Abstract
Introduction
With the advancements in cancer prevention and diagnosis, the proportion of newly diagnosed early-stage cervical cancers has increased. Adjuvant therapies based on high-risk postoperative histopathological factors significantly increase the morbidity of treatment complications and seriously affect patients’ quality of life.
Objectives
Our study aimed to establish a diagnostic nomogram for vaginal invasion (VI) among early-stage cervical cancer (CC) that can be used to reduce the occurrence of positive or close vaginal surgical margins.
Methods
We assembled the medical data of early-stage CC patients between January 2013 and December 2021 from the Fujian Cancer Hospital. Data on demographics, laboratory tests, MRI features, physical examination (PE), and pathological outcomes were collected. Univariate and multivariate logistic regression analyses were employed to estimate the diagnostic variables for VI in the training set. Finally, the statistically significant factors were used to construct an integrated nomogram.
Results
In this retrospective study, 540 CC patients were randomly divided into training and validation cohorts according to a 7:3 ratio. Multivariate logistic analyses showed that age [odds ratio (OR) = 2.41, 95% confidence interval (CI), 1.29-4.50, P = 0.006], prognostic nutritional index (OR = 0.18, 95% CI, 0.04-0.77, P = 0.021), histological type (OR = 0.28, 95% CI, 0.08-0.94, P = 0.039), and VI based on PE (OR = 3.12, 95% CI, 1.52-6.45, P = 0.002) were independent diagnostic factors of VI. The diagnostic nomogram had a robust ability to predict VI in the training [area under the receiver operating characteristic curve (AUC) = 0.76, 95% CI: 0.70-0.82] and validation (AUC = 0.70, 95% CI: 0.58-0.83) cohorts, and the calibration curves, decision curve analysis, and confusion matrix showed good prediction power.
Conclusion
Our diagnostic nomograms could help gynaecologists quantify individual preoperative VI risk, thereby optimizing treatment options, and minimizing the incidence of multimodality treatment-related complications and the economic burden.
Introduction
Although cervical cancer (CC) rates have been declining owing to vaccination and screening efforts, 1 it is still the fourth most common cancer in women worldwide, with approximately 604,000 new cases and 342,000 deaths in 2020. 2 The burden faced by low- and middle-income countries is significantly greater than that faced by high-income countries. 3 With the concepts and knowledge of its prevention, diagnosis, and treatment evolving rapidly, more cases of CC are being diagnosed at an early stage. 4 A nested case-control research found that regular CC screening was associated with a 95% reduction in stage III or worse CC. 5 The Korean National Screening Program has also demonstrated that carcinoma in situ (CIS) was diagnosed in 71.77% of patients with CC who had been screened while 54.78% of those who had never been screened. 6
The standard treatment for early-stage CC (FIGO stages IB-IIA) is radical hysterectomy (RH) with bilateral pelvic lymphadenectomy or radical radiation therapy (RT). 7 Previous studies have confirmed that CC patients with FIGO 2018 stage IB-IIA1 could gain equal survival outcomes either RH or RT. 8 However, two-thirds of CC patients who receive RH treatment still need chemotherapy or combined radiotherapy due to high-risk histopathological factors, such as positive or close surgical margins, parametrial involvement, or lymph node metastasis. 9 The combination of RH and adjuvant RT significantly increases the morbidity of treatment complications and side effects, thereby increasing physical and mental suffering, and seriously affecting the patient’s quality of life. 10 Hence, the importance of meticulous selection of RH or RT should be highlighted for each patient based on preoperative assessment.
Vaginal invasion (VI) can be easily diagnosed by physical examination (PE) due to its unique visible anatomy. According to the International Federation of Gynecology and Obstetrics, PE is considered the main standard of preoperative VI in CC patients. 11 Patients with positive or close vaginal margin after surgery require additional chemoradiotherapy. However, there remains a significant discrepancy between the gynaecological and pathological results. A retrospective study involving 23,933 CC patients demonstrated that VI was the leading cause of stage inaccuracy, with a 62.3% mistaken rate. 12 Moreover, another retrospective study revealed that 8% of CC patients with stage IB turned to positive margins or close vaginal margins with safety margins of less than 5 mm after radical surgery. 13 Another research also found that 16.7% of patients with stage IB-IIA had VI based on pathology. 14 Notably, the recurrence rate was 24% in patients with positive surgical margins compared with 9% in patients with negative surgical margins. 15 It is clear that inaccurate VI assessment is a leading determinant of positive vaginal surgical margins. Hence, the identification of effective biomarkers to aid in the accurate preoperative evaluation of VI status is urgently needed.
Nutrition and inflammation play substantial roles not only in the development of cancer but also in the postoperative outcomes of primary tumor resection. 16 Several pretreatment nutritional and immune indexes, such as the platelet-lymphocyte ratio (PLR), neutrophil-lymphocyte ratio (NLR), prognostic nutritional index (PNI), and controlling nutritional status (CONUT), are closely associated with the pathological outcomes of various malignant tumors, such as pancreatic cancer, gastric cancer, cervical cancer, and breast cancer.17–20 However, their relationship with the VI in CC patients has not yet been elucidated.
We aimed to construct a nomogram based on a clinical and nutritional index with the diagnostic predictive value of VI in early-stage CC using postoperative pathological results as a reference standard. The nomogram showed good predictive accuracy and clinical value. Gynecologists can quickly quantify individual VI risk probabilities and help to optimize treatment plans and surgical procedures to minimize the incidence of multimodality treatment-related complications and economic burden.
Materials and Methods
Patient Selection
Between January 2013 and December 2021, we retrospectively and consecutively reviewed the medical data of CC patients (stages IB-IIA) at the Fujian Cancer Hospital. All patients underwent a standardized staging evaluation before RH with bilateral pelvic lymphadenectomy, including preoperative laboratory tests, abdominal MRI, and thoracic CT. The inclusion criteria were as follows: FIGO stage IB-IIA confirmed by PE and imaging results. An abdominal MRI was performed within 3 weeks before surgery. All included cases and disease stages were confirmed via histopathology. Type III radical hysterectomy with bilateral pelvic lymphadenectomy was performed either via laparotomy or laparoscopy. No prior treatment was administered before surgical resection; regardless of postoperative adjuvant therapy. Our exclusion criteria were neoadjuvant chemotherapy followed by surgery; concurrent chemoradiotherapy; concurrent chemoradiotherapy followed by adjuvant type III radical hysterectomy; patients with primary tumors from other sites; patients with immune disease, liver disease, hematologic disorders, or chronic infectious diseases; and those with incomplete clinical data. Finally, 540 patients were included in our study. All the patient details were de-identified to ensure confidentiality.
Data Collection
Data on demographics, laboratory test (albumin, platelets, neutrophils, lymphocytes, and monocytes), MRI features, PE, and pathological outcomes were gathered. Experiments involving humans were carried out following the ethics policy approved by the Ethics Committee of Fujian Cancer Hospital on 25 October 2023 (approval no. K2023-369-01). An exemption from informed consent was obtained from the ethics committee of Fujian Cancer Hospital, due to the retrospective nature of the study.
Platelet-lymphocyte ratio (PLR) was calculated as the absolute platelet count (109/L) divided by the absolute lymphocyte count (109/L), while neutrophil-lymphocyte ratio (NLR) was calculated as the absolute neutrophil count (109/L) divided by the absolute lymphocyte count (109/L). The prognostic nutritional index (PNI) was defined as albumin level (g/L) + 5 × absolute lymphocyte count (109/L), while systemic inflammation response index (SIRI) was defined as (neutrophil count × monocyte count)/lymphocyte count.
Assessment
The vaginal invasion was defined as cervical tumor disruption of the vaginal wall. 21 VI assessments included preoperative clinical and postoperative pathological assessments. The preoperative evaluation was systematically evaluated by MRI and PE. Two experienced gynecologists performed physical examination and two radiologists evaluated MRI scans. Postoperative pathological evaluation was performed by two senior pathologists. All assessors were blinded to patients' history, clinical diagnosis, and group allocation.
Statistical Analysis
By setting random seeds, the patients were randomly divided into a training set and validation sets in a 7:3 ratio. The receiver operating characteristic (ROC) curve was used to determine the optimal cut-off values for age, PNI, PLR, NLR, and SIRI. The chi-square test or Fisher’s exact test was used to analyze clinicopathological categorical variables. Univariable and multivariable logistic regression analyses were employed to estimate the association between the presence of VI and each of the independent variables in the training set. A forward stepwise method of variable selection was used in multivariate logistic regression analyse. The multivariate model was fitted using the forward selection method. The statistically significant factors (P
Results
Patient Characteristics
The flowchart of this study is detailed in Figure 1. A total of 540 patients were randomly divided into training cohort (70%, n = 378) and validation cohort (30%, n = 162). Table 1 summarizes the clinicopathological features of the training and validation cohorts. The results showed no statistical difference in the clinicopathological features between the two groups. Table 2 shows the accuracy of MRI and PE in preoperatively diagnosing CC with VI. The flow chart of the study. Abbreviations: CC, cervical cancer. Characteristics of Patients With Early-Stage CC, 2013-2021. Abbreviations: SD, standard deviation; n, number; CC, cervical cancer; PNI, prognostic nutritional index; PLR, platelet-lymphocyte ratio; NLR, neutrophil-lymphocyte ratio; SIRI, systemic inflammation response index; SCC, Squamous cell carcinoma; NEC, neuroendocrine carcinoma; AC, adenocarcinoma; ASC, adenosquamous carcinoma; VI, vaginal invasion; PE, physical examination. Accuracy of MRI and PE in Diagnosing CC With VI. Abbreviations: PE, physical examination; CC, cervical cancer; VI, vaginal invasion.


The Univariate and multivariate Analyses
The Univariate and Multivariate Logistic Regression Analysis in the Training Cohort.

Abbreviations: OR, odds ratio; PNI, prognostic nutritional index; PLR, platelet-lymphocyte ratio; NLR, neutrophil-lymphocyte ratio; SIRI, systemic inflammation response index; SCC, squamous cell carcinoma; VI, vaginal invasion; PE, physical examination; others refer to squamous cell carcinoma, neuroendocrine carcinoma, adenocarcinoma and adenosquamous carcinoma.
Development and Validation of a Nomogram
Based on the multivariate analysis results, we constructed a predictive nomogram using the following parameters: age, PNI, histological type, and VI based on PE (Figure 2). As shown in Figure 3A and B, the AUCs were 0.76 (95% CI, 0.70-0.82) and 0.70 (95% CI, 0.58-0.83) in the training and validation cohorts. The nomogram had a moderate ability to predict VI. The calibration curves indicated that the nomogram had a good fit for the diagnosis of VI in training cohort (Brier score: 0.12; calibration slope: 1.00; intercept: 0.00) and validation cohort (Brier score: 0.11; calibration slope: 0.76; intercept: 0.57), res pectively (Figure 3C and D). In both cohorts, DCA demonstrated favourable positive net benefits of the nomogram (Figure 3E and F). Table 4 shows that the accuracy, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of the nomogram were 0.64, 0.59, 0.85, 0.96, and 0.28, respectively, in training cohort, while those of the validation cohort were 0.56, 0.53, 0.77, 0.94, and 0.21 in validation cohort, respectively. Diagnostic nomogram for VI based on pathology prediction in CC patients. Abbreviations: PNI, prognostic nutritional index; VI, vaginal invasion; PE, physical examination; others refer to squamous cell carcinoma, neuroendocrine carcinoma, adenocarcinoma and adenosquamous carcinoma. AUC for diagnostic nomogram in the training cohort (A) and validation cohort (B). Calibration curves in the training cohort (C) and validation cohort (D). DCA in the training cohort (E) and validation cohort (F). Confusion Matrix of Nomogram. Abbreviations: PPV, positive predictive value; NPV, negative predictive value; CI, confidence interval.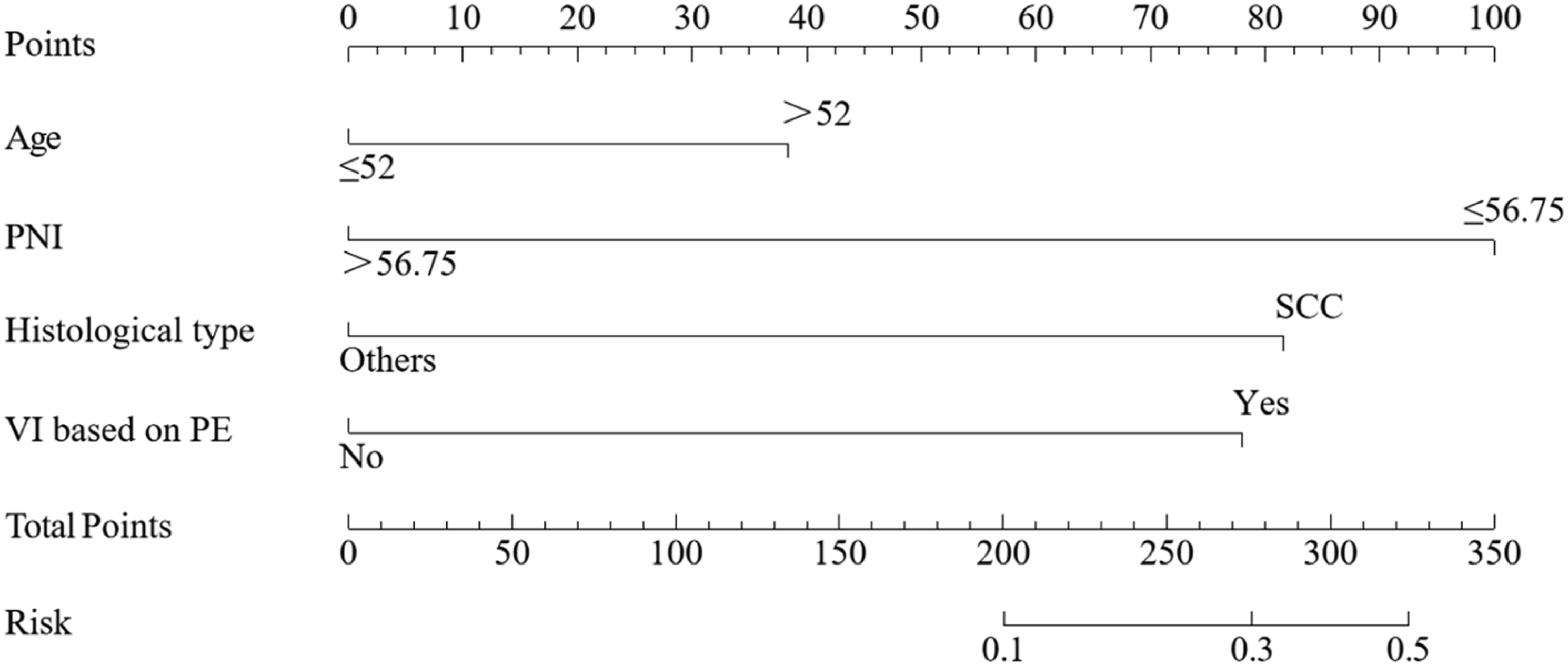


Discussion
The VI is a crucial index for CC tumor staging and prognosis prediction. However, the role of VI has been overlooked in the literature compared to those of lymph node metastasis and parametrial invasion. As far as we know, this is the first study to construct a nomogram based on clinical parameters and nutritional biomarkers for predicting the probability of pathological VI. Our results showed that PNI is a risk factor for VI. Moreover, we found that PE had greater sensitivity than MRI in predicting VI. The diagnostic nomogram based on age, histological type, PNI, and PE had a favourable predictive power for predicting VI. Our nomogram may help gynecologists better and more effectively predict VI in early-stage CC preoperatively to optimize surgical procedures and therapeutic options.
The prognostic nutritional index (PNI) is a nutritional and inflammatory index calculated by albumin and absolute lymphocyte count in the peripheral blood. 23 Previous studies have shown that the PNI is a predictor of the prognosis of several tumors, including melanoma, 24 stomach cancer, 25 and esophageal cancer. 26 A low PNI indicates poor immune and nutritional status, which may be related to the poor prognosis of tumor patients. 25 It’s worth noting that many studies evaluating PNI have focused on disease prognosis, rather than predictors of disease occurrence. Our study is the first to discover PNI’s role in predicting VI in CC patients. Patients with high PNI tended to have VI. In addition, unlike other screening methods, PNI can be easily and quickly detected in laboratory tests during preoperative diagnosis. Hence, it is convenient to use and popularise in clinics.
Age is an independent risk factor for VI in early-stage CC, which is consistent with the result of previous studies.27,28 Diver et al found that older women were more likely to present with higher stage CC (P < 0.001). 29 A SEER database study found that older CC patients have an increased chance of developing regional or distant diseases as they age. 30 Late staging of CC in older patients may be partly attributed to a lack or failure of cervical screening.
Our data demonstrate that an MRI assessment or a single physical examination is insufficient for the diagnosis of VI. The accuracy of MRI in predicting tumor size was 79%–90%, while that of PE was 47%–70%. 31 It seems that MRI has a more substantial predictive power than PE. However, Ditto A. et al 14 showed the overall accuracy of MRI was not superior to that of PE. Moreover, the inter-observer agreement of MRI in the diagnosis of VI was lower than that of parametrial invasion and lymph node metastasis. 32 It is consistent with our study. Our data showed that MRI had poorer statistical power in detecting VI than in detecting PE. This may be related to the lower positive predictive value of MRI for vaginal invasion. VI appears as a segmental disruption of low-signal intensity vagina on T2WI. A large cervical tumor that stretches the vaginal walls and benign reactive changes in the vagina may make distinguishing VI challenging.
Therefore, there is an urgent need to construct an integrated parameters model to increase the predictive accuracy of VI. To date, this is the first study to identify the diagnostic and clinical value of peripheral blood testing in early-stage CC patients with VI and to construct a nomogram based on age, PNI, PLR, histology types, and physical examination to increase the diagnostic accuracy of VI.
Our study has several limitations. First, it is a retrospective, non-randomized design study with only Chinese participants. Second, the influence of different VI assessment methods on the prognosis of early-stage CC has not yet been evaluated. Third, age, histological type, PNI, PLR, NLR, and SIRI were analysed as binary categorical variables owing to the limited number of cases. Therefore, further multicentre prospective studies are needed to confirm our nomogram.
Conclusion
The present study demonstrated five preoperative factors, including age, PNI, PLR, histological type, and physical examination, as novel diagnostic predictors of VI in early-stage CC. These variables can be incorporated into a pre-treatment diagnostic nomogram, which could facilitate individualised diagnostic assessments and treatment tailoring for early-stage patients.
Footnotes
Acknowledgments
We would like to thank all the researchers and patients, as well as recognize Yang Sun, whose contributions significantly enhanced the quality of this publication.
Author Contributions
Writing - original draft, Ning Xie and Jie Lin; Formal analysis, Ning Xie; Methodology, Jie Lin; Investigation, Haijuan Yu; Visualization, Haijuan Yu; Validation, Li Liu; Data curation, Sufang Deng; Software, Linying Liu; Conceptualization, Yang Sun; Writing - review & editing, Yang Sun.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Major Scientific Research Program for Young and Middle-aged Health Professionals of Fujian Province, China(Grant No. 2022ZQNZD008) and the High-level Talents Training Project of Fujian Cancer Hospital (2022YNG04).
Ethical Statement
Data Availability Statement
The data supporting the results of this study can be provided by the corresponding author upon reasonable request.
