Abstract
Background
Percutaneous Hepatic Perfusion (PHP) is a liver directed regional therapy recently FDA approved for metastatic uveal melanoma to the liver involving percutaneous isolation of liver, saturation of the entire liver with high-dose chemotherapy and filtration extracorporeally though in line filters and veno-venous bypass. The procedure is associated with hemodynamic shifts requiring hemodynamic support and blood product resuscitation due to coagulopathy.
Objective
To assess the cardiac safety and subsequent clinically significant sequalae of this therapy.
Methods
Consecutive PHP procedures done at our center between 2010-2022 were assessed retrospectively. Cardiac risk factors, post procedural cardiac enzymes, electrocardiograms, and transthoracic echocardiograms along with 90-day cardiac outcomes were reviewed. All data were reviewed by cardio-oncologists at our institution.
Results
Of 37 patients reviewed, mean age was 63 years and 57% were women. 132 procedures were performed with an average of 3.57 procedures per patient. 68.6% of patients had elevated troponin during at least 1 procedure. No patients were found to have acute coronary syndrome, heart failure, unstable arrhythmias, or cardiac death. No patients had notable echocardiographic changes. 10.8% of patients with positive troponin had asymptomatic transient electrocardiographic changes not meeting criteria for myocardial infarction. One patient had non-sustained ventricular tachycardiac intra-operatively which did not recur subsequently. Three patients died from non-cardiac causes within 90-days. There was no oncology treatment interruption, even in those with troponin elevation. In multivariable analysis, a history of hyperlipidemia was a predictor of postoperative troponin elevation. (P = .042).
Conclusion
Percutaneous Hepatic Perfusion is safe and associated with a transient, asymptomatic troponin elevation peri-operatively without major adverse cardiac events at 90 days. The observed troponin elevation is likely secondary to coronary demand-supply mismatch related to procedural hemodynamic shifts, hypotension, and anemia.
Plain language summary
Percutaneous hepatic perfusion using melphalan in patients with uveal melanoma and liver metastases carries no significant cardiac adverse events.
Introduction
Uveal melanoma (UM) is the most common intra-ocular malignancy in adults, albeit rare, with an age-adjusted incidence of 5.1-6.0 per million. 1 Overall survival at 5-years is a modest 82%. 2 While local disease is potentially curable, approximately 50% of patients will develop metastatic UM (MUM), which has a very poor prognosis. 3 Median survival after detection of MUM is 8-13 months.4,5 The liver is the most common site of first metastasis (93%). 6 When conditions are favorable (disease free interval from primary diagnosis >24 months, <4 liver lesions, absence of miliary disease) an R0 resection (microscopically negative margins) can provide long term survival greater than 2 years. However, only ∼30% of patients undergoing surgery achieve an R0 resection. 7
Systemic therapies have yielded minimal response in metastatic UM. Traditional cytotoxic therapies, Dacarbazine or temozolomide, have very poor response rates, high progression rates, and median OS of 6-12 months.8,9 Molecular targeted therapies have demonstrated minimal response rates, and the few that have provided stability of disease have not translated to prolonged Overall Survival (OS). 10 The most promising systemic therapies to date have been immunotherapies with response rates of 12%–19% and potentially improved OS up to 19 months.11-13 Tebentafusp, a T-cell receptor–bispecific molecule that targets glycoprotein 100 and CD3, is approved for adult patients who are positive for HLA-A*02:01. This drug (tebentafusp) was approved by the FDA in 2022 after a phase 3 trial showed an overall survival in the tebentafusp group of 73% at 1 year vs 59% in the best alternative care control group. PFS was also higher in the tebentafusp group 31% vs 19% at 6 months. It should be noted the median PFS was 3.3 months in the tebentafusp group vs 29 months in the control group, meaning there was about a 2-week median PFS advantage in the tebentafusp group. Additionally, the tebentafusp trial was for treatment naïve patients, first line therapy for metastatic ocular melanoma. Lastly, this drug, now approved by the FDA, is only useful to 50% of patients who are the ones who harbor the correct HLA-A2 phenotype. Updated 3-year survival continued to show a modest benefit for tebentafusp with the median overall survival in the experimental group being 21.6 months vs 16.9 months in the best alternative care/control group. Just slightly less than 5 months PFS benefit. Estimated percent of patients alive at 3 years were 27% in the tebentafusp group vs 18% in the control group, over a doubling of 3-year estimated survival in favor of tebentafusp.14,15
Percutaneous hepatic perfusion (PHP) is a strategy of delivering high dose melphalan to the liver via percutaneous canulation of the arterial inflow and isolation of hepatic venous outflow via specialized percutaneous catheter (Delcath Systems, Inc. Queensbury, NY) with balloon occlusion of the IVC above and below the hepatic veins. A prior randomized control trial on PHP with melphalan vs standard of care chemotherapy yielded significantly improved response rates in the liver (36% vs 2%, respectively) and hepatic PFS (7.0 months vs 1.6 months, respectively). Overall survival was around 10 months in this trial for both arms and deemed likely to be insignificant different due to high crossover in the study. 16
A more recent phase III trial that led to FDA approval for PHP was concluded in 2022. The phase III results showed that PHP was superior to best alternative care (BAC) in terms of disease control rate (DCR), ORR and PFS as follows. The DCR was 73.6% PHP vs 37.5% BAC, PFS was 9.03 months (PHP) vs 3.12 months (BAC) and the ORR was nearly 3 times better in favor of PHP, 36.3% vs 12.5% (BAC).17,18 This led to FDA approval of PHP in August 2023. Grade 3/4 adverse events occurred in 90% of patients peri-procedurally and post-procedure (>72 hrs). There is a high rate of circulating cell sequestration or hemodilution with hemoglobin decrease in 60% and platelet decrease in 74% requiring significant transfusion with a mean of 8.5 units of cryoprecipitate, 3.5 units of fresh frozen plasma, 2.3 units of packed red blood cells, and 7.6 units of platelets. There are less profound, but still significant, effects on liver function peri-procedurally with increased INR in 20%, prolonged aPTT in 25%, AST increase in 20%, and bilirubin increase in 10%. Post-procedure the most significant adverse event is neutropenia, experienced in 86%, frequently requiring bone marrow stimulation in the following weeks. Hypotension on initiation of procedure is common with vasopressors routinely used. Troponin elevation was noted in 9% in this trial, but true myocardial infarction only occurred in 1 of 70 patients.
A prior study at Moffitt Cancer Center from 2008-2013 assessed the general safety outcomes of the procedure when performed on patients with unresectable melanoma and sarcoma to the liver prior to its U.S. Food and Drug Agency (FDA) approval. 19 With PHP’s recent FDA approval in patients with uveal melanoma and metastases to the liver, its use is expected to rise outside of clinical trial settings.
By assessing the acute perioperative cardiac outcomes of this therapy, this study’s purpose is to report on the cardiac profile in patients who have undergone PHP using the second generation catheter. Since troponin elevation can be seen in a variety of conditions including acute coronary syndromes and heart failure, the study aimed to understand the pathology behind troponin elevation in patients treated with PHP in the hope of providing a reference to future studies evaluation this therapy.
Methods
Data on percutaneous hepatic perfusion procedures performed between 2010-2022 at the H. Lee Moffitt Cancer Center & Research Institute were collected in a retrospective fashion. PHP procedures were performed as part of an ongoing trial of a second generation catheter at Moffitt Cancer Center [NCT02678572]. The current study was exempted by the institutional review board due to its retrospective nature (MCC 20896).
Consecutive patients undergoing PHP procedures were included. A series of underlying patient characteristics including cardiac risk factors (age, gender, race, diabetes, hypothyroidism, hypertension, hyperlipidemia, chronic kidney disease, history of known arrhythmias, peripheral vascular disease, coronary artery disease, smoking history), cardiac medication use (at time of procedure), and preceding vitals were collected. Baseline vs post procedural cardiac enzymes, electrocardiograms (ECG), and transthoracic echocardiograms (TTE) along with 90-day clinical outcomes were reviewed. Troponin-T assays were used in our institution for the initial portion of the study. The assay was later changed to conventional troponin-I. The established institutional cutoff values for troponin-T and I were followed to determine an abnormal troponin result. These biomarkers were collected when patients arrived at the post anesthesia care unit, and then every 6-8 hours per the clinical team, until a down-trending troponin value is noted. Patients with no ECG or troponin data were excluded from the analysis. Acute coronary syndrome was defined as acute ST-segment elevation or depression associated with clinical characteristics of chest pain, and a rise or fall in troponin. When angina could not be assessed, echocardiographic evidence of wall motion abnormalities was used for confirmation.
Any abnormal ECGs or TTEs were evaluated clinically at time of treatment with an inpatient cardiology consultation and further reviewed retrospectively by a cardio-oncology physician as part of this study. Clinical outcomes including new post procedure diagnosis of type 1 MI, stroke, and cardiac arrhythmias were collected. Any deaths that occurred within 90 days were reviewed and assessed whether secondary to non-cardiac or cardiac causes. The reporting of this study conforms to STROBE guidelines. 20
Procedural Details
The PHP procedure is performed via arterial chemoperfusion of the liver with melphalan chloride (3 mg/Kg ideal body weight) as a chemotherapeutic agent. The procedure is performed by first obtaining access in the femoral artery with the placement of a sheath (Figure 1). With the assistance of hepatic angiogram, the tip of the microcatheter is adjusted to the hepatic artery, which will be the intended location of the chemotherapeutic infusion. A right internal jugular venous sheath is placed as a blood return catheter. A double balloon venous catheter is placed via the right common femoral vein. The cranial balloon is advanced into the right atrium at which point it is inflated and retracted into the atriocaval junction. The caudal balloon is then inflated in the inferior vena cava with the placement below the hepatic veins, and level of the renal veins. After the placement of all lines, heparin is administered with a goal activated clotting time (ACT) over 400 seconds for the duration of the procedure. Once both balloons are inflated, their positioning is verified by obtaining a venogram through the injection of contrast medium, with the goal of prevention of hepatovenous blood from entering the systemic circulation to minimize systemic melphalan exposure. Catheter fenestrations are present between the 2 balloons which will later allow melphalan rich hepatovenous blood to be aspirated. This double balloon catheter is connected to an extracorporal circulation system, where hepatovenous blood is pumped through a system of hemofiltration cartridges and eventually returned to the systemic circulation via the systemic venous return catheter in the right internal jugular vein. After the melphalan infusion is completed, there is a washout period of approximately 30 minutes during which extracorporeal filtration is continued to ensure clearance of melphalan from the liver. After this, protamine sulfate is administered to reverse the heparin effects. Percutaneous Hepatic Perfusion Procedural Details.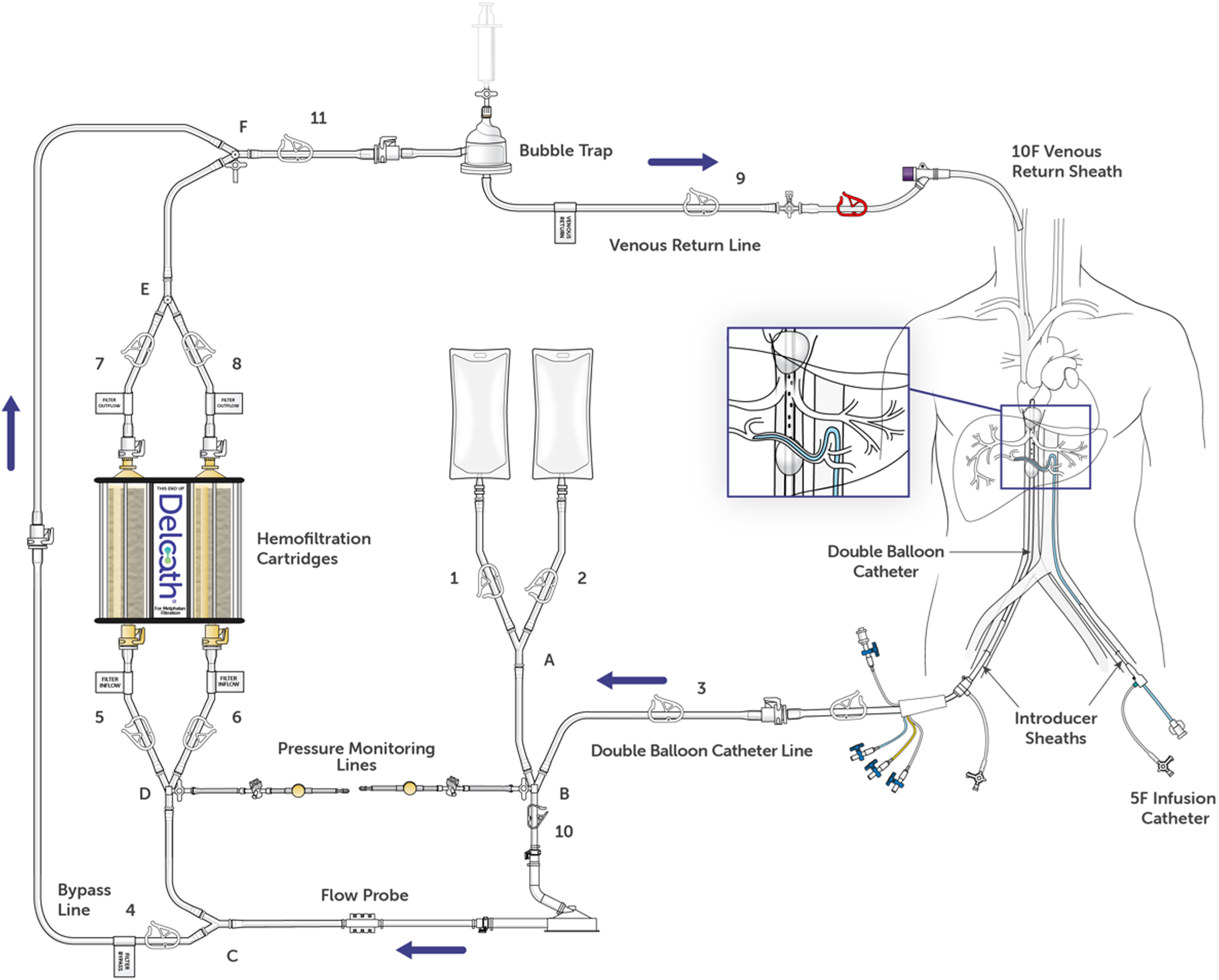
Statistical Analyses
This is a retrospective study of the patients who have undergone PHP procedures between 2010-2022 at H. Lee Moffitt Cancer Center and Research Institute. Descriptive statistics were utilized to present patient and disease characteristics. Categorical variables were denoted as counts and percentages, while continuous variables were presented as medians with their respective ranges. Patients undergoing PHP were classified into 2 cohorts: those with a post-PHP troponin level exceeding .029 ng/mL and those with a level of .029 ng/mL or below. The generalized linear mixed model was utilized to investigate the association between the selected variables and binary outcomes (either elevated or normal post-PHP troponin). To avoid overfitting, we included age and gender in the multivariable model.
Results
Baseline Characteristics.

No patients were found to have acute coronary syndrome (ACS). Mean troponin-T was .1 ng/dL (normal <.029) and mean troponin-I was .49 ng/dL
In the univariable analysis, age (P = .013), a history of hyperlipidemia (P = .011), and current beta-blocker use (P = .042) were found to be associated with higher odds of postoperative elevated troponin levels. Upon multivariable analysis, history of hyperlipidemia (P = .042) remained statistically significant as a predictor of postoperative troponin elevation, after adjusting for age and sex.
An intraoperative drop in mean arterial pressure (MAP) < 50 occurred in 23/68 (33.8%) procedures with available perioperative hemodynamic data (clinical trial procedures). This was found to be not statistically significant on univariable or multivariable analyses.
Three patients (8.1%) died within 90 days from non-cardiac causes (2 from progression of underlying malignancy and 1 from a massive pulmonary embolism). There were no cardiac causes of cancer therapy interruption in any of the 37 reviewed patients.
Discussion
To the best of our knowledge, this is the first study to evaluate the cardiac safety profile of percutaneous hepatic perfusion procedures in patients with uveal melanoma and metastases to the liver. Our study’s main finding is that PHP poses no significant cardiac risks as measured by the lack of major adverse cardiac events in the study’s follow up period. The procedure is associated with a clinically insignificant elevation in troponin during 90 days of follow up. We hypothesize that transient elevations in troponin levels post-procedurally are due to demand related to procedural blood loss, hemodynamic shifts, and low mean arterial pressure, however, this was not linked to a mean arterial pressure below 50 mmHg.
Despite elevated troponin levels, no patients were found to have acute coronary syndrome. There were no notable echocardiographic changes. Importantly, there was no cancer therapy interruption in any of the study’s cohort.
Our study revealed that a statistically significant variable for the development of elevated troponin on both univariate (P = .011) and multivariate analyses (P = .042) was having a history of hyperlipidemia. This is not surprising as hyperlipidemia is a well-recognized risk factor for atherosclerosis. 21
Patients in the elevated troponin group were older, although the difference in age between the 2 groups did not reach statistical significance. This may be due to the small number of patients in our study, rendering it difficult to detect a statistically significant difference.
When compared to women, a higher percentage of men enrolled in the study were found to have an elevated troponin value for any individual procedure (60% vs 45%), though this gender difference was not found to be statistically significant (OR = 2.87, P = .215). Traditionally, men are more likely to have atherosclerotic coronary artery disease, which may explain the observed difference. 22
On review of post procedural ECGs, no sustained malignant arrythmias or ischemic changes meeting criteria for myocardial infarction were noted. Of the patients who underwent post-procedural echocardiograms, no changes in left ventricle ejection fraction or wall motion abnormalities were found. In all patients found to have elevations in troponin-I or troponin-T, rise of cardiac enzymes returned to baseline within 36 hours post-procedure.
Studies have shown that in patients receiving systemic high dose melphalan as many as 11% developed supraventricular tachycardia, most often atrial fibrillation, or atrial flutter. 23 Other studies have shown a 2.7% rate of developing cardiomyopathy as defined by a decrease in left ventricular ejection fraction of >10% to a value of less than 50%. In 1 study, patients who developed heart failure after systemic melphalan responded well to cardiac medical therapy and all recovered normal heart function at follow up. 24 Although other systemic therapies for melanoma have been associated with cardiotoxicity, it was beyond the scope of this study to fully investigate this association. The findings of our study which revealed no patient developed either supraventricular tachycardias or cardiomyopathy demonstrate that PHP was effective in limiting the systemic toxicities from melphalan therapy, at least when viewed from the lens of cardiotoxicity.
As mentioned before these results show that while many patients undergoing PHP may have transient elevations in their troponins, these appear to be clinically insignificant. These findings help to address an important gap in our knowledge as these results and outcomes will help to minimize unnecessary monitoring, testing and procedures for our patients' care, and more importantly, avoid interruptions or delays of cancer therapies.
Study Limitations
The primary limitation of this study is that it comprises a retrospectively collected non-randomized data set. We present data from a single center, Moffitt Cancer Center, which may not be comparable to findings at other cancer centers. Additionally, and most importantly, the low number of patients (n = 37) and the fact that our dataset included multiple samples for each patient limit how widely our data can be interpreted on a larger scale.
Conclusions
Percutaneous hepatic perfusion for uveal melanoma with liver metastases is safe with no significant adverse cardiac effects. Troponin elevation that is observed is likely secondary to demand-supply mismatch related to procedural hemodynamic shifts, hypotension, and anemia.
Clinical Perspectives
Competency in Medical Knowledge
In patients with uveal melanoma and metastases to the liver, percutaneous hepatic perfusion using arterially directed melphalan is frequently associated with asymptomatic troponin elevation, likely due to the hemodynamic shifts caused by the veno-venous extracorporal hemofiltration.
Translational Outlook
Future research with larger trials may be needed to confirm the study’s finding.
Footnotes
Author’s Note
Jonathan Zager: (1) Lead international principal investigator on the FOCUS Phase III trial, (2) Medical advisory board- Delcath Systems.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: None for current study. Original prospective trial was funded by Delcath Systems, Inc. Queensbury, NY.
Ethical Statement
Data Availability Statement
Deidentefied data will be available upon request.
