Abstract
Background
Breast cancer liver metastasis (BCLM) is a severe condition often resulting in early death. The identification of prognostic factors and the construction of accurate predictive models can guide clinical decision-making.
Methods
A large sample of data from the Surveillance, Epidemiology, and End Results (SEER) database was analyzed, including 3711 patients diagnosed with de novo BCLM between 2010 and 2015. Predictive models were developed using histograms, and stepwise regression addressed variable collinearity. Internal validation was performed, and results were compared to similar studies.
Results
In this study of 3711 BCLM patients, 2571 didn't have early death. Out of the 1164 who died early, 1086 had cancer-specific early death. Prognostic factors for early death, including age, race, tumor size, and lymph node involvement, were identified. A nomogram based on these factors was constructed, accurately predicting early all-cause and cancer-specific death.
Conclusions
Valuable insights into the prognosis of BCLM patients were provided, and important prognostic factors for early death were identified. The developed nomogram can assist clinicians in identifying high-risk patients for early death and inform treatment decisions.
Keywords
Introduction
Breast cancer has overtaken lung cancer as the most prevalent cancer in women, with a 2020 incidence rate of 11.7%. 1 Despite advances in medical treatment, breast cancer cells can spread via lymphatic and circulatory systems, leading to metastasis. 2 Encouragingly, the survival rate for breast cancer has surged beyond 90% in recent years, bolstered by novel therapeutic interventions. 3 Yet, nearly a third of patients succumb to distant non-lymph node metastases. On average, 50% of the patients who experience these distant metastases subsequently endure liver metastases, a transition that carries a dismal 5-year survival statistic of just 8.5%.4,5
The term "early death" in cancer patients does not have a standard definition. In research and clinical studies, early death often refers to the death of a patient within a short stipulated time frame after diagnosis or treatment. 6 Referring to a previous study, we define early death in this context as death occurring within 3 months of initial diagnosis. 7 Early death among BCLM patients remains challenging within the breast cancer population, partly due to ineffective treatment options. For instance, immunotherapy involving checkpoint inhibitors shows promising clinical advancements in treating metastatic breast cancer. However, the response rate to PD-1/L1 immunotherapy checkpoint inhibitors is lower in BCLM patients than in patients with metastases in other regions. 8 On the other hand, patients with BCLM tend to have poorer physical and nutritional status than those with non-metastatic breast cancer, which can increase the likelihood of complications during treatment, such as liver failure, persistent ascites, portal vein thrombosis, pain, and malnutrition. This can lead to early death, underscoring the importance of considering cancer stage and characteristics, patient health status, and treatment effectiveness and safety when developing a treatment strategy for BCLM patients. 9
Recognizing the intricate factors contributing to decreased survival outcomes in patients with BCLM is crucial in formulating elevated prevention and treatment tactics. The consequence of early mortality drastically impacts the prognosis of BCLM patients. Yet, notwithstanding its critical essence, there is a paucity of comprehensive research probing the risk factors correlated with this outcome, emphasizing the pressing necessity for a thorough investigation to reveal these pivotal associations.
This study aims to fill a research gap by comprehensively analyzing the clinicopathological characteristics of patients with BCLM. The analysis is based on Surveillance, Epidemiology, and End Results (SEER) database data. This robust analysis successfully pinpointed factors strongly correlated with early mortality in patients with BCLM. In the relentless pursuit of improved patient outcomes, it is imperative to identify these linked factors and predict their incidence with marked precision. Consequently, we instigated the development of a predictive model, specifically for estimating early mortality in BCLM patients, and proceeded to validate this model. This process aimed to augment the precision of early death predictions, thereby furnishing invaluable insights that may positively impact the management and therapeutic tactics for BCLM patients.
Methods
Ethics Approval Statement
All SEER patient data were anonymized, and due to its retrospective nature and patient privacy, this study was terminated with the ethics committee's approval.
Data Source
We conducted a retrospective analysis using the SEER database, which provides demographic, clinicopathologic, and survival data on cancer patients. Our study utilized the SEER Research Plus Data, 17 Registries (2000-2019) version released in November 2021. Data on breast cancer patients were extracted from the SEER database using SEER*Stat software (version 8.4.0). As the SEER database is publicly available and published data do not require patient consent, informed consent was unnecessary for this study.
Patient Selection
This study included breast cancer patients diagnosed between 2010 and 2015, identified using International Classification of Diseases for Oncology, third edition (ICD-O-3) codes for primary breast cancer (C50.0-C50.9) with histological confirmation. In this study, certain patients were excluded based on specific criteria. Those diagnosed based on autopsy or death certificate; those without a histological diagnosis; those with multiple primary tumors; those with Tis, T0, or unknown stage; those with unknown distant metastasis status; and those with incomplete follow-up or anonymous variable information were all excluded from the study. In this study, we analyzed 3711 patients with liver metastasis from breast cancer. To ensure the accuracy of our findings, we randomly divided the patients into two groups: a training group (n = 2,599, accounting for 70%) and a validation group (n = 1,112, accounting for 70%). For a visual representation of our patient selection process, please refer to Figure 1. In this subject paper, we extracted various variables from the SEER database. These variables include age at diagnosis, marital status, race, histology, grade, T stage, N stage, surgical status, radiation status, chemotherapy status, bone metastasis, brain metastasis, lung metastasis, subtype, tumor size, expression of estrogen receptor (ER) and progesterone receptor (PR), cause of death, and survival time. The SEER database collected annual organ metastasis information. This study used publicly available data that did not require informed patient consent. Data selection flow chart for breast cancer liver metastasis patients in the SEER database.
Statistical Analysis
This study examined the relationship between early death in breast cancer patients and demographic, clinicopathological, and survival factors. The patient's baseline characteristics were collected and categorized based on the size and number of metastatic organs. A training cohort was selected randomly, and they developed a nomogram that was then validated. The Pearson chi-square test was utilized to compare the two groups' baseline characteristics. We used logistic regression analysis and backward stepwise selection to identify risk factors associated with early death in BCLM patients. Based on these selected variables, we created early nomograms for all-cause death and cancer-specific prognostic models. To evaluate the performance of our models, we utilized ROC curves on both the training and validation sets, a calibration curve, and the time-correlated area under the DCA. We used R software (version 4.1.3) and SPSS statistical software (version 23.0) for the analysis. We considered a P value less than .05 to be statistically significant.
Results
Characterization of Patients: Demographic and Clinical Features
Demographic and Clinicopathological Characteristics of BCLM Patients With and Without early Death.

Early Death Rates in the Study Population
In a cohort of 3711 breast cancer patients, 1164 (38.4%) died prematurely from various causes, including 1086 from cancer-related causes and 78 from non-cancer-related causes. The most frequently affected organ, other than the primary site, among all patients who died early was bone (65.2%), followed by lung (44.5%) and brain (15.6%). Patients over 60 years of age had a significantly higher early death rate than those under 60 years of age.
Identification of Prognostic Factors Associated With Early Death
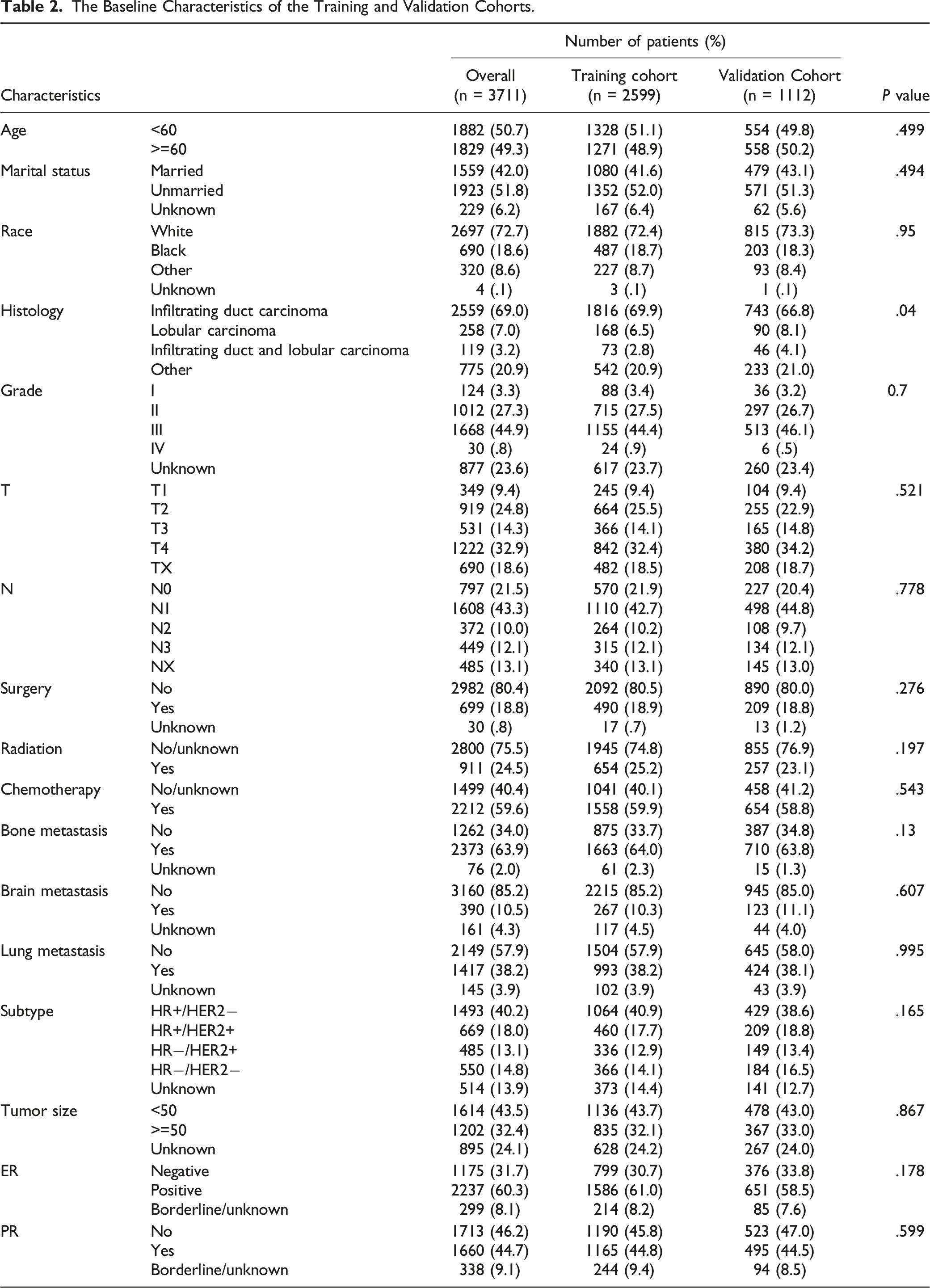
The Baseline Characteristics of the Training and Validation Cohorts.
Univariate Logistic Regression for Analyzing the Prognostic Factors for Early Death.
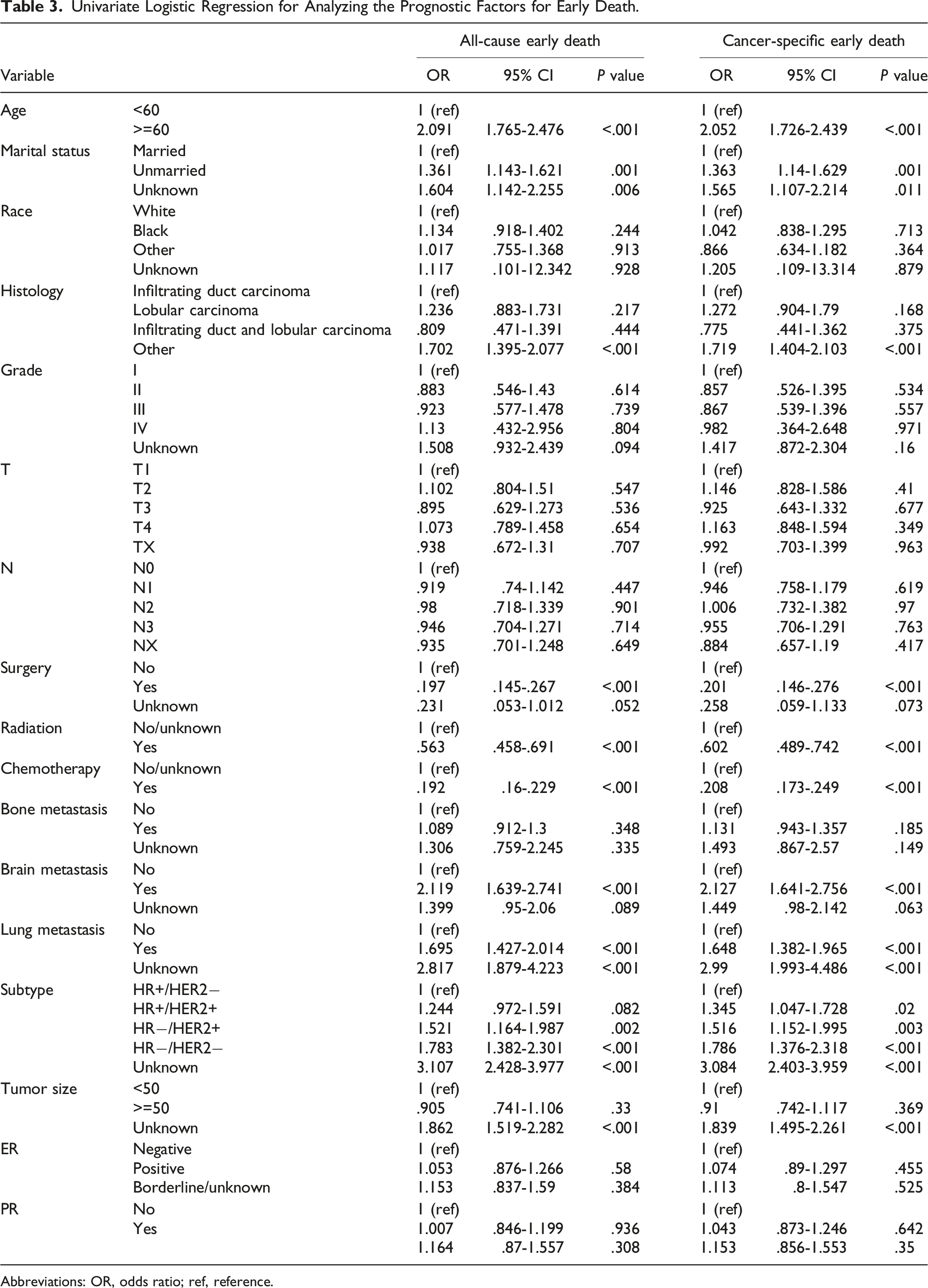
Abbreviations: OR, odds ratio; ref, reference.
Multivariate Logistic Regression for Analyzing the Prognostic Factors for Early Death.

Abbreviations: OR, odds ratio; ref, reference.
Construction of Nomograms
Based on the independent risk factors identified by multivariate logistic regression analysis, two nomograms were developed to predict the risk of all-cause and cancer-specific early death in patients with breast cancer. The risk associated with each factor was assigned a score, and the total score was calculated by adding up the scores of all prognostic factors. The nomograms were constructed using these scores to assist in the prognostic assessment and clinical decision-making (Figure 2(A) and 2(B)). Beneath the probability line, a vertical line is drawn to indicate the probability of early death with coefficients ranging from .1 to .9, although not all points will intersect. The results of the nomogram analysis revealed that surgery was the most crucial prognostic factor in predicting premature death. Nomograms for predicting early death (all-cause in (A) and cancer-specific in (B)) in breast cancer liver metastasis patients.
Nomogram Assessment
The nomogram's ROC (receiver operating characteristic) curves accurately predicted early all-cause death and early cancer-specific death. In the training cohort, the AUCs were .783 and .774, respectively (Figure 3(A) and (A)). In the validation cohort, the AUCs for early all-cause and cancer-specific death were .827 and .825, respectively (Figure 3(C) and (D)). The calibration curves of both nomograms were close to the diagonal, indicating good agreement between the predicted probabilities and the actual observations in both the training and validation cohorts (Figure 4(A)–4(D)). The decision curve analysis (DCA) plots indicated that the nomograms had superior predictive power for all-cause and cancer-specific premature death compared to individual variables, with higher net gains in training and validation groups. Additionally, our findings demonstrate the clinical utility of these nomograms (Figure 5(A)–5(D)). Therefore, these nomograms may be valuable tools for predicting premature death in breast cancer patients. Receiver operating characteristic (ROC) curves for discriminating nomograms in predicting early death (all-cause and cancer-specific) in the training cohort (A, B) and the validation cohort (C, D). Calibration curves for evaluating the calibration of the nomograms in predicting early death (all-cause and cancer-specific) in the training cohort (A, B) and the validation cohort (C, D). Decision curve analysis (DCA) for nomograms in predicting early death (all-cause and cancer-specific) in the training cohort (A, B) and the validation cohort (C, D).


Discussion
Despite the progress made in treating breast cancer, with the development of targeted drugs, optimized surgical approaches, and endocrine therapy, there has been a surge in the incidence of metastases. Liver metastases, the second most common after bone and lung metastases, pose a significant threat to breast cancer patients, with survival rates lower than those for other sites of metastasis. 10 Identifying prognostic factors associated with early death in patients with liver metastases from breast cancer is crucial to recognize those at higher risk of premature death. Hence, attention should be given to the early death of these patients to improve their prognostic evaluation and clinical management. Many studies have explored early death in breast cancer patients, but there are no studies on early death in breast cancer patients with liver metastases.11-13 This study aims to address this gap by exploring prognostic factors and constructing a columnar plot to identify early death in breast cancer patients with liver metastases. This represents the first such study to our knowledge. The results of this study will inform the development of effective treatment strategies for older breast cancer patients at increased risk of early death due to liver metastases.
In this study, among 3711 patients, 2571 did not experience early death. Of the 1164 patients with early death, 1086 were categorized as having cancer-specific early death. This indicates the importance of timely oncologic treatment, particularly for patients with hepatic metastatic breast cancer. Additionally, the significant increase in the proportion of cancer-specific deaths in this study suggests that patients with metastatic liver breast cancer are more likely to die from cancer. These findings highlight the need for personalized treatment strategies and close monitoring of this patient population. It is worth noting that a previous study reported a lower percentage of cancer-specific deaths among patients with stage IV breast cancer who died within one year, with only 80.78% of deaths being attributed to cancer. 12 Further research is needed to fully understand the factors contributing to early death in breast cancer patients with liver metastases.
After analyzing the data, we identified several variables independently associated with all-cause early death and cancer-specific early death in breast cancer patients with liver metastases. Our findings indicate that patients over 60 who have concomitant brain and lung metastases and have the HR−/HER2− subtype are at a higher risk for early death. However, we also found that patients who underwent surgery, radiotherapy, and chemotherapy had a lower risk of early death. These findings highlight the importance of early detection and timely treatment to improve outcomes for breast cancer patients with liver metastases.
In treating BCLM, various interventions, including surgery, radiotherapy, and chemotherapy, have reduced patients' risk of early death. Hepatectomy, radiofrequency ablation, and transcatheter arterial chemoembolization are surgical treatments used in BCLM patients.14,15 Among these, hepatectomy has been shown to provide high survival benefits. A retrospective study showed that the median survival of 85 BCLM patients who underwent surgery was 32 months, better than that of patients treated with medications only (15 months). 16 In another clinical study, 460 BCLM patients who underwent hepatectomy had a median survival of 45 months. 17 However, the clinical evidence supporting hepatic resection as a standard treatment for BCLM is still lacking, especially for patients with extrahepatic metastases. 18 Based on the data from a retrospective study of 167 patients, no statistically significant difference in overall survival was observed between patients who received surgical treatment and those who received medical therapy. Therefore, patients should be evaluated for eligibility before performing hepatectomy, considering factors such as limited visceral tumor load, a long disease-free interval between primary breast and BCLM diagnosis, and effective systemic treatment for HER2-enriched and triple-negative subtypes. 19 Surgical treatment should be cautiously approached for patients not meeting these criteria.
Our study findings suggest that radiation therapy, mainly external radiation therapy (EBRT), may be beneficial in reducing the risk of early death. Selective internal radiation therapy (SIRT), which involves injecting yttrium 90 (90Y) microspheres into the tumor tissue via the hepatic artery, has also been proposed as a treatment option for BCLM.20,21 However, our data analysis only includes EBRT, and previous clinical studies have indicated that EBRT is more effective in patients with small liver metastases. On the other hand, SIRT may lead to increased levels of proteases that promote tumor progression, ultimately leading to early death in patients with BCLM. 22 Therefore, both EBRT and SIRT require further multidisciplinary and large-scale studies to determine their accurate indications for treatment in patients with BCLM.
Chemotherapy is a significant prognostic predictor in BCLM treatment. It should only be given to patients in good general health who can tolerate chemotherapy's adverse effects. 23 Chemotherapy treatment for BCLM requires close monitoring of patients, including patient-reported symptoms, physical status, weight changes, laboratory tests, and radiographic imaging. Clinicians should evaluate and consider various forms of information to minimize the risk of early death.
Based on our findings, we observed that patients aged over 60 years with BCLM have a higher likelihood of experiencing early death. This could be attributed to age-related decline in immune function and coexisting health issues such as diabetes, heart disease, or kidney disease, which can complicate cancer treatment. 24 Furthermore, elderly cancer patients may have poor self-care and social support, negatively impacting their ability to cope with cancer treatment's physical and emotional challenges. 25 Therefore, treatment strategies for older patients should be individualized and tailored to their specific needs and health status. This may involve modifying the dose or frequency of chemotherapy, considering less toxic treatment options, or prioritizing supportive care to manage symptoms and enhance the quality of life, ultimately decreasing the risk of early death. It is imperative to recognize the unique challenges faced by elderly patients with BCLM and provide comprehensive, patient-centered care to optimize clinical outcomes.
Various factors can influence cancer patients' prognosis, including age, cancer type, stage, individual health status, and treatment response. Although older cancer patients may face challenges related to reduced immune system function and other health problems, it is essential to recognize that each patient is unique, and prognosis may vary accordingly. Therefore, it is crucial to consider individual patient characteristics when developing treatment plans and assessing prognosis. Clinicians should work closely with patients to understand their needs, goals, and preferences to provide personalized care that optimizes quality of life and reduces the risk of early death.
In HR−/HER2− subtype, the risk of early death is positively associated. TNBC is a subtype of breast cancer that lacks clinical biomarkers for endocrine or HER2-targeted therapy, and thus, chemotherapy remains the standard of care. 26 However, TNBC is also associated with a high incidence of liver metastasis, and patients with TNBC and liver metastasis have a poorer prognosis than patients with other subtypes. This may be due to the upregulation of genes related to angiogenesis and extracellular matrix in the microenvironment of liver metastases in TNBC, which could contribute to the higher risk of early death in these patients. 27 However, TNBC is also associated with an increased incidence of liver metastasis, and patients with TNBC and liver metastasis have a poorer prognosis than patients with other subtypes. This may be due to the upregulation of genes related to angiogenesis and extracellular matrix in the microenvironment of liver metastases in TNBC, which could contribute to the higher risk of early death in these patients. 28 It is important to note that the prognosis can vary depending on the individual patient's circumstances, such as the stage and type of cancer and their response to treatment.
Our study has confirmed that patients with BCLM who have brain, lung, and bone metastases are at a higher risk of early death. This finding is consistent with a previous study that showed breast cancer patients with bone metastases had the best survival outcome, while brain metastases had the worst prognosis. 29 Although bone metastases can cause pain, decreased physical status, and skeletal-related events, they are generally considered less aggressive than brain or lung metastases. 30 Moreover, the limited permeability of the blood-brain barrier remains a challenge in developing effective treatments for brain metastases from breast cancer. 31 Therefore, the presence of brain and lung metastases should be closely monitored when deciding on treatment approaches for patients with BCLM.
Nomograms have become essential for providing personalized prognostic information to breast cancer patients and their physicians. Nomograms use quantitative data to visually represent predictive models, helping clinicians and patients make informed treatment options and decisions. 32 In this study, we developed a column line chart to predict early death in breast cancer patients with BCLM. Our study utilized a comprehensive and high-caliber sample from the SEER database, incorporating other widely accessible prognostic factors in medical records. Despite several similar studies recently published, our research possesses distinctive features. For example, Aldrick Ruiz et al 33 developed a nomogram to predict post-hepatectomy survival in patients with BCLM, incorporating factors such as post-hepatectomy targeted therapy, post-hepatectomy hormonal therapy, vascular invasion, and triple-negative status.
In contrast, our study focused on early death in BCLM patients and employed stepwise regression to eliminate multicollinearity between variables, resulting in a more convincing prediction model. Molecular subtypes also influence the risk of liver metastasis in breast cancer patients. Molecular subtypes have been found to impact the risk of liver metastasis in breast cancer patients. Studies have shown that molecular subtypes are crucial in determining the risk of liver metastasis in breast cancer patients. In a recent study, Yue et al developed a nomogram to evaluate the impact of multiple factors on the probability of liver metastasis in breast cancer patients with the N + Luminal B (HER2−) subtype. By utilizing this nomogram, clinicians can assess the likelihood of liver metastasis in these patients, allowing for more informed decisions regarding treatment options. 34
While the study provides valuable insights, it is important to acknowledge several limitations. First, the SEER database lacks information on disease recurrence or subsequent sites of involvement, limiting our analysis to patients with de novo BCLM. Second, excluding some cases due to missing data may have introduced selection bias. Third, our nomograms were only validated internally; further prospective studies are needed to validate our findings externally. Thus, the study focuses on the short-term prognosis of patients with BCLM. Finally, in multifactor analysis, reverse stepwise regression is employed to perform regression. This method begins with all predictors in the model and then iteratively eliminates variables based on their statistical significance. However, this approach may exclude significant predictors from the final model, losing valuable information and potential predictive power. These limitations may have led to study bias and weakened the efficacy of the analysis.
Conclusion
BCLM is a devastating condition associated with high early death rates. In this study, we aimed to develop a predictive model for early death in BCLM patients using the SEER database. Our study found that brain and lung metastases, in addition to bone metastases, were independent predictors of cancer-specific and all-cause early death in patients with BCLM. We developed a column line chart incorporating additional prognostic factors that are meaningful for early death in BCLM patients and easily accessible from the medical record. Our convincing prediction model is developed based on stepwise regression to eliminate multicollinearity between variables.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Postgraduate Innovation Research and Practice Program of Anhui Medical University (No. YJS20230086).
Data Availability Statement
The data sets used in this study were obtained from the publicly available SEER database, which can be accessed at ![]() .
.
