Abstract
Introduction
Patients with lung adenocarcinoma not expressing TTF1 and those with a KRAS mutation have worse prognosis. However, available data are limited and sometimes contradictory. Therefore, this retrospective cohort analysis aimed to clarify whether there was a difference in overall survival and progression-free survival between these groups of patients.
Methods
In total, data derived from 181 patients with metastatic lung adenocarcinoma treated at the Martha-Maria Halle-Dölau Hospital from 2016 to 2019 were analyzed. Kaplan-Meier curves were generated, and associated values, such as median survival and its confidence intervals, were determined using the log-rank test.
Results
A benefit in overall survival (OS) (8.4 vs 5.8 months; HR, .8; 95% CI, .53-1.19; P = .267) was associated with positive TTF1 expression, but this was not statistically significant. The same trend was shown with the progressive free survival (PFS) (6.5 vs 4.6 months; HR, .76; 95% CI, .51-1.20; P = .162). In patients with a KRAS mutation, there was no difference in OS compared to those with a wildtype KRAS. The median survival was almost identical at 7.5 months (KRAS mutation, 95% CI, 3.32-11.74) and 7.0 months (KRAS wildtype, 95% CI, 3.59-10.41). Additionally, in PFS, there was no difference between the 2 groups (5.8 vs 6.3 months).
Conclusions
Our analysis did not show a worse prognosis in patients with a KRAS mutation or in those with missing TTF1 expression, which is most likely related to the new therapeutic options. As a result of the administration of immunotherapy in patients with a KRAS mutation and the change from a regimen containing pemetrexed to a regimen containing no pemetrexed, the corresponding patients no longer seem to have a worse prognosis.
Introduction
Lung carcinoma remains the most common cause of cancer-related deaths with almost 1.8 million deaths worldwide every year. 1 At the time of initial diagnosis, half of the patients are already in a metastatic stage, with approximately 5% and 3% being the 5-year survival rate for women and men, respectively (Koch-Institut, Robert and Gesellschaft Der Epidemiologischen Krebsregister In Deutschland E.V. 2019). There are various histological subtypes, with adenocarcinoma (AC) being the most common subtype at approximately 40%. 2 Typical markers of AC include napsin A and thyroid transcription factor 1 (TTF1). 3 TTF1 is particularly relevant as an immunohistochemical marker for the identification of the primary tumor metastasis or differentiation of carcinoma, as it indicates the development of lungs AC. 4 If another primary tumor is ruled out clinically, lung AC can also be TTF1 negative as a result of a dedifferentiation of the tumor. Other studies have shown that approximately 80% of primary pulmonary AC are TTF1 positive.5-7 According to new findings, patients with TTF1-negative tumors have a worse prognosis, partly because they respond poorly to chemotherapy containing pemetrexed. 8
Furthermore, driver alterations occur more frequently in AC than in other histological subtypes, which is why appropriate molecular testing should be performed before initiating systemic therapy for metastatic AC. 9 It is currently recommended to test for at least EGFR mutations in exons 18-21, ALK fusions, ROS1 fusions, and BRAF V600 mutations at the metastatic tumor stage. 9 Targeted therapy for the corresponding driver changes has decisively improved the prognosis of these patients.
The most common mutation in AC is detected in the KRAS gene. In general, KRAS-mutated carcinomas are associated with poor prognosis. Several meta-analyses have reported an increased risk of death in patients with a KRAS mutation.10,11 It is unclear whether this association will continue to be applied in the future, as there are also studies showing that patients with the corresponding KRAS mutation respond better to immunotherapy, especially with TP53 comutation.12,13 Since January 2022, the first targeted therapy for KRAS-G12 C mutations in patients with progressive disease under at least 1 previous systemic therapy has been approved. 14 This could also decisively improve the prognosis of patients.
The aim of this retrospective cohort study was to clarify whether there is a difference in overall survival (OS) and progression-free survival (PFS) between patients with and without a KRAS mutation or TTF1 expression.
Methods
A total of 181 patients with metastatic lung AC were treated at the Martha-Maria Halle-Dölau Hospital between 2016 and 2019 and complete mutation analysis was performed. In addition, age at initial diagnosis (ED), ECOG performance status, sex, stage at initial diagnosis (stages IVA and IVB or in patients with ED in 2016 only stage IV), smoking history, medical history, and possible occupational exposure to asbestos were recorded. The type of first- and second-line therapies (chemotherapy, immunotherapy, immunochemotherapy, tyrosine kinase inhibitors, radiation only, and none) was also noted. The end of the observation period was November 2020. Patients who are lost to follow up at 1 point in time and patients who were still alive or progression-free at the end of the observation period were listed as censored.
The methods used for mutation analysis were nucleic acid amplification by PCR of the sections coding for KRAS exon 2 codon 12/13, EGFR exon 18, 19, 20, 21, and BRAF exon 15 codon 600 with specific probes and subsequent sequencing with direct detection of potential point mutations with a sensitivity of 5% (KRAS, BRAF) or 20% (EGFR) mutated DNA.
To determine ALK and ROS status, chromogenic hybridizations (CISH) were used with the ZytoDot 2C SPEC ALK or ROS DNA probe combinations spanning the corresponding gene loci from Zytovision, showing a translocation by assigning the different colored signals in the sense of a so-called break-apart probe and subsequent counting of at least 50 tumor cells.
Statistical analyses were performed using SPSS 27 (IBM Corp., Armonk, NY, USA). Survival analyses first comprised a descriptive presentation of the cumulative survival functions according to Kaplan-Meier analysis, and differences among the curves were evaluated using the log-rank test. Hazard ratios were determined using Cox regression analysis. The chi-squared test was used to determine whether there could be an association between certain characteristics. The reporting of this study conforms to STROBE guidelines. 15
Results
A total of 181 patients with metastatic lung AC were treated at the Martha-Maria Halle-Dölau Hospital between 2016 and 2019 and complete mutation analysis was performed.
Patient demographics.

Mutation analysis of all 181 patients.

Analysis of the KRAS subtypes.

The most common mutations were G12 V and G12 C in 15 and 14 patients, respectively, accounting for 33% and 31% of all KRAS mutations, respectively. The second most common mutation was G12D detected in 8 patients (18%). These 3 types together accounted for over 80% of all mutations in the KRAS gene in this sample. The remaining 18% were accounted for by G12S, G12 A, G12 F and 2 mutations in codon 13.
There was no association between sex and KRAS mutation (26% of men and 23% of women had KRAS mutations).
First-line therapies with respect to KRAS status.

Among the 181 patients, 81.2% were current or former smokers and 18.8% had never smoked. Over 91% of patients with a KRAS mutation were current or former smokers, while only 78% of those with wildtype KRAS had ever smoked. This difference becomes even clearer when examining these values from a different perspective. The rate of KRAS-mutated tumors at 28% among current or former smokers was about twice as high as that of non-smokers (12%). The odds ratio was 2.90 (95% CI, .96-8.75). Using the chi-square test, a P-value of .05 was determined for the question of a possible association between smoking status and the presence of a KRAS mutation (Fisher: 0.076).
A comparison between patients with and without occupational exposure (OE) in terms of asbestos contact shows a very similar picture. Among the KRAS mutants, the proportion of patients with OE was 13.3%, approximately 2.5 times as high as that among the KRAS wildtypes (5.1%). At 46.2%, occupationally exposed individuals had KRAS mutations almost twice as often as patients without OE. The odds ratio therefore is 2.84 (95% CI, .90-8.93, P = .065; Fisher 0.092). If 1 compares patients with both OE and a history of smoking with nonsmokers without asbestos contact, the difference becomes even clearer. In the group exposed twice, KRAS mutations occurred more than 3 times as frequently. The odds ratio was 6.43 (95% CI, 1.42-29.08; P = .01; Fisher 0.017).
PFS and OS were analyzed according to KRAS status (Figures 1 and 2). Because of the known significantly better prognosis of patients with driver alterations that can be treated as first-line treatment, patients with EGFR, ALK, ROS1 and BRAF alterations were excluded. In total, this corresponded to 154 patients, of whom 45 patients were KRAS mutated. PFS of patients with a KRAS mutation and all patients carrying the wildtype gene without EGFR, ALK, ROS1 and BRAF alteration. OS of patients with a KRAS mutation and all patients carrying the wildtype gene without EGFR, ALK, ROS1, and BRAF alteration.

There were no differences in PFS between patients with or without the KRAS mutation. The median of patients carrying a KRAS mutation progressed at 5.8 months (95% CI, 4.00-7.53) vs 6.3 months (95% CI, 3.24-9.43) for patients carrying with wildtype gene. Even in multivariable regression, no prognostic influence of KRAS could be concluded from the values of this sample.
In terms of OS, there was also no difference between the patients carrying the mutated and wildtype KRAS gene. The median survival times were almost identical at 7.5 months (KRAS mutation, 95% CI, 3.32-11.74) and 7.0 months (wildtype, 95% CI, 3.59-10.41) for patients with the wildtype KRAS.
Finally, OS was analyzed with regard to KRAS subtypes (Figure 3, Table 5). All patients were also included, with the exception of those with EGFR, ALK, ROS1 and BRAF alterations. Among them, there were 109, 15, 14, and 8 patients with the wildtype KRAS, a G12 V mutation, G12 C and G12D mutation, respectively. We combined the remaining 8 mutations in 1 study arm, since these groups would otherwise be too small for analysis. OS with respect to KRAS subtype. OS by KRAS subtype.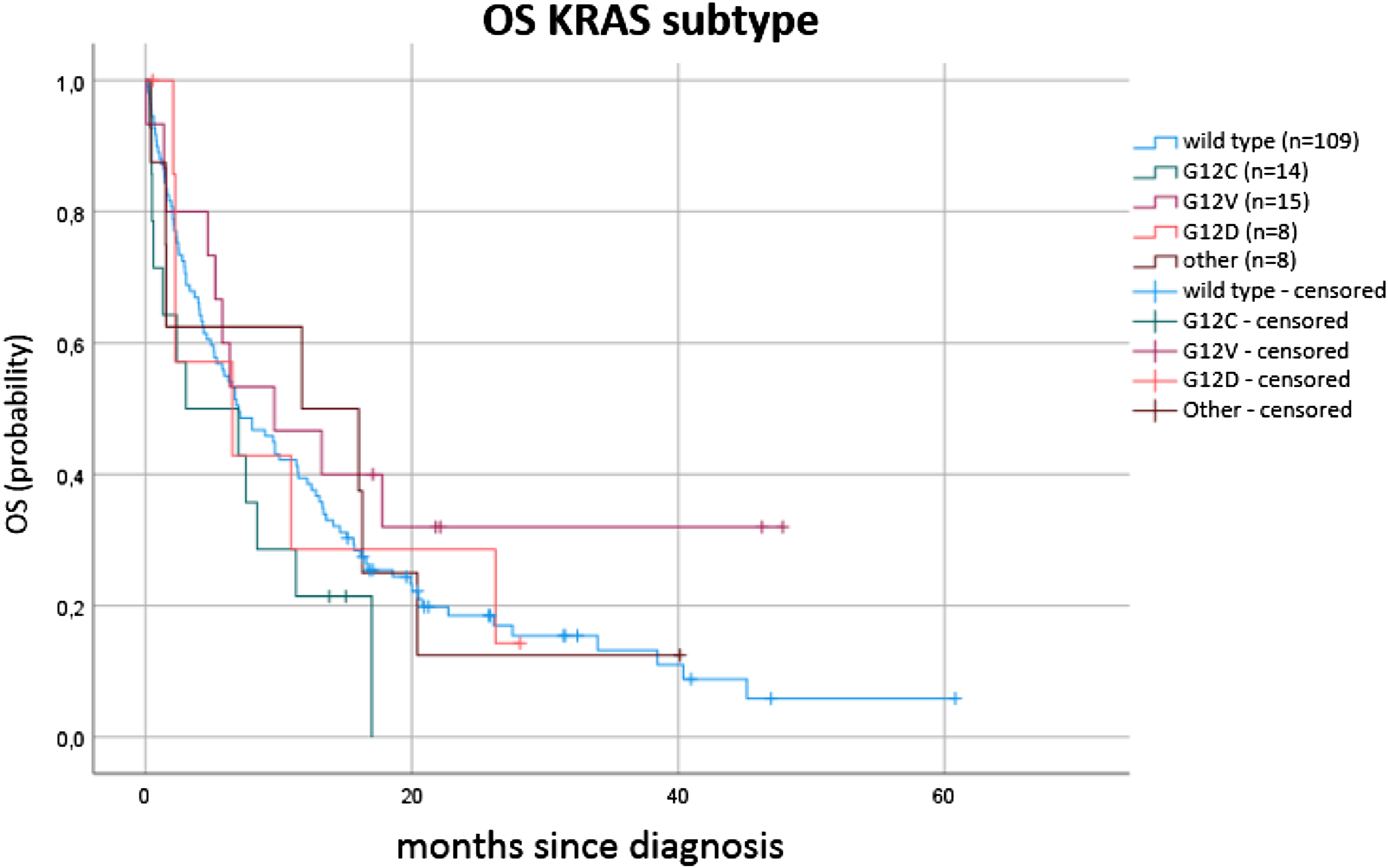

Again, no significant differences between the groups could be determined. However, an interesting trend has emerged. Comparing patients with the G12 C mutation to those with the G12 V mutation yields an approximately twice as high risk of death for patients with G12 C mutation compared to those with G12 V mutation.
The second part of the evaluation involved analysis of TTF1 expression. A total of 146 (80.7%) of patients showed weak TTF1 expression in the nuclei. Among the patients with TTF1 expression, the proportion of women was higher at 35.6% (52 out of 146) than among the those who were negative for TTF1 at 25.7% (9 out of 35). At least 85.2% of all women tested (52 out of 61) were TTF1 positive, but only 78.3% of all men tested were positive (94 out of 120). When tested for the association between sex and TTF1 expression, P = .266 (Chi 2 ), Fisher 0.322. Among non-smokers, the TTF1 rate slightly increased to 85.3% (P = .448; Fisher 0.630). The KRAS-mutated group showed slightly lower TTF1 expression (73.3% vs 83.1%; P = .151; Fisher 0.191). Among the 19 EGFR-mutated subjects tested, all subjects had TTF1 expression (P = .024; Fisher 0.027).
For the analysis of PFS and OS of patients expressing TTF1 compared to those not expressing TTF1, we included all patients except for those with EGFR, ALK, ROS1 and BRAF alterations (Figures 4 and 5). Patients who do not express TTF1 progressed after a median of 4.6 months (95% CI, 3.75-5.45) compared to 6.5 months (95% CI, 4.52-8.54) for patients with proven TTF1 expression. The hazard ratio in this case was .76 (95% CI, .51-1.20) with a P value of .162. PFS with respect to TTF1 status excluding patients with EGFR, ALK, ROS1, or BRAF alteration. OS with respect to TTF1 status excluding patients with EGFR, ALK, ROS1, or BRAF alteration.

While individuals without TTF1 expression died after a median of 5.8 months (95% CI, 3.76-7.78), those with expression did not die until after 8.4 months (95% CI, 5.77-11.03). According to the HR, patients expressing TTF1 showed only .80-fold (95% CI, .53-1.19; P = .267) higher risk of death.
Discussion
In this analysis, the mutation frequency of KRAS, at just under 25%, was slightly lower than that observed in other samples, which is most likely due to demographic and geographical differences.16,17 A clear association between the occurrence of a KRAS mutation and current or former nicotine consumption and exposure to asbestos could be shown. This has also been described in other studies in the past.18,19 The subtype analysis showed the G12 V mutation to be the most common subtype with a third of all KRAS mutations, closely followed by the G12 C mutation with a good 31%. All other subtypes occurred with a significantly lower frequency. Overall, 95% of all mutations were in codon 12 and almost 5% in codon 13. While the distribution between the codons corresponds to that of other studies, this sample shows a lower frequency of the G12 C mutation in favor of the G12 V mutation in comparison with previous work.20,21 Similarly, this could most likely be due to demographic and geographical differences, but for our geographical region, there is no study that has analyzed the KRAS subtypes to a relevant extend. Based on our data, no conclusion can be drawn between nicotine use or asbestos exposure and the subtypes of the KRAS mutation.
When considering the PFS and OS of the patients with and without KRAS mutation, any relevant difference between the groups cannot be determined. Older studies have shown an increased risk of death for patients with a KRAS mutation. For example, in the study by Huncharek et al., patients with a KRAS mutation had a 2.35-fold increased risk of death compared to those carrying the wildtype KRAS. 10 However, in this study, there was no adjustment with the tumor stage, while in 1999, there were no further therapy options, such as immunotherapy. In addition, Meng et al. found a worse prognosis for patients with KRAS mutation in a meta-analysis in 2013 with approximately 7,000 patients (HR, 1.39; 95% CI, 1.24-1.55) 11 ; however, no immunotherapy was used either.
There is already evidence that patients with the KRAS mutation respond better to immunotherapy than those with the wildtype KRAS.12,13 In our analysis, approximately 64% of all patients with KRAS mutation had either first- or second-line immunotherapy either in combination with chemotherapy or as monotherapy. This can thus explain the lack of differences in PFS and OS in patients carrying the KRAS mutants compared to the wildtype gene. Further analyzes could be performed in the future, comparing patients with a KRAS mutation with and without immunotherapy with regard to PFS and OS.
Subtype analysis showed a slightly increased risk of death in patients with a G12 C mutation. The hazard ratio was 1.553 (95% CI, .847-2.849, P = .155), and the median survival was only approximately 3 months compared to 7 months. This shows a clear trend that should be verified using a larger number of cases. With a hazard ratio of .648 (95% CI, .337-1.248, P = .19), G12 V was also a subtype that could be associated with a different prognosis, this time with a risk reduction. Comparing these 2 mutations, the hazard ratio was 2.02 (95% CI, .84-4.87; P = .115), which was approximately twice the risk of death for patients with G12 C compared to those with G12 V.
An association between G12 C and poorer survival was reported in 2014 by Nadal et al. who examined 179 resected AC with known KRAS status in a retrospective study of OS. According to them, G12 C is a negative prognostic factor with a strong risk increase in OS (HR, 2.35; 95% CI, 1.35-4.10, P = .003) compared to the wild type, but also compared to other KRAS mutations. 22 In addition, Svaton et al. described a poorer prognosis for patients carrying a KRAS mutation, especially G12 C, compared to the wildtype gene. 23 In contrast, Cui et al. found no difference between the 65 G12 C mutants and 79 other patients carrying a KRAS mutation in their study of 346 patients with NSCLC (HR, 1.19; 95% CI, .78-1.80, P = 0, 39). 24 Spira et al. found no survival disadvantage for G12 C in a retrospective study of over 7,000 patients with NSCLC. A somewhat longer OS has been observed in this subgroup. 25 Cai et al. found a poorer prognosis for G12D than for G12 C and G12 V (P < .0001). They had 20, 24, and 16 patients at their disposal, respectively. 26
The current data situation is very contradictory, and the individual studies are partly based on a small number of cases, since the respective subtypes comprise only a small proportion of patients. According to our data, there is currently no reliable difference in OS by KRAS subtype, although the trend mentioned above exists for the KRAS-G12 C and G12 V mutations.
Looking at TTF1 expression as the second part of our study, 80% of the patients had at least weak nuclear TTF1 expression, whereby our values correspond to the results of other studies.5,7 It is striking, however, that among the EGFR mutants, no patient tested negative for TTF1 and the rate of the patients tested positive was therefore significantly higher than among those with EGFR-wildtype gene, in which only 78% showed nuclear TTF1 expression (P = .018). This connection has been described by other authors.7,27,28 For example, Somaiah et al. found a high negative predictive value of over 96% in the absence of an EGFR mutation in the case of negativity for TTF1. 27
Regarding PFS and OS, the overall trend was that TTF1-negative patients had a poorer prognosis. In the case of OS, the Kaplan-Meier curve showed a separation of the 2 graphs after a few months, with a higher proportion of survival among the positive patients. This accounts for the largest difference in median OS of 8.4 months (95% CI, 5.77-11.03, positives) vs 5.8 months (95% CI, 3.76-7.78, negatives). However, the curves overlapped and almost matched at the end of the observation period. According to the hazard ratio, positive patients were at a .8-fold (95% CI, .53-1.19; P = .267) lower risk of death. The same trends were observed in the PFS (6.5 vs 4.6 month; HR, .76; 95% CI, .51-1.20; P = .162), but statistically more reliable due to the smaller number of censored cases. The graphs in the Kaplan-Meier curves did not overlap and were separated from each other at the end of the observation period, with a larger proportion of patients with progression-free disease among those who were expressing the TTF1 gene.
A possible cause could be the poor response of patients with missing TTF1 expression to pemetrexed-containing chemotherapy. 8 Almost 63% of TTF1-negative patients in our analysis were treated with a pemetrexed-containing regimen as first-line therapy. Over the course of time, based on the above-mentioned findings, pemetrexed therapy was already dispensed in our institution in this type of patient, and other regimens, such as a combination with a taxane, were selected instead. This is most likely the reason why, although there is a trend towards a poorer prognosis in TTF1-negative patients, it is not statistically relevant.
In summary, our analysis does not show a worse prognosis for patients with KRAS mutation or for those with missing TTF1 expression, which is most likely related to new therapeutic options. As a result of the addition of immunotherapy in patients with KRAS mutation and the change from a regimen containing pemetrexed to a regimen containing no pemetrexed in patients with missing TTF1 expression, the corresponding patients no longer seem to have a worse prognosis. This observation should be verified in larger samples in the future.
Abbreviations
adenocarcinoma
anaplastic lymphoma kinase
rapidly accelerated fibrosarcoma isoform B
chromogenic hybridizations
epidermal growth factor receptor
Kirsten rat sarcoma
occupational exposure
overall survival
polymerase chain reaction
progression-free survival
proto-oncogene tyrosine-protein kinase -1
thyroid transcription factor 1
Footnotes
Acknowledgments
We would like to thank Nancy Kuhn-Friedrich for giving us the anonymized patients information from the archive.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
A positive ethics vote for this retrospective study is available under number 2020-199 from the local ethics committee of Martin Luther University Halle-Wittenberg.
