Abstract
Introduction
Docetaxel plus ramucirumab (DTX + RAM) therapy is a standard treatment for previously treated lung cancer, but many adverse events have been reported. This retrospective study was conducted to examine if the side effects of DTX + RAM therapy can be minimized by the combined use of oral dexamethasone (DEX), and to assess the therapeutic effect of DTX + RAM in patients with recurrent lung cancer.
Methods
Forty patients with relapsed non-small cell lung cancer who underwent DTX + RAM therapy were divided into two groups based on the concomitant use of oral DEX, and the therapeutic effects and toxicities in the two groups were compared.
Results
The objective response rate (ORR) was significantly better in the DEX group (P = 0.0203). The median progression-free survival (PFS) was 5.20 months vs 2.87 months (P = 0.064) in the DEX and non-DEX groups, respectively. However, the median overall survival (OS) was significantly better in the DEX group (15.17 months vs 7.37 months, P = 0.0317). The frequency of fluid retention within six months of the start of treatment was 10.0% vs 42.5% in the DEX and non-DEX groups, respectively, with the fluid retention rate being significantly higher in the non-DEX group (P = 0.039).
Conclusion
Concomitant use of oral DEX during DTX + RAM therapy may facilitate the long-term continuation of treatment and contribute to OS prolongation.
Introduction
Docetaxel plus ramucirumab (DTX + RAM) therapy is a standard treatment for previously treated non-small cell lung cancer (NSCLC). In the REVEL study, patients who were administered DTX + RAM therapy had a median progression-free survival (PFS) of 4.5 months and a median overall survival (OS) of 10.5 months, and significantly prolonged survival compared to the patients who were administered DTX alone (P < 0.0001 and P = 0.023, respectively). 1 In a phase II study conducted in Japan, patients who were given DTX + RAM therapy had a median PFS of 5.22 months and a median OS of 15.15 months, which was better than in patients who were given DTX alone (hazard ratios (HR) 0.83 and 0.86, respectively). 2 But, in several other studies, DTX + RAM therapy showed strong toxicities such as febrile neutropenia (FN), thrombocytopenia, mucositis, and epistaxis.
Corticosteroids are often used to prevent side effects when administering DTX. One of the most important reasons for using steroids is to prevent fluid retention. Edema is one of the most important side effects of DTX. Some past report showed that edema caused by DTX occurred due to increased vascular permeability. 3 Semb et al. demonstrated that DTX induced an initial enhancement of fluid filtration followed by a capillary protein leakage. 4 Peripheral edema was found in 16% of the patients receiving DTX + RAM therapy and 8% of those receiving DTX alone, and the frequency of edema increases with the combined use of anti-vascular endothelial growth factor (VEGF) antibody. 1 Fluid retention including peripheral edema, pleural effusion, and ascites was reported to occur in about 40% of patients at 6 weeks after administering DTX, and in 90% of patient when the cumulative dose was 400 mg/m2. 5 Although oral dexamethasone (DEX) before and after administration of DTX was reported to prevent fluid retention, most of the reports were on breast cancer,5,6 and there are no reports on the impact of oral DEX on the prevention of fluid retention in lung cancer patients.
This study retrospectively examined if the side effects, including fluid retention, could be minimized by concomitant use of oral DEX along with DTX + RAM therapy and evaluated the therapeutic effects of DTX + RAM therapy in patients with recurrent lung cancer.
Materials and Methods
Forty patients with previously treated NSCLC who underwent DTX + RAM therapy between January 2017 and December 2021 were included in this study. The study participants were anonymized and enrolled consecutively. The opt-out method was adopted to obtain informed consent from participant, because existing information based on medical records was used in this study. They were divided into two groups based on the usage of oral DEX: the DEX group and the non-DEX group. The therapeutic effects and toxicities were examined and compared between the two groups.
On the day of DTX + RAM administration (day 1), 6.6 mg of DEX was administered as a premedication in both groups via an intravenous drip. Oral DEX was administered at 8 mg on the day after DTX + RAM administration and also the next day (days 2 and 3).
To study the therapeutic effects, the disease control rate (DCR) and objective response rate (ORR) were compared using Fisher’s exact test, and PFS and OS were compared using the log-rank test in each group. Toxicities, which were evaluated using Common Terminology Criteria for Adverse Events (CTCAE) ver5.0, was compared using Fisher’s exact test. The frequencies of edema and fluid retention were compared using the log-rank test. All statistical analyses were performed using by R software (R Foundation for Statistical Computing, Vienna, Austria). The reporting of this study conformed to STROBE guidelines. 7 The protocol was approved by the Institutional Review Board of Hiroshima Prefectural Hospital (No. R3-28-8).
Results
Patient Characteristics
Patient Characteristics.

Efficacy
Disease Control and Objective Response Rates.

CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; NE, not evaluated; DCR, disease control rate; ORR, objective response rate.
Median PFS showed a positive trend in the DEX group when compared to the non-DEX group (5.20 months vs 2.87 months), but was not significant (P = 0.0640). On the other hand, the median OS from the initiation of DTX + RAM therapy in the patients was significantly better in the DEX group than in the non-DEX group (15.17 months vs 7.37 months, P = 0.0317). (Figure 1a and b). Kaplan-Meier curves of DEX- and non-DEX groups (a) progression-free survival and (b) overall survival.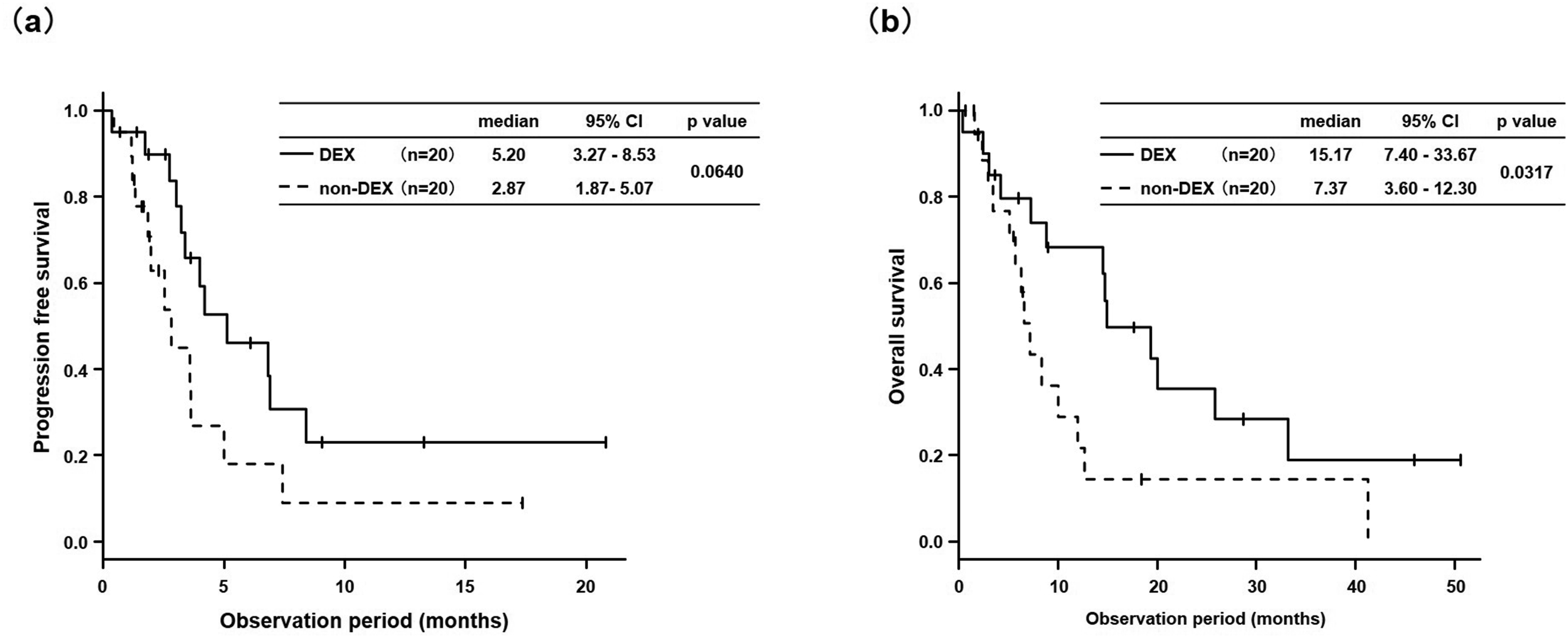
Adverse Events
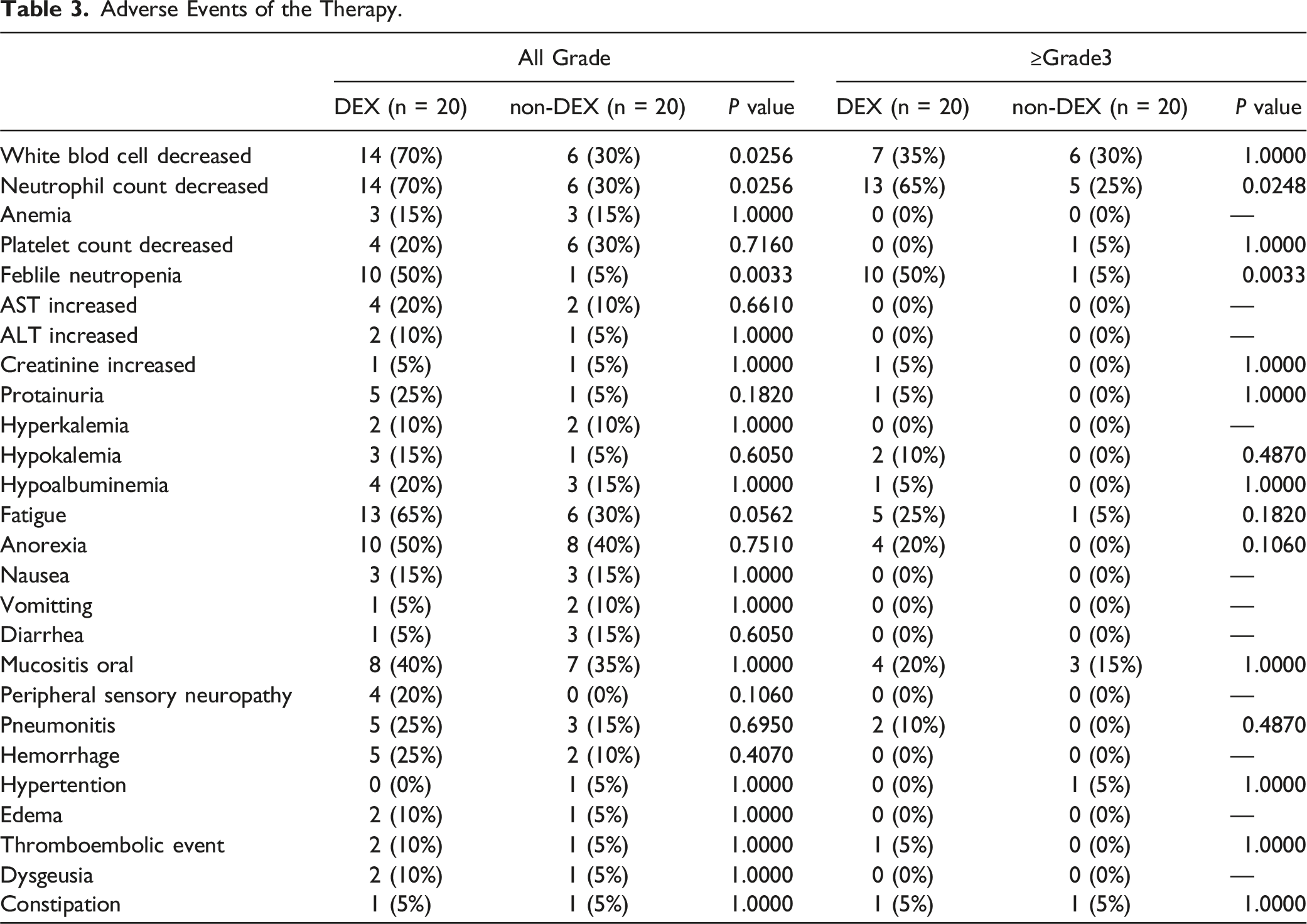
Adverse Events of the Therapy.

Kaplan-Meier curves showing the rate of fluid retention (peripheral edema, pleural effusion and ascites).
Discussion
No study has reported the therapeutic benefit of DTX + RAM therapy in combination with DEX in lung cancer patient. Previous studies on breast cancer have shown the effectiveness of concomitant use of oral DEX before and after administration of DTX, to prevent fluid retention, such as pleural effusion, ascites, and edema.5,6 In this study on lung cancer, the frequency of fluid retention six months after the start of treatment was 10.0% vs 42.5%, which was significantly higher in the non-DEX group. Only one patient in the non-DEX group who was able to continue treatment for more than a year eventually discontinued because of systemic edema and pleural effusion.
The PFS was better in the DEX group than in the non-DEX group, but no significant difference was observed. In contrast, the OS was significantly prolonged in the DEX group. Harada et al. compared the efficacy of DTX + RAM therapy in patients with and without ICIs with prior treatment. The median PFS was significantly better in those with ICIs than in those without (5.7 months vs 2.3 months, P = 0.020), the median OS was also more favorable in the former than in the latter group, at 13.8 months vs 10.5 months (P = 0.065). 8 Moreover, Kato et al. compared the effects of cytotoxic anti-cancer drug injection in the third-line treatment following an ICI with the second-line treatment. The median OS for patients receiving DTX + RAM therapy was reported to be 17.5 months in the third-line vs 13.5 months in the second-line therapy (HR 0.67, 95% confidence interval (CI) 0.41-1.11, P = 0.12). 9 These studies demonstrated that DTX + RAM therapy after ICI treatment was more effective than before ICI treatment. Furthermore, Kasahara et al. stated that the use of pegfilgrastim reduced the incidence of FN to 5%, with a median PFS of 6.6 months and a median OS of 18.4 months. 10 In this study, more patients in the non-DEX group received ICIs before DTX + RAM therapy, and pegfilgrastim compared to those in the DEX group. In the non-DEX group, all patients received ICIs, whereas in the DEX group, only 15 (75%) received ICIs throughout the entire clinical course (P = 0.0471). Notably, this study revealed a significant prolongation of OS in the DEX group, despite their lower pretreatment utilization of pegfilgrastim and ICIs. This finding implies that DTX + RAM therapy, when combined with DEX, may be sustained over an extended period.
The adverse toxic events, leukopenia, neutropenia, and FN were significantly more common in the DEX group. However, it is considered that this is caused by the significant increase in the use of pegfilgrastim in the non-DEX group. The DTX + RAM therapy was continued for a longer period in the DEX group, even though there was no significant difference in the frequency of the other adverse events because, in addition to the significantly longer time to fluid retention, DEX administration might also have improved quality of life (QOL) which cannot be evaluated by CTCAE. However, previous reports on steroid-sparing showed no significant difference in QOL between the standard steroid treatment group and the steroid-spared group. 11 Further studies are needed on this point. Moreover, it is possible that the duration of side effects was shortened by using DEX in combination, even if the grade in CTCAE is similar. However, this was not verified in this study.
This study had several limitations. First, this was a single-center retrospective study. Hence no significant difference in PFS was observed because of the small sample size. Second, patient backgrounds differed between the two groups. In particular, there were significant differences in the use of pegfilgrastim, and the treatment interruption due to myelosuppression was not investigated. Propensity matched analysis was preferable to compare two cohorts with different backgrounds, such as this study. However, it was difficult because of the small sample size. This study showed that the combined use of DEX in DTX + RAM therapy might diminish the side effects and improve the treatment outcomes, including the evaluation of QOL. It was insufficient to discuss the definitive necessity of DEX, so a multicenter prospective study with similar patient backgrounds is warranted.
Conclusions
Concomitant use of oral DEX during DTX + RAM therapy reduced the frequency of fluid retention, and may facilitate the long-term continuation of treatment and contribute to OS prolongation.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Kosuke Hamai. The first draft of the manuscript was also written by Kosuke Hamai and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
