Abstract
Purpose
To explore clinicopathological characteristics and their prognostic value among young patients with cervical cancer (who are aged ≤25 years old).
Methods
The Surveillance, Epidemiology, and End Results Program (SEER) database was used to extract data on cervical cancer patients. They were then stratified by age as young women (≤25 years old) and old women (26–35 years old) and analyzed for clinicopathology characteristics and treatment modalities. Prognosis was analyzed using Kaplan–Meier survival curve, as well as hazard ratios using Cox regression modeling. The nomogram was developed based on Cox hazards regression model.
Results
Compared to 26–35 years old women, patients aged ≤25 years tended to be white ethnicity, unmarried, had earlier stage of disease. There was also a better prognosis among younger cohort. Grade, FIGO stage, histologic subtypes, and surgical modalities influenced the survival outcomes of young patients. Among young cohorts, surgery prolonged the survival time of IA-IIA stage patients while surgical and non-surgical management presented no statistically prognostic difference among patients at IIB-IVB stage. Besides, the nomogram which constructed according to Cox hazards regression model which contained independent prognosis factors including FIGO stage, surgery type, and histologic type of tumor can robustly predict survival of young patients.
Conclusion
Cervical cancer patients ≤25 years old were uncommon and lived longer than the older patients. Among these young patients at IA-IIA stage, surgical treatment could be more effective at preventing death than non-surgery. The nomogram could perfectly predict the prognosis of young adults and adolescents with cervical cancer.
Introduction
Cervical cancer is the fourth most common and lethal cancer among women. It is estimated that there were approximately 570,000 new cases and 311,000 cancer-related deaths in 2018 worldwide, most of which occurred in the developing country. 1 In addition, in the United States, cervical cancer was the second leading cause of cancer related death among women aged 20–39 years, with 10 deaths per week. 2 Furthermore, an increasing number of young women have been diagnosed with cervical cancer according to epidemiologic studies.3-5
As has been proposed from previous studies, the morbidity and mortality of cervical cancer have been marked decreased in the United States over the past few decades6-8 due to the availability of the HPV vaccination and adoption of cervical screening. Although, for young women especially adolescents, starting screening earlier than 21 years old was believed to obtain more harm than benefits. 9 In addition, the treatment options for young adults and adolescents with cervical cancer is complicated because preservation of fertility is of great importance and should be considered to young patients(10-12). Hence, it is of importance to explore the clinicopathological characteristics and find a meaningful method to predict the prognosis of young women with cervical cancer.
Accordingly, in the present study, the clinical characteristics, treatment modalities, and prognosis of cervical cancer patients ≤25 years old were analyzed and their difference with patients at 26–35 years old were evaluated.
Patients and Methods
Patients
The information of the patients was obtained from the latest version of Surveillance, Epidemiology, and End Results (SEER) cancer registry database, which covers nearly 28% of the US population. The incidence rate of cervical cancer (ICD-O-3 C53.0-C53.1, C53.8-C53.9) from 2004 to 2016 was acquired from the rate session of SEER*Stat 8.3.6. Furthermore, case listing session was used to identify all cervical cancer patients who were diagnosed from 2004 to 2016, and patients diagnosed >35 years old were excluded.
Variables
Patients’ characteristics were analyzed under following parameters: age, year of diagnosis, race (white, black, and other), FIGO stage (I, II, III, and IV), grade (well differentiated, moderately differentiated, poorly differentiated, and undifferentiated), tumor histology (squamous, adenocarcinomas, and others including uncommon subtypes such as complex epithelial neoplasms, small cell carcinoma and adenosquamous cell carcinoma), tumor size (≤4 cm and >4 cm), nodal status (node positive, node negative), numbers of positive lymph nodes (1, 2, 3, and ≥4) and survival. Besides, the FIGO stage was classified according to the FIGO cancer report 2018 13 based on the TNM stage provided by SEER database. In addition, treatment modalities such as the administration of site-specific surgery, radiotherapy, and chemotherapy were also included. As for surgical approaches, they were classified into 2 groups: hysterectomy including simple hysterectomy, modified radical or radical hysterectomy, hysterectomy not otherwise specified and pelvic exenteration; local tumor resection including conization alone or trachelectomy. Besides, “total hysterectomy without removal of tubes and ovaries” (surgery code 30) and “hysterectomy without removal of tubes and ovaries” (surgery code 61) were classified into hysterectomy without ovarian removal; on the other hand, “total hysterectomy with removal of tubes and ovaries” (surgery code 40) and “hysterectomy with removal of tubes and ovaries” (surgery code 62) were classified into hysterectomy with ovarian removal. Patients were then stratified by age into young patients (≤25 years old) and old patients (26–35 years old). It is worth mention because of the limitation of rate session of SEER database, which groups patients every 5 years from the age of 0 onward, such as 0–4, 5–9, and 10–14. The age groups were “<25 years old” and “25-34 years old” when analyzing incidence rate of cervical cancer patients.
Statistical Analysis
Further comparison of the qualitative data was done using the chi-squared (χ2) and Fisher’s Exact probability tests. The differences of cause-specific survival (CSS) and overall survival (OS) between the age of ≤ 25 and 26–35 were estimated using the Kaplan–Meier method and the comparison of the CSS and OS between 2 age groups was conducted by the log-rank test. Multivariate Cox proportional hazards model analyses were then performed to explore the risk factors for OS in women who under or at 25 years old. When
Nomogram
The univariate prognostic factors of OS and CSS were determined by the Kaplan–Meier estimates and the log-rank test, respectively. Variables with
The nomogram model was created based on the independent prognostic factors previously obtained by multivariate Cox hazards model and was then validated in 2 ways. First, the performance of prognostic nomogram was evaluated using concordance index (C-index), which ranged from .5 to 1.0 and was positively correlated with the accuracy of the prediction. 14 Second, the calibration curve was employed to display concordance probabilities of prognostic nomogram based on 1000 bootstrap resamples. In an ideal calibration curve, the predicted probabilities were agreed with the actual probabilities. 14 The nomogram and calibration curve were plotted by rms package of RStudio software (version 2.2.5033).
Results
Incidence and Survival of all Cervical Patients Stratified by Age
The incidence of cervical cancer in patients stratified by age from 2004 to 2016 was examined. As it is depicted in the columns (Figure 1), the incidence of patients was in decline in <25 years old patients. Although the fluctuation of case number in <25 years group was slight, the morbidity dropped by over than 50%. Tendency of incidence rate of cervical cancer patients stratified by age from 2004 to 2016.
Data was evaluable for a total of 7,372 patients with cervical cancer ≤35 years old between the years 2004 and 2016. Survival curves for all patients are depicted in Figure 2. A statistically differences according to the age groups was seen, with a better CSS rate and OS rate of young age group than their older counterparts ( Comparison of survival rates of cervical cancer patients between patients ≤25 and 26–35 years old. (a) Cancer-specific survival; (b) overall survival.
Interestingly, when comparing age groups among different histologic type, the results were different (Supplementary Figure 1). Among patients diagnosed with squamous cell cancer, young patients performed a better prognosis compared with old patients, while no statistically significant difference was observed between 2 age groups among patients diagnosed with adenocarcinoma and other rare histologic subtypes. On the other hand, when FIGO stage was controlled, there was no significantly statistically difference in CSS and OS between young and old age group among each FIGO stage (Supplementary Figure 2).
Demographic and Clinicopathological Characteristics of Patients
Distribution of Cervical Cancer Patients’ Demographic and Clinicopathological Characteristics by Age Group.

Compared with patients diagnosed at 26–35 years old, though no statistically significant difference was observed, more patients ≤25 years old were diagnosed at FIGO I stage (71.6% vs 68.1%). The fraction of patients in FIGO I stage was in decline with increasing age among all cervical cancer, which further confirmed that younger patients with cervical cancer were more likely to be diagnosed at earlier stage (Supplementary Figure 3).
The differences of treatment modalities between the 2 groups are presented in Table 1. The younger group had a higher percentage of undergoing surgery than the older group (78.0% vs 73.9%,
Univariate and Multivariate Survival Analysis for Young Cervical Cancer Patients
Univariate Analysis of Overall Survival in Cervical Cancer Patients ≤25 Years Old.

Multivariate analysis was performed with variables including grade, FIGO stage, histologic type, and surgery type of the patients with cervical cancer patients using Cox proportional hazards model and revealed that histologic type, FIGO stage, and surgery were independent prognostic factors for the OS rate of cervical cancer patients ≤25 years old (Figure 3). Specifically, compared to squamous cell neoplasms, other subtypes demonstrated a hazard ratio of 2.67 ( Forest plot based on Cox hazards model of adolescent and young adult patients with cervical cancer.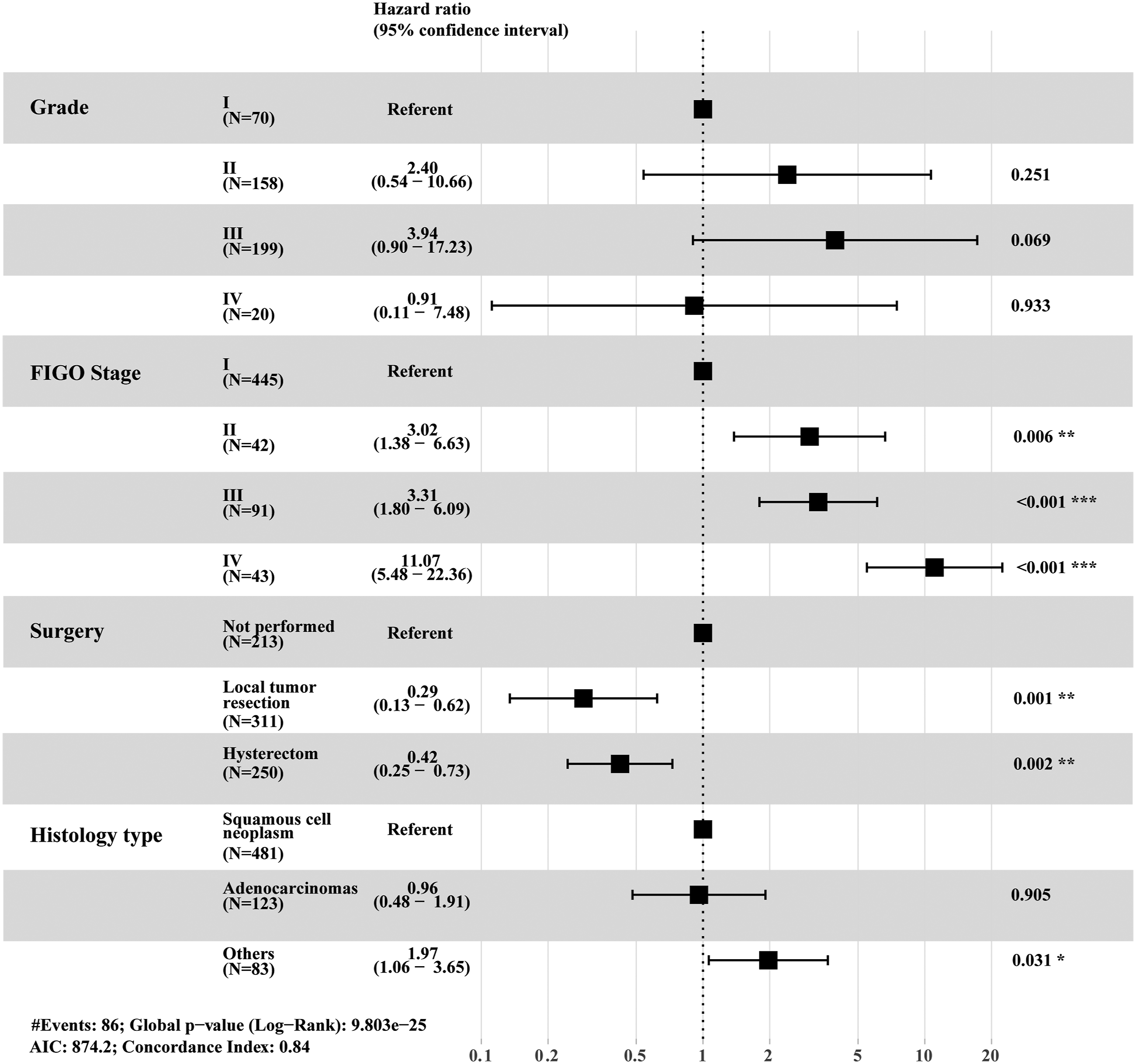
Influence of Surgery on Overall Survival Rate Among Young Cervical Patients
Influence of surgery on overall survival rate among young cervical cancer patients in multivariate Cox hazards model.

*

Comparison of survival rates of overall survival (OS) rates according to different surgical approaches in adolescent and young adult patients with IA-IIA stage cervical cancer who only received operation (

Comparison of survival rates of overall survival (OS) rates according to whether ovarian removal was conducted among young adult patients with hysterectomy (
Construction and Validation of Nomogram of Young Patients
According to the previous Cox proportional hazards model, the independent prognostic factors were FIGO stage, histologic type, and surgery. Therefore, a nomogram was developed with these variables to predicting OS of cervical patients ≤25 years old (Figure 6). The nomogram illustrated that FIGO stage had the greatest impact to the prognosis, followed by tumor grade and histologic type. Each prognostic factor determined a score on the point scale. By positing the total score on the total point scale, the probability of 1-year, 3-year, and 5-year OS can be estimated. For instance, a young cervical cancer patient diagnosed squamous cell cancer at FIGO II stage has undergone hysterectomy. The total score of her was 102 (27 from histologic type, 32.5 from surgery type and 42.5 from FIGO stagerespectively); by positing the total score on the scale, we got the predicted survival probabilities of 1 year-, 3 year-, and 5 year- (>90%, 82%, and 76.4%, respectively). Overall survival (OS)–associated nomogram for adolescent and young adult patients with cervical cancer.
The C-index for the nomogram was .84 (95% CI, .82–.86), which implied that the probability of the agreement between forecast and actual was 84%. To further validate the performance of the prognostic predicted model, the calibration curves were plotted. It can be observed that the 1 year-, 3 year-, and 5 year-actual calibration was very close to the perfect calibration, which suggested that there was a satisfactory agreement between prediction and observation (Supplementary Figure 4).
Discussion
This research represented the largest series to date analyzing the prognostic factors and their prognostic value of adolescents and young adult patients with cervical cancer.
In this study, time trend analyses revealed that the morbidity of young patients was in decline from 2000 to 2016, which is similar to the decreased occurrence rate and mortality of cervical cancer patients in many populations worldwide.15,16 It mostly attributed to the development of HPV vaccine 1 and cervical cancer screening. 9 In many countries, it is currently recommended that women started routinely cervical screening at 25 years old.17-21 Particularly, some guidelines also recommend that women should started screening at the age of 20 or 219,22-24. However, what age cervical cancer screening should begin at remains further discussion. Previous study revealed that cervical screening in women aged 20–24 has no significant effect on decreasing incidence rate. 25 Also, the benefit of screening before 25 years old cannot balance against the potential harms and the false-positive results may cause overtreatment. 21 Therefore, it needs more research to identify the optimal age when starting screening.
As for the survival rate of young women with cervical cancer, some previous studies showed that age does not affect the prognosis of cervical cancer patients,26-28 while other studies argued that younger patients show a better survival rate.12,29 In this study, young cervical cancer patients live longer than the older. The differences might ascribe to the different age boundaries among different research.
In further exploring the prognostic factors of cervical cancer among patients ≤25 years old, univariate and multivariate analysis revealed that grade, FIGO stage, histologic type, tumor size, nodal status as well as patterns of treatment could influence the survival outcomes of young cervical cancer. These findings were consistent with those among older patients according to several previous reports.30-32 Interestingly, we found that although histologic type did influence the survival outcome of cervical cancer patients ≤25 years old, there were no significant survival differences between adenocarcinoma and squamous cell cancer women. It was contrast to previous researches where squamous cell cervical cancer had a better prognosis than the adenocarcinoma presented.33-35 This result may be explained by the fact that the majority of the patients in this age group were diagnosed at FIGO I stage. Among women at FIGO I stage, the squamous cell carcinoma showed no survival advantage over adenocarcinoma according to several previous findings.36-39
Findings from previous studies showed that regardless of age, women ≤35 years old generally presented a higher frequency of adenocarcinoma compared with patients >35 years old,40,41 while the most common histological type was squamous cell carcinoma.40-43 However, these previously published series did not mention the distribution of histologic type in patients at 26–35 years old. Our study showed that adenocarcinoma presented a lower incidence in patients ≤25 years old. In addition, squamous cell cancer was indeed the most frequent type of histology in cervical cancer both in patients at ≤25 and 26–35 years old.
In the distribution of stages, no statistically significant difference was found between patients ≤25 and 26–35 years in a cross-sectional study by
Our nomogram indicating that among variables that impact the prognosis of young cervical patients, FIGO stage had the most significant influence, followed by histologic type and surgery, successively. The C-index was very close to 1.0, and the calibration curve was perfectly in agreement with actual prediction curve, which indicated that these 4 factors can precisely predict the survival outcome of young cervical patients.
The optimum treatment for cervical cancer patients diagnosed at early stage has remained a source of controversy. Previous study conducted in cervical cancer patients at Ib stage by
As for stage IIB-IVB of the disease, it is well acknowledged that definitive chemoradiotherapy is the standard initial treatment for cervical cancer, 49 and combined therapy was not recommended by the FIGO cancer report 2018 since surgery with adjuvant radiotherapy is believed to increase morbidity. 13 Consistent with these viewpoints, in our multivariate analysis of the advanced cohort, surgery was not an independent prognostic factor, which proved that no significant difference of the OS was found between patients received combined treatment and patients who did not undergo any type of operation but received radiation or (and) chemotherapy. In addition, there were nearly half of the patients at stage IIB-IVB who underwent surgery in our research. With the publication of the new FIGO classification for cervical cancer, patients at stage I and II with lymph node metastases were reclassified into stage III. 50 Most of them had received hysterectomy, which may explain the reason for the large proportion of patients at stage IIB-IVB undergoing surgery in this study.
In the current study, there are a number of strengths and limitations that warrant careful consideration. First, this is currently the largest study concentrating on female cervical cancer ≤ 25 years old, including clinical factors such as FIGO stage, histology, and grade. Second, we used data from the SEER, the largest tumor registry in the United States, which provide valuable evidence about the treatment of cervical cancer in young women. One of the primary limitations of this study is its retrospective nature, causing difficulty to rule out selection bias. In addition, patients with unclear FIGO stage were not included in our cohort, which may lead to bias to our results. Because the SEER database lack of the HPV infection information, further work is required to better understand the relationship of HPV infection and the characteristics as well as modalities of younger patients with cervical cancer.
Conclusions
The incidence rate of cervical cancer was in decline from 2004 to 2016. Although there was a low proportion of patients aged ≤ 25 in cervical cancer patients, this group tended to be less aggressive, with a better prognosis than their older counterparts. Moreover, greater attention should be paid to cervical carcinoma patients with unusual histologic subtypes, higher clinical stage, poor differentiation grade, and lymph node metastasis, as these factors predict significantly shorter survival times. In addition, different surgical approaches had no significant effect on prognosis for stage IA-IIA among young cervical cancer patients. Furthermore, we developed a well validated nomogram to predict the prognosis of patients aged ≤ 25 years old.
Supplemental Material
sj-pdf-1-ccx-10.1177_10732748211051558 – Supplemental Material for Clinicopathological Features and Survival of Adolescent and Young Adults with Cervical Cancer
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748211051558 for Clinicopathological Features and Survival of Adolescent and Young Adults with Cervical Cancer by Shuya Pan, Wenxiao Jiang, Shangdan Xie, Haiyan Zhu and Xueqiong Zhu in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Science and Technology Planning Project of Wenzhou city (No.ZS2017006) and Center for Uterine Cancer Diagnosis & Therapy Research of Zhejiang Province. The sponsor had no involvement in the collection, analysis, and interpretation of data or in the writing of the article.
Ethics Approval
All data in this article are achieved from SEER database, which have passed the ethical application.
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
