Abstract
Background
Pancreatic cancer is a challenging disease, often requiring invasive procedures for diagnosis. Reliable tumour markers are essential for ensuring early detection and better patient outcomes. Although Carbohydrate Antigen 19-9 is the most commonly used marker, it is marred by low predictive accuracy and high false positivity. Carcino Embryonic Antigen also has limited practical use. A novel antigen, Cytokeratin fragment 21-1, is gaining significance for its diagnostic value in various tumours.
Materials and Methods
This prospective study aimed to evaluate the potential of Cytokeratin fragment 21-1 in comparison with Carbohydrate Antigen 19-9 and Carcino Embryonic Antigen in diagnosing pancreatic cancer. From January 2016 to December 2019, 45 patients with confirmed pancreatic ductal adenocarcinoma were included in this cross-sectional study.
Results
Carbohydrate Antigen 19-9 was raised in 22 patients, Carcino Embryonic Antigen was elevated in 17, and Cytokeratin fragment 21-1 was elevated in 30 cases. Carbohydrate Antigen 19-9 was found to be elevated in the presence of jaundice. Both Carbohydrate Antigen 19-9 and Cytokeratin fragment 21-1 had good correlation with stage of cancer, while Carcino Embryonic Antigen had very minimal correlation.
Conclusion
In this study, Cytokeratin fragment 21-1 was elevated in a higher number of cases than Carbohydrate Antigen 19-9 and Carcino Embryonic Antigen. Both Cytokeratin fragment 21-1 and Carbohydrate Antigen 19-9 correlated well with cancer stage. Also Cytokeratin fragment 21-1 was not affected by jaundice, unlike Carbohydrate Antigen 19-9. Therefore, Cytokeratin fragment 21-1 has the potential to be an effective individual tumour marker in pancreatic cancer.
Keywords
Introduction
Pancreatic tumours stand out from other gastrointestinal malignancies due to their heightened likelihood of surgical inoperability and considerably low survival rates. Prompt detection is of utmost importance, as surgical intervention remains the sole curative measure for pancreatic cancer. Nonetheless, a mere 10%–30% of patients with pancreatic tumours are eligible for curative surgery, and out of those, only half are able to undergo R0 resection. 1 Even those patients who are able to undergo R0 resection with additional adjuvant treatment achieve a 5-year survival rate of approximately 25% only. 2 Those diagnosed with unresectable stage III and IV carcinomas are ineligible for any potentially curative treatment, leaving them with a dismal median survival period of around 5-12 months only. 3
The non-specific nature of early-stage symptoms, coupled with the high biological aggressiveness of pancreatic malignancies, contributes to the significant delay in diagnosis and increased mortality. Due to the deep-seated location of the organ, obtaining tissue for definitive diagnosis is often challenging. Therefore, improving methods for early detection is crucial to increase the number of resectable carcinomas and to enhance patient outcomes. Various imaging modalities for pancreatic cancer include endoscopic ultrasound (EUS), computed tomography (CT), endoscopic retrograde cholangio-pancreatography (ERCP), magnetic resonance imaging (MRI), and magnetic resonance cholangio-pancreatography (MRCP). However, many of these are prohibitively expensive or come with drawbacks such as radiation exposure and infrequent usability. Apart from the abovementioned methods, invasive methods like explorative laparoscopy and laparotomy may sometimes be needed for diagnosis and staging.
The commonest serum marker used in pancreatic cancer is the sialylated Lewis blood group antigen known as Carbohydrate Antigen 19-9 (CA 19-9). 4 It has been shown that CA 19-9 levels after surgery or radiation treatment directly affect the prognosis of pancreatic cancer cases.5,6 The National Comprehensive Cancer Network (NCCN) recommendations continue to advocate the use of CA 19-9 as the biomarker in routine clinical practice. 7 However, inflammation, cholangitis, and biliary obstruction are common complications in advanced pancreatic cancer patients, and these factors can significantly affect CA 19-9 levels. Furthermore, CA 19-9 serum levels are significantly increased in both benign and malignant biliary diseases, such as choledocholithiasis, gall bladder cancer, and cholangiocarcinoma. Elevated CA 19-9 serum levels in 10%–50% of cases are found in benign or precursor pancreatic lesions such as acute or chronic pancreatitis, intraductal pancreatic mucinous neoplasm (IPMN), and pancreatic intraepithelial neoplasia (PANIN).8-10 Even other benign conditions such as ovarian cysts, heart failure, Hashimoto’s thyroiditis, rheumatoid arthritis, and diverticulitis have been associated with elevated serum CA 19-9 levels.11,12
Only approximately 40% of patients diagnosed with early stage I pancreatic cancer experience an increase in CA 19-9 levels. 13 CA 19-9 blood levels are unable to detect early or small tumours or precancerous lesions in 10%–15% of patients who have a higher risk of pancreatic cancer, such as those with hereditary pancreatitis, a family history of the disease, or Peutz-Jeghers syndrome. 14 Due to the fact that CA 19-9 production requires the presence of sialylated Lewis (Le)a blood group antigen, individuals with a Lea-b-phenotype (absence of Lewis antigen glycosyl-transferase) are unable to synthesize CA 19-9. This leads to a false-negative result when testing for CA 19-9 in these individuals (approximately 5%–10% of the population). 15 Additionally, due to its poor positive predictive value (PPV) of around .9% only, CA 19-9 cannot be used as an appropriate screening test in asymptomatic patients.12,16
CEA is a glycoprotein with established roles in predicting time to progression and overall survival in colorectal and lung cancer, as well as in detecting tumour recurrence. However, its use in pancreatic cancer diagnosis is not as evident, and although some data have been published, the diagnostic relationship is limited. 17
Cytokeratin Fragment 21-1 also known as CYFRA 21-1 is a soluble structural protein fragment of cytokeratin 19 (CK19), an acid type cytokeratin, with a molecular weight of 40,000d. 18 Cytokeratin 19 (CK19) is a protein found in the intermediate filament proteins that are necessary for maintaining epithelial cell integrity. This antigen is recognized by 2 mouse monoclonal antibodies, KS 19-1 and BM 19-21, which were developed against the MCF-7 cell line. Normal and proliferative epithelium both express CYFRA 21-1, and monoclonal antibodies can be used to identify specific epitopes on cytokeratin 19 to characterize it.
There are various postulates explaining the possible effects of K19 in malignant tissues. It has been demonstrated that the release of CK19 fragment is closely related to the mRNA expression for CK19, and there is a possibility that the genomic change of CK19 DNA thus down-regulates the expression of mRNA for CK19. 19 The levels of CYFRA 21-1 are also found to be increased significantly in TNF-alpha-treated cells whilst displaying higher percentage of apoptosis, granular-like aggregation of CK19, as well as elevated activity of caspase-3 than non-treated cells. 20 Also, the levels of CYFRA 21-1 are found to decrease significantly when caspase-3 was inhibited in the TNF-alpha-treated cells. Hence, the release of CYFRA 21-1 could be reflective of cellular apoptosis during the process of tumour growth.
CYFRA 21-1 is already being developed as a tumour marker for squamous cell carcinoma, adenocarcinoma, and large cell carcinomas of non-small cell lung cancer (NSCLC). In addition to lung cancer, CYFRA 21-1 has also shown promise as a useful marker for monitoring tumour recurrence and predicting overall prognosis in various malignancies including those of the liver, cervical, oesophageal, breast, gastric, and bladder cancer.21,22
An ideal biomarker for cancer would allow for early detection and assist in treatment decisions, as well as predict overall outcomes and assess recurrence. However, finding such a biomarker can be challenging, and multiple biomarkers may need to be used in combination to achieve the desired level of accuracy. The search for new biomarkers for pancreatic cancer is ongoing and formed the basis of this study also.
Materials and Methods
The primary objective of the study was to compare the diagnostic efficacy of CYFRA 21-1 and CA 19-9 in patients with pancreatic cancer, while the secondary objective was to assess the correlation between the values of these markers and the stage of the cancer. The study was conducted using a cross-sectional design.
This study was conducted prospectively over a period of 3 years, from January 1, 2016, to December 31, 2019. Before beginning the study, permissions from the Institutional Review Committee and the Ethics Committee were obtained. The study was approved by the Human Ethics Committee, xxxx, vide IEC order no. 04/18/2015/MCT dated 31/07/2015. Adult cases with pancreatic ductal adenocarcinoma, as determined by cytology or histology, were eligible to be chosen as participants. To prevent interference with tumour marker readings, patients with any previously diagnosed malignancies or other causes of jaundice were removed from the study. The subjects were instructed on the study protocol, and written informed consent with signature was obtained from them for the blood collection. Before the initiation of definitive surgery or chemotherapy, blood samples were taken. There was no change in the management of the patients, and they underwent appropriate treatment based on multi-disciplinary tumour board decisions.
The sample size was calculated using a standardized formula for sample size estimates in diagnostic test studies, 23 with the sensitivity of the novel and established tests derived from reference studies. The study was designed with a sample size of 45, aiming for a power of 80% and an alpha error of 5%. Consecutive patients meeting the selection criteria were included. The levels of CEA and CA 19-9 were measured using a solid-phase, two-site chemiluminescent enzyme immunometric assay, while commercially available enzyme-linked immunosorbent assay kits were used to determine the levels of CYFRA 21-1. The normal range for CA 19-9 was set as below 37 U/mL, while for CYFRA 21-1 it was set below 1.96 ng/mL. The normal range of CEA was set at below 3 micrograms per litre, with a higher cutoff of 7 among smokers. Clinical staging was based on the AJCC staging system.
A comprehensive questionnaire was used to record clinical information for each case. Pearson’s correlation coefficient was used to assess the correlation between the 3 tumour markers themselves and the correlation between age and tumour marker levels. Spearman’s Rho test was used to examine the relationship between cancer stage and the tumour markers. The association between jaundice and the markers as well as between gender and markers was evaluated using the Chi-square test. Statistical analysis was conducted using Microsoft Office Excel and Epi Info software (CDC) version 6. Data are reported as arithmetic means (AM) with standard deviation (SD) or percentages, and statistical significance was considered whenever P-values were less than .05. All patient details are presented de-identified. We have followed relevant Equator guidelines, and the study is reported according to the STROBE checklist. 24
Results
Demographic Distribution of the Study Participants.

Among the 45 patients, 22 had elevated levels of CA 19-9, 17 had elevated levels of CEA, and 30 had elevated levels of CYFRA 21-1 (Chart 1). The mean values of CA 19-9, CEA, and CYFRA 21-1 were 940.62 (+/−2597.34) U/mL, 7.41 (+/−8.93) mcg/L, and 12.24 (+/−17.11) ng/mL, respectively, indicating that these tumour markers were present in measurable quantities in the pancreatic cancer patients under study. The range of values for these markers varied significantly, with CA 19-9 having the widest range of 2597.34 U/mL and CEA having the narrowest range of 8.93 mcg/L. It is interesting to note that while all patients had pancreatic cancer, the levels of tumour markers varied significantly, which could suggest that tumour markers may be influenced by the biological characteristics of individual patients. Distribution of positivity of the 3 tumour markers among study participants.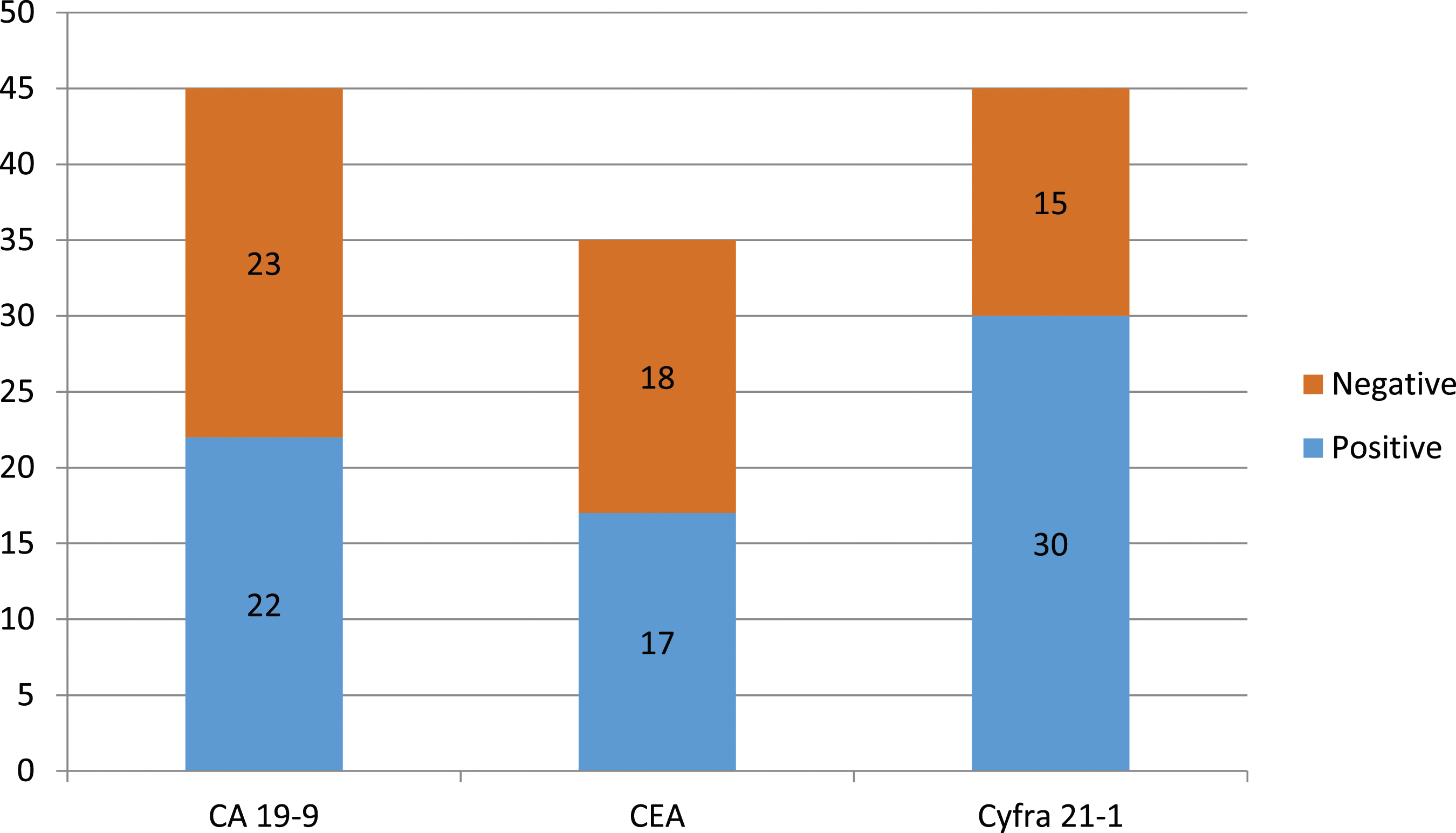
We analysed the relationships between the 3 tumour markers. Pearson’s correlation coefficient revealed a small negative relationship between CEA and CA 19-9, with an R-value of −.0179. In contrast, CEA and CYFRA 21-1 showed a weak positive association, with a correlation coefficient of .2555. CA 19-9 and CYFRA 21-1 showed a poor correlation, with an R-value of .2731. These findings suggest that the different tumour markers may have distinct biological implications in pancreatic cancer.
We analysed the relationship between gender and tumour marker positivity. For CA 19-9, the Chi-square statistic was .3429, with a P-value of .558185. In the case of CEA, the Chi-square statistic was .8229, the P-value being .364346. For CYFRA 21-1, the Chi-square statistic was found to be .3302, with a P-value of .565534. None of the markers had any significant association with respect to gender.
We checked for any relationship between age and tumour marker levels with Pearson’s correlation. In the case of CA 19-9, the R-value was .2125, with a P-value of .147043. For CEA, the R-value was found to be −.0181, the P-value being .982. For CYFRA 21-1, the R-value was found to be −.0019, with a P-value of .994618. None of the tumour markers had any significant correlation with the patients’ age.
We also analysed the relationship between the tumour markers and jaundice by using the Chi-square test. We found a significant link between jaundice and CA 19-9, with a Chi-square statistic of 7.7816 and a significant P-value of .05. However, there was no link between CEA and jaundice, with a Chi-square statistic of 2.7011 and a P-value of .10028. Similarly, there was no relationship between CYFRA 21-1 and jaundice, with a Chi-square statistic of 1.6071 and a P-value of 2.0489.
To investigate the relationship between tumour markers and cancer stage, we used Spearman’s correlation. We found a significant positive correlation between CA 19-9 and cancer stage, with a Rho value of .52348 and a P-value of .00022. In contrast, there was no significant correlation between CEA and cancer stage, with a Rho value of .02249 and a P-value of .8834. Interestingly, we found a significant positive correlation between CYFRA 21-1 and cancer stage, with a Rho value of .51269 and a P-value of .0032.
These results indicate that tumour markers may have distinct associations with different stages of pancreatic cancer. CA 19-9 was found to be negative in all AJCC stage 1 patients, whereas CYFRA 21-1 was shown to be positive in all stages (Chart 2). Lastly, we observed that all 3 tumour markers had lower values in stage 4 patients, which is surprising since this stage is generally considered the most advanced and severe. TNM stage-wise distribution of the 3 tumour markers.
Discussion
Our study, which included a sample of 45 patients diagnosed with pancreatic cancer, yielded some anticipated results as well as some unexpected ones concerning CA 19-9 and CYFRA 21-1. Our findings indicate that CYFRA 21-1 is more closely associated with pancreatic cancer than CA 19-9 while both CA 19-9 and CYFRA 21-1 showed a good correlation with the stage of pancreatic cancer. Also we observed that elevated levels of CA 19-9 were significantly linked to jaundice, a common symptom of pancreatic cancer.
Several studies have found a correlation between pre-treatment CYFRA 21-1 levels and overall survival, and as a potential marker for predicting treatment responsiveness to chemotherapy. 25 A comparison study of the diagnostic efficacy of CEA, CA 19-9, and CYFRA 21-1 found that the combination of all 3 markers resulted in the best outcomes, with the highest number of instances correctly identified. 26 Another study on pancreatic cancer indicated that CYFRA 21-1 demonstrates better sensitivity and predictive values than CA 19-9. 27 In this trial involving 59 patients, pre-treatment levels of CYFRA 21-1 showed a significant correlation with overall survival, regardless of the imposed cutoff (at a mean value of 4.9 ng/mL). Along with the presence of mutKRAS ctDNA, higher levels of CYFRA 21-1, CA 19-9, and CEA before the initiation of first-line chemotherapy have been significantly correlated to adverse overall survival. 28 Lee assessed multiple tumour markers among patients with primary pancreatic malignancy and those with benign pancreatic cysts and found CA 19-9 to have good diagnostic performance. 29 Based on 2 cutoff values, CYFRA 21-1 (≥2.0 and 1.83 ng/mL) was found to have an acceptable sensitivity of 80.4% and 82.3%, respectively, and was also much more significant than other tumour markers. A weak significant relationship was found between fluorodeoxyglucose (FDG) uptake by the tumour and CYFRA 21-1 or CA 19-9. Additionally, by multivariate analyses, both CA 125 and CYFRA 21-1 levels demonstrated independent prognostic significance for predicting overall survival.
CA 19-9 cannot be solely relied upon as a screening indicator in asymptomatic patients, but it can aid in differentiating between benign pancreatitis and pancreatic cancer to a certain extent. Studies by Liao et al, Groblewska et al, Banfi et al, and Jiang et al have reported excellent diagnostic performance in distinguishing between carcinoma and control groups, with high sensitivity and specificity rates. For instance, Liao et al found superior diagnostic performance in differentiating between 58 carcinomas and 102 controls, with a sensitivity rate of 81% and a specificity rate of 91%. 30 Similarly, Groblewska et al reported a sensitivity rate of 74% and a specificity rate of 100% in a study involving 62 carcinomas and 65 controls. 31 Banfi et al and Jiang et al also obtained high sensitivity rates of 79% and 85%, respectively, and a specificity rate of 100% in their studies involving carcinomas and control groups.32,33
Although the correlation is not strong, there is a link between serum CA 19-9 and tumour load, resectability, and overall survival. Patients with postoperative CA 19-9 levels exceeding 180 U/mL had a notably lower survival rate than those with lower values. 34 In patients with stage II–III cancer, where the accurate extent of disease spread may be challenging to determine before surgery, preoperative serum levels of CA 19-9 are considered clinically relevant in evaluating prognosis. 35 Independent predictors of survival in patients include low preoperative serum CA 19-9 levels, a significant postoperative decrease in levels, and stand-alone levels of less than 200 U/mL. 36
In an interesting study, increasing the CA 19-9 serum cutoff to above 100 U/ml or 1000 U/ml for the purpose of identifying pancreatic cancer resulted in a higher specificity of 98% and 99.8%, respectively. 37 However, this was accompanied by a decrease in sensitivity, which declined to 68% and 41%, respectively. According to other studies, CA 19-9 levels have been found to increase in approximately 40% of Stage I pancreatic tumours.38,39 In only approximately 33% of patients, CA 19-9 shows a modest increase in localized tumours (T1 and T2) with values greater than 120 U/mL. 40 While some researchers suggest that CA 19-9 levels above 300 U/mL should be carefully assessed for the presence of malignancy, others propose adjusting CA 19-9 values by incorporating C-reactive protein or bilirubin to enhance diagnostic accuracy.41,42
CEA has been validated as a useful tumour marker for staging and prognosis in colon cancer, but it is not an effective biomarker in pancreatic cancer. The main problem with CEA in pancreatic cancer is its very low sensitivity, which ranges from 25% to 56%, for distinguishing between carcinoma and controls (in spite of a high specificity of 82% to 100%).43-46
There are several limitations to this research. Firstly, it was conducted in a single centre which may limit the generalizability of the findings to other settings. Secondly, the study design did not allow for follow-up to assess prognosis and survival. Additionally, because only confirmed cases were included, it was not possible to calculate the sensitivity and specificity values and area under the curve for each marker. Moreover, the study did not include information on the patients’ Lewis status, which could have impacted CA 19-9 levels. Finally, the sample size was relatively small, which may have reduced the statistical power of the analysis.
Conclusions
An ideal tumour marker should be able to provide both diagnostic and prognostic information, as well as the ability to stratify patients. Both CYFRA 21-1 and CA 19-9 correlate with the stage of pancreatic cancer and therefore provide information on the patient’s prognosis. However, CA 19-9’s low sensitivity makes it unsuitable as a screening tool for pancreatic cancer. Furthermore, CA 19-9 is limited by its false-negative results in Lewis-negative individuals and false-positive results in cases of obstructive jaundice and other benign diseases. In contrast, CYFRA 21-1 is more strongly associated with pancreatic cancer and is not impacted by jaundice. Therefore, CYFRA 21-1 shows significant promise as a tumour marker in pancreatic malignancies. Future studies may aim to address the limitations of this study and further investigate the potential of CYFRA 21-1 as a tumour marker in pancreatic cancer using high-quality sample cohorts and standardized multiplex formats.
Supplemental Material
Supplemental Material - Cyfra 21.1: A Useful Tumour Marker in Pancreatic Ductal Adenocarcinoma: Cross-Sectional Study
Supplemental Material for Cyfra 21.1: A Useful Tumour Marker in Pancreatic Ductal Adenocarcinoma: Cross-Sectional Study by Krishnakumar G. Kuttanchettiyar, Viswanathan Kollengode V, and Meer M. Chisthi in Cancer Control
Footnotes
Acknowledgements
The authors are grateful to Dr KB Harikumar, Scientist, Rajiv Gandhi Centre for Biotechnology, Trivandrum, for carrying out CYFRA 21-1 assays and Dr KL Jayakumar, former Professor, Department of Oncology, Government Medical College, Trivandrum, for helping with data collection.
Author Contributions
Krishnakumar G Kuttanchettiyar: Conception and design of the work and acquisition of data for the work; Viswanathan Kollengode V: Drafting the work and reviewing it critically for important intellectual content; Meer M Chisthi: Analysis and interpretation of data for the work and final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by funding from the State Board for Medical Research, Government Medical College, Trivandrum, Kerala, India.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
