Abstract
Patients with advanced cancers and their oncologists are often faced with difficult treatment decisions, especially when there are borderline situations of expected benefit or increased risk of complications. In this narrative review, we will explore the decision-making process for patients with advanced cancers and provide insights on how to approach this complex task, while didactically dividing the oncologist’s assessments according to a mnemonic rule of the ABCDE of therapeutic decision-making. Part A (advanced cancer) recalls that the rule is to be used specifically for advanced cancers. Parts B (potential benefits) and C (clinical conditions and risks) represents the traditional risk vs benefit scale. In Part D, we discuss ways to identify and understand patients’ desires, values, preferences, and beliefs. The prognostic estimation, from Part E, may function as an “adjust” for the antineoplastic treatment decision-making. Treatment decisions need to be conducted by skilled oncologists, in a patient-centered care, aiming to promote valuable oncology with lower rates of aggressive care.
Introduction
Clinical oncology has come a long way in recent decades, moving from a phase focused on cytotoxic antineoplastics to the era of personalized medicine, which includes numerous new molecularly targeted drugs, such as a focus on several driver mutations and signaling pathways (such as BRAF, HER2, ALK, PI3K, PTEN, FGF). 1 Other examples are the effective use of ADP ribose polymerase inhibitors (PARPi) for the treatment of ovarian and breast BRCA-mutated cancers 2 and the new antibody-drug conjugate drugs, 3 such as Trastuzumab Entamsine, Sacituzumab Govitecan, and Trastuzumab deruxtecan, for the treatment of breast cancer. In addition, the use of immune checkpoint inhibitors is revolutionizing the treatment of several types of malignant neoplasms and keeps moving forward. 4 Despite enormous therapeutic advances, most treatments are still offered to prolong and facilitate patient survival, hence called palliative antineoplastic treatment.
In this context of multiple systemic therapeutic options, the oncologist, who has an optimistic nature and often personal difficulty in dealing with situations of finitude, finds himself in the difficult situation of suspending an antineoplastic treatment. The “availability bias,” 5 a mental shortcut that relies on immediate examples that come to mind, for example with the memory of anecdotal cases of significant clinical improvement with chemotherapy, probably interferes in the medical decision to try a new cancer treatment. This situation is often stimulated by the patient’s inclination to want to be treated and to seek a new source of hope. The communication of the absence of new systemic antineoplastic treatment is often complex, demanding a lot from the oncologist, who may not have had adequate training for this task. 6
Chemotherapy in the last days of life may cause harm by decreasing quality of life and raising costs, and it does not improve survival times.7-9 As a result, death within 14 and 30 days of chemotherapy has been used as a quality indicator for cancer care.10,11 Although not a well-established definition, overly aggressive care is associated with more than 10% of patients who have received chemotherapy in the past 14 days. 12 When a patient receives chemotherapy treatment at the end of life, it suggests that the oncology service has failed to adequately integrate the philosophy of palliative care into cancer care.
There has been a significant advance in oncology support strategies during antineoplastic treatment and after it has ceased. Several clinical trials have shown the benefit of early inclusion of palliative care concomitant to oncologic treatment.13-17 There are improved health-related quality of life indices, reduced symptoms of depression, improved symptom control, and greater satisfaction with care by patients and caregivers, as well as reduced costs and less aggressive end-of-life care.18,19 Thus, when integrated early into a service with well-structured palliative care and adequately trained staff, it is possible for patients to receive more time in oncology treatment, including more intensity of treatment, without significant impact on their qualities of life. It is important to note that with the advance of new cancer therapies, for example, with the advent of oral therapies that can be used for a prolonged time and with high efficacy, active and palliative care have blurred boundaries.
In this article, we will didactically divide the oncologist’s assessments according to a mnemonic rule of the ABCDE of therapeutic decision-making (Figure 1). Conceptual flowchart of the suggested therapeutic decision-making process. A: Advanced cancer. The first step in applying these concepts is to identify whether the disease in question is indeed an advanced and incurable cancer. B: potential benefits. Discuss and share with the patient the potential benefits of the antineoplastic treatment. C: clinical conditions and risks. Discuss and share with the patient the clinical conditions (functionality, comorbidities) and the potential risks of the antineoplastic treatment. D: desire, values, preferences, and beliefs. Identify and understand patients’ desires, values, preferences, and beliefs. E: prognostic estimation. The prognostic estimation may function as an “adjust” for the antineoplastic treatment decision-making.
A:
B:
C:
D:
E: Prognostic
What Is the Disease? (A: Advanced Cancer)
The first step in applying these concepts is to identify whether the disease in question is indeed an advanced and incurable cancer. Throughout this article, we will consider as advanced disease that which is locally recurrent or with distant metastases, not amenable to surgical resection. Thus, the mnemonic rule below was designed to be used where the expected clinical outcomes of treatment are symptom control, improved quality of life, and/or improved survival times. In early disease cancers, the primary focus is on increasing cure rates and survival times; thus, often with “acceptable” increases in treatment toxicity. In incurable disease, the focus is ultimately on promoting quality of life and, if possible, increasing lifespan.
What Are the Potential Clinical Benefits of This Treatment? (B: Potential Benefits)
This is perhaps the most important step to consider during the process of deciding whether or not to treat a patient with an advanced cancer. The medical literature is extensive regarding new anticancer therapies. However, most clinical trials have not included real-life patients, but rather patients in good clinical condition and not fully representative of the general patient population. 20 When a patient perfectly fits the eligibility criteria of a previous positive clinical trial, the decision about treatment is often easy since the potential benefit can be generalized and explained to the patient during the shared decision process. The difficulty, which often translates into the art of oncology, is treatment decisions in situations outside the contexts of clinical trials, for example, in patients with comorbidities or inadequate functional performance.
Currently, there are tools developed to help measure the potential benefit of a treatment, such as the European Society for Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS) 21 and the American Society of Clinical Oncology (ASCO) Value Framework—Advanced Disease. 22 In the ASCO Value Framework—Advanced Disease, for instance, 4 sections are evaluated: Clinical Benefit; Toxicity; Bonus Points; and Net Health Benefit. In the Clinical Benefit section, overall survival (OS), progression-free survival (PFS), and response rate (RR) are measured with distinct weights; the authors suggest multiplying OS, PFS and RR benefits by 16, 11, and 8, respectively. Interestingly, in the Bonus Point section, symptom palliation and treatment-free intervals are measured. These tools were developed considering the vast universe of new cancer therapies, with both patients’ and healthcare systems’ economic impact. Although the tools cited are extremely useful in defining therapeutic protocols, they are probably of little use in real-life shared decision-making processes.
Palliative antineoplastic treatments can vary significantly depending on the economic region of the world in which a patient lives, and the type of access to public or private healthcare services. Factors such as the availability of specific drug protocols, medical expertise, and funding can impact the type and quality of care provided. 23 In some countries or regions, access to palliative antineoplastic therapies may be limited, while in others, patients may have a wider range of treatment options available to them. 24 In addition, racial and ethnic minorities are underrepresented in oncology clinical trials, potentially reducing the generalizability of findings. 25 These differences highlight the importance of considering individual circumstances when determining the most appropriate treatment plan for patients with advanced cancer.
In daily practice, what the clinical oncologist needs is to know the details of clinical trials that guide his/her patients’ treatment indications and know how to conduct shared decision-making using good communication skills. In addition to defining the potential benefit of treatment based on the literature using tools that measure value in health care, it is of utmost importance to identify the value of treatment from the patient’s perspective. 26
What Are the Clinical Conditions and Potential Risks of the Treatment in Question? (C: Clinical Conditions and Risks)
Weighting the risks and benefits of prescribing an antineoplastic treatment for older adults, with comorbidities, or borderline performance status is challenging for the oncologist.
Previous studies in the general population, have suggested an association between measures of physical performance and all-cause mortality.27,28 As in the general population, physical functioning is associated with the prognosis of patients with advanced cancer.29-31 One of the traditional ways of evaluating the functionality of a cancer patient is through the measurement of his functional performance by means of the Karnofsky Performance Status 32 and the Eastern Cooperative Oncology Group Performance Status (ECOG-PS). 33
Most clinical trials have included patients in better clinical condition, with ECOG-PS 0 or 1 and, in less than 10% of the time, ECOG-PS of 230. In the meta-analysis by Cheng et al, 34 only one clinical trial out of 62 evaluated (<2%), included patients with an ECOG-PS of 3. Thus, day-to-day therapeutic decisions in patients with poor functional conditions (ECOG-PS 2 or 3) are mostly made outside of clinical trial best evidence. While prognosis is clearly associated with patient performance status (PS), the benefit of cancer treatment may not be related. The relative benefit of treatment appears to have no interaction with performance status, both in studies that included older treatments regimens 30 and studies with targeted drugs included. 34 In contrast, non-small cell lung cancer patients with impaired PS status were, on average, twice less likely to achieve a clinical response when exposed to immune checkpoint inhibitors when compared with ECOG-PS ≤1 population. 35
The association of antineoplastic treatment with the risk of adverse events in the different PS subgroups needs to be further explored in new studies. Severe nausea and vomiting, for instance, were much more pronounced in ECOG-PS 2 patients than in ECOG-PS 0 to 1 patients. 30 In addition, it is important to remember that cancer patients may have their PS influenced by the cancer itself, but also as a function of some comorbidity. Such information can be important for the oncologist while defining the treatment. For example, in patients with cancer symptoms and reduced PS, antineoplastic treatment may be indicated in an attempt to therapeutic response, improvement of symptoms and PS; however, patients with poor PS due to comorbidity may even worsen the PS by oncologic treatment.
Another variable that interferes in the decision about cancer treatment is advanced age and the consequent vulnerability of the patient. In patients aged 65 and older, receiving antineoplastic treatments, geriatric assessment (GA) should be used to identify vulnerabilities or geriatric impairments. The GA comprises the evaluation of functional status, falls, comorbidities, depression, social activity/support, nutritional status, and cognition. 36 Elderly cancer patients are particularly prone to “over-treatment,” with a high likelihood of complications/toxicity, or “undertreatment,” that is, not receiving desirable treatment that they would be fit for, for fear (on the part of the oncologist or the patient themselves) of complications.37,38 Furthermore, traditional oncology performance measurement tools, such as KPS or ECOG-PS, are not accurate in predicting risk of complications in elderly patients. 39
Validated Geriatric Assessment Tools.
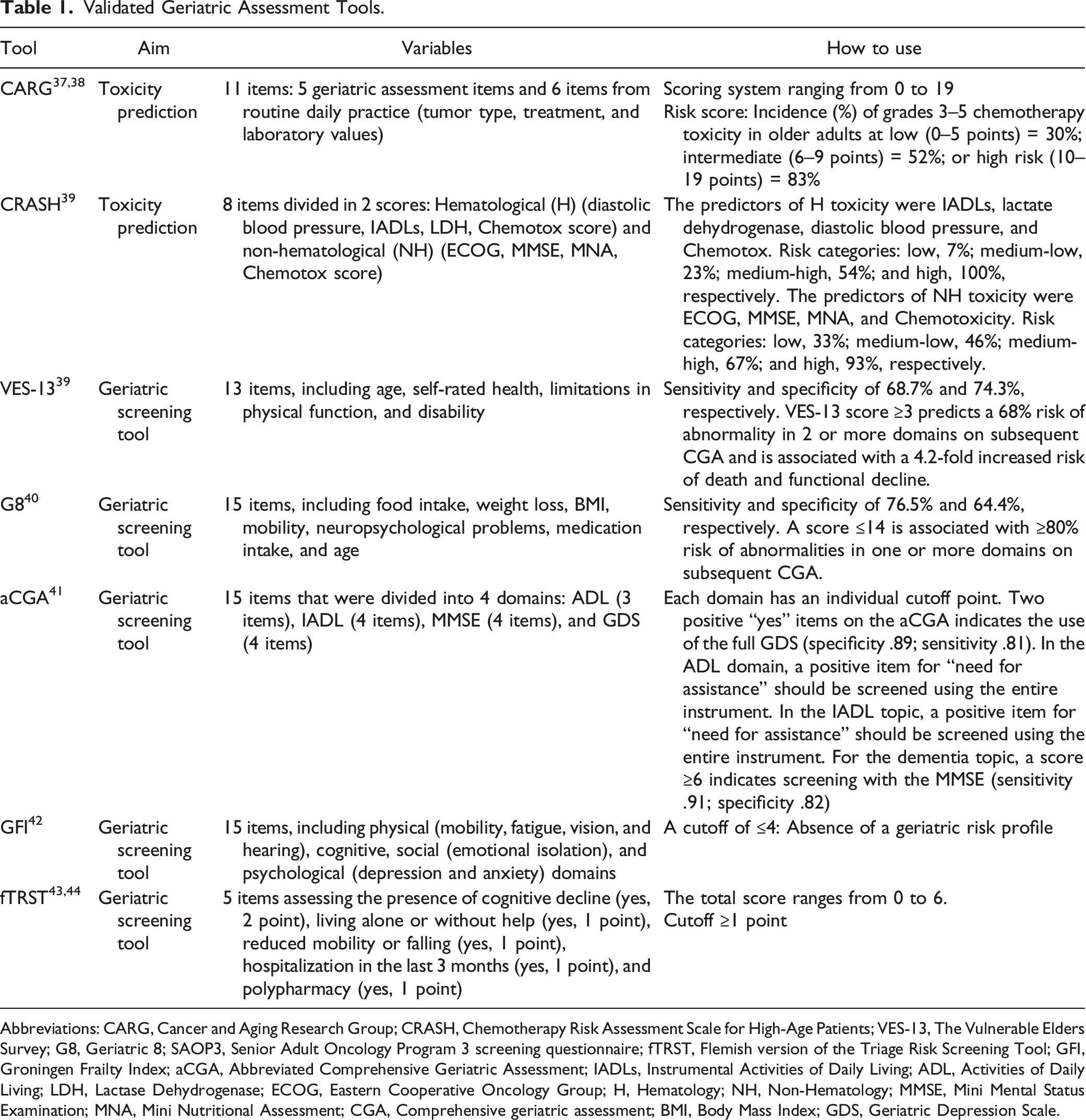
Abbreviations: CARG, Cancer and Aging Research Group; CRASH, Chemotherapy Risk Assessment Scale for High-Age Patients; VES-13, The Vulnerable Elders Survey; G8, Geriatric 8; SAOP3, Senior Adult Oncology Program 3 screening questionnaire; fTRST, Flemish version of the Triage Risk Screening Tool; GFI, Groningen Frailty Index; aCGA, Abbreviated Comprehensive Geriatric Assessment; IADLs, Instrumental Activities of Daily Living; ADL, Activities of Daily Living; LDH, Lactase Dehydrogenase; ECOG, Eastern Cooperative Oncology Group; H, Hematology; NH, Non-Hematology; MMSE, Mini Mental Status Examination; MNA, Mini Nutritional Assessment; CGA, Comprehensive geriatric assessment; BMI, Body Mass Index; GDS, Geriatric Depression Scale.
The Geriatric 8 (G8), 42 the Vulnerable Elders Survey-13 (VES-13), 43 the abbreviated Comprehensive Geriatric Assessment (aCGA), 44 the Groningen Frailty Index (GFI), 45 and the Flemish version of the Triage Risk Screening Tool (fTRST)46,47 are among the screening tools that have been associated with adverse outcomes. These tools should be used to identify frail and/or vulnerable patients who are most likely to benefit from a geriatric assessment. 48 The G8 and the VES-13 are the screening tools with the most scientific evidence for clinical use 49 (Table 1).
Classic Comorbidity Assessment Tools and its Weighed Cancer-Specific Versions.

Abbreviations: CCI, Charlson Comorbidity Index; NCI, National Cancer Institute Combined Index; ECI, Elixhauser comorbidity index.
Considering that a comorbidity measure developed or adapted for a specific disease population is likely to perform better than a generic score, 57 the CCI has been further reviewed and adapted for use in oncology populations. Of note, the original CCI includes as comorbidities some hematological neoplasms and metastatic cancer. Thus, in 2000, a cancer-specific National Cancer Institute Comorbidity Index (NCI-CI) was developed by using SEER derived breast and prostate cancer cases cohorts, excluding solid tumors, leukemias, and lymphomas as comorbid conditions. 58 Subsequently, the NCI comorbidity score evolved into the NCI Combined Index Models, which outperformed the original CCI in predicting 2-year survival in cancer patients. 59
Another tool for assessing the impact of comorbidities is the Elixhauser comorbidity index (ECI), which includes a total of 30 comorbidities. 60 Originally, each comorbidity has equal weight; modifications of the ECI with different weights for each comorbidity have already been described.61,62 A previous systematic review 63 found that the ECI performs better than the CCI in predicting mortality beyond 30 days. It has already been adapted for use in cancer patients, 64 achieving higher C-Index values when compared to the NCI Combined Index Model.
In patients with lung cancer, the Scottish Comorbidity Scoring System (SCSS), 65 which classifies 9 commodities according to their severity (nil: 0; low: 1–6; severe: 7 or more), proved more useful than the CCI in predicting overall survival in patients with localized (stages I–IIIA) and advanced disease (stages IIIB and IV).
As mentioned earlier, the most commonly used way to assess comorbidities in studies is through the CCI or one of its variations. However, there is no gold standard instrument for this. 66 As a practical suggestion, there are easy-to-use Excel applications that can be obtained for free and used to calculate comorbidity scores.67,68 Despite the availability of the tools mentioned, in practice, the oncologist generally evaluates comorbidities in an individualized manner within a typical anamnesis. When planning systemic antineoplastic therapy, hepatic, renal, cardiovascular, and hematologic functions need to be carefully considered. In addition, uncontrolled active infections should be ruled out before the initiation of antineoplastic therapies since most of treatments can course with a drop in WBC and neutrophil levels and changes in the immune system.
Although not well defined, the decision to treat with antineoplastics can differ significantly among various medical oncology subspecialties, particularly those that specialize in specific tumor types, such as breast, gynecologic, lung, or colorectal vs those that treat a wide range of tumors in general oncology. Interdisciplinary communication and collaboration among all involved disciplines, including palliative care, general practitioners, and geriatric oncology, are crucial to ensuring that patients receive the most appropriate care for their unique circumstances.69–71
Patients with hematological malignancies carry a significant burden of illness, often receiving more aggressive end-of-life treatments and experiencing a high level of symptom distress. Despite this, they tend to underutilize palliative care, especially those with leukemia.72,73 It has been reported that hemato-oncologists are more inclined than oncologists to prescribe systemic therapies that carry moderate toxicity and do not confer any survival benefits to patients who present low performance status and an expected survival of one month. 74
Physicians without specialized knowledge in palliative medicine tend to make more aggressive decisions regarding end-of-life care, such as administering higher levels of intravenous hydration, nasogastric tube insertion, and blood transfusions. 75 Additionally, a previous systematic review found that younger and less experienced oncologists were more likely to continue palliative antineoplastic treatment. 6
In the decision to treat with antineoplastics, and particularly in the choice of the type of antineoplastic, the oncologist needs to measure the possible adverse events of the treatment, even if not severe enough to be life-threatening, but that may interfere with the quality of life of the patients. For example, a professional pianist with peripheral neuropathy may have to stop work. Similarly, patients who need their body image intact to work (such as models, TV journalists) will strongly wish to avoid alopecia. Thus, just as treatment needs to be customized considering the molecular characteristics of the tumor, it is also necessary to customize treatment considering the treatment’s potential interference with the patient’s personal and work life.
D: How can we identify the Patients’ Wishes, Preferences, Beliefs, and Values That May Influence the Choice of Treatment? (D: Desires, Values, Preferences, and Beliefs)
In our mnemonic model of therapeutic decision, after evaluating parts B and C, which basically defines a decision scale (risks vs benefits), we move to the next phase that uses the patient’s perception of the treatment, taking into consideration their personal wishes, values, and beliefs; part D is broad and interferes with the discussion on parts B and C (Figure 1). Here it is important to discuss ways of extracting such information from the patient. The question “do you want to try a new treatment?” is generally inappropriate, as most patients wish to live and be cured and will answer the question in the affirmative.
Patients with cancer must cope with a variety of stressors, including the need for treatment on an urgent basis, the management of adverse effects of treatment, the sharing decision-making processes, the numerous physical and emotional challenges while dealing with a life-threatening illness, and having to tolerate uncertainty regarding their future.76,77 Thus, one cannot expect patients to be consciously positive if they don’t have a space to meet and talk about their care preferences and express their distress, or without providing them the necessary time to adapt to the cancer experience and enhance their repertoire of coping strategies, 77 a gap that can be reduced with the help of an Advance Care Planning (ACP).
ACP is a process of planning for future health and personal care that helps patients make decisions and identify their values and preferences for care at a time when they are unable to make or communicate their decisions. This type of planning is intended to align the care the patient will receive with their preferences and priorities. 78 ACP develops in an interactive and communicative process where patients, healthcare staff, and family members have conversations about preferences for care, particularly those at the end-of-life. These preferences may even be recorded in a legal document, called advance directives.79,80
Advance care planning is a crucial aspect of cancer care, especially when making decisions about new anticancer treatments. A previous ACP can help patients and their families make informed decisions about treatment options, considering the potential benefits and harms of each intervention, as well as the patient’s overall health status and prognosis. However, patients with advanced cancer often have not previously participated in ACP discussions. In such situations, serious illness goals of care conversations need to be conducted by trained oncologists. While ACP focuses on preparing the patient for future healthcare decisions, goals of care discussions focus on current healthcare decision-making.
In order to help patients with advanced cancer articulate their goals of care, oncologists need to assess patients’ knowledge of their disease and prognosis. Often, patients with advanced cancers undergoing chemotherapy report an inaccurate perception of curability.81,82 Prognostication is a dynamic and multi-step process encompassing the sequence of prognostic prediction, prognostic disclosure, prognostic awareness, prognostic acceptance, and prognostic-based decision-making. 83 In fact, when patients are aware of their disease stage and prognosis and, preferably, emotionally accept the situation, the clinical decisions to treat or not to treat them are facilitated by sharing decisions in a mature way and with potentially less distress.
Below are some examples of open, in-depth questions, oncologists may use to identify and understand the patient’s wishes, preferences, beliefs, and values84–86:
Identifying Patient Values
“What does ‘living well’ mean to you? What are your goals right now?” “What are your biggest fears and worries about the future with your health?” “What gives you strength as you think about the future with your illness?” “What abilities are so critical to your life that you can’t imagine living without them?” “How much does your family know about your priorities and wishes?”
Exploring Disease Understanding and Treatment Goals
“What are your most important goals if your health situation worsens?” “What do you understand about the current status of your illness?” “What is your understanding now of where you are with your illness?” “How much information about what is likely to be ahead with your illness would you like from me?”
Identifying Future Care and Treatment Preferences
“If you become sicker, how much are you willing to go through for the possibility of gaining more time?” “Tell me a little bit about your main future wishes” “Would you like to actively participate in your advance care planning?” “When you no longer have the ability to make decisions, what care or treatment would you not want in your end-of-life process?”
To understand the profile of patient care preferences and to serve as a stimulus for further discussion about cancer treatment, we have been using in practice the following trade-off item from a previous publication 81 : “If you could choose only one of the options, would you prefer... (A): a treatment that was designed to prolong life as much as possible, even if the treatment caused more pain and discomfort; or (B): a care plan to relieve pain and discomfort as much as possible, even if it meant not living as long.” Other ways of assessing health care preference in a similar way, particularly in the elderly with cancer, have been described.87,88
To take into consideration the wishes and values of patients is to respect their autonomy, and consequently, their dignity.89,90 The conduction of dignity-promoting activities in the process of illness and end-of-life of cancer patients brings to the family members the possibility of discussions about the resolution of pending issues, whether affective, financial, and even bureaucratic (insurance, contracts, inventories), deliberation of care for dependents, better development of resources for coping with the imminent loss of loved ones, and elaboration of the bereavement process. 91
The practical use of tools that allow the health team to identify the wishes of patients during the dying process and subsequent development of conducts that promote their dignity, is a promising strategy to improve the care of cancer patients. A potentially useful strategy to facilitate end-of-life conversations is the Go Wish Cards Game (GWCG), composed of 36 cards, which display a variety of end-of-life wish preferences. The cards are separated into 3 piles considering the ten not so important, the ten more or less important, and the ten very important wishes; the most important wishes are further discussed with the patients. The GWCG method is one of the ways of optimization in dignity therapy for patients in advanced disease process. The use of the cards brings important ways to initiate dialogues and broaden engagements between patient, family members, and multidisciplinary team, through simple vocabulary and variety of possible wishes that extend to the essence of well-being of the human being.92,93 A study carried out with patients with advanced cancer compared a random wish list developed by the patients and the GWCG and found that 62% of the patients preferred the GWCG for its ease of understanding and for presenting a wider range of wish options. Additionally, it was easy to apply. 94
In general, the oncologist needs to be open and trained to identify patients’ wishes, values, beliefs, and preferences. The introduction of processes into routine care (such as ACP and goals of care discussions) and the use of practical tools such as the GWCG can be extremely helpful to the oncologist in the therapeutic decision process. It is important to note that the treatment decision does not necessarily have to occur in a single moment but is often the result of sequential medical consultations.
E - What Is the Patient’s Estimated Lifetime? (E: Prognosis Estimation)
After evaluating the potential benefits (part B), the clinical conditions/risks of the treatment (part C), and the importance of a new therapeutic attempt in the patient’s view (part D), it is essential to “adjust” the clinical decision considering the patient’s life expectancy (part E) (Figure 1). As mentioned earlier, the parts are divided here from a didactic point of view. Thus, prognostication is intrinsically related to part D and can be omitted from the process. We suggest part D as an adjustment in the shared decision, particularly in situations where treatment decisions are incongruent from the oncologist’s point of view. For example, patients where it is proposed to treat with new chemotherapy, but the estimated prognosis is only a few months.
The prognosis estimation is not able to independently establish the indication of a new treatment, however it can assist in the decision making. Furthermore, expectations are not usually communicated to patients routinely, but only in specific situations. The prognosis is somewhat dynamic and can be modified by the treatment itself. 95 We strongly suggest that its communication, when provided to the patient, should be in the form of intervals and never with specific time values or mean/medians, such as “hours to days,” “days to weeks,” “weeks to months,” “months to years.”
Clinical Prediction of Survival
Among the strategies for estimating the prognosis of an advanced cancer patient, one of the most classic and instituted ways is the clinical prediction of survival (CPS), in which the opinion of the health professional, mainly the physician, is considered. It is often overestimated, due to excessive optimism in predicting the survival, particularly those patients who die early in the terminal phase and could benefit from earlier palliative care. 96
The low concordance between clinical predictions and actual survival times is evidenced in systematic reviews of the literature, with 83% of prediction errors being overestimated by physicians 97 and an estimate of 4 weeks beyond observed in 27% of cases. 98 The accuracy of physicians in estimating the prognosis of advanced cancer patients in survival intervals, for example, varies around 60% and the professional’s experience does not seem to interfere on the accuracy of prediction. 99 Even palliative care specialists are unable to predict survival in 32% of cases if a patient is alive or dead at 4 weeks. 100
Functionality Scales
The functionality assessment is another way to predict prognosis, using functional performance scales, such as the Eastern Cooperative Oncology Group Performance Status (ECOG-PS), 33 the Karnofsky Performance Status (KPS) 32 and the Palliative Performance Scale (PPS). 101 These instruments have been widely used as prognostic tools, with an important role in the scenario of survival estimates, through the interpretation of a patient’s dependence on the environment in which they live, measuring their ability to perform activities of daily living, without assistance.
Symptoms of Advanced Disease and Laboratory Parameters
With the awareness of the potential limitations of functional performance scales (such as, low inter-observer reproducibility and cross-sectional evaluation), the assessment of clinical symptoms in combination with KPS and physicians’ clinical estimates may improve survival prediction. Symptoms such as anorexia, cachexia, weight loss, dysphagia, and pain may be independently associated with a worse prognosis in advanced cancer patients. In exclusive palliative care setting, the mental confusion, fatigue, cachexia, weight loss, dyspnea, dysphagia and nausea predict a shorter survival. 102 It is important to point out that the clinical assessment of symptoms is also subjective and often influenced by psychosocial factors, which leads to some inaccuracy in its measurement, even with the use of visual scales.103,104
Laboratory parameters are possible prognostic markers in patients with advanced-stage cancer. The presence of leukocytosis, as well as lymphopenia, is associated with a worse prognosis in patients with advanced cancer. 105 Lactic dehydrogenases (LDH), a serum marker that rises in cases with high disease volume, and the c-reactive protein, a marker related to inflammation, are more nonspecific, but in the context of advanced cancer they can be associated with progressive disease.106,107 Hypoalbuminemia, in addition to reflecting the nutritional status of patients, also expresses a negative influence on the prognosis of critically ill patients. 105
Prognostic Tools
Knowing the limitations of predicting survival based on clinical estimates by physicians, assessment of symptoms and biochemical parameters, there is robust evidence that the combination of all parameters in one model may improve the prediction of survival in advanced cancer patients. 96 Prognostic models have been created in the palliative care setting, improving the accuracy, and helping healthcare professionals to plan the patient journey.
The Palliative Performance Scale (PPS), 101 Palliative Prognostic Score (PaP),108,109 Palliative Prognostic Index (PPI), 110 Objective Prognostic Score (OPS) 111 and Prognosis in Palliative Care Study Predictor (PiPS) 112 were developed and validated for patients with few months of life expectancy. In contrast, the modified Glasgow Prognostic Score (mGPS),113,114 the PRONOPALL prognostic tool, 115 the Spanish Prognostic Nomogram (SPN), 116 the Barretos Prognostic Nomogram (NPB) 117 and the HAprog (clinical and full versions) 118 were developed in patients with advanced cancer with an expectation of several months of survival (Table 3). It is interesting to note that the prediction accuracies increase the closer the patient is to the end of life.
There may be a myriad of combinations between prognostic variables; each model has its peculiarity, according to the profile of patients evaluated. However, some variables are more frequently contemplated, such as the functional status of the patient. Almost all the main models, regardless of life expectancy, include performance status as a clinical variable, with KPS being the most used way. Only models related to a shorter life expectancy (days to weeks) include symptoms and clinical signs as prognostic factors, with “delirium,” “dyspnea,” and “anorexia” being the most frequent observed.
Inflammatory laboratory variables were included in some prognostic models, among which “total leukocytes,” “serum albumin,” and “LDH” stand out as the most frequently related. Among these models with longer life expectancy, only the NRF and HAprog clinical version models did not assess laboratory variables.118,119 On the other hand, the mGPS is the only model that presents only laboratory variables in its composition (c-reactive protein, albumin), however better accuracy is achieved when evaluated together with the ECOG-PS.113,114 Laboratory variables can indeed increase the prognostic ability of a tool, but it can be a barrier to its applicability in the clinical routine, regarding the request for complementary tests in patients under palliative care.
The quick access to prognostic tools for use in clinical practice is essential. PredictSurvival.com is an online platform, free to use, which includes grouped classic prognostic tools, being particularly useful for cases of patients in the last 6 months of life. 120 Its use by health professionals has increased prognostic confidence and accessibility to discuss the prognosis with patients. 121 In this same context, the HAprog models have been transcribed into a digital version for use in smartphones. 118 Interestingly, in addition to calculating the probabilities of survival at 30, 90, and 180 days, HAprog shows the probable survival times in days in the expected, worst-case, and best-case scenarios. The practical implication of these results still needs to be evaluated in subsequent studies.
Despite the availability of validated predictive tools in the current literature, there are still barriers to their use in daily clinical practice. Knowing the importance of estimating an accurate prognosis, choosing which tool to use for each patient, knowing how to interpret and communicate the estimated survival value and managing time during patient care are issues that must be still on the discussion.120,122 It is important to emphasize that the use of a prognostic tool does not have the intention of standardizing or “mechanizing” the process of evaluating the patients’ survival, but it increases the clinical evaluation of the physician with less subjectivity and greater accuracy.
Future Perspective
Communicating difficult news in oncology is a complex and stressful task for oncologists 123 that should be taught, learned, and retained. The SPIKES protocol, 124 the elements of which are Setting, Perception, Invitation, Knowledge, Emotions, and Strategy, is the most commonly used framework in the education of medical students, residents, and oncologists. 125 A recent systematic review 126 identified 3 communication models validated through randomized clinical trials; that is, the Serious Illness Conversation Guide, 127 the 4 Habits Model, 128 and the acronym ADAPT. 129 Another very useful mnemonic when discussing goals of care is the framework REMAP (Reframe, Expect emotion, Map outpatient goals, Align with goals, and Propose a plan). 130
Prognostic Tools for Use in Advanced Cancer Patients.

Abbreviations: PPS, Palliative Performance Scale; PaP, Palliative Prognostic Score; D-PaP, Palliative Prognostic Score with Delirium; PPI, Palliative Prognostic Index; OPS, Objective Prognostic Score; mGPS, Modified Glasgow Prognostic Score; NRF, Number of Risk Factors; SPN, Spanish Prognostic Nomogram; BPN, Barretos Prognostic Nomogram; ECOG, Eastern Cooperative Oncology Group; KPS, Karnofsky Performance Status.
Conclusions
The therapeutic decision process of whether or not to treat patients with advanced cancer with systemic antineoplastic therapies is complex. Oncologists need to know in detail the therapeutic protocols to which they will submit their patients, mainly its potential clinical benefits and risks. In addition, they need to know how to capture and adequately comprehend their patients’ wishes, values, and personal beliefs. Accurate prognostic estimates can also help in “fine-tuning” therapeutic decisions.
Footnotes
Appendix
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ Note
CEP and BSRP are Research Productivity Fellows Level 2 from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil).
