accounting for 80% of all cases. 2 Previous reports have shown a strong association between ITAC and exposure to hardwood dust, while non-ITAC is considered unrelated to wood dust exposure. 3 Due to the non-specific and relatively mild nature of early symptoms, the diagnostic latency of SNAC is long, and approximately 40.6% of patients are diagnosed at clinical stage IV at the time of initial diagnosis. 4 Surgery is the first-line treatment for SNAC. 5 In recent years, the treatment modality of postoperative adjuvant radiotherapy or chemotherapy has also been gradually explored, but the studies are mainly based on single-centre case reports, lack of large-sample evidence of evidence-based medicine, and have a limited role in the study of the epidemiological characteristics of SNAC and its prognostic factors.
The Surveillance, Epidemiology, and End Result (SEER) database is a cancer registry system based on the US population, covering approximately 30% of the US population. It includes demographic variables such as age at diagnosis, sex, race, and clinical variables such as clinical diagnosis, staging, pathological type, tumor size, and treatment methods. Due to the extensive and representative nature of the data in the SEER database, as well as the relative completeness of the variables, the data from the SEER repository are widely used in constructing nomograms for the prognosis of malignant tumors. A nomogram is a graphical tool that facilitates the understanding of statistical prediction models and can predict the probability of specific clinical events. 6
The objective of this study is to utilize the SEER database from 2000 to 2020 to conduct a comprehensive analysis of the epidemiological trends of SNAC patients and to delve into the related risk factors that influence the prognosis of SNAC. Subsequently, based on the identified independent prognostic factors, a nomogram model for the prognosis of SNAC will be constructed, which is expected to facilitate personalized treatment for SNAC and enhance therapeutic efficacy.
Materials and methods
Patient Selection
We conducted a retrospective study. Firstly, we obtained permission to access the SEER database. We used SEER*Stat software (v. 8.4.2) to collect data, downloaded data from 17 centers, and the data was uploaded in November 2022, including clinical and demographic data. The inclusion criteria were as follows: (1) the diagnosis time was between 2000 and 2020; (2)Tumor histological code was adenocarcinoma (8140) and adenocarcinoma with intestinal type (8144); 3) the primary tumor site was malignant neoplasm of the nasal cavity (C30.0), paranasal sinuses (C31.0, C31.1, C31.2, C31.3, C31.8, and C31.9), and the code of the primary site was based on the International Classification of Diseases for Oncology, 3rd Edition (ICD-O-3). The exclusion criteria were patients who were diagnosed only by autopsy or death certificate.
For the study of SNAC epidemic trends, all cases were included regardless of whether the pathology or survival data were complete.
For the study of prognostic risk factors of SNAC and the impact of different treatment methods on SNAC prognosis, cases with incomplete clinical staging, survival status, or survival information were excluded. Then, the included cases were randomly divided into a Training set and a validation set in a ratio of 7:3. The Training set was used to screen for independent risk factors for adverse prognosis of SNAC, develop prognostic prediction nomograms using independent risk factors, and analyze and evaluate their performance by measuring consistency indexes (C-index), Receiver Operating Characteristic (ROC) Curve, Area Under Curve (AUC), calibration curves, etc. Finally, the performance of the model was further validated in the validation set. The specific operation process is shown in Figure 1. The reporting of this study conforms to TRIPOD guidelines. Flowchart of the study. SEER, surveillance, epidemiology and end results.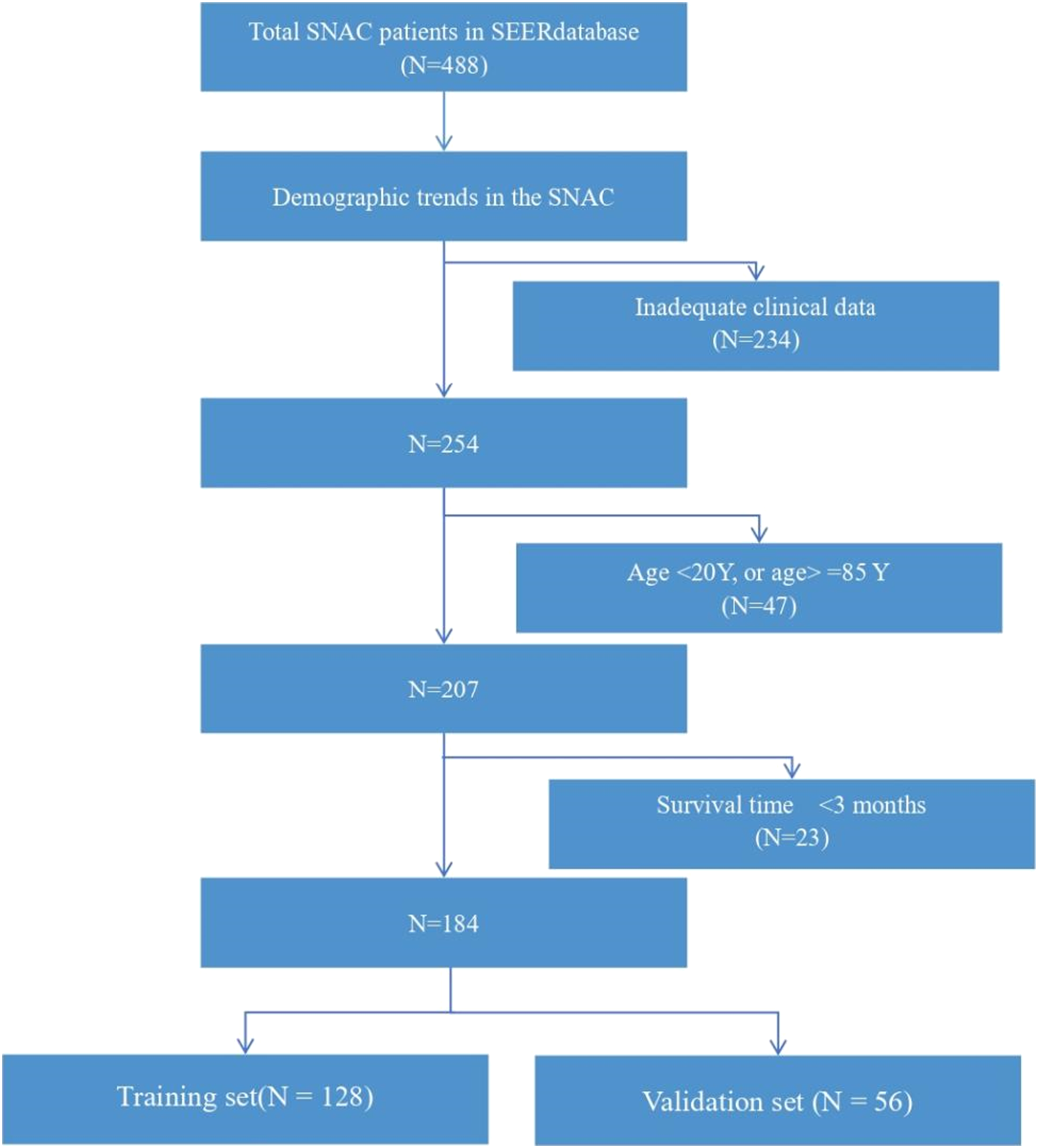
Statistical Analysis
This study used SPSS 26.0 and R software (v. 4.3.1) for statistical analysis. The chi-square Training or Fisher’s exact Training was used to compare the distribution of two groups of variables. Univariate and multivariate Cox regression analysis were used to screen for risk factors of adverse prognosis of SNAC. To control for bias, variables with P < 0.1 in univariate Cox regression analysis were included in stepwise multivariate Cox regression analysis. We also used Kaplan-Meier survival curve analysis to compare survival differences between different treatment methods. All statistical analyses were performed using two-tailed Trainings, and P < 0.05 was considered statistically significant.
Results
Epidemiologic Feature
We retrieved 488 SNAC cases with demographic data from the SEER database. Demographic data included age, gender, race, tumor primary site, histopathological type, and time to diagnosis. The results showed an overall upward trend in the incidence of SNAC, with the lowest number of new diagnoses in 2006 and the highest number in 2020 (Figure 2). For those aged 60 to 69 years, accounting for 25% of the total cases, the gender ratio was 1.5:1,60.25% (294 cases) of male and 39.75% (194 cases) of female. The vast majority of SNAC reports were from white patients (80.74%, 394), vs 11.07% (54). Most of the lesions involved the nasal cavity (50.61%, 247 cases), followed by the maxillary sinus (21.52%, 105 cases) and the ethmoid sinuses (15.57%, 76 cases), while the frontal sinus was the rarest primary site currently reported, with only 1.23% (6 cases). Demographic data of 488 patients with nasal sinuses adenocarcinoma are shown in Table 1. Annual diagnosis of SNAC. Demographic Data of the 488 SNAC Patients.

Prognosis-related risk factors
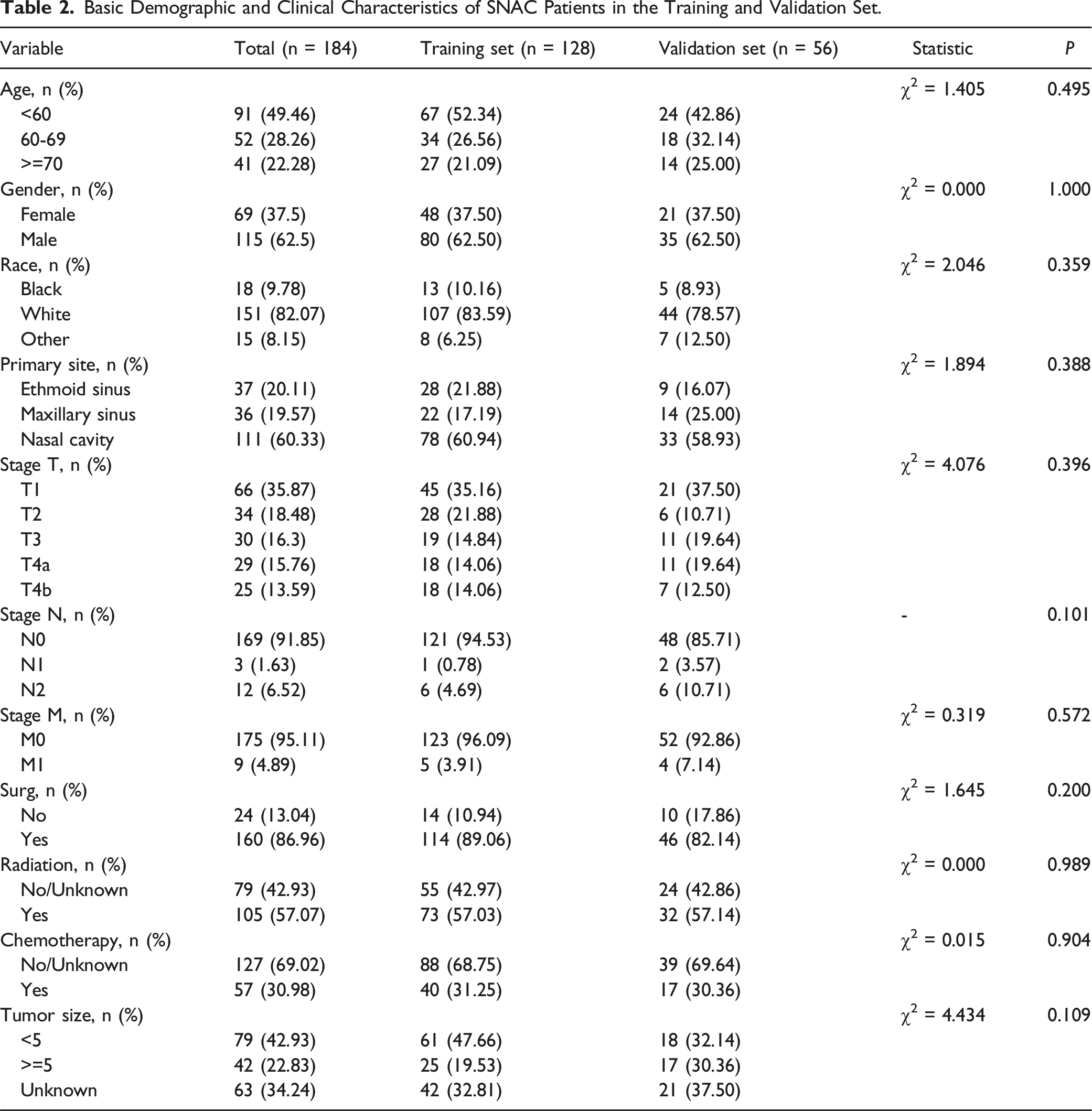
Basic Demographic and Clinical Characteristics of SNAC Patients in the Training and Validation Set.
Results of Univariate Cox Regression and Multivariate Cox Regression Analysis.

Performance and validation of the nomogram for predicting SNAC prognosis
In the multivariate Cox regression model, the predictive nomogram for SNAC prognosis was integrated, and it is shown in Figure 3. The 1, 3, and 5-year AUCs of the nomogram in the training set were 0.997 (95%CI: 0.989-1.004), 0.945 (95%CI: 0.897-0.994), and 0.969 (95%CI: 0.939-1.000) (Figure 4(A)–(C)), respectively. These high AUC values indicate that the model has very high prediction accuracy and can well distinguish patients with different risks. The corresponding validation sets at the same time points in the training set had AUCs of 0.776 (95% CI: 0.610-0.941), 0.841 (95%CI: 0.718-0.964), and 0.821 (95%CI: 0.689-0.954) (Figure 4(D)-(F)). Although the AUC values of the validation set are lower than those of the Training set, they still show good prediction performance. The consistency C-index of the Training set was 0.904 (95%CI: 0.848-0.960), while that of the validation set was 0.789 (95%CI: 0.701-0.877), indicating that the model has good discrimination and further confirms the accuracy of model prediction. The calibration curve shows good consistency between the predicted probability of SNAC prognosis and the observed probability (Figure 5). nomograms predicting 1-year, 3-year, and 5-year OS in SNAC patients. The ROC curves for predicting patients’ OS at 1-year, 2-year, and 3-year for the training (A-C), validation (D-F) cohorts. ROC, receiver operating characteristic; AUC, area under the curve; OS, overall survival. Calibration curves for predicting patients’ OS at 1-year, 2-year, and 3-year for the training (A-C), validation (D-F) cohorts. OS, overall survival.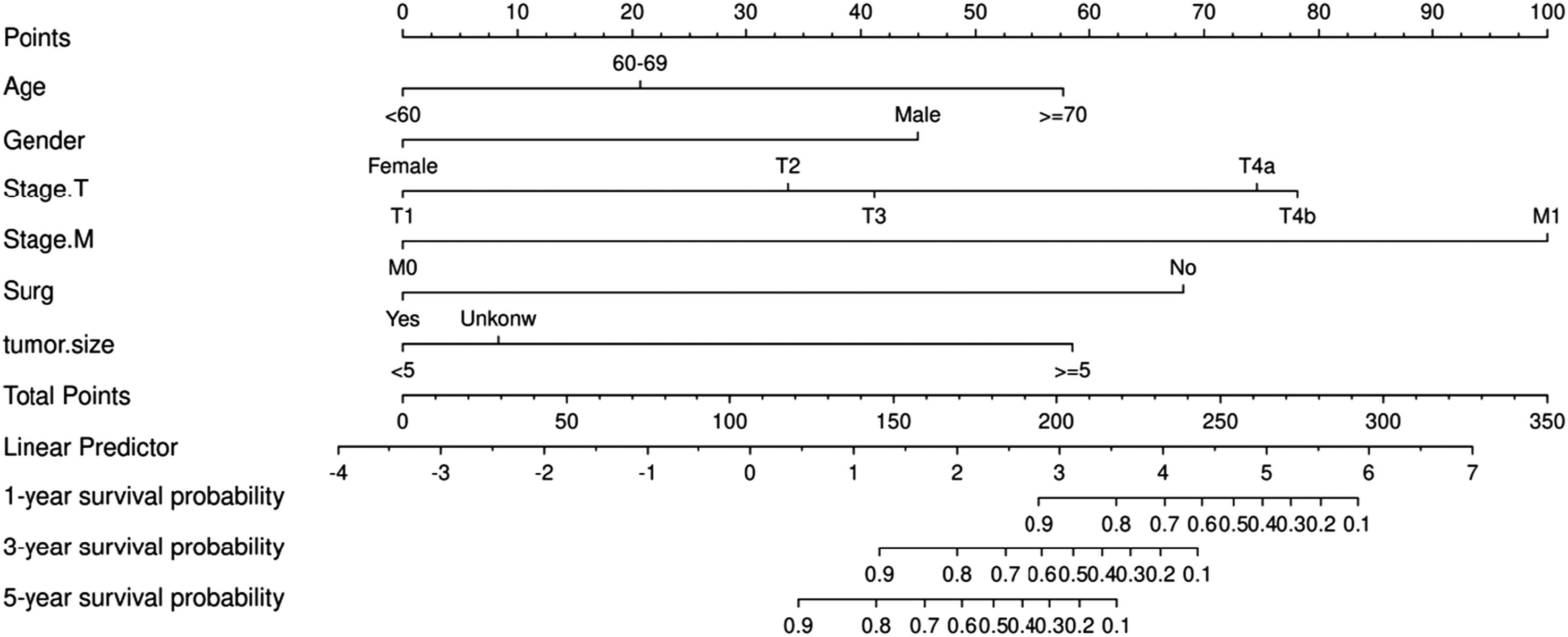


Discussion
Malignant tumors of the sinonasal cavity are relatively rare, accounting for 1% of all malignancies and 3% of all head and neck malignancies, SNAC is the second most common malignant tumor of the sinonasal cavity after squamous cell carcinoma, comprising approximately 13% of all sinonasal malignancies. 7 In a population-based study of SNAC from 1973 to 2009, more than half of the patients were diagnosed at the age of over 55 years, and the male-to-female ratio was slightly higher, at 1.06:1.00. The majority of SNAC cases were reported in white individuals, with most lesions involving the sinuses, followed by the nasal cavity. 8 We updated the population-based demographics and survival trends of SNAC from 2000 to 2020. Our study found that the highest incidence of SNAC occurred in the age group of 60-69 years, with a male-to-female ratio of approximately 1.5:1, showing an increase compared to previous studies. Similar to the 1973-2009 findings, SNAC was more commonly reported in white individuals, followed by black individuals. Furthermore, our study provided further insight into the primary sites of SNAC, with over half of the cases occurring in the nasal cavity, followed by the maxillary sinus and ethmoid sinus, while the frontal sinus was the rarest primary site, consistent with the findings by Patel et al, in 2020 based on the National Cancer Database (NCDB). 1
Clinical symptoms of SNAC are not typical and early symptoms often include nasal obstruction, bloody nasal discharge, epistaxis, and reduced sense of smell, which are not easily distinguishable from rhinitis, In advanced stages, patients may experience proptosis, diplopia, or neurological symptoms. 1 Approximately 40% of patients have presented with relevant symptoms for more than 6 months before diagnosis, 9 and at the time of initial diagnosis, most patients are already at stage IV. 2 Surgery remains the standard treatment for SNAC. The 5-year survival rate for SNAC exhibits heterogeneity, with previous reports indicating a range from 63% to 85% for 5-year survival in patients with SNAC10,11 It is crucial to actively explore adverse risk factors that influence the prognosis of SNAC for monitoring and personalized treatment. The study reports that achieving maximum resection of tumors through surgery and ensuring negative surgical margins are critical factors influencing the prognosis of SNAC. 12 Given the often subtle early symptoms of SNAC, the majority of cases are detected at advanced stages, where the disease extensively invades vital structures such as the orbit and skull base, as well as the dura mater, which constitutes key determinants of the prognosis for SNAC patients. 13 Previous reports have shown that poorly differentiated SNAC, advanced stage at diagnosis (T4), lymph node involvement, or involvement of the meninges or brain parenchyma tend to have worse prognosis.7,14 We used a Cox proportional hazards model for analysis and found that age ≥70 years, male, late T stage (T4b and T4a), distant metastasis, tumor size ≥5 cm, and lack of surgical treatment were independent risk factors for poor prognosis in SNAC. The study by Huang H et al categorized tumor size into three groups: <40 mm, ≥40 mm, and unknown. The research found that the larger the tumor size, the worse the survival rate (≥40 mm, HR, 1.783, P = 0.047; unknown, HR, 1.806, P = 0.044). This is similar to the conclusion of the present study, indicating that tumor size is an independent factor affecting the prognosis of SNAC patients. 15
Surgery is considered the primary treatment method for SNAC, 5 and our study also confirmed that surgery is a significant variable influencing the prognosis of SNAC. Previously, open craniofacial resection and maxillary resection were the main treatment methods for sinus tumors. 16 At present, endoscopic surgery has become the main surgical treatment for malignant sinus tumors.17-20 This is because endoscopy can not only ensure the complete resection of the tumor, but also can more directly approach the tumor without altering or damaging healthy tissues. In addition, the patient’s recovery is usually faster and simpler, avoiding facial incisions, thereby obtaining better cosmetic effects and potentially preventing further complications, including nasal cutaneous fistula.21-23 A systematic review including all types of sinus tumors showed that the 5-year overall survival rate of patients receiving endoscopic surgery was significantly higher than that of patients treated with open surgical methods. 24 Furthermore, studies have shown that in addition to resecting the primary tumor, combined bilateral ethmoidectomy can maximize the reduction in the occurrence of secondary tumors, as tumor nests have been pathologically proven to exist in healthy mucosa distant from the tumor. 25 The need for adjuvant therapy after surgery is still controversial. Li 26 found that the overall survival (OS) in the surgery-only group was slightly higher than in the surgery plus radiotherapy group. Blanch 27 proposed that the addition of radiotherapy to surgery in early-stage lesions did not provide a survival benefit, while retrospective analyses by Antognoni 28 and Huber GF 29 indicated that endoscopic surgery combined with postoperative radiotherapy was a safe, minimally invasive, and most effective treatment option for SNAC. Similar to radiotherapy, the role of chemotherapy in the treatment of SNAC remains unclear. In the study by Roux FX, cisplatin and 5-FU may have an effect in the treatment of SNAC. 30 Tachino H reported a case of complete pathological and clinical remission in a patient with locally advanced SNAC who underwent neoadjuvant concurrent chemoradiotherapy followed by surgical treatment, his study suggested that a regimen involving super-selective intraarterial infusion of cisplatin (CDDP) combined with conventional fractionated radiotherapy followed by salvage surgery may be beneficial for the treatment of SNAC. 31 In our study, univariate regression analysis indicated that chemotherapy was associated with the prognosis of SNAC, but multivariate regression analysis revealed that chemotherapy was not an independent risk factor for the prognosis of SNAC.
In conclusion, we have summarized the epidemiological characteristics of SNAC over the past 20 years and identified the independent prognostic risk factors affecting SNAC, we identified that being aged 70 or above, male, with T staging of T4b and T4a, distant metastasis, tumor size of 5 cm or above, and no surgical treatment were independent risk factors for poor prognosis of SNAC, which significantly contributes to our understanding of SNAC. In addition, this study also established a nomogram model for predicting the overall survival (OS) of SNAC patients at 1, 3, and 5 years. The model not only improved the accuracy of predicting the overall survival rate of SNAC patients but also facilitated personalized risk assessment and treatment planning. Moreover, our findings underscore the importance of surgical intervention in improving prognosis and have enhanced the current knowledge of the effectiveness of treatment modalities for SNAC.
However, this study still has some limitations: 1. This is a retrospective analysis, which may be subject to selection bias in data selection. 2. The study population is derived from the SEER database, which mainly reflects data from Western countries. 3. Due to the rarity of the tumor, even though data from 2000-2020 were collected, the data are still limited, and it is not possible to analyze the impact of treatment method on prognosis.4. In the 5th edition of the World Health Organization (WHO) classification in 2022, SNAC was divided into two subtypes: ITAC and non-ITAC, the histological subtype plays a significant role in constructing predictive models for SNAC, however, the SEER database utilizes the International Classification of Diseases for Oncology, Third Edition, Revised (ICD-O-3) coding system, which does not fully align with the recently released WHO cancer classification criteria. Under the ICD-O-3 coding framework, the SEER database categorizes 8140/3 as “Adenocarcinoma, Not Otherwise Specified (NOS),” and 8144/3 as “Adenocarcinoma, intestinal type.”, Consequently, due to the limitations of the available data and the specificity of the ICD-O-3 coding used in the SEER database, the histological subtypes (ITAC/non-ITAC) were not included as variables in our prognostic model.
Conclusion
This investigation, leveraging the SEER database, elucidates the epidemiology of SNAC and pinpoints key adverse prognostic determinants: age ≥70, male gender, T stages T4b and T4a, presence of distant metastasis, tumor size exceeding 5 cm, and absence of surgical intervention. Moreover, it introduces a novel nomogram capable of accurately forecasting 1-, 3-, and 5-year OS for SNAC patients, thereby enhancing disease comprehension and facilitating the formulation of tailored therapeutic strategies by clinicians.
Footnotes
Acknowledgments
The authors are grateful to all the patients, researchers, and institutions that participated in the SEER database.
Author Contributions
Lu Yang and Yufeng Gu co-designed the study and were responsible for data collection, organization and preliminary analysis and manuscript writing. Lei Yu conducted in-depth data analysis and interpretation, particularly in the construction of the prognostic nomogram. Qiang Zhou, Li Liu, Ying Mao, and Ji Ma participated in data collection and organization and discussed the initial results. Yong Li and Na Li provided overall guidance and supervision, ensuring the scientific rigor and robustness of the research.All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
