Abstract
Objectives
CpG ODN is a Toll-like receptor 9 agonist with immunotherapeutic potential for many cancer types, including aggressive breast cancers. There is strong interest in utilizing CpG ODN as an adjuvant to improve clinical efficacy of current treatments and immunogenicity of breast cancers not traditionally responsive to active immunotherapy, such as those that are human epidermal growth factor receptor 2 (HER2)-positive. This study aimed to study the efficacy and safety of combination CpG ODN plus anti-HER2 antibody trastuzumab treatment in patients with advanced/metastatic breast cancer.
Methods
This single-arm, open-label phase II clinical trial treated patients (n = 6) with advanced/metastatic HER2-positive breast cancer with weekly subcutaneous CpG ODN and trastuzumab. Patients may have received any number of prior therapies to be enrolled (most enrolled at median 1 prior line of chemotherapy). Peripheral blood was collected at baseline and weeks 2, 6, 12, and 18 for immune analyses. Six patients were enrolled and 50% achieved stable disease (SD) response.
Results
Median PFS was 8.3 months. Three of the six patients enrolled opted to stop treatment due to tolerability issues. Multiplex assay for cytokine measurements revealed significantly higher VEGF-D levels at week 2 compared to baseline. Peripheral blood mononuclear cells analyzed by flow cytometry showed a significant increase in monocytic MDSC between weeks 6 and 12. Patients with progressive disease tended to have higher levels of week 6 monocytic MDSC and PD-1+ T cells than patients with SD. NK cell populations did not significantly change throughout treatment.
Conclusions
CpG ODN and trastuzumab treatment of metastatic HER2 + breast cancer was safe but was not tolerable for all patients. This combination did induce potentially predictive immune profile changes in treated patients with metastatic HER2 + breast cancer, the significance of which needs to be further explored.
Plain language summary
Why was the study done? Breast cancer that has metastasized (moved outside of the breast and local lymph nodes) is currently considered incurable and can be difficult to treat. Treatments that can stimulate the immune system to recognize cancer cells have been found to be useful for many types of cancers, including some types of breast cancers. This study tested a new immune stimulator (CpG ODN) in combination with a currently on-the-market antibody treatment for breast cancer (trastuzumab). What did the researchers do? The research team enrolled patients who had metastatic breast cancer and treated them all with a combination of trastuzumab and CpG ODN for 12 weeks. These patients were monitored for any side effects/toxicity, monitored for how long their breast cancer responded to this treatment, and monitored for how long they lived after beginning this treatment. Patients also had their blood drawn at different time points to observe how their immune cells and immune proteins (e.g. cytokines) changed on treatment. What did the researchers find? The research team enrolled six patients and found that the treatment was safe and that 50% of the patients treated did not have any breast cancer growth when given CpG ODN plus trastuzumab. Looking at the immune cells in the patient blood samples, some cells that are known to decrease the immune response to cancers (myeloid-derived suppressor cells) did increase towards the end of treatment. What do the findings mean? Overall, CpG ODN and trastuzumab treatment was found to be safe and potentially effective in preventing breast cancer growth.
Keywords
Introduction
Approximately 15%–20% of all breast cancer cases are human epidermal growth factor receptor 2 (HER2)-positive and historically represent a group of aggressive tumors with poor outcomes. 1 The advent of HER2-targeting agents has revolutionized the treatment of HER2-positive breast cancer and has conferred significant improvements in survival rates among affected patients. 2 The first of these drugs was trastuzumab, a humanized monoclonal IgG1 antibody that binds to the extracellular domain of HER2. Trastuzumab has been successfully combined with chemotherapy, other HER-directed monoclonal antibodies, and tyrosine kinase inhibitors to further amplify its benefit.3-7 Moreover, new HER2-targeting antibody-drug conjugates have shown impressive response rates in pre-treated patients with metastatic disease.8,9 Despite the immense benefits of these treatment options, HER2-positive metastatic breast cancer remains problematic with a median overall survival rate of less than 5 years.1,10 Therefore, there is a need for novel and more effective therapeutic options for such patients.
Several studies have shown that the antitumor effects of trastuzumab are dependent on the presence of immune effector cells that bear FcγR, such as natural killer (NK) cells.11,12 One method to further promote the anti-HER2 immunity of trastuzumab is to add an agent that can act as an immunostimulant. A possible candidate would be a Toll-like receptor (TLR) agonist. Human TLRs are a family of ten membrane-bound receptors that recognize unique pathogen-associated molecular patterns. 13 TLRs can be found on the cell membrane or intracellularly where they are localized to endosomes, lysosomes, or the endoplasmic reticulum. TLR activation can promote a robust innate immune response and produce anti-tumor responses. For example, TLR agonists can increase tumor antigen-specific cytotoxic T cell killing,14-16 lead to anti-tumor Th1 polarization,17,18 and boost anti-PD-1 adaptive immunity.19,20 There has been considerable interest in TLR9, a receptor that recognizes unmethylated CpG oligodeoxynucleotides (ODN) commonly found in bacterial and viral genomes. 21 TLR9 is expressed by a variety of antigen-presenting cells, T cells, and NK cells.12,22 TLR9 is a receptor of particular interest in the breast cancer field as high levels of TLR9 expression in breast carcinomas is independently associated with improved patient outcomes. 23 TLR9 stimulation leads to a MyD88-dependent signaling cascade that induces IRF7, MAPK, and NF-kB transcription factors. TLR9 signaling in the setting of cancer can increase the amount of tumor-infiltrating lymphocytes, 24 inhibit angiogenic signaling, 25 prevent immunosuppressive T regulatory cell activities, 26 and improve the efficacy of immune checkpoint inhibitors.20,24
Importantly, NK cell activity can be augmented by TLR9 activation. Our group has shown that co-stimulation through the NK cell Fc receptor and addition of a secondary activating agent (e.g. interleukin-12 (IL-12), IL-21) can lead to a robust NK cell killing response against antibody-coated tumor cells.27-30 Specifically, we have demonstrated in murine models that activation of TLR9 on NK cells with CpG ODN can improve responses to anti-HER2 antibody-coated tumor cells leading to enhanced cytotoxicity and the secretion of interferon γ (IFN-γ), IL-8, and MIP-1α. 12 Other studies have shown that repeated administration of a TLR9 agonist and anti-CD20 monoclonal antibodies yields improved anti-CD20 activity compared with a single administration, 31 suggesting that dual treatment may be beneficial in the clinical setting.
Summary of Prior Oncology Clinical Trials Using TLR9 Agonists.

PF03512676 safety has been studied in combination with trastuzumab within phase I trials of patients with metastatic breast cancer and has been shown to be safe in doses up to 0.16 mg/kg. 55 Initial efficacy measurements from these trials suggest that the best initial responses were seen with the higher doses (0.12 and 0.16 mg/kg) of PF03512676, however, final results from these studies are not currently available from the sponsor. In the present clinical trial, the combination of subcutaneous CpG ODN and intravenous trastuzumab was studied in patients with advanced/metastatic HER2 + breast cancer who had trastuzumab-responsive disease. The primary objective was to assess the safety and tolerability of this regimen, with secondary objectives being to evaluate the effect of the drug combination on progression-free survival (PFS) and levels of immune markers including circulating cytokines and immune cells.
Materials and Methods
Study Design
A single arm, open-label phase II clinical trial was conducted at the Ohio State University Comprehensive Cancer Center under an IRB-approved protocol (IRB protocol no: 2008C0116). This was registered within the publicly accessible database ClinicalTrials.gov. The ClinicalTrials.gov ID is NCT00824733 and the registered title is “Agatolimod and Trastuzumab in Treating Patients with Locally Advanced or Metastatic Breast Cancer” (https://beta.clinicaltrials.gov/study/NCT00824733?id=NCT00824733). The sample size/accrual goal estimated was 15 patients due to limited availability of the CpG ODN agent (PF-03512676) and time constraints (formalized sample size calculation not completed). Patient accrual occurred between March 2009 and April 2014 at which point the study was terminated due to slow patient accrual. The primary objective was to determine the safety and tolerability of trastuzumab and CpG ODN treatment. Secondary objectives included the assessment of progression free survival (PFS) and measurement of cytokines and immune cell populations. Written informed consent was obtained from all study participants. Peripheral blood for correlative studies was collected at baseline and at the beginning of weeks 2, 6, 12, and 18. These timepoints were correlated with treatment schedule to include collections prior to receiving study drug, middle of treatment, end of treatment, and 6 weeks after completion.
Eligibility
Eligible patients were ≥18 years old and diagnosed with biopsy-proven metastatic, HER2 + breast cancer. HER2 overexpression was defined as 3+ by immunohistochemistry (IHC) or +2 IHC with HER2/CEP17 ratio of ≥2.2 or ≥6 copies of HER2 per nucleus. At time of enrollment, patients were required to have stable disease on trastuzumab therapy for up to 6 months prior to receiving their first dose of CpG ODN. Patients who had been receiving trastuzumab for greater than 6 months or those with disease that progressed immediately prior to enrollment were not eligible. There were no restrictions in enrollment regarding the number of prior systemic therapies given for metastatic and/or non-metastatic disease. Other eligibility criteria included Eastern Cooperative Oncology Group (ECOG) performance status of 0-2, normal organ and bone marrow function, left ventricular ejection fraction of 50% or greater, no prior history of autoimmune or antibody-mediated disorders, and absence of unstable brain metastases. Patients with treated and stable brain metastases were eligible but were required to be off any systemic corticosteroids and anti-convulsants for at least 4 weeks prior to enrollment in the study. Patients with uncontrolled human immunodeficiency virus (HIV), hepatitis B, or hepatitis C were not eligible.
Treatment Schema and Study Procedures
Patients received intravenous (IV) trastuzumab at a dose of 2 mg/kg and subcutaneous (SQ) CpG ODN at 0.16 mg/kg once weekly for 12 weeks. Patients were permitted to continue IV trastuzumab alone following completion of 12 weeks of CpG ODN. Patients who had not received trastuzumab within 4 weeks of the treatment start date or more received a loading dose of trastuzumab at 4 mg/kg in week 1. Three patients opted to continue oral anti-HER2 therapy (lapatinib) while on trial. Patients were also permitted to continue lapatinib and endocrine therapy while on study; they were not required to have progressive disease to enroll. The response to therapy was measured by performing computed tomography of the chest, abdomen, and pelvis at 12-week intervals. Assessment of left ventricular ejection fraction by echocardiogram or multi-gated acquisition scan was performed at baseline and every 12 weeks. Complete blood counts, chemistries, and hepatic function tests were obtained weekly for 12 weeks and at week 18. History and physical examination were performed at baseline (week 1) and prior to weeks 2, 6, 12, and 18. Safety signals were evaluated using the Common Terminology Criteria for Adverse Events (CTCAE) version 3.0 in use at the time.
Correlative Blood Studies
Peripheral blood mononuclear cells (PBMC) were isolated from peripheral venous blood via density gradient centrifugation with Ficoll-Paque (Cytiva) as previously described. 56 PBMC and plasma were cryopreserved and stored at −80°C until batch analysis could be performed. PBMCs were analyzed by multi-color flow cytometry to quantify the presence of immune cell subsets as previously described.57,58 The specific fluorescent-tagged antibodies employed are listed in Supplemental File 1. NK cells were defined as CD56+ cells and were further categorized by expression of CD69, CD16, and/or PD-1. All T cell subsets were CD3+, with naïve T cells being CD44-/CD62 L+, memory T cells being CD44+/CD62 L+, and effector T cells being CD44+/CD62L-. PD-1 expression was also measured on CD3+ T cells. Myeloid derived suppressor cells (MDSC) were defined as the CD33+/CD11b+/HLA-DR low PBMC population. Monocytic MDSC (M-MDSC) were further phenotyped as CD14+/CD15-, polymorphonuclear MDSC (PMN-MDSC) were defined as being CD14-/CD15+, and double-positive MDSC were defined as being CD14+/CD15+. Single color staining was performed for compensation. All samples were run on a BD LSR II flow cytometer and were subsequently analyzed with FlowJo software (TreeStar, Inc). Plasma was analyzed for the presence of several cytokines thought to be associated with CpG administration/NK cell activation (GM-CSF, IFNγ, IL-1α, IL-1β, IL-4, IL-5, IL-6, IL-8, IL-10, IL-13, IL-15, IL-17, IL-18, IP-10, MCP-1, MCP-2, MDC, MIG, MIP-1α, MIP-1β, RANTES, TNFα, VEGF-A, VEGF-C, VEGF-D) via custom-designed multiplex U-Plex and V-Plex assays (Meso Scale Discovery) which were performed per manufacturer’s instructions. Multiplex plates were read on a MESO QuickPlex SQ 120 instrument.
Statistics
Linear mixed effect modeling was used to model repeated measures on percentage of various immune cells/cytokines over time and between multiple time points. Data distribution comparing immune cell and cytokine levels over time were determined by linear mixed modeling to allow for multiple testing corrections. To decrease the chance of false-positive results with this testing, Tukey’s method was also used to adjust P-values for multiple comparisons between time points. All P-values were double sided and statistical significance was set at alpha of 0.05. Adverse reactions were tabulated based on grade using CTCAE version 3.0 and attribution to study therapy. Response was assessed using Response Evaluation Criteria in Solid Tumors (RECIST) criteria version 1.1. Overall survival times were measured from date of first study treatment to date of death. Overall survival of stable and progressive disease patient groups were compared by log-rank (Mantel-Cox) analysis.
Results
Patient Demographics
Patient Demographics and Tumor Characteristics.

Safety
Summary of Adverse Events. All Grade 2 or Higher Adverse Events Deemed to be at Least Possibly Related to Study Treatment are Included.

Clinical Responses
Of the six patients enrolled on this study, there were no objective responses, but three patients (50%) experienced stable disease (SD) at 12 weeks as their best response. Median progression free survival of study patients was 8.3 months (Figure 1A). Of note, all three stable disease patients remained free of disease progression for at least 12 months from the start of study treatment. One patient with stable disease was also on endocrine therapy, one patient with stable disease was on lapatinib (no endocrine therapy), and the last patient with stable disease was on neither additional medication. Median overall survival (OS) of all patients was 84.6 months (Figure 1B). Median OS for patients with SD has not been reached, while median OS for patients with progressive disease (PD) was 40.5 months (P = 0.0588). Survival analysis. Kaplan-Meier graphs are shown demonstrating the (A) progression-free survival and (B) overall survival rates of study patients. The overall survival graph also contains lines denoting patients who had either stable or progressive disease as their best treatment response.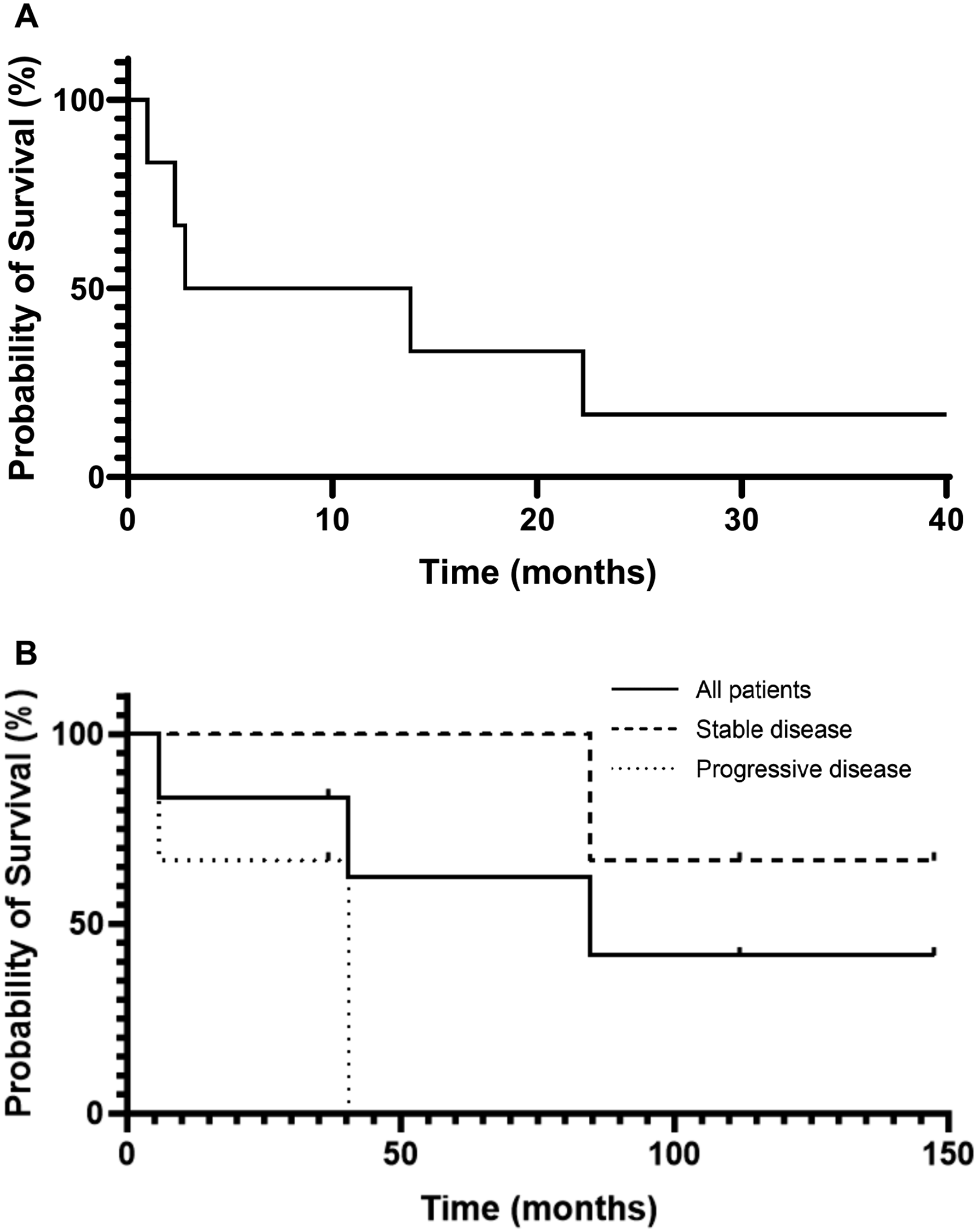
Circulating Plasma Cytokine Levels
Peripheral blood for correlative studies was available from five patients. Plasma cytokine multiplex analysis revealed that VEGF-D levels significantly increased among patients from off-treatment baseline to week 2 on treatment (P = 0.0392) (Figure 2). CpG-TLR9 signaling and NK cell activation-related cytokines including MCP-1, MCP-2, MIP-1α, MIP-1β, and RANTES levels were also measured, however, no significant trends between time points were elucidated. No statistically significant changes were observed in plasma levels of the other cytokines included within the multiplex panel, including GM-CSF, IFNγ, IL-1α, IL-1β, IL-4, IL-5, IL-6, IL-8, IL-10, IL-13, IL-15, IL-17, IL-18, IP-10, MDC, MIG, TNFα, VEGF-A, VEGF-C, and VEGF-D (Supplemental File 2). Plasma cytokine levels of patients receiving CpG ODN and trastuzumab. Blood specimens were collected from patients at the beginning of weeks 1 (baseline), 2, 6, 12, and 18 while they were on study. Plasma was isolated and batch analyzed by multiplex cytokine analysis. Individual lines/shapes denote different patients’ cytokine trajectories. Data distribution comparing cytokine levels over time were determined by linear mixed modeling to allow for multiple testing corrections. Tukey’s method was used to adjust P-values for multiple comparisons between time points. Statistical significance was set at an alpha of .05. P-value of <.05 is denoted by *.
Immune Cell Subset Measurements
PBMC were isolated, stained, and analyzed by flow cytometry to determine immune cell populations over the course of treatment. Several T cell subsets were measured, including naïve CD8+ T cells, effector CD8+ T cells, memory CD8+ T cells, and PD-1+ T cells (Figure 3A). No significant trends were noted in the level of these subsets while patients were on treatment. NK cell subsets were also analyzed by flow cytometry within patient PBMC samples. Activated CD16+ and CD69+ NK cells were quantified as were exhausted PD-1+ NK cells (Figure 3B). There were no significant differences in levels of NK cell populations across the different trial time points. Total MDSC levels were measured as were the PMN-MDSC and M-MDSC subtypes (Figure 3C). Total MDSC tended to increase towards the end of overall CpG ODN treatment, but this finding did not reach statistical significance. However, levels of M-MDSC were significantly higher at week 12 compared to week 6 of treatment (P = 0.0099) and appeared to further increase at week 18 (6 weeks off CpG ODN treatment). Double-positive (CD14+/CD15+) MDSC were also identified, however, no appreciable differences in these cells were found between time points (data not shown). Measurement of peripheral blood immune cell subsets from patients receiving CpG ODN and trastuzumab. PBMC were isolated from patient peripheral blood specimens then stained and measured for immune cell subsets by flow cytometry. (A) T cell, (B) NK cell, and (C) MDSC. (D) PD-1+ T cell and total MDSC levels were evaluated in the context of clinical outcome where patients were grouped as having either stable disease or progressive disease as their best clinical response. Individual lines/shapes denote different patients’ cytokine trajectories. Statistical significance was set at an alpha of .05. P-value of <.01 is denoted by **.
Trends comparing immune cell levels of patients who had stable disease versus progressive disease were also conducted. While not powered for statistical comparison, patients with progressive disease on treatment tended to have higher levels of PD-1+ T cells and total MDSC (Figure 3D).
Discussion
Combinations of CpG ODN with a variety of chemotherapeutics and biologic therapies have shown it to be a safe and tolerable treatment option for most patients; however, there is no combination that has been proven effective in a late-stage disease trial. In the present study, we report the results of a phase II clinical trial which enrolled patients with metastatic HER2 + breast cancer and treated them with the HER2 monoclonal antibody trastuzumab and CpG ODN. Unfortunately, this trial closed after recruiting only 6 of the planned 10-15 patients due to slow accrual, in part due to patient concern over the frequency of drug administration. There were also limitations on the correlative studies performed due to only two of the patients completing all the pre-specified blood draw time points. Nevertheless, several important findings can be gleaned from this study.
Patients experienced adverse events that were similar in type and frequency to those previously described in other CpG ODN clinical studies. 45 The most common symptoms were injection site reaction, fatigue, and vomiting. These symptoms were all short-lived, but for a small number of patients they were significant enough for the patient to stop treatment. Of the patients on this study, half experienced stable disease as their best clinical disease response, while the other half had progressive disease. All three patients with stable disease were able to continue with trastuzumab treatment and they all experienced stability for at least 12 months prior to disease progression. This outcome is particularly encouraging when it is noted that all study patients had received trastuzumab treatment in the past. Compared to those with progressive disease following CpG ODN/trastuzumab treatment, patients with stable disease also tended to have longer durations of overall survival.
It was observed that patient serum VEGF-D levels significantly increased within one week of receiving CpG ODN and trastuzumab compared to baseline. VEGF expression has been found to be upregulated by activation of several TLR, including TLR9 within an in vitro model of pancreatic carcinoma and an in vivo model of lung carcinoma.59,60 While a TLR9-stimulated increase in VEGF expression was generally found to promote angiogenesis and metastasis, it should be noted that TLR9 expression has been identified as a poor prognostic marker in several cancers. However, increased TLR9 expression indicates an improved prognosis in patients with breast cancer. 23 It is not known if this brief elevation of VEGF-D expression is enough to confer additional risk for tumor progression, particularly as the spike returned to baseline levels after the initial week. Many other cytokines were found to fluctuate over the course of CpG ODN/trastuzumab treatment, but it was difficult to identify specific cytokine expression trends within any group of patients on this study.
Patient peripheral blood M-MDSC increased significantly between weeks 6 and 12 of CpG ODN/trastuzumab treatment. While not statistically significant, this trend continued into the post-treatment period at week 18. Also, patients with stable disease tended to have overall lower amounts of MDSC compared to those with progressive disease throughout the course of treatment. M-MDSC are typically considered to be an immunosuppressive subset of cells whose presence are negatively associated with cancer survival outcomes, including breast cancer. 61 Notably, M-MDSC are known to express TLR9 and it has been found that CpG treatment of M-MDSC leads to their differentiation into tumoricidal macrophages and promotion of a Th1 response. 62 While M-MDSC levels are often thought to be associated with poorer cancer-associated prognoses, it is possible that the increase in M-MDSC populations towards the end of CpG ODN treatment is indicative of disease progression more than CpG ODN effects or whether this portends that the therapy is having unintended immune consequences. In future studies, it would also be interesting to study the tumor microenvironment changes that occur after CpG ODN treatment (e.g. tumor-infiltrating lymphocyte characterization) as these may be unique compared to trends seen in peripheral blood PBMCs.
Patients with stable disease also tended to have lower amounts of circulating PD-1+ T cells than patients with progressive disease. Our group has previously shown increased levels of intratumoral PD-1+ T cells to be associated with improved HER2 + breast cancer outcomes. 63 Alternatively, it has been suggested that circulating PD-1+ T cells are in fact a negative prognostic marker in breast cancer. 64
All patients who were enrolled in this trial had metastatic hormone (estrogen and progesterone) receptor-positive (HR), HER2-positive breast cancer. Patients with metastatic HR-positive, HER2-positive breast cancers tend to have longer rates of overall survival than those with HR-negative, HER2-positive breast cancer. 65 With the exception of adding endocrine therapy to HR-positive, HER2-positive breast cancer, there are no current differences in treatment management of these patients compared to those with HR-negative, HER2-positive metastatic disease. 66 It is important to highlight that recent research suggests that the tumor microenvironment of HER2-positive breast cancers may differ depending on HR receptor expression. For example, in the CALGB 40601 and PAMELA trials was noted that HER2-positive breast cancers that were also HR-negative had higher proportions of tumor infiltrating lymphocytes than HR-positive tumors. 67 This may suggest that HR-negative HER2-positive tumors have a more immunogenic environment than their HR-positive counterparts and thus may be more responsive to immunotherapeutic responses.
Ultimately the results of this study show that the regimen of trastuzumab and CpG ODN was found to be safe and associated with fluctuations in circulating cytokine and immune cells levels. Discontinuation rates were higher than other CpG ODN-based clinical studies, but the true proportion of patients who would tolerate this treatment regimen may be higher if expanded to a larger study size. An encouraging overall survival rate was observed among treated patients; however, due to this study’s small sample size this combination requires further investigation for verification of outcomes and correlative results. Of note, it is unclear what the effect of concomitant treatments (e.g. lapatinib, endocrine therapy) could have on the efficacy of these agents. With the current HER2 + breast cancer landscape in mind, CpG ODN would be a relevant adjuvant to study in combination with current immune checkpoint inhibitors (ICI) or other novel immunotherapies to expand options for breast cancer treatment. It is known that HER2 + breast cancers are not typically responsive to chemotherapy plus ICI alone, 68 therefore the addition of an agent that can further prime/boost the immune response may be beneficial. Additionally, CpG ODN may be provide a stronger response to earlier lines of metastatic HER2-positive breast cancer treatment, such as adding it to standard-of-care docetaxel plus trastuzumab and pertuzumab. Within the larger treatment context for breast cancer, many opportunities to improve upon anti-tumor immunity remain.
Supplemental Material
Supplemental Material - An Open-Label Study of Subcutaneous CpG Oligodeoxynucleotide (PF03512676) in Combination with Trastuzumab in Patients with Metastatic HER2+ Breast Cancer
Supplemental Material for An Open-Label Study of Subcutaneous CpG Oligodeoxynucleotide (PF03512676) in Combination with Trastuzumab in Patients with Metastatic HER2+ Breast Cancer by Dionisia Quiroga, Robert Wesolowski, Sara Zelinskas, Ashley Pinette, Brooke Benner, Emily Schwarz, Himanshu Savardekar, Courtney Johnson, Andrew Stiff, Lianbo Yu, Erin Macrae, Maryam Lustberg, Ewa Mrozek, Bhuvaneswari Ramaswamy, and William E. Carson in Cancer Control
Supplemental Material
Supplemental Material - An Open-Label Study of Subcutaneous CpG Oligodeoxynucleotide (PF03512676) in Combination with Trastuzumab in Patients with Metastatic HER2+ Breast Cancer
Supplemental Material for An Open-Label Study of Subcutaneous CpG Oligodeoxynucleotide (PF03512676) in Combination with Trastuzumab in Patients with Metastatic HER2+ Breast Cancer by Dionisia Quiroga, Robert Wesolowski, Sara Zelinskas, Ashley Pinette, Brooke Benner, Emily Schwarz, Himanshu Savardekar, Courtney Johnson, Andrew Stiff, Lianbo Yu, Erin Macrae, Maryam Lustberg, Ewa Mrozek, Bhuvaneswari Ramaswamy, and William E. Carson in Cancer Control
Footnotes
Acknowledgments
The authors would like to thank the patients and their families, the investigators, research nurses, study coordinators, and operations staff who contributed to this study. The authors would like to thank the Shared Resources Core at The Ohio State University James Comprehensive Cancer Center.
Authors Note
The original research in this manuscript was accepted and presented as a poster presentation at the National Comprehensive Cancer Network 2022 Annual Meeting. This original research and manuscript have not otherwise been submitted or accepted for publication in another journal.
Author’s Contribution
Conceptualization/supervision (BR, WEC); formal analysis (DQ, LY); funding acquisition (BR, WEC); investigation (DQ, RW, SZ, AP, BB, ES, HS, AS); methodology (DQ, BR, WEC); resources/patient enrollment (EO, ML, EM, BR); visualization (DQ, CJ); writing – original draft (DQ); writing – review and editing (DQ, CJ, BR, WEC).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH Grants P01 CA095426 (M. Caligiuri, WEC III), P30 CA016058 (M. Caligiuri, R. Pollock), R21 CA084402 (WEC III), K24 CA093670 (WEC III), T32 CA009338 (AP), and T32 CA247815 (DQ). Pfizer provided drug support for PF03512676.
Ethical Statement
Clinical Trials Registration
ClinicalTrials.gov Registration: Agatolimod and Trastuzumab in Treating Patients with Locally Advanced or Metastatic Breast Cancer (NCT00824733) URL: https://beta.clinicaltrials.gov/study/NCT00824733?id=NCT00824733&rank=1
Off-Label Use/Unapproved Drugs or Products
This manuscript discusses a clinical trial in which the investigational drug PF03512676 (CpG ODN) is being tested in combination with trastuzumab. PF03512676 is not currently an approved treatment for breast cancer and was used in the scope of a clinical trial.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request. It is confirmed that other researchers may reproduce the methodology presented in this manuscript from the description given in the Methods section and that corresponding author is available for assistance on reasonable request.32-44,46-54
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
