Abstract
Introduction
Malignant wounds are lesions caused by metastasis from distant primary cancers or by direct invasion of the cutaneous structures of a primary cancer, and are most common in patients with breast or head and neck cancers. Malignant wounds not only cause physical symptoms, but also affect survival. Recognizing prognosis in terminal-stage cancer patients is necessary for both patients and health care providers. The prognostic impact of malignant wounds in patients with head and neck cancer has been poorly investigated.
Methods
This is a secondary analysis of the results of a prospective cohort study that investigated the dying process in patients with advanced cancer in 23 palliative care units in Japan. The primary outcome of this study was the prognostic impact of malignant wounds in patients with head and neck cancer. The difference in survival between patients with head and neck cancer who had malignant wounds and those who did not was compared using the log-rank test.
Results
Of 1896 patients admitted to palliative care units, 68 had head and neck cancer, and 29 of these had malignant wounds. Overall survival was significantly shorter in patients with malignant wounds than that in those without (median: 19.0 days vs 32.0 days, P = 0.046).
Conclusion
Patients with head and neck cancer who had malignant wounds had worse overall survival than those who did not.
Keywords
Introduction
Cancer is a leading cause of death globally.1,2 Alleviating symptoms such as fatigue, loss of appetite, pain, and dyspnea is crucial for patients with advanced cancer. 3 Malignant wounds, which are also referred to as ulcerating tumors or fungating wounds, 4 are lesions caused by metastasis from distant primary cancers, or by direct invasion of a primary cancer into the cutaneous structures. 5 Malignant wounds are found in 5%–15% of all cancer cases,6,7 most commonly in patients with breast cancer or head and neck cancer.8,9 Malignant wounds results in a loss of vascularity, and therefore the loss of nourishment to the skin, leading to tissue death and necrosis, 10 which are associated with specific findings such as exudate, bleeding, odor, itching, and appearance changes.8,9 A previous report showed that 67.7% of patients with malignant wounds had at least 1 of these conditions. 9 Malignant wounds not only cause various signs and symptoms, but also affect survival. Previous reports showed that malignant wounds were significantly associated with the occurrence of sudden, unexpected death. 11 Accurately recognizing the prognosis of patients in the terminal stage is necessary for both patients and health care providers, because it helps determine the treatment plan. Few previous studies have reported the prognostic impact of malignant wounds in each cancer type. Among patients with breast cancer, survival time was not significantly different between patients with and without malignant wounds. 12 However, there are no reports on the prognostic impact of malignant wounds in patients with head and neck cancer. Thus, we investigated the prognosis of malignant wounds in patients with head and neck cancer.
Multiple prognostic scores have been developed to provide clinicians with more accurate information on the prognosis of advanced cancer patients. 13 The Palliative Prognostic Index (PPI), the Palliative Prognostic Score (PaP score), the Delirium-Palliative Prognostic Score (D-pap score), and the modified Prognosis in Palliative Care Study predictor model (PiPS model) are all used as prognostic instruments. Although they differ in terms of the components used to predict prognosis, their accuracy is similar. 13 Among them, the PPI was initially developed in a hospice inpatient unit in Japan and is a well-known scoring system for predicting the survival of terminally ill cancer patients. 14 Only a small proportion of patients with head and neck cancer were included when the scores were developed. As such, the validity of these scores for patients with head and neck cancer is unknown. It is also unclear whether prognostic scores can be used for patients with malignant wounds.
The aim of this study was to evaluate the prognostic impact of malignant wounds in patients with head and neck cancer, as well as to investigate the utility of the PPI as a prognostic tool in this context.
Methods
Study Cohort
This study consisted of a secondary analysis of the results of a large prospective cohort study known as the East Asian Collaborative Cross-cultural Study to Elucidate the Dying Process (EASED), which investigated the dying process in patients with advanced cancer in 23 palliative care units (PCUs) in Japan (Aso Iizuka Hospital, Eiju General Hospital, Eikoh Hospital, Gratia Hospital, Hiroshima Prefectural Hospital, Hyogo Prefectural Kakogawa Medical Center, Japan Baptist Hospital, Japanese Red Cross Medical Center, JCHO Tokyo Shinjuku Medical Center, Kawasaki Municipal Ida Hospital, Komaki City Hospital, Mitsubishi Kyoto Hospital, National Cancer Center Hospital East, Osaka City General Hospital, Osaka General Hospital of West Japan Railway Company, Seirei Mikatahara General Hospital, St Luke’s International Hospital, Suita Tokushukai Hospital, Tohoku University Hospital, Tokyo Medical and Dental University Hospital, Tokyo Metropolitan Cancer & Infectious Diseases Center Komagome Hospital, Tsukuba Medical Center Hospital, and Yodogawa Christian Hospital) between January 2017 and June 2018. 15
The inclusion criteria of the present study were (1) adult age (18 years of age or older), (2) a diagnosis of locally advanced (cancer that spreads to nearby tissues, but has not yet metastasized to distant organs) or metastatic head and neck cancer (cancer that spreads from its site of origin to another part of the body), and (3) current admission to a PCU. Eligible inpatients newly admitted to the participating PCUs during the study period were consecutively enrolled in the study. During the study period, all study patients were followed for 6 months or until death. Patients with missing data for outcome variables were excluded.
All participating centers received ethical approval for the primary study, the protocol of which had assumed future secondary analyses of de-identified data of the study. Review of such secondary analyses by the Institutional Review Boards was waived as per the Ethics Board of the central study site, Seirei Mikatahara General Hospital (Hamamatsu, Japan) (No. 16-22). Informed consent was waived in accordance with local regulations. Patient registration was based on an opt-out model. This study was conducted according to the principles of the Declaration of Helsinki. The reporting of this study conforms to STROBE guidelines. 16
Procedure
The attending palliative care physicians and nurses recorded all data and symptoms on admission using the structured questionnaire. Patients’ clinical characteristics (ie, sex, age, the Eastern Cooperative Oncology Group-Performance Status [ECOG-PS], presence of metastasis, antidepressant use, laboratory data of albumin and CRP), the status of malignant wounds (reddening [redness of the skin], skin defects [wounds which has lost epithelium], necrosis [death of the cells in skin], and fistula [an abnormal connection between skin and another structure]), and specific manifestations of malignant wounds (exudate, bleeding, odor, and pain) were obtained. The frequency of dressing changes was recorded as well. Patients’ pain was assessed using the numerical rating scale (NRS)-pain and oral morphine equivalent daily dose (OMEDD).17,18 NRS-pain is an 11-point numeric scale (ranging from 0 to 10, from no pain to worst possible pain) that is used by patients to evaluate their own pain. 17 Overall survival (OS) was calculated from the day of PCU admission until all-cause mortality or censoring at the last date of confirmed survival.
Primary Outcome
The primary outcome of this study was the prognostic impact of malignant wounds in patients with head and neck cancer. The survival time of patients with head and neck cancer was compared between patients with and without malignant wounds. Subsequently, the utility of the PPI as a prognostic instrument was assessed in those with malignant wounds.
PPI
The PPI was measured as the sum of the scores of performance status (evaluated using the Palliative Performance Score [PPS]), oral intake, and the presence or absence of dyspnea, edema, and delirium. 19 The PPS was categorized as 10-20, 30-50, and 60 or more (partial score values: 4.0, 2.5, and 0). Oral intake was categorized as severely reduced, moderately reduced, or normal (partial score values: 2.5, 1.0, and 0). The following were categorized based on presence or absence: dyspnea at rest (partial score values: 3.5 and 0), edema (partial score values: 1.0 and 0), and delirium (partial score values: 4.0 and 0). The PPI total score ranged from 0 to 15. A PPI score of ≥6 was adopted as the cut-off.
Statistical Analyses
Comparisons between patients with and without malignant wounds were made using Fisher’s exact test or t test as appropriate. OS in patients with and without malignant wounds was estimated using the Kaplan-Meier method, and the survival difference was compared using the log-rank test.
All statistical analyses were performed using SPSS version 22.0 (IBM Corporation, Armonk, NY, USA) and GraphPad Prism version 8.0 (GraphPad Software, San Diego, California, USA). The results were considered statistically significant at a two-sided P-value of <0.05.
Results
Patient Characteristics
Patient Characteristics.

ECOG: Eastern Cooperative Oncology Group; NRS: numerical rating scale; OMEDD: oral morphine equivalent daily dose; PS: performance status.
Among the 68 patients with head and neck cancer, 29 (42.6%) had malignant wounds and 39 (57.4%) did not. There were no significant differences in the characteristics of patients with and without malignant wounds (Table 1).
Characteristics of Malignant Wounds
Conditions and Manifestations Caused by Malignant Wounds.
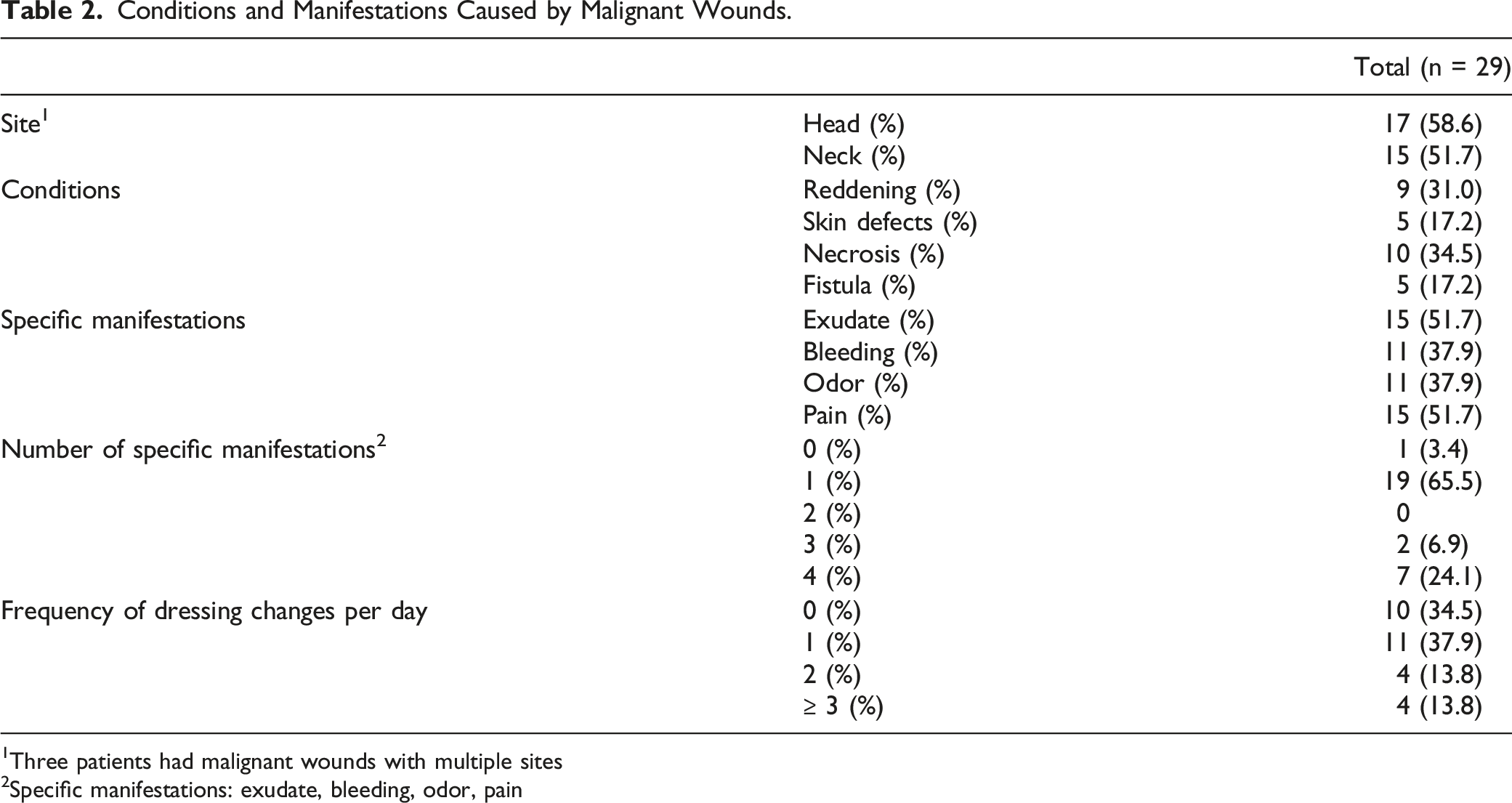
1Three patients had malignant wounds with multiple sites
2Specific manifestations: exudate, bleeding, odor, pain
The frequency of dressing changes was once a day or never in 21 patients (72.4%), and twice or more a day in 8 patients (27.6%) (Table 2). Patients with multiple manifestations were more likely to require dressing changes than those with zero or 1 manifestation (P = 0.009).
Overall Survival
Among patients with head and neck cancer, OS was significantly shorter in patients with malignant wounds than in those without (median OS: 19.0 days vs 32.0 days, respectively, P = 0.046) (Figure 1). The presence of malignant wounds was therefore associated with a worse prognosis. Overall survival of patients with head and neck cancer stratified by the presence or absence of malignant wounds. Overall survival was significantly shorter in patients with malignant wounds than in those without (median overall survival; 19.0 days vs 32.0 days, respectively, P = 0.046).
Among patients without malignant wounds, the OS of patients with a PPI score ≥6.0 was significantly shorter than that of patients with a PPI score <6.0 (median OS: 10.0 days vs 43.0 days, respectively, P = 0.003) (Figure 2). High PPI scores were therefore associated with a poor prognosis in this group. Overall survival of patients with head and neck cancer without malignant wounds, stratified according to the Palliative Prognostic Index score. Overall survival was significantly shorter in patients with a Palliative Prognostic Index score ≥6.0 than in those whose score was <6.0 (median overall survival; 10.0 days vs 43.0 days, respectively, P = 0.003).
In patients with malignant wounds, the OS of patients with a PPI score ≥6.0 was not significantly shorter than that of patients with a PPI score <6.0 (median OS: 14.0 days vs 23.0 days, respectively, P = 0.54) (Figure 3). High PPI scores were therefore not significantly associated with a poor prognosis in this group. Overall survival of patients with head and neck cancer with malignant wounds, stratified according to the Palliative Prognostic Index score. Overall survival was not significantly shorter in patients whose Palliative Prognostic Index score was ≥6.0 than in those whose score was <6.0 (median overall survival; 14.0 days vs 23.0 days, respectively; P = 0.54).
PPI
Palliative Prognostic Index.

Discussion
Among 68 end-of-life patients with advanced head and neck cancer, 29 (42.6%) had malignant wounds. OS was significantly shorter in patients with malignant wounds than in those without. High PPI scores were associated with a poor prognosis in patients without malignant wounds, while they did not significantly predict a poor prognosis in patients with malignant wounds.
The burden of malignant wound manifestations, such as exudate, bleeding, odor, and pain, can negatively affect prognosis. Almost all patients with malignant wounds in this study had these specific manifestations. Previous reports have shown that malignant wounds may be risk factors for bacterial infection or candidemia.20,21 Another report showed that patients with malignant wounds often suffer from poor general conditions, such as anemia and malnutrition. 22 We previously reported that malignant wounds involving the breast cause physical and psychological symptoms and are not associated with poor prognosis. 12 In contrast, malignant wounds involving the head or neck had a poor prognostic impact in this study. Patients with head and neck cancer often develop in the vicinity of the airways and major vessels, which are vital organs, and thus their prognostic impact is likely to be different from that of patients with breast cancer. Malignant wounds affecting the head or neck may also specifically worsen the prognosis by causing fatal hemorrhage involving large blood vessels, asphyxia due to airway narrowing, poor oral intake due to esophageal compression, pneumonia due to aspiration, and infection of the wounds themselves. However, it was not possible to analyze causes of death because these are often simply classified as cancer progression in PCUs in this study. The symptom burden of malignant wounds may also influence the cause of admission to PCUs. Over 50% of the 29 patients with malignant wounds in this study did not have metastases on admission.
Recognizing a poor prognosis is necessary for patients and health care providers, because it helps in determining the treatment plan. Predicting prognosis using a scoring system, as done in this study, has also been performed to determine the appropriateness of palliative sedation therapy. 23 (PPI)s are often used in clinical practice in Japan to predict the prognosis of terminally ill cancer patients. High PPI scores in this study did not significantly predict poor prognosis in patients with malignant wounds. Among such patients, prognosis is probably defined by factors not included in the PPI, such as specific manifestations of malignant wounds (exudate, bleeding, odor, and pain) or conditions caused by such wounds (necrosis, fistula).
This study has several strengths. First, this was a large, prospective, multicenter study. Out of nearly 2000 patients with cancer, we included 68 with head and neck cancer. Additionally, a range of information was predefined and prospectively obtained, including clinical characteristics, the status of malignant wounds, and wound manifestations. Finally, the large amount of data obtained and the fact that the minimal data were missing provide a high degree of generalizability.
This study has several limitations. First, this was a post-hoc analysis of a prospective cohort, and all data were obtained from Japanese patients in PCUs. The present results are not applicable to patients with head and neck cancer other than those in the terminal stage who are currently admitted to PCUs. Second, although there are a variety of head and neck cancer subtypes, information on cancer types (eg, oral, oropharyngeal, and hypopharyngeal) was not collected in this study. Subtypes might influence prognosis and become a confounding factor. Third, although OS was calculated from the day of PCU admission, not from the day of appearance of malignant wounds, admission timing varies between different PCUs and physicians. As such, OS might not have accurately reflected prognosis. In addition, the number of patients with head and neck cancer who had malignant wounds was relatively small, and therefore multivariate analysis could not be performed. The small sample size of this study may affect the reliability and generalizability of the results. The confidence intervals for the results may be wide. However, patients with head and neck cancer who had malignant wounds were selected from nearly 2000 patients. In the absence of such a large study, the selection bias of this study is small and the selection process is desirable in terms of generalizability. Despite these limitations, this study conveys valuable data on the prognosis of patients with head and neck cancer who have malignant wounds.
Conclusions
This study showed that the prognosis of patients with head and neck cancer was worse among those with malignant wounds than among those without. Although it is valuable for health care providers to predict poor prognosis, prognosis is probably defined by factors not included in the PPI. Hence, the PPI may not be useful as a prognostic tool in patients with head and neck cancer who have malignant wounds.
Footnotes
Acknowledgments
We thank all the patients whose data were used in the study. Editage (Cactus Communications) provided editorial support in the form of medical writing, table assembly, collation of author comments, copyediting, fact-checking, and referencing based on the authors’ detailed directions.
Author Contributions
All authors contributed significantly to the paper and approved the final manuscript as submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported in part by a Grant-in-Aid from the Japan Hospice Palliative Care Foundation.
Ethical Statement
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
