Abstract
Objective
To develop a prognostic model for optimizing management of colorectal liver oligometastases (CLOM) patients with different primary tumor locations who underwent thermal ablation (TA).
Materials and Methods
The reporting of this retrospective study conforms to STROBE guidelines. A total of 525 CLOM patients who underwent TA from 3 hospitals between 2011 and 2021 were enrolled. Firstly, intra and extrahepatic disease-free survival (DFS) and overall survival (OS) for CLOM patients with different primary tumor locations were analyzed. Then, cox regression models were used to identify independent factors predicting OS. Finally, a prognostic score was developed to identify CLOM patients benefiting from TA. All patient details were de-identified.
Results
A total of 423 eligible patients were identified, with 762 CLOM (121 male, median age 59 years) and a median follow-up of 45.8 (IQR, 7.3-114.8) months. Independent predictors of OS were identified, including multiple liver metastases (P = .0085), right-sided colon cancer (P = .0210), tumor size ≥2 cm (P = .0273), and lymph node metastasis of primary colorectal cancer (P = .0302), termed as the “MRSL” score. On the basis of the best separation of MRSL score, patients were divided into high-risk (cutoff value ≥8) and low-risk groups (cutoff value <8). Further stratified analysis indicated that right-sided CLOM patients had shorter OS than left-sided patients in the high-risk group (54.9 vs 92.5 months, P = .0156). However, no significant difference in OS was observed between right-sided and left-sided CLOM patients in the low-risk group (97.7 vs 102.2 months, P = .28).
Conclusion
The MRSL score-based model helps in selecting potential right-sided CLOM patients who benefit from TA.
Keywords
Introduction
Colorectal cancer (CRC), ranking third in incidence and second in cancer-related mortality worldwide,1,2 represents a major health concern. About half of CRC patients will develop liver metastases during their disease course,3-5 significantly elevating mortality rates. Colorectal liver metastases (CLM) appears to be a serious disease progression, but for those presenting with colorectal liver oligometastases (CLOM)—a state of limited metastatic tumor burden—there is potential for cure, 6 a prospect that has garnered increasing interest in CLM research.
Although systemic therapy is the default treatment strategy and often the first line of defense, for the majority patients with CLM who received palliative chemotherapy, the median overall survival (OS) is approximately 24-30 months, with a 5-year OS of 10%. 7 Recently, accumulating evidence suggests that aggressive local treatments, such as thermal ablation (TA) or hepatic resection (HR), can significantly prolong survival in CLOM patients.8-10 Especially, ultrasound-guided microwave ablation (MWA) is a new percutaneous TA procedure that has similar benefits of the radiofrequency ablation (RFA), perhaps it has more theoretical technique advantages, such as a greater volume of cellular necrosis and higher temperatures delivered to the target lesion. 11 In addition, recent studies have revealed that for tumors ≤3 cm, TA has a comparable effect to HR, providing not only complete ablation but also minimally invasive procedures and fewer postoperative complications.12-14 Consequently, TA for liver metastasis is now recommended in both US and European guidelines for radiologic management of CLOM.15-19
However, despite current guidelines, some questions persist, 20 including the definition of biological phenotype for the oligometastatic in colorectal cancer, the selection of the patients for potentially benefit from TA. Therefore, optimize patient selection for TA is an urgent priority. Existing research establishes primary tumor location as a crucial factor in determining CLOM treatment outcomes due to disparate molecular characteristics.21,22 Patients with right-sided colon cancer, in particular, have been associated with worse survival outcomes post-TA and HR.23,24 But these findings primarily draw from heterogeneous cohorts with extrahepatic metastases, leaving a gap in prognostic assessment for right-sided CLOM patients without extrahepatic metastasis post TA therapy. This raises the question: might some right-sided CLOM patients stand to benefit from TA.
To address this knowledge gap, this study only included CLOM patients without extrahepatic metastasis who have undergone complete radiographic TA. The aim of this study was to develop a prognostic evaluation model to help identify right-sided CLOM patients who may significantly benefit from TA.
Study Design and Population
In this retrospective cohort study, patients with CLOM treated via ultrasound-guided MWA or RFA were included from January 2011 to October 2021 across 3 central hospitals in China. Of the 525 CLOM patients screened, 423 eligible patients with 762 CLOM were selected for inclusion (121 male, median age 59 years). The study workflow is presented in Figure 1. The institutional review boards of these hospitals approved this study (Approval number: S2023-266-01), and the requirement for informed consent for data publication was waived due to the retrospective data collection and anonymization protocols. The reporting of this retrospective study conforms to STROBE guidelines.
25
Flowchart diagram.
Inclusion and Exclusion Criteria
To be included, patients needed to meet the following criteria: (1) pathologically confirmed CRC and primary site resection; (2) pathological diagnosis or at least 2 imaging diagnoses of liver metastases; (3) CRC oligometastasis with maximum tumor size (diameter) ≤3 cm and ≤5 liver metastases; (4) not candidates for surgery or refusal of surgery for potentially resectable lesions; (5) percutaneous TA of liver metastases (RFA or MWA); (6) minimum 6 months of peri-operative chemotherapy, with oxaliplatin, irinotecan, and fluorouracil as the primary systemic treatment drugs (Detailed information were shown in Supplementary Table 1); (7) stable tumor disease; (8) age >18 years; (9) ECOG score of 0 to 2; and (10) expected survival time of >6 months (Refer to Figure 1).
Exclusion criteria encompassed: (1) patients with primary intestinal tumors at unclear locations or presenting with 2 primary sites of colorectal tumors; (2) co-occurrence of other malignancies; (3) the presence of extrahepatic metastases or recurrence at the primary intestinal site; (4) liver metastases with a maximum tumor size (diameter) of >3 cm or >5 liver metastases; (5) tumors of indeterminate size or number; (6) undergoing cryoablation or other ablation treatments; (7) TA combined with TACE or incomplete TA followed by surgical resection; (8) local tumor progression (LTP) within 1 month post-TA; (9) incomplete TA or failed TA therapy; or (10) loss to follow-up (Refer to Figure 1).
TA Procedures and Quality Assurance
MWA or RFA were performed using Sequoia US system (Acuson, Mountain View, CA, USA) or GE systems (E9, GE, United States), with 3.5-5.0 MHz linear multifrequency transducer. A cooled-shaft microwave system (KY-2000; Kangyou Medical, China) was used in the TA procedures. Details information . presented in the previous studies. 26 All percutaneous hepatic TA procedures were performed using real-time ultrasonic imaging system to ensure adequate ablation. In addition, percutaneous ultrasound guided bipolar TA was applied due to complex anatomical location of liver metastasis. In addition, considering the influence of hospital volume and surgeon volume on the quality assurance of TA procedures, 27 all percutaneous hepatic TA procedures were executed by experienced interventional radiologists with a minimum of 5 years in the field. Each of the 3 participating medical centers carries out over 200 liver metastasis TA procedures annually. All the TA procedure were carried out in accordance with the International Society of Multidisciplinary Interventional Oncology’s consensus for liver ablation.
Follow-up and post-TA assessment of tumors involved contrast-enhanced magnetic resonance imaging or CT conducted 4 weeks post RFA or MWA, establishing a baseline for subsequent imaging and assessing the procedure’s efficacy. Complete tumor TA, defined as an ablation area that wholly encapsulates the targeted tumor with ablative margin of at least 5 mm (Figures 2(A)–2(D)), was used as the benchmark to determine the success of liver metastasis TA. Images in a 65-year-old-man who underwent MVA. (A) Two-dimensional gray scale US image before MWA showing indicator tumor (arrow). (B) Contrast-enhanced ultrasonography (CEUS) image before MWA shows that the index tumor (arrow). (C) Axial MRI T2-weighted images shows a 2.3-cm lesion of high signal intensity (arrow) in segment IV before MWA. (D) The patient underwentMWA and obtained technical effectiveness 1 month after MWA. MWA. Microwave ablation; CRLM, colorectal liver metastases; US, ultrasound.
Follow-Up and Data Collection
Patients were tracked from the start of liver metastasis TA through telephone assessments and reviews of medical records. The follow-up endpoint was set for October 1, 2022, or until the event of death or loss to follow-up.
Follow-up assessments were scheduled every 3 months during the first 2 years post-treatment and then every 6 months thereafter. The median follow-up period extended to 45.8 months, ranging from 7.3 to 114.8 months. Records of LTP, intra- and extra-hepatic disease-free survival, and OS were meticulously maintained (Refer to Supplementary Text 1).
Statistical Analysis
To address the missing data typically present in retrospective studies, multiple imputation based on 5 replications and a chained equation approach via the RMI procedure were performed. Variables with over 30% missing values were excluded from the analysis, including the Rat sarcoma viral oncogene (RAS) mutation status. A sensitivity analysis of multiple imputation was assessed to minimize sources of bias and heterogeneity.
Baseline variables differences between groups with CLOM of different primary tumor location were analyzed. Categorical variables are expressed as percentages and were compared using chi-square and Fisher’s exact tests. Continuous variables are expressed as median with interquartile range (IQR) and were compared using Student’s 2-sample t test or ANOVA, as appropriate.
Sample size calculation is provided in Supplementary Text 2. Intra- and extra-hepatic DFS and OS for CLOM of distinct primary tumor location post-TA were compared by log-rank test . The Cox regression models were used to analyze both unadjusted and adjusted cohorts (eg, peri-ablation chemotherapy pattern and recurrent liver metastasis). Univariate and multivariate analyses of intrahepatic recurrence, extrahepatic recurrence, and OS were conducted. Then, variables with values of <0.1 in the univariate analysis or with clinical significance for prognosis were included in the final multivariate analysis to construct a multidimensional score-based prognostic model. And, the risk score formula for each patient was weighted by its estimated regression coefficient on COX regression analysis. On the basis of the best separation of the MRSL score, patients were divided into high-risk-score (the cutoff value ≥8) and low-risk-score groups (the cutoff value <8).
To evaluate the predictive performance of the model, the area under the receiver operating characteristic (ROC) curve (AUC) was performed by R software, then, the internal bootstrap method was used to verify the model. Additionally, individual prediction nomogram and KM curve analysis were assessed in this study. And, the Logarithmic likelihood ratio test was used to evaluate the interaction analysis between covariates and MRSL score by R software.
E values are an approach to exploring the sensitivity of non-experimental study results to unobserved confounding factors. To address unobserved confounding, the robustness of the results of the primary analyses to unmeasured confounding was assessed using E-values.
All statistical analyses were executed using SPSS (version 20.0; IBM, Armonk, NY, USA) and R software (version 4.0.2). Moreover, all statistical tests were two-tailed, with statistical significance established as P < .05.
Results
Study Design and Workflow Overview
Of the total 525 patients with CLOM screened, 89 were excluded due to various reasons, leaving 436 patients. Next, after excluding 13 due to loss to follow-up, 423 patients were finally included in the study (Figure 1).
Of the 423 eligible patients with 762 CLOM (121 male, median age 59 years), 335 were diagnosed with CLOM of left-sided colon rectum cancer (mean age: 58.23 ± 11.70 years, range: 25 to 89), and 88 with CLOM of right-sided colon cancer (mean age: 57.41 ± 11.47 years, range: 18 to 84). The median follow-up periods for patients with CLOM of left-and right-sided colon cancer were 48 (range: 9.3 to 114.8) and 40.3 (range: 7.3 to 85.8) months, respectively. Complete tumor TA was confirmed through reexamination 1 month after TA, with a success rate of 98.3% (516/525).
Baseline Characteristics of CLOM Patients in Different Primary Tumor Location.

Note.—Data for continuous variables are median with interquartile range (IQR), and data for categorical variables are number of patients (%). NA stands for missing data. DFI = disease-free interval from primary resection to the diagnosis of liver metastasis.
Baseline Characteristics of Treatment for CLOM Patients in Different Primary Tumor Location.
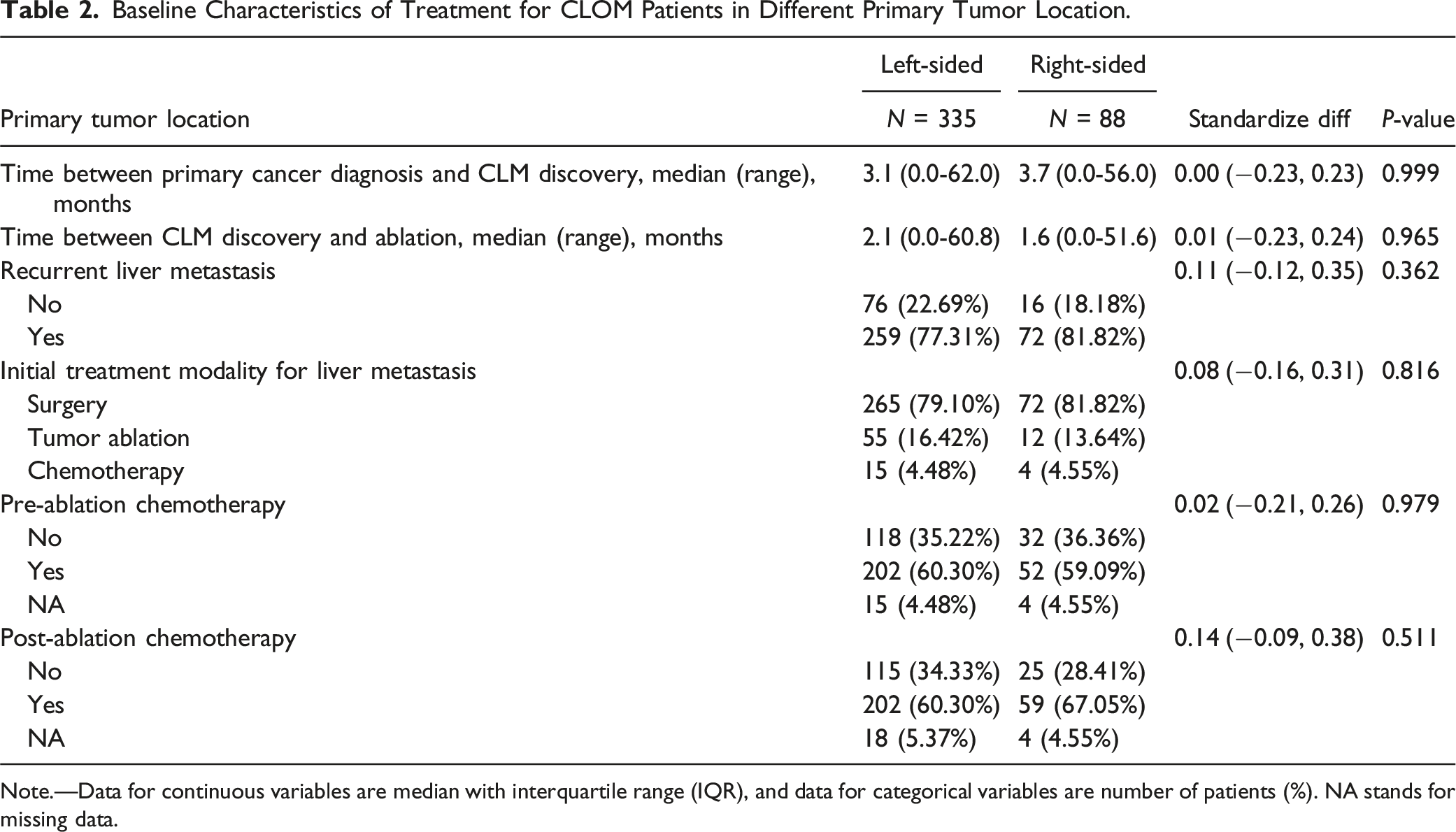
Note.—Data for continuous variables are median with interquartile range (IQR), and data for categorical variables are number of patients (%). NA stands for missing data.
In addition, the results found no significant differences in the baseline characteristics of the patients with CLOM selected from the 3 centers (Supplementary Table 2). Also, no significant differences in OS were observed between the patients with CLOM from the 3 centers.
Analysis of Missing Variables in Baseline Characteristics of Patients
The results indicated no significant difference between the CLOM patients of different primary tumor location in the RAS missing data group (Supplementary Table 3). In particular, the results noted no significant difference in OS between the 2 groups (Supplementary Figure 1).
For other variables with less missing information, applying multiple imputation method, the sensitivity analysis revealed a coefficient of 0.47 and β value of 1.61 (95% CI: 1.03 to 2.51; Supplementary Table 4). Moreover, the results noted no significant differences in baseline characteristics between patients with complete data and those with imputed data (Supplementary Table 5).
Clinical Outcomes of CLOM of Distinct Primary Tumor Locations Post-TA
First, for the incidence of LTP, the results revealed no significant difference in cumulative LTP between the patients with CLOM from left-sided and right-sided (12.01% vs 13.64%, P = .68).
Comparison of Median Survival Time and 1-Year, 3-Year, and 5-Year Survival Rate of CLOM Patients in Different Primary Tumor Location.

Note.—DFS = disease-free survival.
Finally, using KM curve analysis ,the 1-year, 3-year, and 5-year OS rates of the patients with CLOM of right-sided were 98.7%, 87.2%, and 72.8%, respectively, and those of the patients with CLOM from left-sided were 99.4%, 80.1%, and 59.9% (P = .007, .041, and .066), respectively (Supplementary Figure 3A). Using landmark analysis, there was still a statistical difference between the 2 groups, except for CLM who died within 2 years (Supplementary Figure 3B).
Univariate and Multivariate Cox Regression Analysis of Clinical Outcomes Post-TA
Univariate and Multivariate OS Analysis of CLOM Patients.

Non-adjusted model adjust for: None; Adjust I model adjust for: Recurrent livermatas; Initial treatment modality; Adjust II model adjust for: Age; Pre-ablation chemotherapy; Post-ablation chemotherapy.
Additionally, factors influencing intra- and extra-hepatic recurrence were further examined, and the detailed results were presented in Supplementary Tables 6 and 7
Construction and Evaluation of the MRSL Prognostic Scoring Model
A prognostic scoring model, named the MRSL (multiple liver metastases, right-sided colon cancer, tumor size ≥2 cm and lymph node metastasis of primary CRC), was constructed for predicting OS. And the MRSL score model can be expressed as: 0.51*(Multiple liver metastases) + 0.52*(Right-sided colon cancer) + 0.59*(Tumor size ) + 0.32*(Lymph node metastasis of primary CRC).
Using time-dependent Receiver Operating Characteristic (ROC) analysis the Area Under Curve (AUC) values for OS at 3, 5, and 8 years were 0.771, 0.780, and 0.757, respectively. The stability of this model was further validated through internal bootstrap verification (Figure 3(A)). Evaluation of the MRSL scoring model. (A) Time ROC analysis of the MRSL scoring model. (B) Nomogram analysis of the MRSL scoring model. (C) KM curve analysis of CLOM patients between low-and high risk MRSL score in the total population. (D) KM curve analysis of CLOM patients between low- and high-risk MRSL scores in the left-sided colon rectum cancer patients. (E) KM curve analysis of CLOM patients between low- and high-risk MRSL scores in the right-sided colon cancer patients.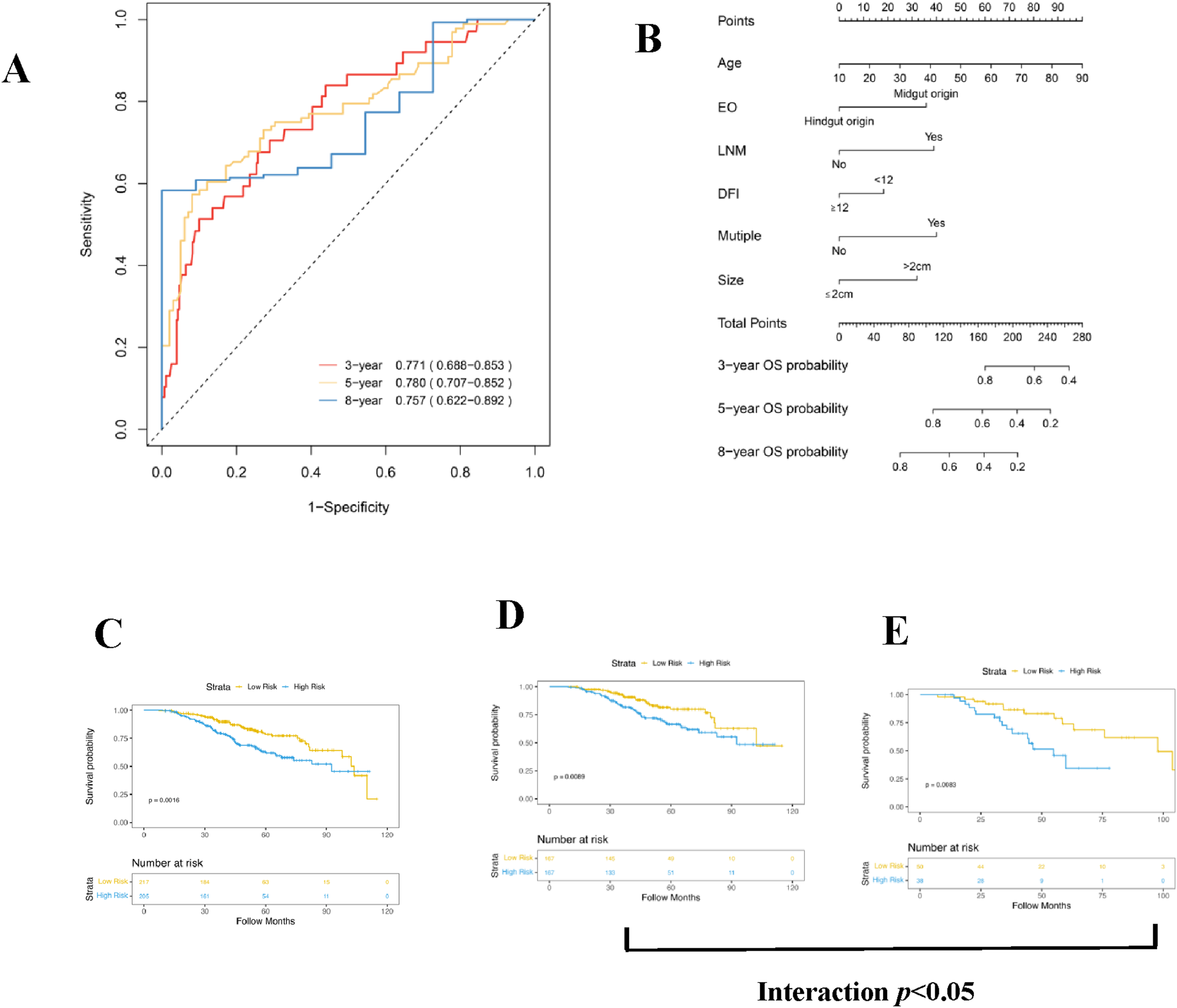
The robustness of the prognostic model was further confirmed, using a nomogram and Kaplan-Meier (KM) curve analyses (Figures 3(B), (C) and (E)). The KM curve showed that the low-risk group, as per the MRSL score, had a longer OS compared to the high-risk group in the total population (Figure 3(C)). Similar results were observed for patients with CLOM from both left- and right-sided colon cancer (Figures 3(D),(E)). An interaction was observed between the primary tumor location and the MRSL score (Figures 3(D), and (E)), and all differences were statistically significant.
Stratified Analysis Based on the MRSL Scoring Model. The results of stratified analysis showed that, in the high-risk group, patients with CLOM from left-sided colon rectum cancer exhibited significantly longer OS post-TA compared to those with CLOM from right-sided colon cancer (Figure 4A). However, no significant difference in OS post-TA between patients in the low-risk group (Figure 4B). Stratified analysis of KM curve was performed according to MRSL scoring model. (A) Comparison of OS between the patients with CLOM in the high-risk group. (B) Comparison of OS between the patients with CLOM in the low-risk group.
The Mode of Recurrence Underlying the Poor Prognosis of High-Risk Patients with CLOM
The mode of recurrence contributing to the poor prognosis observed in high-risk CLOM patients were further investigated, by examining intrahepatic and extrahepatic recurrence rates. The analysis showed no significant difference in the risk of intrahepatic recurrence between CLOM patients from different primary tumor locations (Supplementary Figure 4A). Nevertheless, patients with CLOM from right-sided colon cancer displayed a higher rate of extrahepatic recurrence compared to those with CLOM from left-sided colon rectum cancer (Supplementary Figure 4B), a difference that was statistically significant.
Further investigation of the sites of extrahepatic recurrence revealed a higher likelihood of lung metastasis in patients with CLOM from left-sided colon rectum cancer, whereas abdominal and pelvic metastasis were more commonly observed in patients with CLOM from right-sided colon cancer (Supplementary Table 8).
Discussion
Current European and US radiologic management guidelines endorse the use of TA for patients with colorectal liver oligometastasis (CLOM). NCCN guidelines also suggest a category 2A recommendation for TA in patients with oligometastatic disease. 11 However, patients with CLOM from right-sided colon cancer tend to exhibit poorer survival rates. The main aim of this study was to provide a comprehensive understanding of the prognostic factors influencing survival outcomes in right-sided CLOM patients undergoing TA. This endeavor could potentially facilitate the development of personalized treatment strategies, optimize patient selection for TA, and ultimately enhance the management of this patient subgroup.
Few studies have specifically explored patients with CLOM from distinct primary tumor locations post-TA. However, their study included a small sample size (74 patients) and incorporated CLOM patients with extrahepatic metastases, 24 which may have biased their results. Given that those previous studies have focused on heterogeneous cohorts with extrahepatic metastases, the potential benefits of TA for specific CLOM patients with right-sided colon cancer necessitates further exploration. Hence, this multicenter study addressed the issue by excluding patients with CLOM who also had extrahepatic metastases. By adhering to these strict inclusion and exclusion criteria, a multidimensional risk stratification for liver metastasis scoring system was developed to identify right-sided colon cancer patients most likely to benefit from TA.
The results demonstrated differences in the prognosis of CLOM patients from different primary tumor locations over a 10-year follow-up period. Hence, there’s a need for further refinement of management strategies for CLOM patients to determine their potential benefit from TA. The study additionally provides a comprehensive evaluation of the overall efficacy of TA for CLOM patients by distinguishing between intrahepatic and extrahepatic recurrence. These findings contribute valuable insights for post-ablation management strategies.
One of the strengths of this study is its pioneering role in providing a prognostic risk stratification for CLOM from right-sided colon cancer post-TA. The multidimensional MRSL scoring system takes into account recurrence risk, 28 tumor burden, 29 and the primary tumor location in colorectal cancer. This could potentially improve decision-making accuracy regarding the appropriateness of TA for patients with CLOM from different primary tumor locations. In light of the observed interaction between the primary tumor location in colorectal cancer and the MRSL score, both these factors should be incorporated into the decision-making process when choosing an appropriate therapeutic approach (TA) for CLOM patients
Another strength of this study is its exploration of potential reasons for poor outcomes in high-risk patients with CLOM from right-sided colon cancer. The research indicates that these patients might not benefit from TA alone due to a higher likelihood of developing extrahepatic metastases, especially peritoneal metastases. 30 Those previous research corroborates these findings.
Despite its contributions, this study has limitations. Firstly, it employed a retrospective design, warranting further verification through prospective studies. And, this study spanned from 2011 to 2021, during which significant advancements in diagnostic and therapeutic modalities for colorectal cancer have occurred. Additionally, evolving clinical guidelines could have influenced treatment decisions. These factors could potentially introduce heterogeneity in patient management, potentially impacting outcomes. Secondly, previous studies have shown that RAS mutation in colorectal cancer could influence the prognosis of CLOM. 31 To mitigate its impact, the baseline characteristics was analyzed between the general population and patients with missing RAS data, concluding that RAS status had a minimal effect on OS outcome. Thirdly, percutaneous hepatic TA procedure is minimally invasive with low complications that is currently being adopted as alternatives to HR for CLOM, however, assessment of quality of life is also an important outcome measure, especially for patients with stage IV colorectal cancer. Our study did not incorporate quality-of-life metrics, which is critical in assessing the full impact of TA vs HR. Finally, the MRSL model has not been externally validated, but its robustness was further evaluated, using multiple metrics including AUC, nomogram, and Kaplan-Meier curve analyses.
Conclusion
In summary, a robust MRSL score-based model was developed, and aimed at improving the decision-making process for determining the suitability of Thermal Ablation (TA) in patients with Colorectal Liver Oligometastases (CLOM) originating from right-sided colon cancer. These findings suggest that high-risk patients with CLOM from right-sided colon cancer might not solely benefit from TA. Given the inherent aggressiveness of right-sided colon cancer, additional therapeutic approaches, such as chemotherapy, might be essential for these high-risk patients to minimize the probability of extrahepatic recurrence post-TA.
Supplemental Material
Supplemental Material - Decision-making for Ablation of Colorectal Liver Oligometastases Patients: A 10-year retrospective study of survival Outcomes Based on Right- Versus Left-Sided Primary Tumor Location
Supplemental Material for Decision-making for Ablation of Colorectal Liver Oligometastases Patients: A 10-year retrospective study of survival Outcomes Based on Right- vs Left-Sided Primary Tumor Location by Xiao-Guang Qi, Jian-Ming Li, Jian-Ping Dou, Fang-Yi Liu, Zhen Wang, Zhao-He Zhang , Ping Liang, Jie Yu in Cancer Control.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
