Abstract
Patients with unresectable hepatic metastases, from uveal or ocular melanoma, are challenging to treat with an overall poor prognosis. Although over the past decade significant advances in systemic therapies have been made, metastatic disease to the liver, especially from uveal melanoma, continues to be a poor prognosis. Percutaneous hepatic perfusion (PHP) is a safe, viable treatment option for these patients. PHP utilizes high dose chemotherapy delivered directly to the liver while minimizing systemic exposure and can be repeated up to 6 times. Isolation of the hepatic vasculature with a double-balloon catheter allows for high concentration cytotoxic therapy to be administered with minimal systemic adverse effects. A detailed description of the multidisciplinary treatment protocol used at an institution with over 12 years of experience is discussed and recommendations are given. A dedicated team of a surgical or medical oncology, interventional radiology, anesthesiology and a perfusionist allows PHP to be repeatedly performed as a safe treatment strategy for unresectable hepatic metastases.
Keywords
Introduction
The liver is a frequent site of malignancy. It may be affected by primary malignancies, such as hepatocellular carcinoma and cholangiocarcinoma, and metastases from a variety of malignancies including cutaneous and uveal melanoma, soft tissue sarcomas, breast and ovarian cancer, neuroendocrine, pancreatic, and colorectal tumors. In certain circumstances, these hepatic lesions arise as the sole sites of disease. There are several therapeutic options for isolated hepatic disease, yet surgical resection remains the option with the best long-term outcomes. For patients with unresectable disease, chemoembolization, cryoablation, radiofrequency ablation, transarterial radioembolization with Yttrium-90, and hepatic artery infusion are possible treatment pathways.
Treatment of Unresectable Melanoma Hepatic Metastases
In the treatment of metastatic cutaneous melanoma, surgical resection for limited hepatic involvement has been reported to improve overall survival (OS) in selected patients, however due to high recurrence rates and procedural morbidity, the overall benefit of metastatectomy is limited. 1,2 For patients with isolated liver metastasis, the ability to achieve negative margins with surgical resection can improve median OS up to 39 months, yet historically only 2-13% of patients have been found to be eligible for treatment. 2,3 Uveal melanoma continues to represent a treatment challenge. While the primary site is easily treated, up to 50% of patients will develop metastases, most commonly to the liver, with limited median survival of up to 12 months. 4 -6 Surgical resection for limited hepatic involvement has been reported to improve OS in selected patients, however due to a miliary pattern of spread, high recurrence rates, and procedural morbidity, the overall benefit of metastatectomy in patients with metastatic uveal melanoma is limited. 1,2 Patient eligibility for liver resection been reported to be higher in other malignancies such as intrahepatic cholangiocarcinoama, approximately 30% with up to 36 month median OS. 7
The use of radioembolization with Yttrium-90 (Y90) in the treatment of metastatic cutaneous and uveal melanoma has been reported as safe, with median OS ranging from 8-10 months. 8 -10 As the majority of liver metastases derive their blood supply from the hepatic artery, high doses of glass microsphere based irradiation are able to be administered while sparing the non-diseased hepatic parenchyma supported by the portal vein. 11 Transarterial chemoembolization (TACE) also takes advantage of this anatomy to deliver a mixture of emulsified chemotherapy, instilled alongside embolic particles to create drug stasis in the region of disease. Median OS with this method has been reported of up to 4-9 months. 12 -16
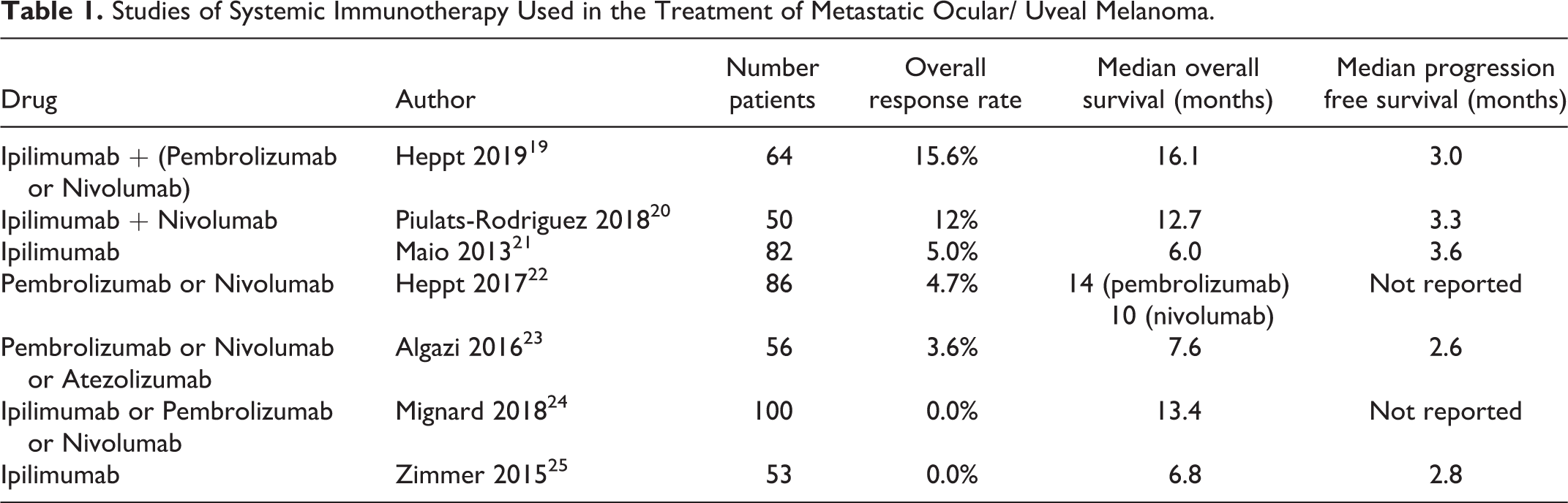
In terms of systemic therapy, chemotherapy has not been shown to be effective for treating metastatic uveal melanoma. 17,18 The discovery of immune checkpoint inhibitors has led to investigations of its benefit for metastatic uveal melanoma, though unfortunately numerous studies have yet to demonstrate appreciable clinical benefit. Among studies evaluating > 50 patients, survival outcomes reported median overall response rate (ORR) of 4.7 - 16%, median OS of 7.6 - 16 months, and median progression-free survival (PFS) of 2.6 - 3.6 months (Table 1).
Studies of Systemic Immunotherapy Used in the Treatment of Metastatic Ocular/ Uveal Melanoma.
Hepatic perfusions of the liver were initially performed via isolated hepatic perfusion (IHP), a surgical procedure requiring open laparotomy to cannulate the hepatic artery for direct administration of melphalan hydrochloride. 26 The inferior vena cava (IVC) was clamped above and below the hepatic venous return and melphalan drained via a retrohepatic IVC venous cannula. The patient was placed on veno-veno bypass to enable a hyperthermic, high-flow liver perfusion. Although IHP was associated with response rates of 59-68%, 27 it carried a high perioperative mortality of approximately 22% due to post-procedural liver failure and complications associated with major abdominal surgery limited the duration of response. 28 -31
The percutaneous hepatic perfusion (PHP) is a minimally invasive alternative to the IHP (see Table 4 by Broman and Zager 32 ). Chemotherapy is selectively delivered to the liver using a percutaneous catheter system (Hepatic CHEMOSAT® Delivery System, Delcath Systems, Inc., Queensbury, NY, USA). While not yet approved in the United States by the Food and Drug Administration, it received CE mark approval in the European Union in April 2011. The procedure has been studied for isolated hepatic malignancies, such as metastatic uveal melanoma, which have limited effective treatment options. An ORR of 47% has been observed for the treatment of metastatic uveal melanoma, cutaneous melanoma, sarcoma, breast cancer and cholangiocarcinoma. 6,33 -37 Severe AEs (≥ grade 3) including liver failure are infrequent, and the procedure can be repeated up to 6 times. Although no direct comparison exists between the different treatment methods for hepatic metastasis, one retrospective study which evaluated the outcomes of metastatic cutaneous and uveal melanoma found significantly prolonged hepatic progression free survival (PFS), overall PFS, and OS in patients treated with PHP versus Yttrium-90 and chemoembolization. 35 Unfortunately, patients tend to develop extrahepatic disease no matter the treatment method. In a phase Ib/II trial of radio-frequency ablation combined with ipilimumab, response was seen in the extrahepatic metastases. 38 With the higher response rate seen in PHP, combination with immune checkpoint inhibition using ipilimumab and nivolumab is currently being investigated (NCT04283890).
Institutional Experience
In an ongoing phase III clinical trial (NCT02678572), 107 PHPs were attempted among 31 patients between 2008-2020. Median patient age was 64 (IQR 54.4-69) and 65% were women. 105 (98%) perfusions were successfully completed Each patient received a median of 3 perfusions (range 1-6). Diseases treated included uveal melanoma (n = 24), cutaneous melanoma (n = 3), melanoma of unknown primary (n = 1) and sarcoma (n = 3) (Table 2). The treatment team consisted of anesthesiologist, certified registered nurse anesthetist, certified anesthesiologist assistant, interventional radiologist, certified clinical perfusionist, and surgical oncologist.
Characteristics of Patients Receiving Percutaneous Hepatic Perfusion.

* Other: gastrointestinal stromal tumor, leiomyosarcoma.
Abbreviations: IQR, interquartile range; PHP, percutaneous hepatic perfusion.
Patient Selection
Patients diagnosed with unresectable primary or metastatic isolated hepatic disease must undergo a complete staging workup with full body computed tomography (CT) to rule out extrahepatic disease. Liver directed CT or magnetic resonance imaging (MRI) are necessary to report the position of hepatic lesions relative to their size, proximity to major vasculature, and overall tumor burden. Patients with a tumor burden > 50% of hepatic mass are ineligible for treatment at this time. Also ineligible are patients with an Eastern Cooperative Oncology Group (ECOG) score > 1, body weight < 35 kg (due to physical limitations concerning the size of the double-balloon catheter), Child-Pugh score B or C cirrhosis, liver cirrhosis associated with portal hypertension or encephalopathy, congestive heart failure, total bilirubin > 1.5 times the upper limit of normal, enzymes AST/ALT > 5 times the upper limit of normal, hemoglobin < 10 g/dL, absolute neutrophil count < 1.5 cells/µL, platelet count < 100,000 per µL, serum creatinine > 1.5 mg/dL unless creatinine clearance is > 40 mL/min/1.73m 2, and any significant disorder or medical comorbidity which would preclude the administration of general anesthesia and systemic heparinization. Other prohibitive reasons to consider include current use of immunosuppressive therapy other than oral corticosteroids ≤ 10 mg/day and history of allergic reaction or known hypersensitivity to heparin, angiographic contrast medium, natural rubber latex component of the double-balloon catheter, or melphalan.
Protocol
Pre-perfusion
After induction of general anesthesia, catheters are placed using the Seldinger technique with ultrasound and fluoroscopic guidance: A) triple-lumen central venous catheter (CVC) into the internal jugular vein (IJ), B) 10 Fr venous sheath into IJ contralateral to the triple-lumen CVC (A), C) 5 Fr arterial sheath into femoral artery, D) 18 Fr venous sheath into the femoral vein. For the first 2 infusions, 18 Fr femoral sheath (D) and 5 Fr arterial sheath (C) are placed in the right groin (Table 3). Femoral catheter placements then alternate between the left and right groins for subsequent perfusions. Visceral angiography (superior mesenteric artery and celiac artery angiograms) is performed prior to melphalan infusion to evaluate for variant arterial anatomy, specifically replaced and/or accessory hepatic arteries and patency of the portal vein with hepatopedal flow on venous phase imaging. A hepatic artery angiogram is then performed to evaluate for systemic connections to the gastrointestinal tract. When necessary, these vessels are embolized to prevent systemic infusion of melphalan. The gastroduodenal artery is frequently embolized. The gastric arteries are also routinely evaluated for systemic anastomoses with the right gastric artery more commonly embolized based on the proximity to the infusion catheter placement. In the event vessels cannot be embolized (e.g.: small vessel caliber), whether the infusion catheter can be placed distally into the left and right hepatic arteries is immediately assessed. Patients that cannot safely receive isolated hepatic melphalan perfusion are deemed ineligible for PHP treatment. Angiography is repeated on the day of each subsequent procedure to ensure the hepatic circulation remains isolated. After placement of arterial catheter and venous sheaths, the patient is systemically anticoagulated with heparin sulfate 300 U/kg for a target activated clotting time (ACT) of greater than 400 seconds prior to placement of the double balloon catheter. Additional heparin is given during the procedure to maintain ACT > 400 seconds.
Vascular Access and Catheter Placements for Percutaneous Hepatic Perfusion.

Procedure
Prior to isolation of hepatic circulation, an arterial infusion catheter (Chemofuse® Catheter, Delcath Systems, Inc.) is introduced through the 5 Fr femoral arterial sheath (C) into the proper hepatic artery. A 16 Fr double-balloon hepatic isolation and aspiration catheter (Isofuse® Isolation and Aspiration Catheter, Delcath Systems, Inc.) is introduced through the 18 Fr femoral venous sheath (D) into the retrohepatic IVC (Table 3). The isolation catheter has 2 balloons (cephalad, caudal) spaced either 50 mm or 62 mm apart. The catheter is fenestrated between the balloons allowing for drainage of hepatic venous outflow to extracorporeal filtration via the main catheter lumen. There are 4 accessory ports, 2 are used to inflate the balloons and the third allows for access to the main catheter lumen for blood aspiration or contrast injection to confirm retrohepatic IVC isolation. The fourth port allows for placement of the catheter over a wire which also serves as a bypass channel for venous return distal to the occluded IVC segment.
The hemofiltration circuit is a veno-veno bypass circuit that collects blood from the fenestrated isolation catheter and returns it through a catheter placed through the IJV venous return sheath (B). The bypass machine is turned on at this point while the chemofilters are clamped and bypassed. The pump is initially cycled at 1,000 rpm and gradually increased to 2,500 rpm for maximum flow through the isolation catheter. Blood flow rate is set using a Medtronic BIO-CONSOLE® 560 system (Medtronic plc, Dublin, Ireland) for a target flow rate of approximately 750-800 mL/min. Inline pressure monitors are present to evaluate flow dynamics for both the circuit and filters. Pump inlet negative pressure is monitored to ensure that there is no disruption of venous return to the circuit. A high negative pressure indicates that there is some obstruction to pump inflow, either due to obstruction of the hepatic vein, or a kink in the pump inflow line. Additionally, pre-filter and post-filter pressures are monitored to ensure filter competency as well as total system pressure. An elevated pre-filter pressure gradient may indicate that the filters have clotted, while an elevated post-filter pressure gradient indicates a blockage in the circuit after the filters. An elevated post-filter pressure is highly likely secondary to clotting or problesm with inflow and the IJ catheter. A Medtronic 540 T External Drive Unit is used to drive the BP50 Bio-Pump® Centrifugal Blood Pump and the Medtronic BIO-PROBE® TX50/DP38 blood flow monitoring system and transducer is used to monitor rate of blood flow in the circuit (Figure 1).

Diagram of the complete percutaneous hepatic perfusion circuit for isolation of the retrohepatic inferior vena cava and extracorporeal filtration of melphalan-infused blood via veno-veno bypass. Reproduced with persmission from Delcath Systems, NY, NY.
The cephalad balloon is inflated in the right atrium under fluoroscopic guidance with approximately 30-35 (maximun 38mL) mL of dilute contrast and retracted to occlude the IVC. The caudal balloon is inflated in the IVC superior to the renal veins until its form appears flattened against the IVC, typically requiring approximately 10-15 mL dilute contrast. A retrohepatic IVC venogram is performed to ensure isolation, which requires momentary interruption of the veno-veno bypass circuit to prevent evacuation of the contrast medium during IVC venography (Figure 2).

Venogram demonstrating percutaneous isolation of the retrohepatic inferior vena cava with a double balloon isolation catheter and retrograde IVC venogram showing no leak around either balloon. Clips are in the gastroduodenal artery embolizing it.
Once hepatic venous return is isolated, the isolation catheter will require steady traction for the duration of the procedure to maintain cephalad balloon occlusion of the inferior atriocaval junction. The double-balloon catheter is marked with felt pen at the hub of the venous sheath to ensure appropriate catheter positioning and traction are kept throughout the infusion period. (Figure 3) Balloon positioning is also confirmed fluoroscopically intermittently during the procedure per the discretion of the proceduralist. The chemofilters are then engaged one at a time while the bypass line is left open. Once both chemofilters are engaged and there is appropriate flow through the system, the bypass line is clamped. Proper communication with the anesthesiology team is especially important at this point in the procedure.

Photograph illustrating how the double balloon isolation catheter is marked at the hub of the venous sheath to ensure appropriate traction during the procedure. Identification of this mark throughout the procedure allows the surgeon to ensure catahter is not slipping into sheath and therfore upper balloon migrating out of its wedged position in the artrial- IVC junction.
Anesthesiology considerations
The procedure results in a significant degree of hemodynamic instability, which has been largely attributed to the filtration of endogenous and exogenous catecholamines with engagement of the extracorporeal chemofilters, although McEwan et al. have suggested that melphalan infusion may also be contributory. 39 The most significant decrease in blood pressure occurs once both chemofilters are fully engaged and the bypass circuit is clamped. Observation using the Vigileo monitor (Edwards Lifescience, Irvine, CA) indicates that the hypotension is characterized by high stroke volume variation (SVV) and high cardiac output, which is consistent with a vasodilatory state. These hemodynamic derangements are treated with intravascular volume expansion with crystalloids and colloids during the pre-perfusion phase and the administration of vasopressors during the bypass phase. At Moffitt Cancer Center, we routinely will increase the mean arterial pressure (MAP) using titrated vasopressors to a target of approximately 150 mm Hg immediately prior to chemofiltration in order to mitigate the precipitous decrease in MAP once the bypass circuit is clamped. The primary vasopressor agentsare used to achieve this are norepinephrine and vasopression. In patients requiring high doses of norepinephrine, or when undesirable levels of tachycardia result from the infusion, incrementally titrated doses of vasopressin are helpful at restoring the MAP to target level. However, with increasing amounts of vasopressin administered, the more likely arterial vasospasm is to be encountered on the hepatic angiogram. The melphalan infusion is begun once patients are hemodynamically stable with a target MAP of 75 mm Hg or higher.
Melphalan infusion
Once hemodynamic stability is achieved, the arterial infusion catheter is advanced into the left hepatic artery. A hepatic angiogram is performed to ensure proper flow. If arterial spasm is observed, nitroglycerin (100 µg) is infused directly into the hepatic artery and a hepatic angiogram is repeated to ensure resolution of spasm. If arterial spasm cannot be resolved, significant retrograde flow from the hepatic artery is assumed and the procedure is terminated to prevent extrahepatic delivery of melphalan. Upon confirmation of good antegrade hepatic artery blood flow without spasm, melphalan infusion is initiated. A Bayer MEDRAD Mark V ProVis (Bayer HealthCare LLC, Whippany, NJ, USA) is used. The melphalan dose is 3 mg/kg ideal body weight (maximum dose 220 mg) and is divided into 5 infusions of 100 mL each over 4 minutes (25 mL/min). At Moffitt Cancer Center, we routinely start with the left hepatic artery (when the left and right hepatic arteries are selectively catheterized for lobar infusions), the infusion is divided in a 2:3 ratio between the left and right hepatic circulation based on liver volume. Prior to each 100 mL infusion, a hepatic arteriogram is repeated to ensure infusion catheter placement and patency of the hepatic artery (checking for arterial spasm), with 100 ug of nitroglycerin administration prior to each infusion if needed for relief of hepatic artery arterial spasm. After completion of melphalan infusion, extracorporeal filtration is continued for 30 minutes for passive washout. Appropriate balloon occlusion of the retrohepatic IVC is maintained during this time. Upon completion of washout, the balloons are deflated then bypass is terminated. No residual blood in the circuit is returned to the patient. All sheaths are left in place and the isolation catheter is removed and replaced with an obturator. Protamine sulfate is administered to reverse anticoagulation, based on total heparin dose. A final ACT level is obtained to ensure appropriate reversal. The patient is extubated and brought to the recovery room.
Post-operative interventions
Coagulation status is normalized by starting with the transfusion of 10 U cryoprecipitate for all patients upon immediate arrival into the post-anesthesia care unit. Further correction of coagulation and platelet derangements is managed with transfusion of fresh frozen plasma and platelets as indicated. Transfusion of fresh frozen plasma is based on coagulation studies and given until a normal international normalized ratio (INR) is attained. Platelets are transfused to a count of greater than 75,000/mm3 and packed red blood cells to a hemoglobin level greater than 9 g/dL. After the coagulation derangements are corrected, the arterial and venous catheters are removed the same day of the procedure. Direct pressure over the catheter sites for 45 minutes is held and patient activity is limited to bedrest until the following morning. The patient is monitored in the intensive care unit overnight with plan for discharge on the first post-operative day. The patient is scheduled for an outpatient visit 3-4 days later to draw blood work assessing for liver toxicities and early bone marrow suppression, and to receive pegfilgrastim injection.
Toxicity
The major immediate post-procedure adverse events are anemia and thrombocytopenia due to filtration of blood components by the chemofilters. 40,41 This is routinely corrected in all patients as described above. After discharge, typically 1-2 days after the procedure, bone marrow suppression is the most common adverse event and is associated with melphalan infusion, typically manifesting as neutropenia. Some degree of low-grade bone marrow suppression is seen in all patients. All patients are treated with pegfilgrastim injection 3 days after PHP and transfusions if needed. There were no mortalities related to the procedure. The 2 aborted PHPs were discontinued for the following reasons: one patient experienced ventricular tachycardia shortly after the chemofilters were turned on and the procedure was promptly aborted prior to melphalan administration. In the other patient, the cephalad balloon could not be wedged in the atriocaval junction due to anatomical issues. Neither of these patients received melphalan infusion.
Discussion
A detailed institutional protocol for performing PHP was described and recommendations from experience at a high-volume center that has completed 107 procedures since 2008 were given. The safety of this procedure is demonstrated with no procedure-related mortalities and expected AEs treated in the outpatient setting. In the EU, PHP has been applied to nearly all malignancies that affect the liver. Aside from metastatic melanomas, PHP has been used to treat primary hepatocellular carcinoma and cholangiocarcinoma, and metastatic breast, gastric, colorectal, renal and pancreatic cancers. 42 This institutional experience is primarily in the treatment of metastatic uveal melanoma, but good outcomes for metastatic cutaneous melanoma and sarcoma have also been demonstrated. 43,44 PHP has significantly improved ORR, OS, and PFS for patients with metastatic uveal melanoma while minimizing the morbidity from major abdominal surgery. Current studies have reported high response rates of up to 83%, median hepatic PFS of 11 months and OS of 27 months. 6,44 -46 A single institution retrospective review of 60 patients treated for ocular melanoma or cholangiocarcinoma reported an ORR of 33%, higher for patients with ocular melanoma (42%), and median OS of 9 months from first PHP, numerically longer for ocular melanoma (12 months). 47 The results of a phase III, single-arm, multicenter trial are eagerly awaited, which will represent the largest prospective trial of metastatic uveal melanoma patients to date (NCT02678572).
The main source of PHP success relies on the isolation of the hepatic IVC using a double-balloon catheter, with leakage of high concentration melphalan around the balloons one of the main sources of procedural toxicity. In evaluation of 18 procedures with leakage compared to 15 controls (no leakage), the use of venogram post-processing using 2D-perfusion angiography was able to identify and quantify flow of contrast past the balloons in real time during all 18 procedures, providing a tool which may prove beneficial and deserves further evaluation in a larger, multi-institutional setting. 48
Conclusion
A PHP requires a multi-disciplinary team comprised of a surgical or medical oncologist, interventional radiologist, anesthesiologist, and a perfusionist. It is a safe treatment modality for isolated hepatic metastases which obviates the need for morbid laparotomy based liver perfusions and can be repeated in patients who derive hepatic control of disease.
Footnotes
Abbreviations
Authors’ Note
Acknowledgments
Special thanks to Moffitt Cancer Center Clinical Trials Coordinator Neelam Lal for her dedication to the patients who undergo this clinical trial.
Declaration of Conflicting Interests
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JSZ advisory board for Sanofi/Regenron, Novartis and Merck. Speaker’s bureau for Array Biopharma and Sun Pharma. Research funding from Amgen, Delcath Systems, Philogen, Provectus, Castle Biosciences and Novartis. Consultation for Amgen, Alkermes, Castle Biosciences and Philogen. Medical advisory board for Delcath Systems.
Ethics Statement
Written consent was obtained from the Institutional Reviewer Board via expedited review, IRB#00000971.
Moffitt Cancer Center—MCC #20186, MCC 20186: Percutaneous Hepatic Perfusion for Metastatic Uveal Melanoma (Pro00037427)—Via Expedited Review, IRB# 00000971.
Written consent to perform each procedure was obtained from each of the patients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
