Abstract
Epidermal growth factor receptor (EGFR) exon 20 insertion (ex20ins) mutations are the third most frequent EGFR mutation type, following only exon 19 deletions and exon 21 L858R point mutations. EGFR ex20ins mutations are found in approximately 4%-12% of all EGFR-positive non-small cell lung cancers (NSCLCs). Unlike classical EGFR mutations, EGFR ex20ins mutations display remarkable subtype diversity and heterogeneity. Patients harboring these mutations generally have an inferior prognosis because of insensitivity to conventional treatment approaches such as immunotherapy, chemotherapy, and targeted therapy. Consequently, there remains a significant unmet medical need for efficacious treatments. Recently, amivantamab and sunvozertinib have demonstrated notable efficacy as first-line treatments, and several other promising novel targeted drugs are also challenging the status quo of traditional first-line platinum-based chemotherapy regimens. These developments are anticipated to further improve survival outcomes for NSCLC patients with EGFR ex20ins mutations. Hence, this review summarizes the epidemiology, molecular attributes, detection methodologies, and therapeutic advancements for EGFR ex20ins mutations in NSCLC, and briefly discusses the mechanisms of drug resistance.
Introduction
As the third most common EGFR mutation in NSCLC patients, EGFR ex20ins mutations account for 4% to 12% of all EGFR-mutated NSCLCs.10-12 The clinical characteristics of NSCLC patients with EGFR ex20ins mutations resemble those of patients with classical EGFR mutations; notably, EGFR ex20ins mutations are more common in women (47% of ex20ins vs 28% of EGFR wildtype (WT), P = 0.003), 13 Asian patients (15% of ex20ins vs 4% of EGFR WT, P = 0.02), 10 and never-smokers (56% of ex20ins vs 20% of EGFR WT, P < 0.000110 and 67% of ex20ins vs 26% of other uncommon EGFR mutations, P < 0.01).14,15
Unfortunately, due to the absence of effective targeted therapies, patients with EGFR ex20ins mutations have notably shorter median overall survival (mOS, ranging from 11.04 to 24.4 months) than patients with classical EGFR mutations (approximately 38.6 months).16-19 Recent strides in precision medicine and biopharmaceutical technology have illuminated multiple exon 20 mutation types and sparked promising research endeavors focused on novel targeted drugs and bispecific antibodies. These advancements hold immense potential to facilitate significant breakthroughs in the diagnosis and treatment of EGFR ex20ins mutations. This review focuses primarily on molecular characteristics related to EGFR ex20ins mutations, highlighting variations among detection methods. Furthermore, the review provides a thoughtful summary of advancements regarding treatment options for patients with EGFR ex20ins mutations, briefly overviews the intricate relationship between different insertion sites and targeted therapy effectiveness, and discusses potential drug resistance mechanisms and unique challenges faced by patients with brain metastases. The ultimate goal of this review is to assist patients with EGFR ex20ins mutations in selecting the most appropriate treatment strategies to improve quality of life.
The Molecular Characteristics and Heterogeneity of EGFR ex20ins Mutations
EGFR, a receptor tyrosine kinase, plays a pivotal role in transducing extracellular signals into the cell interior, triggering a cascade of signaling pathways, such as the PI3K/AKT/mTOR and RAS/RAF/MEK pathways, ultimately promoting cell growth, proliferation, and migration. Mutation-induced aberrant activation of EGFR further promotes tumor growth by continuously stimulating downstream signaling pathways.
Structurally, EGFR comprises 28 exons, with mutations predominantly occurring within exons 18-21 of the intracellular tyrosine kinase domain. Among these, EGFR exon 20 encodes amino acid (AA) at the E762–K823 position, which contains 2 important regions spanning across codons E762–C775: the regulatory αC-helix domain (E762–M766) and the adjacent loop that follows it (A767–C775). The loop region can be further divided into the near-loop region (A767–P772) and the far-loop region (H773–C775) according to the different inserted sites (Figure 1).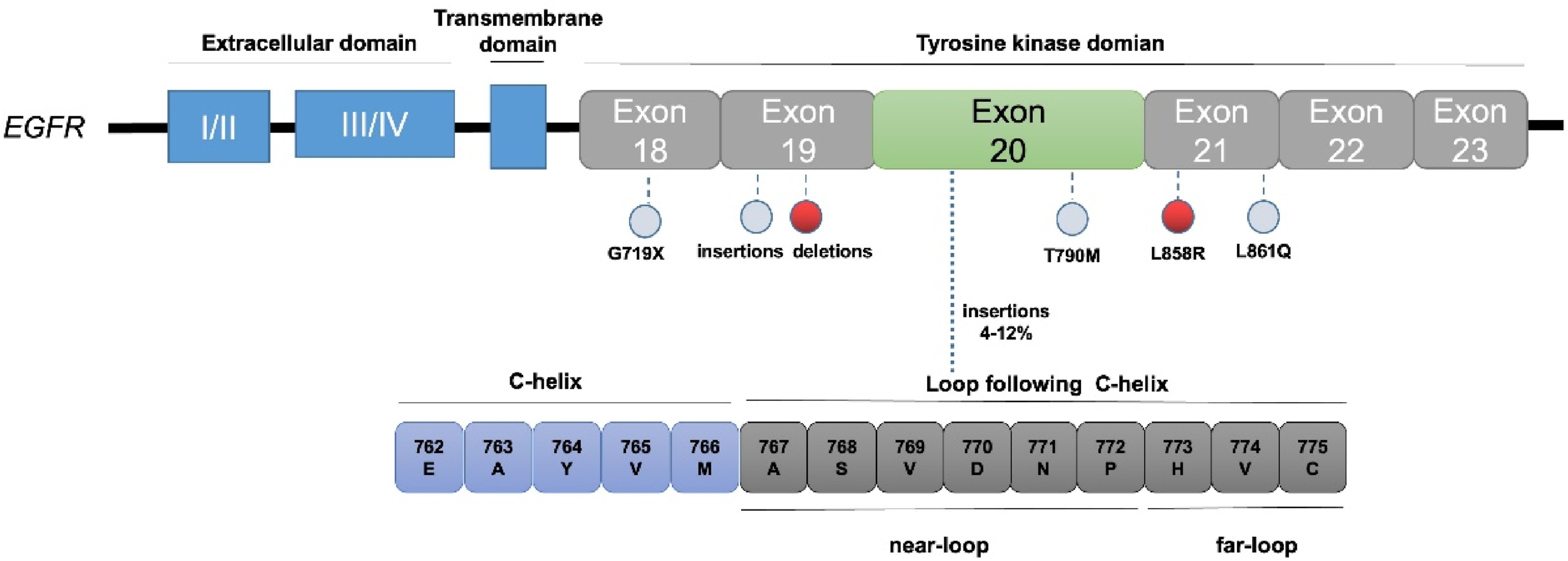
EGFR ex20ins mutations typically involve the insertion or duplication of 3-21 base pairs (equivalent to 1-7 codons) within codons E762–C775 of the EGFR gene. This alteration causes a shift in the α-C helix and phosphate-binding loop (P-loop) into the drug-binding pocket from two directions, inducing steric hindrance and reducing drug-binding pocket size. 23 Notably, over 90% of EGFR ex20ins mutations manifest in the loop region, with near-loop and far-loop insertions accounting for approximately 70% and 30% respectively, while a minor fraction (0%-7%) occurs within the C-helix.20,24-26 Representative near-loop insertion mutations include S768dupSVD, A767dupASV, D770insNPG, and D770delinsGY, whereas far-loop insertion mutations include H773insNPH, H773dupH, V774insAV, and V774insPR. 27
Intriguingly, EGFR ex20ins mutations are similar to EGFR T790 M in terms of alignment of the gatekeeper residue Thr790, which results in increased affinity for ATP and reduced binding of traditional EGFR-TKIs. They are thus recognized as a significant mechanism of acquired resistance to noncovalent traditional EGFR-TKIs. 23
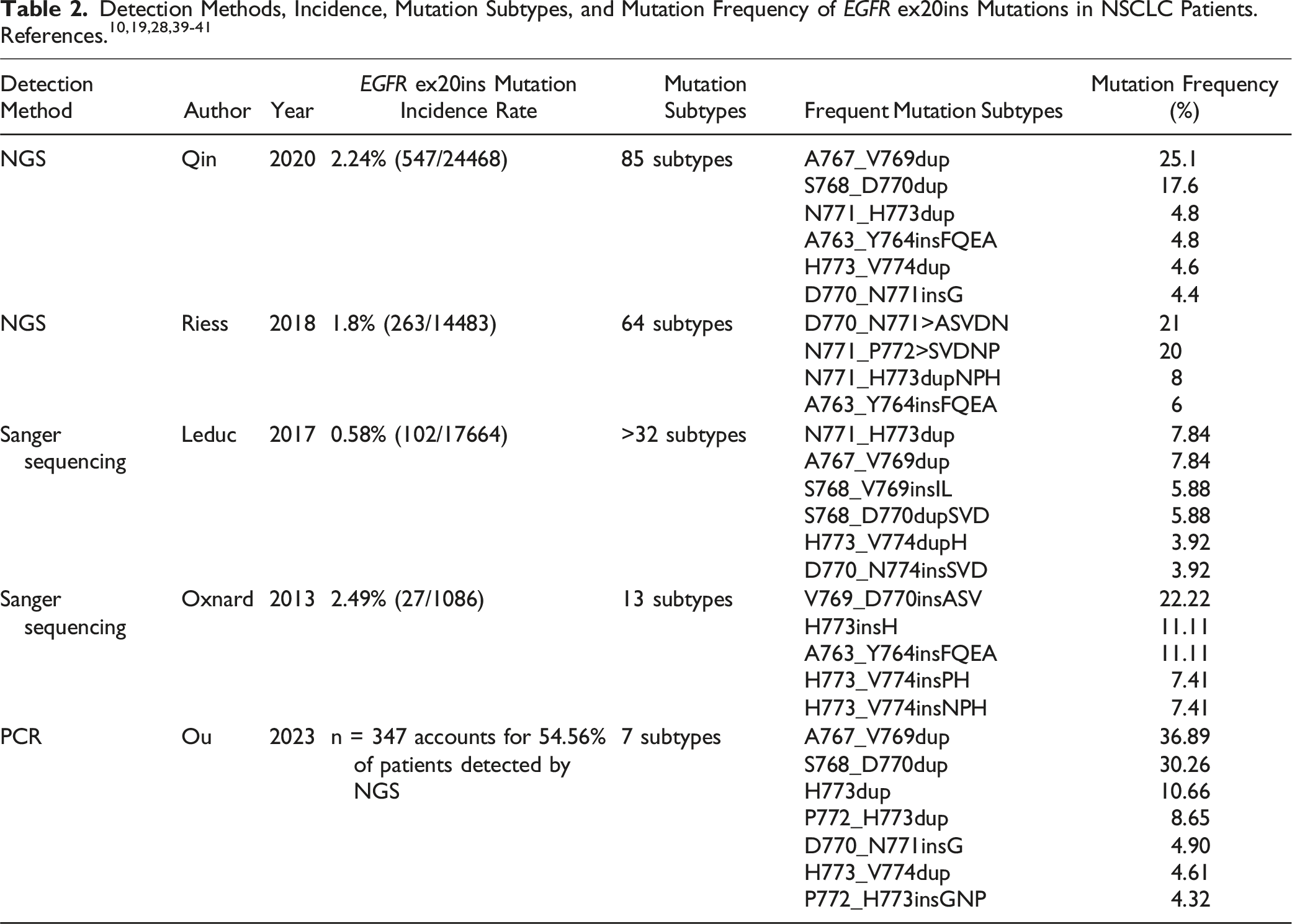
In contrast to classical EGFR mutations, EGFR ex20ins mutations exhibit remarkable subtype diversity and high heterogeneity. A comprehensive analysis of three United States next-generation sequencing (NGS) genomic datasets on a total of 627 patients with EGFR ex20ins mutations revealed up to 102 distinct mutation types. The most prevalent subtypes are V769-A770 (21.1%-25.4%), A770-D771 (28.9%-35.0%), and H773-V774 (22.0%-26.1%). 26 Another study of 636 patients with EGFR ex20ins mutations from five sources revealed 104 distinct mutation subtypes. 28
Moreover, EGFR ex20ins mutations frequently coexist with other genetic alterations. Notably, studies indicate that these mutations are mutually exclusive with other targeted driver mutations in 95% of cases.8,19 The most common cooccurring mutations include those in tumor protein p53 (TP53, 56%), cyclin-dependent kinase inhibitor 2A (CDKN2A, 22%), cyclin-dependent kinase inhibitor 2B (CDKN2B, 16%), and NK2 homeobox 1 (NKX2-1, 14%). 19 Future clinical approaches can leverage these molecular characteristics to personalize treatments more effectively for patients with EGFR ex20ins mutations.
Detection Methods for EGFR ex20ins Mutations
In practical applications, NGS testing often faces significant challenges such as high costs, long turnaround times, insufficient suitable testing samples, and limited laboratory resources. With advancements in genome sequencing technology, automation processes, and computational techniques, the price of genome sequencing is decreasing. Cost analysis studies on NGS have been conducted by researchers from multiple countries and indicate that the cost of NGS testing NSCLC patient ranges from US$440 to US$4932.33-38 The variation in costs is related not only to the sequencing methods and the number of genes coverage for diagnostic purposes (ranging from 10-400 genes) but also to differences in medical costs and the items included in cost estimations in different regions. Owing to the complexity of factors influencing cost calculations, there is currently a lack of large-scale international studies comparing cost or cost-effectiveness evaluations between different countries. Furthermore, for underdeveloped or impoverished countries, NGS technology remains relatively new in clinical practice, limited by the availability of NGS laboratory resources and the lack of additional testing technologies such as liquid biopsies, further decreasing the demand for NGS in these regions.
Therapeutic Advances in EGFR ex20ins Mutations
Recently, various new drugs have undergone baptism by wind and rain. In exploratory studies related to second-line and later-line treatments for patients with EGFR ex20ins mutations, targeted therapies represented by amivantamab, sunvozertinib, and CLN-081 have demonstrated promising antitumor efficacy.42,43
First-Line Treatment Strategies
Amivantamab (JNJ-372) + Chemotherapy
Amivantamab (JNJ-372) is a novel dual-specific monoclonal antibody that targets EGFR and c-MET 44 and can induce EGFR and c-MET receptor downregulation and tumor cell death.45,46 Capitalizing on the significant efficacy demonstrated in second-line and subsequent treatment regimens, the PAPILLON study (NCT04538664), a randomized, open-label phase III clinical trial, evaluated amivantamab in combination with chemotherapy as a first-line therapeutic option for patients harboring EGFR ex20ins mutations. 16 Compared with the chemotherapy alone group (n = 155), the amivantamab + chemotherapy group (n = 153) demonstrated significantly better therapeutic outcomes. According to blinded independent central review (BICR), the two groups had objective response rates (ORRs) of 73% (95% confidence interval [CI], 65∼80) and 47% (95% CI, 39∼56), respectively, with median PFS (mPFS) times of 11.4 months (95% CI, 9.8∼13.7) and 6.7 months (95% CI, 5.6∼7.3), median duration of response (mDOR) times of 9.7 months (95% CI, 8.2∼13.5) and 4.4 months (95% CI, 4.1∼5.6), and mOS times of not reached and 24.4 months (95% CI, 22.1∼not estimable), respectively. This early separation in the PFS curve underscores the benefits of initiating amivantamab + chemotherapy as a first-line treatment for timely disease control.
The most common Grade 3 or higher adverse events (AEs) associated with amivantamab + chemotherapy were neutropenia (33%), leukopenia (11%), and rash (11%). Approximately 7% of patients discontinued treatment due to AEs. Compared with the chemotherapy-only group, the amivantamab + chemotherapy group presented a slightly greater incidence of Grade ≥3 AEs.
While the majority of AEs can be effectively managed through preventative measures and symptomatic treatment, particular attention should be given to infusion-related reactions, which were observed more frequently in the combination group (1% vs 0%). 16 Ongoing studies, such as the phase 2 SKIPPirr trial and PALOMA studies are exploring strategies such as prophylactic glucocorticoid use and subcutaneous formulations to mitigate infusion-related reactions.
On March 1st, the FDA approved the combination of amivantamab with carboplatin and pemetrexed for first-line treatment of NSCLC with EGFR ex20ins mutations. Future research may explore whether amivantamab monotherapy as a first-line treatment has comparable or superior efficacy and investigate different formulations to further reduce the incidence of AEs.
Sunvozertinib (DZD9008)
Sunvozertinib (DZD9008), a small molecule inhibitor, selectively and irreversibly inhibits EGFR with ex20ins mutations. The most recent findings pertaining to its use as a first-line therapeutic in patients with EGFR ex20ins mutations (n = 28), derived from the WU KONG 1 and WU KONG 15 studies, were unveiled at the 2023 ESMO conference. 47 The ORR for all dose groups reached 78.6%, and all patients (100%) achieved shrinkage of target lesions. The mPFS was 10.2 and 12.4 months for the 200 mg and 300 mg groups, respectively. The mDOR was 9.2 months for the 200 mg group and not reached for the 300 mg group. The most common ≥ Grade 3 drug-related treatment-emergent adverse events (TEAEs) included increased blood creatine phosphokinase (17.5%), diarrhea (7%), increased lipase (5.3%), and anemia (5.3%). Only 2 (3.5%) and 4 (7%) patients reduced or terminated treatment, respectively, due to drug-related TEAEs. While these preliminary outcomes underscore the promising potential of sunvozertinib as a frontline therapeutic for patients with EGFR ex20ins mutations, it is crucial to acknowledge the study’s limited sample size and need for further validation. Current phase III clinical trials (WUKONG 28) are evaluating the efficacy of sunvozertinib as a first-line treatment strategy compared with platinum-based chemotherapy. If the phase III investigation confirms the anticipated therapeutic benefits, sunvozertinib, a small-molecule targeted drug that can be administered orally with acceptable safety and without the need for combination chemotherapy, may emerge as the preferred first-line treatment option for patients with EGFR ex20ins mutations.
Second-Line and Above Treatment Drugs
Amivantamab
In addition to its efficacy as a first-line treatment, amivantamab also has notable advantages in second- and later-line treatments for patients with EGFR ex20ins mutations in NSCLC. The CHRYSALIS study (NCT 02609776) 42 included a total of 81 patients with EGFR ex20ins mutations among NSCLC patients who experienced disease progression after platinum-based chemotherapy, all of whom were treated with amivantamab. The BICR-assessed ORR was 40% (32/81), the DOR was 11.1 months, the mPFS was 8.3 months, and the mOS was 22.8 months. Common AEs associated with EGFR inhibition included rash (86%) and infusion-related reactions (66%), whereas those associated with MET inhibition included hypoalbuminemia (27%) and peripheral edema (18%). Most AEs were Grade 1-2, with the most common Grade ≥3 AEs being hypokalemia (5%), rash (4%), pulmonary embolism (4%), and diarrhea (4%). Approximately 13% of patients required dose reductions or discontinued treatment due to adverse events. In May 2021, amivantamab was granted accelerated approval by the FDA on the basis of the promising outcomes of the CHRYSALIS study. 48 In conclusion, amivantamab has demonstrated good efficacy in the context of EGFR ex20ins mutation-positive NSCLC, positioning it as a viable second-line treatment option following the failure of initial therapeutic interventions.
Sunvozertinib
Drawing upon preclinical evidence, Wang et al conducted two phase I clinical studies (WU KONG 1 and WU KONG 2), and demonstrated that, among 56 evaluable patients with EGFR ex20ins mutations, the best ORR at all dose levels was 41.1%. At the recommended phase 2 doses of 200 mg and 300 mg, the confirmed ORRs were 45.5% and 41.9%, respectively, with disease control rates (DCRs) of 81.8% and 90.3%, respectively. 49 As a result of the subsequent WU-KONG 6 study, 50 97 patients with NSCLC harboring EGFR ex20ins mutations were treated with once-daily sunvozertinib at 300 mg, achieving a confirmed ORR of 61% and a DCR of 88%, as assessed by the IRC. In addition, the key results of the WU-KONG 1B study were presented at the 2024 American Society of Clinical Oncology (ASCO) conference. A total of 184 patients with EGFR ex20ins mutations were randomly assigned for treatment with 200 mg or 300 mg of sunvozertinib. The best ORR was 54.3%, with 2.9% of patients achieving a confirmed complete response (CR), and the DCR was 90.8%. The primary endpoint met its predefined target, with statistical significance. 51 Several clinical studies indicate that the most common AEs of sunvozertinib are diarrhea, rash, and elevated creatine kinase (CK), most of which are Grade 1-2 and can be effectively controlled through corresponding supportive treatment. On January 31, 2022, the FDA granted sunvozertinib breakthrough therapy designation (BTD) for the treatment of locally advanced or metastatic NSCLC patients with EGFR ex20ins mutations who were previously treated with at least one prior systemic chemotherapy. Additionally, China has approved second-line treatment targeting EGFR ex20ins mutations for advanced NSCLC. A meta-analysis of pooled data from the WU-KONG series of studies was performed to assess biomarkers of EGFR ex20ins mutations in a large sample size, and the results were reported at the 2024 ASCO conference. In patients with detectable EGFR ex20ins mutations in baseline plasma ctDNA, the vast majority of patients’ mutant allele gradually decreased over time (12/14, 85.7%) during treatment with sunvozertinib. Compared with patients who were positive for baseline plasma EGFR ex20ins mutations, those who tested negative for EGFR ex20ins mutations in baseline plasma ctDNA after sunvozertinib treatment had a higher ORR (68.0% vs 45.8%) and longer mPFS (7.4 months vs 5.5 months), indicating a potential association between the presence of driver mutations in plasma ctDNA and the benefits of sunvozertinib treatment. 52 In light of the current evidence from studies investigating second- and later-line treatments for EGFR ex20ins mutations, sunvozertinib has emerged as the most effective targeted therapy in this class, highlighting its potential for broad clinical application.
CLN-081 (TAS6417/zipalertinib)
CLN-081 (TAS6417) is an innovative small-molecule EGFR inhibitor characterized by a core structure of 6-methyl-8,9-dihydroimidazo [5,4-b] indolizine. This compound covalently modifies the C797S residue within the EGFR ATP binding site, selectively targeting EGFR ex20ins mutations while sparing wild-type EGFR, thereby reducing adverse drug reactions. 53 Preclinical investigations have robustly demonstrated the efficacy of CLN-081 in inhibiting various EGFR ex20ins mutation types in cell models and human NSCLC cell lines. 53 The phase I/IIa clinical trial of CLN-081 (NCT04036682) 54 enrolled a total of 73 patients with EGFR ex20ins mutations (≥second-line treatment) and were treated with CLN-081 at different doses (30/45/65/100/150 mg) twice daily. The confirmed ORR for all dose groups was 38.4%, with an mDOR of 10 months and an mPFS of 10 months. The ORR and mPFS for the 100 mg BID dose group (n = 39) were 41% and 12 months, respectively. The mPFS for the ≤65 mg BID group was 8 months. Notably, CLN-081 exhibited a rapid onset of action, with a median time to response of 1.5 months, and 54 patients (74%) experienced tumor shrinkage in the initial 6-week disease assessment. The most frequently observed AEs associated with CLN-081 included rash (80%), paronychia (32%), and diarrhea (30%). The majority of the AEs were Grade 1/2 in severity, with no cases of Grade ≥3 drug-related rash or diarrhea reported at daily doses of 100 mg or below. CLN-081 was granted BTD by the FDA in January 2022.
Furmonertinib
Furmonertinib is a novel third-generation irreversible, selective EGFR-TKI designed for the treatment of NSCLC patients with EGFR-sensitizing mutations. Preclinical studies have demonstrated the effective inhibition of EGFR ex20ins mutations in BaF3 cells, with an average IC50 ranging from 11 to 20 nM. 55 Updated data presented at the World Conference on Lung Cancer (WCLC) Congress 2023 from the FAVOUR study highlighted the robust antitumor activity of furmonertinib in both treatment-naive and previously treated NSCLC patients with EGFR ex20ins mutations. 56 As assessed by the IRC, the confirmed ORRs for the treatment-naive 240 mg cohort (n = 30), previously treated 240 mg cohort (n = 28), and previously treated 160 mg cohort (n = 28) were 78.6%, 46.2%, and 38.5%, respectively, and the DCRs were 100%, 92.3%, and 84.6%, respectively. The mDOR was 15.2 months, 13.1 months, and 9.7 months, respectively. The incidence rates of Grade ≥3 AEs in the treatment-naive 240 mg cohort, previously treated 240 mg cohort, and previously treated 160 mg cohort were 13%, 29%, and 18%, respectively. The three most common AEs were diarrhea, anemia, and oral ulcers, with the overall safety profile being well tolerated. Current phase II and III clinical trials (FURMO-003, FURVENT/FURMO-004) are ongoing to investigate the efficacy of furmonertinib in NSCLC patients with EGFR ex20ins mutations. In October 2023, furmonertinib was granted BTD by the FDA for treating previously untreated NSCLC patients with EGFR ex20ins mutations.
Osimertinib
As a third-generation EGFR-TKI, osimertinib is a preferred first-line treatment option for NSCLC patients with EGFR-sensitizing mutations. Preclinical studies have demonstrated the inhibitory effects of osimertinib on both EGFR ex20ins-mutant and wild-type cells. 57 Nevertheless, clinical trials have revealed that administering osimertinib at the standard dose of 80 mg has limited efficacy in patients harboring EGFR ex20ins mutations, resulting in an ORR ranging from 0% to 6.5%.58-60 Consequently, higher doses of osimertinib have been assessed in subsequent investigations. The ECOG-ACRIN 5162 study 61 evaluated the therapeutic response of patients with EGFR ex20ins mutations (≥second-line treatment) with oral osimertinib (160 mg), and the results revealed an ORR of 25% (5/20), a DCR of 85% (17/20), and an mPFS of 9.7 months. Similarly, findings from the POSITION20 study reported an ORR of 28% (7/25) for osimertinib 160 mg, with an mPFS of 6.8 months and an mOS of 15.2 months. 62 The current standard osimertinib dose for patients with EGFR ex20ins mutations, and even double this dose, does not have high efficacy.
New Drugs under Development
For drug developers, EGFR ex20ins mutations represent a significant unmet need. Several drugs are currently in development. Should subsequent investigations validate their excellent therapeutic efficacy, these drugs are poised to emerge as groundbreaking treatments in the imminent future (Table 3).
YK-029A
YK-029A is an oral, irreversible third-generation EGFR-TKI that irreversibly inhibits EGFR signaling by forming a covalent bond with amino acid residues on the EGFR protein. Preclinical studies have demonstrated the potent antitumor activity of YK-029A against EGFR ex20ins mutations. 63 Notably, a multicenter, open-label phase I clinical trial (n = 26) presented at the 2023 ASCO conference reported promising results 64 ; the results assessed by the IRC showed that the ORR was 73.1%, the mPFS was 9.3 months, the DCR was 92.3%, and the mDOR was 7.5 months. Grade ≥3 AEs occurred in 27.8% of patients, leading to treatment discontinuation in three patients. Currently, a phase III clinical trial is underway to compare the efficacy of YK-029A as a first-line treatment with that of platinum-containing chemotherapy regimens.
Luminespib (AUY922)
Luminespib is an inhibitor of heat shock protein 90 (Hsp90, a molecular chaperone that regulates the posttranslational folding and stability of client proteins), and Hsp90 inhibition leads to the degradation of client proteins via the ubiquitin-proteasome pathway. 65 Preclinical studies have shown that EGFR ex20ins mutations depend on Hsp90 and can be inhibited by the Hsp90 inhibitor luminespib. 66 A phase II study (NCT01854034) included a total of 29 patients with EGFR ex20ins mutations in advanced NSCLC. The ORR was 17%, with an mPFS of 2.8 months and an mOS of 13 months, resulting in a total survival period of 9.9 months. The Grade ≥3 AEs included ocular toxicity (13.3%), hypophosphatemia (27.7%), and hypertension (3.10%), all of which were reversible. 67
PLB1004
PLB1004 is a novel mono-anilino-pyrimidine small-molecule inhibitor of EGFR that effectively and irreversibly targets ex20ins mutations as well as EGFR-sensitizing mutations, with the ability to penetrate the blood‒brain barrier.
The phase I clinical trial results for PLB1004 in second- and above-line treatment of advanced NSCLC patients carrying EGFR ex20ins mutations were presented at the 2023 American Association for Cancer Research (AACR) annual meeting. The results indicated that the ORR, as evaluated by the investigator (INV), was 57.7% (15/26) across all dose groups, with a DCR of 100% (26/26). Notably, 40% of the responders maintained a response duration exceeding 6 months. The most common Grade ≥3 AEs were diarrhea (18%), rash (11%), and aspartate aminotransferase elevation (3%). 68 Currently, a phase II clinical study of PLB1004 in patients with EGFR ex20ins mutations in locally advanced/metastatic NSCLC is ongoing.
MCLA-129
MCLA-129 is an antibody-dependent cell-mediated cytotoxicity (ADCC)-enhanced IgG1 bispecific antibody that targets EGFR and c-MET. Preclinical studies have indicated that MCLA-129 results in significant growth inhibition of patient-derived EGFR ex20ins mutant NSCLC tumors in a mouse PDX model. 69 The 2024 ASCO conference announced the ongoing first-in-human phase 1/2 study of MCLA-129. A total of 72 patients with EGFR ex20ins mutations were enrolled in cohort B and treated with intravenous injections of 1500 mg or 2000 mg MCLA-129 every 2 weeks. The results showed that the confirmed ORR for the 1500 mg dose group (n = 63) was 55.6%, the DCR was 84.1%, and the mDOR was 7.2 months. The 2000 mg dose group (n = 9) had a confirmed ORR of 28.6% and a DCR of 88.9%. The most common TEAEs were infusion-related reactions (71.9%), hypoalbuminemia (54.8%), and decreased neutrophil count (46.1%). The incidence rates of ≥3 Grade TEAEs and drug-related TEAEs were 51.6% and 39.2%, respectively. 70
ORIC-114
ORIC-114 is a brain-penetrant, orally irreversible small molecule inhibitor designed to target exon 20 insertions in EGFR and HER2. Preclinical studies of ORIC-114 have demonstrated significant tumor regression in various patient-derived xenograft models with EGFR ex20ins mutations and EGFR-driven intracranial lung cancer models. Additionally, it has demonstrated greater brain exposure than other targeted drugs for EGFR ex20ins mutations. 71 Based on this, a phase I clinical trial (NCT05315700) 72 is currently underway to evaluate the use of ORIC-114 in patients with advanced solid tumors harboring EGFR or HER2 mutations.
Blueprint-451
BLU-451 is a selective, central nervous system (CNS)-penetrant investigational EGFR ex20ins mutation inhibitor. It has shown promising CNS penetration and antitumor activity in intracranial xenograft models, with a brain-plasma ratio of 0.62. 73 A phase I/II clinical trial (NCT05241873) 74 for the treatment of advanced tumors with EGFR ex20ins mutations is ongoing, and relevant results have not yet been published.
Drugs that Have Failed in Research
Mobocertinib
Mobocertinib (TAK-788) is a novel oral selective, irreversible EGFR ex20ins mutation TKI drug. It received accelerated FDA approval in September 2021 on the basis of phase I/II study results (NCT02716116), 75 positioning it as a targeted therapy for EGFR ex20ins mutations after first-line chemotherapy. 76 The PPP cohort had an ORR of 28%, as assessed by the IRC, an mPFS of 7.3 months, an mDOR of 17.5 months, and an mOS of 24 months. Similarly, in the EXCLAIM expansion cohort of the same study, the ORR as assessed by IRC was 25%, and mPFS was 7.3 months. However, the subsequent EXCLAIM-2 (NCT04129502) study compared the efficacy and safety of mobocertinib with those of platinum-based chemotherapy in first-line treatment for locally advanced or metastatic EGFR ex20ins-positive NSCLC, and revealed no significant therapeutic advantage for mobocertinib over chemotherapy (ORR: 32% vs 30%), ultimately prompting Takeda Pharmaceutical Company to voluntarily withdraw its marketing authorization.
Poziotinib
Poziotinib is an irreversible oral EGFR inhibitor designed for the treatment of advanced NSCLC characterized by EGFR exon 20 point mutations or insertions. Elamin et al conducted a phase II study (NCT03066206) in which 50 patients with EGFR exon 20 point mutations or insertions in advanced NSCLC were treated with poziotinib. The ORRs confirmed by the INV and IRC were 32% and 31%, respectively, with an mPFS of 5.5 months. 24 The subsequent multicohort, multicenter phase II trial ZENITH20-1 77 also confirmed the antitumor activity of poziotinib in NSCLC patients with EGFR ex20ins mutations, but the ORR was only 14.8%, and the mPFS was 4.2 months. Despite demonstrating acceptable antitumor effects, the FDA rejected poziotinib’s marketing application in September 2022 because of its inferior efficacy and safety profile compared with those of FDA-approved drugs.
Cetuximab in Combination with EGFR-TKIs
Recent studies have explored the potential of combining EGFR-TKIs with cetuximab as a therapeutic approach for patients harboring EGFR ex20ins mutations. Hasegawa et al 79 revealed that the combination of cetuximab with afatinib or osimertinib can effectively inhibit the proliferation of EGFR ex20ins mutant Ba/F3 cells transduced with A764 V765insHH, A767_V769dupASV, and D770_N771insNPG compared with the effects of cetuximab, afatinib, and osimertinib monotherapy (P < 0.05). A retrospective clinical report featuring four NSCLC patients with EGFR ex20ins mutations treated with cetuximab combined with afatinib reported an ORR of 75% (3/4) and an mPFS of 5.4 months. 80
Building upon these preliminary successes, in a single-arm phase 2 trial (NCT03727724), 37 patients were treated with afatinib in combination with cetuximab, and the ORR was 43%, with a confirmed ORR of 32%. The DCR was 54%, the mPFS was 5.5 months (95% CI, 3.7-8.3 months), and the mOS was 16.8 months (95% CI, 10.7-25.8 months). The most common AEs were diarrhea (70%), rash (65%), dry skin (59%), and paronychia (54%). Among all patients, 54% of them experienced grade 3 AEs. 81 Although the combination of cetuximab and EGFR-TKIs holds promise as a potential new treatment strategy for patients with NSCLC harboring EGFR ex20ins mutations, the toxicity associated with this combination therapy remains a significant barrier to its utilization.
Immunotherapy
Recently, immune checkpoint inhibitors (ICIs) have made significant breakthroughs in the field of driver-gene-negative NSCLC patients, profoundly altering the treatment landscape for these patients. Several clinical trials, such as the CheckMate-722 and KEYNOTE-789 studies, have shown that, after patients with classical EGFR mutations acquire resistance to EGFR-TKIs, the use of immunotherapy combined with chemotherapy does not result in survival benefits compared with chemotherapy alone (PFS: 5.6 vs 5.4-5.5 months; OS: 15.9-19.4 vs 14.7-15.9 months).82,83 The addition of antiangiogenic therapy to immunotherapy and chemotherapy or the use of the bispecific antibodies from anti-vascular endothelial growth factor (VEGR) and anti-PD-(L)1 antibodies in combination with chemotherapy has shown PFS benefits over chemotherapy alone (7.2-8.48 vs4.3-5.62 months); however, conclusions regarding OS benefits are still pending.84,85 Currently, most experts believe that individuals with classical EGFR mutations benefit less from immunotherapy. Unlike classical EGFR mutations and driver-gene-negative NSCLC patients, the effects of immunotherapy in patients with EGFR ex20ins mutation remain unclear. One report indicates that the tumor mutational burden (TMB) and the proportion of patients with PD-L1 expression in ≥1% of tumor cells is lower among patients with EGFR ex20ins mutations than among those with EGFR exon 19 deletion/exon 21 L858 R mutation (median TMB 3.4 vs 3.5, P = 0.001; PD-L1 expression ≥1%: 39% vs 22%, P = 0.02) and those without EGFR ex20ins mutations (median TMB 3.4 vs 5.9, P < 0.001; PD-L1 expression ≥1%:60% vs 22%, P < 0.001). 86 This implies that the efficacy of immunotherapy may be suboptimal in this patient population. Treatment efficacy for NSCLC patients with EGFR ex20ins mutations in the real world was reported at the 2021 ASCO conference. Eleven patients were treated with first-line monoimmunotherapy, and sixteen patients were treated with first-line immunotherapy combined with chemotherapy (ORR, 9.1% vs 18.8%; mPFS 3.1 vs 4.5 months; and mOS, 11.0 vs 11.3 months). Among 32 patients treated with immunotherapy as the second-line treatment, the ORR was only 3.1%, with an mPFS of 3.3 months and an mOS of 8.1 months. 87 Another real-world study yielded similar results; 44 patients treated with chemotherapy and 15 patients treated with immunotherapy combined with chemotherapy were ultimately included in the first-line treatment analysis. Compared with that of chemotherapy alone, the ORR was improved in the first-line chemotherapy combined with the immunotherapy group (40.0% vs 18.2%), but the mPFS was not increased (6.53 vs 5.93 months, hazard ratio (HR) = 0.73, 95% CI: 0.38-1.39, P = 0.48). 88 Additionally, analysis by Metro et al of 15 patients with EGFR ex20ins mutations treated with monoimmunotherapy or combination therapy revealed that immunotherapy was associated with a poor clinical outcome. The response rate was observed in 1 out of 15 patients (6.7%), the mPFS was 2.0 months, and the OS was 5.3 months. 89 It appears that NSCLC patients with EGFR ex20ins mutations do not benefit significantly from immunotherapy; even with first-line immunotherapy combined with chemotherapy regimens, the treatment efficacy in these patients (mPFS: 4.5-6.53 months, ORR: 18.8%-40.0%)87,88,90 is significantly lower than that in driver-gene-negative NSCLC patients (mPFS: 9.0 months, ORR: 48.3%). 91 Although the sample size of the above studies is limited, the control groups in current first-line clinical trials for EGFR ex20ins mutations often employ only chemotherapy regimens rather than combined with immunotherapy. Overall, the prevailing opinion is that patients with EGFR ex20ins mutations have limited therapeutic effects from immunotherapy.
Correlation Between the Insertion Site of Exon 20 and Therapeutic Efficacy
Efficacy in NSCLC Patients with Brain Metastasis
Currently, new drugs under development, such as ORIC-114, BLU-451, and PLB1004, exhibit strong central nervous system penetration alongside the targeting of EGFR ex20ins mutations. These agents offer additional treatment options and hope for lung cancer patients with brain metastases. Given the high prevalence and impact of lung cancer brain metastases, further clinical research is essential to improving the survival outcomes and quality of life of affected patients.
Exploration of the Mechanisms of Acquired Drug Resistance
The current state of research delving into the intricate mechanisms of drug resistance in EGFR ex20ins mutations remains predominantly exploratory in nature. Pacini et al 97 postulated that the emergence of resistance to targeted therapies aimed at EGFR ex20ins mutations can be attributed to a trifecta of factors: secondary mutations, the activation of bypass signaling pathways, and the epithelial‒mesenchymal transition (EMT). Subsequent investigations have further elucidated specific resistance mechanisms associated with individual therapeutic agents. The secondary mutations responsible for acquired resistance to mobocertinib and poziotinib depend on the type of EGFR ex20ins mutation, which is associated mainly with T790 M or C797S. T790M-related resistance can be partially attenuated by furmonertinib, sunvozertinib, and zipalertinib, whereas C797S mutations confer resistance to all these EGFR-TKIs.98,99 Furthermore, Elamin et al reported that24,100 the mechanism of acquired resistance to poziotinib may involve EGFR T790 M, MET amplification, and EMT. Nishino et al 101 's recent work confirmed that the EGFR C797S and T790 M mutations are likely culprits in tarloxotinib-E resistance, and these mechanisms are contingent on the original EGFR ex20ins mutation subtypes. 101 A study provided preliminary evidence that C797S mutation acquisition is a potential mechanism of acquired resistance to CLN-081. 102 Additionally, the mechanism of resistance to sunvozertinib may be related to acquired EGFR C797S mutation, EGFR G724S mutation, and abnormalities in other gene mutations in the downstream signaling pathway of EGFR 52 However, the acquired resistance mechanisms of other drugs (such as amivantamab, etc.) have not been extensively studied. Future clinical trials can expand on these findings to comprehensively elucidate specific resistance pathways, thereby guiding the development of innovative therapeutic strategies designed to overcome acquired resistance in patients with the EGFR ex20ins mutations.
EGFR ex20ins Combined with Classical EGFR Mutations
The incidence of EGFR ex20ins combined with classical EGFR mutations appears to be low, with only a few studies reporting related cases. Some reports indicate that the incidence of EGFR ex20ins combined with classical EGFR mutations in patients with EGFR ex20ins mutations is approximately 1.3%-2.6%.19,40 On the basis of the Geno1.2-CLICaP platform, Cardona et al's study indicated that the incidence of EGFR ex20ins combined with classical EGFR mutations was not low (36.4%) among 88 patients with EGFR ex20ins mutations from six Latin American countries. Further efficacy analysis revealed a positive impact on OS (17.3 months vs 14.6 months, P = 0.017) when EGFR ex20ins mutation combined with a classical EGFR mutation compared to patients with only EGFR ex20ins mutation. 103 In general, EGFR ex20ins mutations presented primary resistance to EGFR-TKIs and poor survival outcomes. 104 The EGFR ex20ins combined with classical EGFR mutation seems to improve the response of such co-mutation patients to EGFR-TKIs in an unknown manner, these results may offer novel therapeutic approaches for patients harboring such co-mutation, while this requires further validation.
Summary
EGFR ex20ins mutations have been recognized as an independent molecular subtype for treatment selection in domestic and international guidelines, but there are some clinical challenges related to this subtype, such as high heterogeneity, overlooked rare mutation types, and lack of highly effective and low toxicity targeted EGFR ex20ins inhibitors. Consequently, addressing the unmet therapeutic needs of NSCLC patients with EGFR ex20ins mutations represents a paramount challenge that necessitates urgent attention. Currently, an increasing number of clinical trials are focused on such mutations. With the notable efficacy of representative targeted drugs such as amivantamab and sunvozertinib as first-line treatment, the existing therapeutic dilemma for patients with EGFR ex20ins mutations has been broken, significantly improving the survival benefits for this patient population. Future endeavors should prioritize exploring first-line treatment strategies, elucidating the most suitable treatment paradigm for patients with baseline brain metastases, improving the accessibility and utilization of NGS testing, and fostering the development of novel therapeutic agents. This article presents a comprehensive review of the latest developments in research on EGFR ex20ins mutations, aiming to serve as a valuable reference for guiding future clinical trials and assisting in personalized treatment selection for this patient population.
Footnotes
Acknowledgments
This review was translated in its entirety by several coauthors, including Haoli Liu (Postgraduate Training Base Alliance of Wenzhou Medical University (Zhejiang Cancer Hospital), Zhejiang, China), Jing Qin (Department of Thoracic Medical Oncology, Zhejiang Cancer Hospital, Zhejiang, China), Xinyu Qian(Department of Oncology, Hangzhou Cancer Hospital, Zhejiang, China).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81903981), and the Zhejiang Provincial Natural Science Foundation of China (No.LY21H290002).
Ethics Statement
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.