Abstract
Background
Handgrip strength (HGS), a simple and practical indicator of skeletal muscle function, has emerged as a potential prognostic marker, yet its predictive value in lung cancer remains to be fully elucidated. This study aimed to evaluate the prognostic significance of HGS for Overall Survival (OS) in patients with lung cancer, and to develop a prognostic model integrating HGS and hematologic parameters.
Methods
This prospective cohort study was based on the Investigation on Nutrition Status and its Clinical Outcome of Common Cancers (INSCOC). A total of 3604 pathologically confirmed lung cancer patients were included. Demographic, clinical, and nutrition-related data were collected. The association between HGS and OS was assessed using Cox proportional hazards models. A nomogram was developed, and its predictive performance was evaluated using receiver operating characteristic (ROC) curves and the concordance index (C-index).
Results
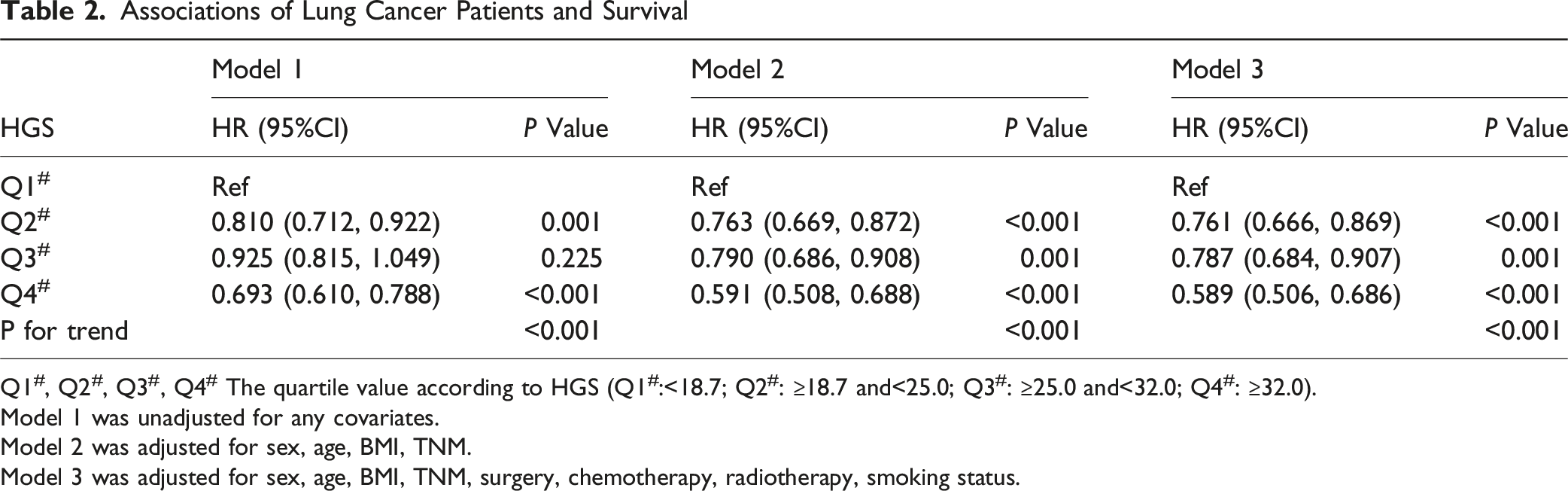
HGS was significantly associated with OS in patients with lung cancer. Compared with the lowest quartile (Q1), patients in Q2, Q3, and Q4 showed 23.9% (HR = 0.761, 95% CI: 0.666-0.869, P < 0.001), 21.3% (HR = 0.787, 95%CI: 0.684-0.907, P = 0.001), and 41.1% (HR = 0.589, 95% CI: 0.506-0.686, P < 0.001) reductions in mortality risk, respectively. In multivariate analysis, sex, HGS, and hemoglobin were identified as independent protective factors, while elevated serum glucose and platelet count were independent risk factors for poor prognosis. The nomogram model constructed based on these variables yielded area under the curve (AUC) values of 64.2%, 61.3%, and 59.8% for predicting 1-, 2-, and 3-year OS, respectively, with a C-index of 0.607 (95% CI: 0.593-0.621), outperforming HGS alone (C-index = 0.541) or any single hematological parameter.
Conclusions
HGS is an independent predictor of survival in lung cancer. Combining HGS with hematologic indicators enhances prognostic accuracy. The developed nomogram may serve as a practical tool for individualized prognostication and nutrition-based interventions.
Plain Language Summary
This study looked at how handgrip strength and routine hematological tests
Highlights
Handgrip strength (HGS) is a robust independent prognostic factor for lung cancer survival. A novel nomogram integrating HGS and hematologic markers effectively predicts OS. Combined assessment of muscle function and nutrition improves prognostic stratification.
Introduction
Despite extensive research and advancements in treatment, lung cancer continues to be one of the most prevalent cancers, with the highest mortality rate worldwide.1,2 Recent data indicated that lung cancer is responsible for approximately 2.09 million new cases and about 1.8 million deaths each year.1,3,4 The impact of lung cancer goes beyond health concerns, imposing significant economic strain on societies. 5 The liver, brain, bone, and kidneys are the most common sites of metastasis from lung cancer, and cases of lung cancer metastasizing to the shoulder have also been reported. 6 While there has been significant progress in understanding lung cancer’s pathogenesis, molecular characteristics, and treatment methodologies, the overall survival (OS) rates remain disappointingly low. 7 Precise prognosis prediction is crucial for determining the appropriate treatment strategies and offering personalized care to lung cancer patients. 8 This highlights the critical need for developing effective prognostic tools. Presently, the prognostic predictors for lung cancer are currently limited, emphasizing the urgent need for the identification and validation of reliable prognostic indicators.
Traditionally, the survival outcomes of lung cancer patients have been assessed using their pathological stage and histological subtype. However, patients with the same stage or subtype often show varying outcomes, highlighting the necessity for more refined prognostic tools. 9 In addition to cost, the inconvenience and significant time investment required are additional drawbacks of these evaluation methods. Cancer-associated cachexia is a multifactorial syndrome characterized by significant weight loss, primarily in skeletal muscle. 10 It is known as a leading cause of death in cancer patients, accounting for up to 20% of cancer-related deaths. Besides, approximately 50%–80% of cancer patients undergo various degrees of this debilitating syndrome. 11 A key feature of cancer-associated cachexia is muscle wasting, which is responsible for low physical performance and functional impairments. 12 Several studies have linked reduced muscle mass to inflammation, 13 immune response, 14 mortality, 15 and complications, 16 demonstrating the deleterious effects of muscle depletion across various cancers.17,18 Handgrip strength (HGS) is a fundamental indicator for assessing muscle function and overall physical capability. 19 Higher HGS has been associated with a range of favorable health outcomes and may enhance the predictive accuracy of outpatient risk scores. Further research is warranted to explore the utility of HGS in risk stratification and screening, in order to determine its potential clinical applications. 20
Previous studies have shown that hematological parameters are closely associated with lung cancer. The systemic immune-inflammation index (SII) has been found to be significantly correlated with lung cancer. 21 Elevated levels of neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), systemic immune-inflammation index (SII), as well as increases in ΔNLR and ΔSII after neoadjuvant immunotherapy, have been significantly associated with poorer OS and event-free survival in patients with non-small cell lung cancer. 22
In this study, we explored the association between HGS and the prognosis of patients with lung cancer. Furthermore, we developed a nomogram model based on HGS and hematologic parameters. We believe that this nomogram can address the limitations of using HGS alone for prognostic assessment and offer a more accurate prediction of patient outcomes.
Method and Materials
Population
We abstracted data from the Investigation on Nutrition Status and its Clinical Outcome of Common Cancers (INSCOC), which was a national multicenter prospective cohort study. Patients in the INSCOC database were enrolled using a consecutive recruitment approach. This study was conducted in accordance with the principles of the Declaration of Helsinki. All participating units and institutions were approved by the Medical Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University [Approval number: Medical Research Ethics Review (2013) 82; Trial registration number: ChiCTR1800020329]. All participants or their legal guardian signed written informed consent before the study. All patient information was de-identified, and no individual could be identified in any way. Patients were included if they satisfied the following inclusion criteria: a) Patients who were pathologically diagnosed with lung cancer at the participating medical centers; b) Age of 18 years or older; c) Absence of other conditions that could potentially cause cachexia or significant weight change, such as HIV infection, pregnancy, organ transplantation or end-stage renal disease, besides lung cancer; d) Have the ability to communicate clearly and complete the study questionnaire; e) Were willing to participate in the study. Patients who met the following criteria were excluded: a) Patients with missing or incomplete clinicopathological data; b) Patients who were lost to follow-up. This work was reported in line with the STROCSS criteria. 23
Data Collection
Patients who met the included criteria were from 29 medical centers. Patient demographic, clinical characteristics, and oncological data were collected by experienced physicians within 48 hours of admission from 29 medical centers. The baseline data encompassed age at diagnosis, gender, weight and height at admission, body mass index (BMI), smoking history
Statistical Analysis
Data were presented as mean ± standard deviation (SD) for normally distributed continuous variables, and as median (interquartile range) for non-normally distributed variables. Categorical variables were expressed as frequency (percentage). Baseline characteristics were compared using Fisher’s exact test or the chi-square test, as appropriate. For continuous variables, the Student’s-t test was used for normally distributed data, while the Mann–Whitney U test was applied for non-normally distributed data. OS probabilities were estimated using the Kaplan–Meier method, and survival differences between groups were assessed with the log-rank test. Both univariate and multivariate survival analyses were performed using Cox proportional hazards regression models to estimate hazard ratios (HRs) and 95% confidence intervals (CIs). Based on the results of multivariate Cox regression, 1-, 2-, and 3-year survival nomograms were constructed. The prognostic performance of the nomogram and conventional lung cancer prognostic factors was evaluated using the area under the receiver operating characteristic curve (AUC) and the concordance index (C-index). The predictive value of the nomogram and HGS-based staging system was further assessed using C-index, AUC, and time-dependent receiver operating characteristic (ROC) curves. All statistical analyses were conducted using R software, version 4.2.0 (R Foundation for Statistical Computing). A two-sided P-value <0.05 was considered statistically significant.
Results
Demographic and Oncological Data of Patients with Lung Cancer
Characteristics of Patients With Lung Cancer Stratified by HGS

Q1#, Q2#, Q3#, Q4# The quartile value according to HGS (Q1#:<18.7; Q2#: ≥18.7 and<25.0; Q3#: ≥25.0 and<32.0; Q4#: ≥32.0).
Association between HGS and OS in lung cancer patients
eFigure 2 showed that HGS was a protective factor for OS in patients with lung cancer. Compared with Q1, Q4 showed a significantly higher OS (Figure 1). Table 2 showed the associations between HGS quartiles and survival outcomes. After adjusting for sex, age, BMI, TNM stage, surgery, chemotherapy, radiotherapy, and smoking status (Model 3), patients in Q2 had a 23.9% lower risk of death compared with those in Q1 (HR = 0.761; 95% CI: 0.666-0.869; P < 0.001). Patients in Q3 showed a 21.3% reduction in mortality risk (HR = 0.787; 95% CI: 0.684-0.907; P = 0.001), and those in Q4 had a 41.1% lower risk of death (HR = 0.589; 95% CI: 0.506-0.686; P < 0.001). In addition, a significant linear trend was observed between HGS and mortality risk (P for trend <0.001), indicating that higher HGS was associated with better survival outcomes. These associations remained consistent across subgroup analyses stratified by sex, tumor stage, age, and presence of metastasis (eTables 1–4). The results remained robust in sensitivity analyses after excluding patients who died within 6 months (eTable 5). KM Curves Showing Associations Between the HGS and OS in Patients With Lung Cancer Associations of Lung Cancer Patients and Survival Q1#, Q2#, Q3#, Q4# The quartile value according to HGS (Q1#:<18.7; Q2#: ≥18.7 and<25.0; Q3#: ≥25.0 and<32.0; Q4#: ≥32.0). Model 1 was unadjusted for any covariates. Model 2 was adjusted for sex, age, BMI, TNM. Model 3 was adjusted for sex, age, BMI, TNM, surgery, chemotherapy, radiotherapy, smoking status.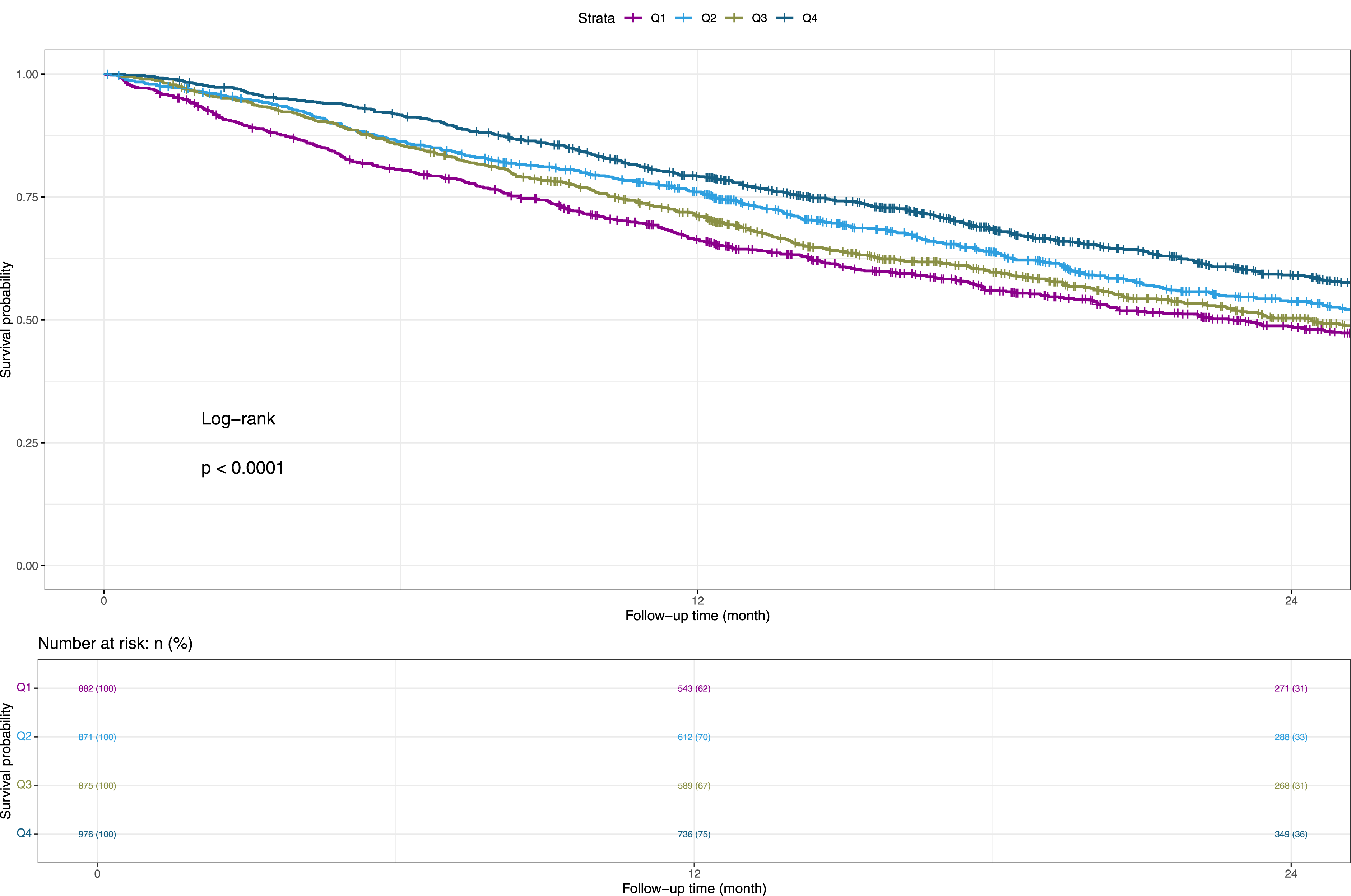
Associations of HGS and hematological indicators with OS in lung cancer patients
eFigure 3 illustrated the correlations between HGS and hematological indicators, with the strongest correlation observed between HGS and hemoglobin (r = 0.25). eTable 6 presented the results of univariate and multivariate Cox regression analyses examining the associations between selected variables and OS in lung cancer patients. In univariate analysis, variables such as sex, age, HGS, total protein, albumin, serum glucose, hemoglobin, and platelet count were significantly associated with survival. In multivariate analysis, sex (HR = 0.62; 95% CI: 0.55-0.69; P < 0.001) and HGS (HR = 0.75; 95% CI: 0.69-0.82; P < 0.001) remained independent protective factors for OS, associated with a 38% and 25% reduction in mortality risk, respectively. Hemoglobin also showed a significant protective effect (HR = 0.90; 95% CI: 0.87-0.92; P < 0.001). In contrast, elevated serum glucose was identified as an independent risk factor for mortality (HR = 1.47; 95% CI: 1.16-1.87; P = 0.001), and platelet count was mildly associated with increased mortality risk (HR = 1.01; 95% CI: 1.00-1.02; P = 0.007).
Construction and evaluation of the nomogram
The nomogram was constructed based on sex, HGS, hemoglobin, serum glucose, and platelet count (Figure 2). It estimated the 1-, 2-, and 3-year OS probabilities for patients with lung cancer. The calibration curves showed good agreement between the predicted and observed survival at 1, 2, and 3 years (eFigure 4). As shown in Figure 3, the nomogram demonstrated superior predictive performance for OS compared to HGS alone. According to the time-dependent ROC curves, the AUCs of the nomogram at 1, 2, and 3 years were 64.2%, 61.3%, and 59.8%, respectively, while those of HGS were 55.8%, 53.1%, and 53.1% (eFigure 5). eTable 7 presented the C-indices of the nomogram and various hematological indicators for predicting prognosis in lung cancer patients. The results showed that the nomogram had the highest discriminative ability, with a C-index of 0.607 (95% CI: 0.593-0.621), which was clearly superior to that of HGS or any other individual hematological marker. A Proposed Nomogram for Predicting Survival Time and Survival Probability in Patients With Lung Cancer AUCs of the Nomogram and the HGS in Patients With Lung Cancer. AUC, Area Under the ROC Curve; ROC, Receiver Operating Characteristic

Discussion
This study, based on a large multicenter prospective cohort in China, systematically evaluated the association between HGS and prognosis in patients with lung cancer. HGS, as a key indicator of muscle function, was found to be significantly and positively associated with OS. Patients with higher HGS had a significantly lower risk of mortality, suggesting that muscle function status had independent prognostic value in lung cancer. A major innovation of this study was the first-time integration of HGS with hematological indicators to construct a combined nomogram model for predicting long-term survival outcomes in lung cancer patients. Although previous studies had separately demonstrated associations between HGS or certain hematological parameters and lung cancer prognosis, few had combined the two to systematically assess their integrated predictive power. The findings showed that the nomogram incorporating HGS, sex, hemoglobin, serum glucose, and platelet count had significantly better prognostic discrimination than HGS or any individual hematological marker alone. Specifically, the model achieved AUCs of 64.2%, 61.3%, and 59.8% for predicting 1-, 2-, and 3-year OS, respectively, whereas the corresponding AUCs for HGS alone were only 55.8%, 53.1%, and 53.1%. Moreover, the nomogram yielded a C-index of 0.607 (95% CI: 0.593-0.621), which was markedly higher than that of HGS alone (C-index = 0.541) or other conventional hematological indicators.
Cancer cachexia is a severe lethal wasting syndrome, notably affecting up to 50%-80% of cancer cases and account for up to 20% of cancer deaths.25,26 This syndrome is not just a symptom, but a complex metabolic disorder characterized by loss of skeletal muscle mass, with or without fat loss. Such loss leads to a decline in overall physical function, severely undermining the quality of life and treatment effectiveness for cancer patients. 27 Sarcopenia is a factor influencing the outcomes of cancer immunotherapy. 28 The relationship between cancer cachexia and lung cancer prognosis has been extensively studied, revealing a substantial impact on patient outcomes.29,30 However, despite its significance, there is a lack of a standardized cachexia parameter for the prediction of lung cancer prognosis. This gap in clinical assessment tools presents a major challenge in the effective management and treatment of lung cancer patients. 31 BMI has been widely used to assess nutritional status of patients and has shown a significant correlation with tumor prognosis. 32 However, the rise in the number of cancer patients with sarcopenic obesity—a condition where obesity coexists with reduced muscle mass—casts doubt on the effectiveness of BMI and other traditional cachexia parameters as reliable indicators. 33 Furthermore, the accuracy of BMI as an assessment tool is compromised by various confounders. For example, dystrophic edema can artificially increase weight without reflecting true nutritional status. Malignancy-associated pleural effusion can alter body weight and composition, complicating the assessment of patients’ nutritional and health status. Increases in visceral adiposity, the accumulation of fat around the internal organs in the abdominal cavity, can skew BMI interpretations and mask muscle loss and sarcopenia. All these factors challenge the conventional approaches to evaluating the nutritional and health status of cancer patients, underlining the need for more sophisticated and nuanced assessment tools.
HGS offers a quick and non-invasive method to evaluate muscular strength, which is a key component of physical health and an important factor in the quality of life, especially for patients with cancer. Cheng - Le Zhuang et al. found that low HGS was associated with lung cancer in men. 34 Compared with absolute HGS, height - adjusted HGS has a better predictive effect, especially in the context of lung cancer. 35 A study by Peixin Dong et al. demonstrated that elevated hemoglobin levels were linked to improved survival outcomes in patients with lung cancer. 36 Wahyu Wulaningsih et al. found a positive association between blood glucose levels and overall cancer risk. 37 Yuan Yuan et al. found that elevated pretreatment platelet counts were associated with poorer OS compared to lung cancer patients with normal platelet counts. 38
The American Society of Clinical Oncology (ASCO) guidelines highlight the uncertain outcomes of pharmacological interventions aimed at treating cancer cachexia. Nonetheless, dietary strategies such as nutritional counseling, increasing protein consumption, and supplementing diets with omega-3 (n-3) fatty acids are advisable and beneficial for patients.39,40 In addition, combined muscle-strengthening and aerobic activities were associated with reduced risk and mortality rate. 41 Recent foundational studies are actively investigating new potential therapeutic targets to combat muscle wasting in lung cancer patients. For example, the LCN2 secreted by tissue-infiltrating neutrophils was a potential target in the treatment of cancer cachexia. 42 Furthermore, antagonists that target RAGE (Receptor for Advanced Glycation End-products) are emerging as a promising therapeutic approach to prevent or alleviate the symptoms associated with cachexia syndrome. 43 At present, these studies are predominantly in the preclinical phase, emphasizing the necessity for more extensive research. Looking ahead, the execution of clinical randomized controlled trials (RCTs) is essential to thoroughly assess and confirm the therapeutic efficacy of these potential treatments.
The integrated assessment of muscle function and systemic nutritional-inflammatory status provided a more comprehensive reflection of patients’ overall physiological reserve and tumor-related stress response. This approach helped identify high-risk individuals more accurately and offered both a theoretical basis and practical tool for precision therapy and nutritional intervention. The construction of this model not only improved the accuracy of prognostic prediction in lung cancer patients but also offered a feasible pathway for developing future individualized and integrated management strategies, with strong potential for clinical application and research relevance. Our study, while comprehensive, had several limitations that should be acknowledged. Firstly, the exclusive inclusion of Chinese individuals in our cohort may limit the generalizability of our findings to other ethnic populations. This limitation is important, as it restricts the applicability of our conclusions in broader, more diverse contexts. Secondly, as this was a prospective study, potential biases in study design and data collection may exist. Furthermore, grip strength measurement might have been affected by comorbidities such as neuromuscular diseases and arthritis. These factors, however, were not completely excluded in the inclusion and exclusion criteria. Future research should involve more refined analyses and external validation in populations of different ethnic backgrounds to clarify the comparative prognostic value of these indicators.
Conclusion
This study, based on a large national multicenter prospective cohort, systematically assessed the relationship between HGS and survival in patients with lung cancer. It identified HGS as not only an independent protective factor for OS but also closely linked to various nutritional and inflammatory status indicators. On this basis, we innovatively integrated HGS with hematological markers to construct a simple and practical nomogram model, which significantly improved the accuracy and clinical utility of prognostic assessment. These findings highlight the importance of a comprehensive evaluation of muscle function and systemic nutritional-inflammatory status in the clinical management of lung cancer, offering valuable guidance for individualized treatment and supportive interventions. This approach provides a reliable tool for advancing precision medicine in lung cancer.
Supplemental Material
Supplemental Material - Prognostic Value of Handgrip Strength in Lung Cancer: Findings From a Multicenter Prospective Cohort Study
Supplemental Material for Prognostic Value of Handgrip Strength in Lung Cancer: Findings From a Multicenter Prospective Cohort Study by Xin Wang, Hong Zhao, Lu Liang, Ying Gao, Lei Song, Hanping Shi in Cancer Control
Supplemental Material
Supplemental Material - Prognostic Value of Handgrip Strength in Lung Cancer: Findings From a Multicenter Prospective Cohort Study
Supplemental Material for Prognostic Value of Handgrip Strength in Lung Cancer: Findings From a Multicenter Prospective Cohort Study by Xin Wang, Hong Zhao, Lu Liang, Ying Gao, Lei Song, Hanping Shi in Cancer Control
Footnotes
Acknowledgement
We sincerely thank the INSCOC project members for their substantial work on data collection and patient follow-up.
Ethical Considerations
This study was conducted in accordance with the principles of the Declaration of Helsinki. All participating units and institutions were approved by the Medical Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University [Approval number: Medical Research Ethics Review (2013) 82; Trial registration number: ChiCTR1800020329]. All patients signed a written informed consent form.
Author Contributions
X.W. and H.Z. wrote the main manuscript text. H.Z. and L.L. developed the software. X.W., Y.G. and H.S. curated the data. X.W., L.S. and H.S. conducted the formal analysis. X.W. acquired the funding. X.W., H.Z., and L.L. conceptualized and developed the methodology. L.S., Y.G. and H.S. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Beijing Hospitals Authority Youth Programme. Grant number QMS20220726.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
Disclaimer
The statements in this article are solely the responsibility of the authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
