Abstract
Keywords
Introduction
Human epidermal growth factor receptor (HER2)-positive (HER2+) breast cancer has historically been associated with an aggressive clinical phenotype, high rates of recurrence, and poor survival.1,2 Around 15% of patients diagnosed with early-stage breast cancer, and 26% with metastatic breast cancer (MBC), are HER2+. 1 Moreover, brain metastases develop in up to 50% of patients with HER2 + MBC and are associated with an even poorer prognosis. 3
The treatment landscape for HER2 + MBC is evolving rapidly, with the emergence of several new HER2-targeted therapies over recent years requiring further investigation of optimal treatment sequencing of emerging treatments. Tucatinib is a highly selective oral tyrosine kinase HER2-receptor inhibitor and was approved by the European Medicines Agency (EMA) in February 2021 for use in combination with trastuzumab and capecitabine (tucatinib combination) for the treatment of adult patients with HER2 + locally advanced or MBC who have received ≥2 prior anti-HER2 treatment regimens. 4 In addition, trastuzumab deruxtecan (T-DXd), which also targets the HER2 receptor, was approved by the EMA in July 2022 for the treatment of adult patients with HER2 + MBC who have received ≥1 prior HER2-directed regimen. 5 As T-DXd was only recently approved, there are limited data on the efficacy or effectiveness of other HER2-targeting agents such as tucatinib when administered after prior therapy with T-DXd.
In a previous study, the first to assess the real-world effectiveness of tucatinib in the HER2 + MBC setting, 6 we utilized a United States (US)-based electronic medical record database to assess patient characteristics, treatment patterns, and clinical outcomes of patients with HER2 + MBC with and without brain metastases who were treated with tucatinib-based therapy in real-world clinical practice. 6 That study also assessed outcomes in the subgroup of patients who received tucatinib in accordance with the US Food and Drug Administration (FDA) label, where tucatinib is approved in combination with trastuzumab and capecitabine for patients with HER2 + locally advanced or MBC, with or without brain metastases, previously treated with ≥1 prior HER2-directed regimens in the metastatic setting. However, the real-world effectiveness of tucatinib in patients treated in a manner consistent with the EMA label, that is after ≥2 prior HER2-directed regimens, has not been specifically evaluated.
This study was a new analysis to further describe the subset of patients with HER2 + MBC who received tucatinib after ≥2 prior HER2-directed therapies in the metastatic setting in the real-world setting, including in those patients receiving tucatinib-based therapy immediately following T-DXd. To assess the effectiveness of tucatinib when sequenced after T-DXd, we also included patients receiving tucatinib-based therapy immediately following T-DXd.
Methods
Study Design and Data Source
The study design and data source have been described previously. 6 Briefly, US patients aged ≥18 years from the nationwide, de-identified electronic health record-derived Flatiron Health MBC database who were diagnosed with HER2 + MBC between January 1, 2017 and December 31, 2022, were included. The Flatiron Health database includes structured data (eg, demographics, laboratory values, and prescribed drugs) and unstructured data (eg, biomarker levels) collected via technology-enabled chart abstraction from physicians’ notes and other unstructured documents.7,8 Patients were excluded if they did not receive any systemic treatment in the metastatic setting, did not have activity in the database within 90 days of MBC diagnosis, or had evidence of prior nonbreast cancer within 6 months before MBC diagnosis. This analysis included patients receiving the tucatinib combination after ≥2 HER2-targeted metastatic therapies. In the Flatiron Health MBC Enhanced Datamart, information before metastatic diagnosis was limited and, as a result, patients in this analysis received ≥2 HER2-directed therapies in the metastatic setting, which is more conservative than the EMA label (wherein prior HER2-directed therapies are not limited to the metastatic setting).
As this was a retrospective database study that did not involve human tissue/samples, ethical review and approval were not required.
Line of Therapy Definitions
The oncologist-defined, rule-based line of therapy (LOT) started on the first administration or abstracted treatment which occurred on or after diagnosis of metastatic cancer; any treatment initiated ≤28 days of the first treatment date was considered part of the same LOT. A treatment switch was defined as a claim for a new HER2-directed therapy, tyrosine kinase inhibitor, or chemotherapy ≥29 days after the start date of the previous LOT. If there was no record of a treatment switch, treatment discontinuation was defined as a gap of >60 days between the last medication date and a new regimen.
Outcomes
This study assessed patient baseline demographics, clinical characteristics, and clinical outcomes. Outcomes assessed were real-world time to treatment discontinuation (rwTTD; time from treatment initiation to discontinuation), real-world time to next treatment (rwTTNT; time from treatment initiation to the start of the subsequent LOT; rwTTNT is used as a proxy for progression-free survival9,10), real-world overall survival (rwOS; time from treatment initiation to death), and treatment persistence (the proportion of surviving patients staying on therapy at 6, 12, 18, and 24 months). Patient demographics and clinical characteristics were captured during the baseline period (any time before tucatinib initiation), and clinical outcomes were assessed from the tucatinib initiation date. All patients were followed until the end of the study period, loss to follow-up, or death, whichever occurred first.
Statistical Methods
Mean, standard deviation (SD), and interquartile range (IQR) were used to describe continuous variables, while frequency (number of cases) and percentages were reported for categorical variables. Time-to-event analyses were carried out for rwOS, rwTTNT, and rwOS with the Kaplan-Meier method. The time-to-event analyses included information on all patients, and patients who lacked recorded events were censored at their last activity date, while only patients with sufficient follow-up were included in the persistence analysis. SAS 9.4 and R 4.2.0 were used to conduct all analyses.
Results
Patient Demographics and Clinical Characteristics
Of 16 733 patients diagnosed with MBC during the study period, 3449 had evidence of HER2 + disease. Of these patients, 89 received the tucatinib combination after ≥2 prior HER2-targeted regimens and were included in the analysis (Figure 1). Median (range) age of included patients was 57 (30-84) years; the majority (60.7%, n = 54) were White, had Eastern Cooperative Oncology Group performance status (ECOG PS) ≤1 (66.3%, n = 59), and had brain metastases (57.3%, n = 51) (Table 1). In patients who received the tucatinib combination in third line (3L) setting, 64.3% (n = 18/28) had brain metastases; for those in fourth line or later (4L+) setting, 54.1% (n = 33/61) had brain metastases. The median (IQR) LOT for patients who received the tucatinib combination in 4L+ was 4.0 (4.0-6.0). Patient selection and attrition counts. Abbreviations: HER2, human epidermal growth factor receptor 2; MBC, metastatic breast cancer. Baseline
a
Demographics and Clinical Characteristics of Patients With HER2 + MBC Receiving Tucatinib With Trastuzumab and Capecitabine After ≥2 Prior HER2-Targeted Therapies in the Metastatic Setting. Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; HER2, human epidermal growth factor receptor 2; LOT, line of therapy; MBC, metastatic breast cancer; T-DM1, ado-trastuzumab emtansine; T-DXd, trastuzumab deruxtecan. aBaseline refers to the period before tucatinib treatment initiation. bNot mutually exclusive.

Most patients (68.5%, n = 61) initiated the tucatinib combination after receiving ≥3 prior LOTs. The most common therapies received at any time before tucatinib initiation were trastuzumab (100.0%, n = 89) and pertuzumab (88.8%, n = 79), followed by trastuzumab-emtansine (87.6%, n = 78), chemotherapy (52.8%, n = 47), T-DXd (38.2%, n = 34), and lapatinib/neratinib (28.1%, n = 25). The median (IQR) follow-up from tucatinib initiation was 11.5 (6.2-18.0) months (Table 1).
Among patients who received the tucatinib combination, 33.7% (n = 30) received this regimen immediately following T-DXd. Median (range) age in this subgroup was 60 (38-84) years, the majority (73.3%, n = 22) had ECOG PS of ≤1, and 20% (n = 6) had brain metastases. In this subgroup, median (IQR) follow-up from tucatinib initiation was 6.7 (3.8-12.5) months.
rwTTD, rwTTNT, rwOS, and Treatment Persistence
Median rwTTD, rwTTNT, rwOS, and Persistence in Patients With HER2 + MBC Receiving Tucatinib With Trastuzumab and Capecitabine After ≥2 Prior HER2-Targeted Therapies in the Metastatic Setting.
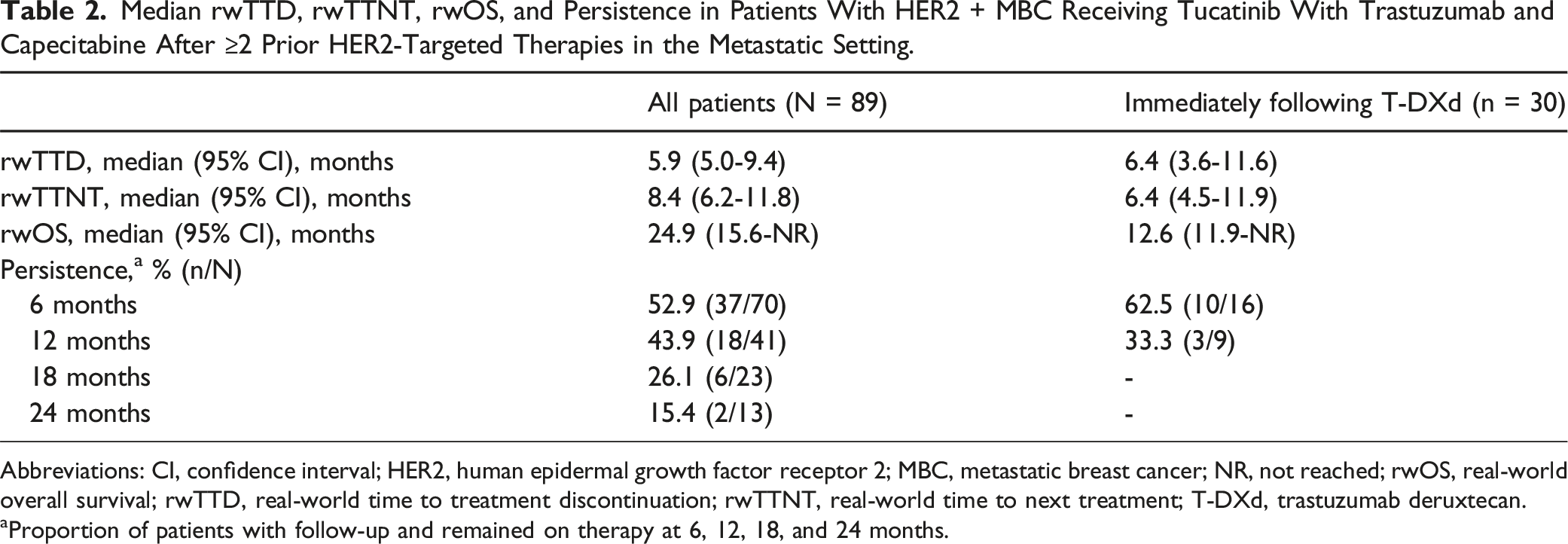
Abbreviations: CI, confidence interval; HER2, human epidermal growth factor receptor 2; MBC, metastatic breast cancer; NR, not reached; rwOS, real-world overall survival; rwTTD, real-world time to treatment discontinuation; rwTTNT, real-world time to next treatment; T-DXd, trastuzumab deruxtecan.
aProportion of patients with follow-up and remained on therapy at 6, 12, 18, and 24 months.

(A) rwTTD, (B) rwTTNT, and (C) rwOS for patients with HER2 + MBC receiving tucatinib in combination with trastuzumab and capecitabine after ≥2 HER2-targeted therapies in the metastatic setting. Abbreviations: HER2, human epidermal growth factor receptor 2; HER2+, human epidermal growth factor receptor 2 positive; MBC, metastatic breast cancer; NR, not reached; rwOS, real-world overall survival; rwTTD, real-world time to treatment discontinuation; rwTTNT, real-world time to next treatment.
Among patients with sufficient follow-up, 44% (n = 18/41) were still receiving the tucatinib combination at 12 months and 15.4% (n = 2/13) at 24 months (Table 2). Persistence was 56.3% (n = 9/16) at 12 months and 12.5% (n = 1/8) at 24 months in the 3L setting, and 36.0% (n = 9/25) and 20.0% (n = 1/5) in the 4L + setting.
In the subgroup of patients who received the tucatinib combination immediately following T-DXd, median (95% CI) rwTTD was 6.4 (3.6-11.6) months, median (95% CI) rwTTNT was 6.4 (4.5-11.9) months, and median rwOS (95% CI) was 12.6 (11.9-NR) months (Table 2). Median (95% CI) rwTTD for the prior T-DXd therapy was 5.9 (4.3-11.1) months.
Discussion
This study demonstrated the real-world effectiveness of the tucatinib combination in patients with HER2 + MBC who received ≥2 prior HER2-directed regimens in the metastatic setting, including in patients who received tucatinib immediately following T-DXd.
Our previous analysis was the first to assess clinical outcomes with tucatinib in a real-world US patient population. 6 As the patient cohort in this study (N = 89) overlaps with that in the previous analysis (N = 216), but restricted to the subgroup of patients with ≥2 prior HER2-directed therapies, the findings in this study add to our prior analysis by providing evidence of the real-world effectiveness of tucatinib specifically after ≥2 HER2-directed therapies in the metastatic setting.
The outcomes with the tucatinib combination in this patient population were numerically comparable with those in the pivotal HER2CLIMB trial, which formed the basis for EMA approval. 11 Patients in this study had a median rwTTD, rwTTNT (a proxy for progression-free survival [PFS]), and rwOS (95% CI) of, respectively, 5.9 (5.0-9.4) months, 8.4 (6.2-11.8) months, and 24.9 (15.6-NR) months, whereas the duration of tucatinib exposure (range), PFS, and OS (95% CI) were, respectively, 7.3 (<0.1-35.1) months, 7.8 (7.5-9.6) months, and 21.9 (18.3-31.0) months in patients in HER2CLIMB. 12 Notably, the population in this study was more racially diverse, and a greater proportion of patients had ECOG PS 2+ (at least 9% vs 0%) and brain metastases (57% vs 48%), although were treated with fewer prior LOTs (median [range]: 3 [2-10] vs 4 [2-14]), compared with HER2CLIMB. 12 This analysis also demonstrated that the real-world effectiveness of tucatinib was similar when administered in a setting consistent with the EMA label as with our previous analysis of patients who received tucatinib as indicated in the FDA label (median rwTTNT [95% CI]: 8.8 [7.1-11.8] months and median rwOS [95% CI]: 26.1 [18.8-NR] months). 6 Patients in this study had lower rwTTD compared with those who received tucatinib as indicated in the FDA label, who had a median rwTTD (95% CI) of 8.1 (5.7-9.5) months; this may be attributed to patients in this study being more heavily pretreated (with a median [range] of 3 [2-10] prior LOTs) than patients receiving tucatinib as indicated in the FDA label. While all patients were included in the survival analyses with censoring incorporated, only patients who had sufficient follow-up were included in the persistence analysis. Treatment persistence largely correlated with survival outcomes in this study, being higher in the overall population from 12 months compared with the sub-population who received tucatinib following T-DXd, with the overall population also having longer rwTTNT and rwOS. Overall, persistence data add to the findings on survival outcomes and provide additional insight into the effectiveness of tucatinib.
In addition, this study supports the effectiveness of tucatinib when used in patients previously treated with T-DXd, with comparable clinical outcomes (rwTTD: 6.4 [3.5-9.4] months, rwTTNT: 8.1 [4.5-11.5] months, rwOS: 13.9 [12.2-NR]) to patients who received tucatinib as indicated in the FDA label as reported in our prior analysis. Given that T-DXd was only recently approved in patients with HER2 + MBC,5,13 this finding is important as there are limited data on the efficacy or effectiveness of other HER2-directed agents after treatment with T-DXd. The different values in this subgroup likely reflect some patients receiving both newly approved therapies (tucatinib following T-DXd) in later LOTs when the disease is more advanced compared with the overall population. This finding is also reflected in the rwTTNT of this subgroup (6.4 [4.5-11.9] months), which was consistent with a median (95% CI) rwPFS of 4.7 (3.9-5.6) months in a prior French cohort study of patients who received the tucatinib combination following T-DXd, 14 in which the median LOT was 5 (compared with 4 in this study). HER2CLIMB was conducted prior to availability of T-DXd; there are a lack of clinical data on the efficacy of the tucatinib combination following T-DXd, which further highlights the value of these studies of real-world effectiveness.
Currently, tucatinib in combination with trastuzumab and capecitabine, and T-DXd, are recommended treatment options for patients with HER2 + MBC in the second line (2L) and 3L settings according to European Society for Medical Oncology guidelines. 11 The present study extends the findings from the HER2CLIMB trial and our prior real-world analysis, demonstrating the durable effectiveness of tucatinib in combination with trastuzumab and capecitabine across multiple LOTs and varying stages of disease progression. Further studies will be beneficial to determine the optimal positioning of tucatinib in the treatment pathway for HER2 + MBC, including sequencing of tucatinib with T-DXd and other HER2-targeted therapies.
Limitations
Limitations of this study include the availability and completeness of clinical records and medical documentation; however, these limitations are inherent to real-world studies of electronic health record data in general. This study was of patients in the United States from the Flatiron Health database with HER2 + MBC receiving tucatinib consistent with the EMA label; therefore, the results may not be fully generalizable to European patients and practices. As this study also included patients who were diagnosed with HER2 + MBC before the approval of tucatinib in the United States, it is likely that a greater proportion of patients received tucatinib-based therapy in later LOTs than more recently diagnosed patients, who would be more likely to receive tucatinib-based therapy in the guideline-recommended 2L or 3L setting. In addition, as it was not possible to reliably identify treatments received in the premetastatic setting in the database, this analysis was limited to patients who received ≥2 HER2-directed therapies specifically in the metastatic setting, which is more conservative than the EMA label indicates. As a result, survival outcomes reported in this study may represent more conservative estimates compared with current real-world practice under the EMA label. Due to the relatively recent adoption of T-DXd in clinical practice, patients included in the post–T-DXd cohort may be those with shorter durations of T-DXd therapy, and outcomes with tucatinib in this group may not be representative of all patients receiving tucatinib post–T-DXd.
Conclusions
The clinical outcomes observed in this study were comparable with those in our previously published real-world analysis of patients receiving tucatinib according to the FDA label. This study demonstrated that tucatinib in combination with trastuzumab and capecitabine is an effective treatment option for patients with HER2 + MBC consistent with the EMA label, including for those patients previously treated with T-DXd.
Footnotes
Acknowledgments
Medical writing support was provided by Yasmin Lau, PhD, of Envision Value & Access, a division of Envision Pharma Group, and funded by Pfizer Inc.
Statements and Declarations
Author Contributions
CA: Conceptualization, methodology, manuscript writing, manuscript review and editing.
EN: Conceptualization, methodology, manuscript writing, manuscript review and editing.
KB: Conceptualization, methodology, manuscript writing, manuscript review and editing.
BTP: Conceptualization, methodology, manuscript writing, manuscript review and editing.
AS: Conceptualization, methodology, manuscript writing, manuscript review and editing.
SW: Conceptualization, methodology, data curation, formal analysis, manuscript writing, manuscript review and editing.
PK: Conceptualization, methodology, manuscript writing, manuscript review and editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Seagen Inc., which was acquired by Pfizer in December 2023.
Informed Consent
Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CA declares research funding from AstraZeneca, Caris Life Sciences, Elucida Oncology, Inc., G1 Therapeutics, Incyclix Bio, Lilly, Merck, Nektar, Novartis, Pfizer, Puma Biotechnology, Seagen Inc., Tesaro, and Zion Pharma; a compensated consultant role with AstraZeneca (3/2020-), Athenex (2/2021-2/2023), Eisai (1/2019-), Elucida (9/2020), Genentech (1/2019-), Immunomedics (10/2020-9/2021), Ipsen (2/2019-), Novartis (5/2020-5/2022), Roche, and Seagen Inc. (11/2019-11/2020); honoraria from AstraZeneca, Athenex, Eisai, Elucida Oncology, Inc., Genentech, Immunomedics, Ipsen, Novartis, and Seagen Inc.; and royalties from UpToDate and Jones and Bartlet. EN and BTP are employees of, and own stock in, Pfizer Inc. KB and AS were employees of Pfizer Inc at the time of the study. SW is an employee of Genesis Research, which was a paid consultant to Seagen (acquired by Pfizer in December 2023) in connection with the development of this manuscript. PK reports financial relationships with Amgen, AstraZeneca, Eisai, H3 BioMedicine, Lilly, Macrogenics, Pfizer, Polyphor, Roche/Genentech, and Sanofi.
Data Availability Statement
The data underlying this study are included in the article; further inquiries can be directed to the corresponding author/s.
