Abstract
Background
It is unclear whether the addition of chemoradiation (CRT) to adjuvant chemotherapy (CT) following upfront resection of pancreatic ductal adenocarcinoma (PDAC) provides any benefit. While some studies have suggested a benefit to combined modality therapy (CMT) (adjuvant CT plus CRT), it is not clear if this benefit was related to increased CT usage in patients who received CMT. We sought to clarify the use of CMT in patients who underwent upfront resection of PDAC.
Methods
Patients with non-metastatic PDAC were retrospectively identified from the linked SEER-Medicare database. Those who underwent upfront resection were identified and divided into two cohorts – patients who received adjuvant CT and patients who received adjuvant CMT. Cohorts were compared. Univariate analysis described patient characteristics. Kaplan-Meier and multivariable Cox proportional hazards modeling were used to estimate overall survival (OS).
Results
3555 patients were identified; 856 (24%) received CT and 573 (16%) received CMT. The median number of CT doses was 11 for both groups. Patients who received CMT were younger, diagnosed in the earlier time frame, and had fewer comorbidities. The median OS was 21 months and 18 months for those treated with CMT and CT (P < .0001), respectively, but when stratified by nodal status, the association with improved OS in the CMT cohort was only observed in node-positive patients. On multivariable analysis, receipt of CMT and removal of >15 lymph nodes decreased the risk of death (P < .05).
Discussion
Receipt of CMT following upfront resection for PDAC was associated with improved survival, which was confined to node-positive patients. The role of adjuvant CMT in PDAC with nodal metastases warrants further study.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC), the fourth leading cause of cancer-related mortality in the United States, is a very aggressive disease with an estimated an estimated 49,830 deaths in 2022 1 , and a 5-year overall survival (OS) of only 11.5%. 2 While surgical resection offers the only potentially curative treatment, even amongst patients who are eligible for resection, survival is poor. Distant recurrence following upfront surgical resection remains the most common cause of death, but autopsy series demonstrate that a significant proportion of patients die with or from local recurrence. 3 Since surgical resection alone does not provide adequate distant or locoregional control for pancreatic cancer – multimodality therapy is needed.
Multiple randomized controlled trials (RCT) have investigated the role of adjuvant chemotherapy (CT) and/or chemoradiation (CRT) to determine the optimal treatment strategy in resected PDAC, some which have reported conflicting results,4, 5 as is evidenced by the fact that the current NCCN guidelines recommend adjuvant CT or CRT for resected PDAC.6, 7 The Gastrointestinal Study Group (GITSG) was the first study to evaluate adjuvant CRT for PDAC and found that adjuvant CRT improved OS compared to observation alone. 8 The European Study Group of Pancreatic Cancer (ESPAC)-1 trial demonstrated a survival benefit with adjuvant CT alone, but actually found a negative impact on survival for adjuvant CRT.9, 10 A Radiation Therapy Oncology Group (RTOG) trial later suggested possible efficacy of CRT. 11 Several subsequent adjuvant CT RCTs have further supported the efficacy of adjuvant CT following upfront surgical resection.12, 13 It is unlikely that adjuvant CRT will ever replace adjuvant CT, 14 yet, a clinically relevant questions remains: does the addition of CRT to adjuvant CT – combined modality therapy (CMT) – impact survival for patients with surgically resected PDAC?
To clarify the impact of CMT vs CT on the survival of patients with surgically resected PDAC, we performed a retrospective review of the National Cancer Institute’s (NCI) Surveillance Epidemiology and End Results (SEER)-Medicare linked database. This database was chosen because it contains claims codes which detail the type and quantity of adjuvant CT received. This allowed us to control for specific details of CT as a potential confounder in survival analysis, when examining the benefit of adjuvant CMT. We hypothesized that patients who received CMT would receive a higher number of CT doses, or would receive CT for a longer duration, than those who received CT alone. We hypothesized that this extended use of CT could be associated with improved survival in patients with more substantial locoregional disease burden (i.e. node-positive).
Methods
Data
The NCI’s SEER-Medicare linked database was used to identify patients diagnosed with PDAC from 2004-2013. The linkage between the SEER Database and Medicare was performed on a person-level. This allowed for analysis of the 1.6 million patients with cancer in the SEER database who are ≥65 years of age and enrolled in Medicare. The SEER database contains patient demographics, tumor characteristics, and vital status for 28% of the US population. The Medicare Provider Analysis and Review and the National Claims History files contain claims for hospitalizations and inpatient procedures. Office visits are captured using a combination of provider charges from the National Claims History files and facility charges from the outpatient Standard Analytical Files. 15 Using claims data, we calculated Charlson Comorbidity Index (CCI), considering the International Classification of Diseases, Ninth Revision (ICD‐9) diagnosis codes, ICD‐9 procedure codes, and Healthcare Common Procedure Coding System procedure codes on the claims. 16 Because cancer was the disease of interest, it was not included in the Charlson Comorbidity Index (CCI).
Patient Population
Patients ≥65 years of age, who were diagnosed with PDAC and underwent upfront surgical resection followed by adjuvant therapy were included. Patients who had metastatic disease at the time of diagnosis, or who had history of another primary malignancy were excluded. Patients who had received any neoadjuvant therapy were also excluded. Although neoadjuvant therapy is entering mainstream practice for patients with borderline resectable and locally advanced PDAC, it is not yet standard of care for resectable PDAC.7,17,18, 19 We therefore excluded these patients in an attempt to more clearly isolate the effect of adjuvant CMT on OS.
All multivariable models included patient age, sex, race, NCI-status of the treatment facility, patient CCI, tumor size, T stage of disease, lymph node status, lymph node number evaluated, and treatment. Age was defined as a categorical variable. The designation of the treating facility as an NCI designated cancer center was derived from the Hospital File and linked via hospital ID. T stage of disease was derived from the American Joint Committee on Cancer (AJCC), Cancer Staging Manual, editions 6 and 7. Radiation treatment receipt was derived from the SEER Patient Entitlement and Diagnosis Summary File (PEDSF) file, since there may be underreporting by Medicare sources if patients receive care outside of the Medicare system. 20 Surgical procedures performed were categorized as pancreaticoduodenectomy (Whipple), total pancreatectomy, or other (including distal pancreatectomy). Receipt of adjuvant CT and adjuvant CRT was characterized using Medicare claims codes and the SEER database. 21 Specifically, Medicare claims codes were used to quantify the number of doses of adjuvant CT administered to each patient and to provide insight into which chemotherapeutic agents were prescribed.
Statistical Analysis
Patients who met inclusion criteria were grouped into two cohorts – those who received adjuvant CT alone, and those who received CMT. Demographics, tumor characteristics, and treatment received were evaluated and compared between cohorts using the chi-squared test. Median OS was analyzed using the Kaplan-Meier method and multivariable Cox proportional hazard modeling. Results were considered statistically significant for a two-tailed P-value ≤.05 and a 95% confidence interval (CI). SAS statistical software, version 9.3 (SAS Institute, Cary, NC) was used for all statistical analyses.
Sensitivity analysis was performed on untreated patients, excluding those who died within two months of diagnosis. These were excluded to reduce confounding by early mortality, based on the assumption that these patients may have had undiagnosed distant metastases, or were too ill to receive a full course of treatment. These alternative analyses had no meaningful effect on our conclusions and are therefore not presented in the following results.
The study was approved by the Institutional Review Board at the University of Minnesota, reference number STUDY00004830. All procedures in this study were conducted in accordance with the University of Minnesota IRB’s (STUDY00004830) approved protocols. A waiver of the consent process was granted by the above-named IRB because the study involved no more than minimal risk. Supplemental information such as programming code available upon reasonable request to the corresponding author.
Results
Characteristics of all Patients Diagnosed with PDAC a who Underwent Upfront Surgical Resection, from Surveillance Epidemiology and End Results-Medicare 2004-2013, N = 3555.

aAbbreviations: PDAC, pancreatic ductal adenocarcinoma; NCI, National Cancer Institute; CT, chemotherapy; CMT, combined modality therapy.
aAbbreviations: PDAC, pancreatic ductal adenocarcinoma; CT, chemotherapy; CMT, combined modality therapy; NCI, National Cancer Institute.
bP-value not applicable since median number of CT doses equivalent between cohorts.
The median OS for patients who received adjuvant treatment after upfront surgical resection is shown in Figure 1A. The median OS for those who received adjuvant CT was 18 months, while the median OS for those who received CMT was 21 months (P < .0001). Survival of the cohort was also stratified by nodal status (Figure 1B and 1C). In the node-positive cohort, the median OS for those who received adjuvant CT was 17 months while the median OS for those who received CMT was 20 months (P < .0001). Conversely, in the node-negative cohort, the median OS for those who received adjuvant CT was 23 months while the median OS for those who received CMT was 25 months (P-value .09). To adjust for competing risks, survival was further described using a Cox proportional hazards model multivariable analysis (Table 3). Receipt of CMT was significantly associated with decreased risk of death (Hazard Ratio [HR] .80, 95% confidence interval [CI] .71-.91, P = .001). The only other factor found to significantly decrease risk of death was evaluation of greater than 15 lymph nodes (HR .88, CI 0.78-.99 P = .04). On the other hand, more recent diagnosis (HR 1.37 CI 1.22-1.55, 2009-2013 vs 2004-2008), T stage 4 (HR 1.53 CI 1.01-2.31), and node-positive disease (HR 1.46 CI 1.28-1.67) significantly increased the risk of death. (a) Survival curve, patients who underwent upfront surgical resection for PDAC and received adjuvant CT vs adjuvant CMT in SEER-Medicare, 2004-2013. Median OS 18 months CT, 21 months CMT (P < .05). (b) Survival curve, node-negative patients who underwent upfront surgical resection for PDAC and received adjuvant CT vs adjuvant CMT in SEER-Medicare, 2004-2013. Median OS 23 months CT, 25 months CMT (P = .09). (c) Survival curve, node-positive patients who underwent upfront surgical resection for PDAC and received adjuvant CT vs adjuvant CMT in SEER-Medicare, 2004-2013. Median OS 17 months CT, 20 months CMT (P < .05). Abbreviations: PDAC, pancreatic ductal adenocarcinoma; CT, chemotherapy; CMT, combined modality therapy; OS, overall survival. Multivariable Cox Proportional Hazard Model. Hazard of Death for Patients Who Received Adjuvant Therapy After Upfront Surgical Resection for PDAC
a
in Surveillance Epidemiology and End Results-Medicare from 2004-2013, N = 1429. aAbbreviations: PDAC, pancreatic ductal adenocarcinoma; REF, Reference (Hazard Ratio = 1.00); NCI, National Cancer Institute; CT, chemotherapy; CMT, combined modality therapy.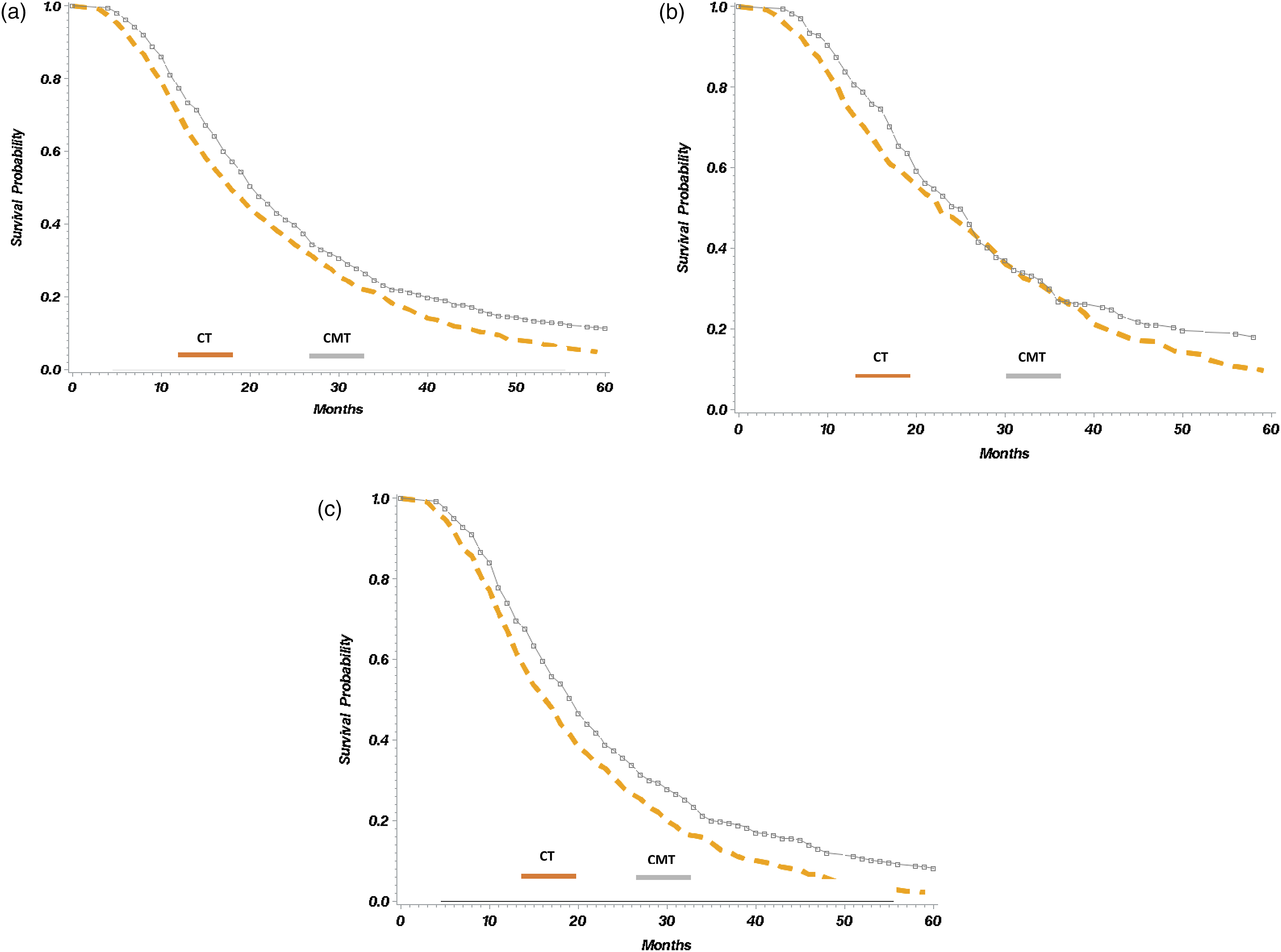
Discussion
The role of adjuvant CMT after surgical resection for pancreatic cancer has remained uncertain given discrepant results of clinical trials. The present evaluation of patients who received adjuvant therapy for PDAC in the SEER-Medicare linked database found that CT and CMT patients received a similar number of doses of systemic chemotherapy (median, 11 doses) and that CMT was associated with improved median OS, which appeared to be confined to patients with node-positive disease. Notably, patients who received CMT tended to be younger, diagnosed in the earlier time period, and had fewer comorbidities.
Early trials that evaluated the role of adjuvant CRT included the Gastrointestinal Study Group (GITSG), 8 the European Organisation for the Research and Treatment of Cancer (EORTC) 10 and the European Study Group for Pancreatic Cancer-1 study (ESPAC-1).9, 10 The GITSG trial first evaluated adjuvant CRT and found an improvement in OS from 11 months to 20 months when compared to observation alone; however, the trial ended prematurely due to low accrual of patients. 8 The EORTC trial attempted to replicate these results in 218 patients, however found no survival benefit to adjuvant CRT. 10 The subsequent ESPAC-1 trial used a 2 × 2 factorial design to evaluate CT and CRT vs observation, and found a survival detriment with CRT (14 months vs 17 months) and a survival benefit with adjuvant CT (20 months vs 16 months). 9 However, interpretation and application of the results of these early trials is difficult in the modern era due to issues with trial design, the use of split course CRT, heterogenous patient populations, and concerns for selection bias.
The rationale for adding radiation to CT in pancreatic cancer is in part due to observed patterns of disease recurrence following resection. In autopsy series of patients who underwent pancreatectomy for PDAC, 8-30% of patients died with only locoregional recurrence of disease.3,22, 23 Specifically, in one study of 76 patients, 30% of patients died with local-only recurrence, while the remaining 70% died with evidence of metastases. 22 CRT, it is thought, may improve local control of disease. In a phase II study designed to evaluate the feasibility and tolerability of a gemcitabine-based CRT protocol in patients who underwent R0 resection for PDAC, the rate of first recurrence as local-only recurrence was lower in those treated with CRT vs CT (11% vs 24%). 24 Furthermore, Parikh et al studied 1130 patients who underwent resection for PDAC and either receieved no adjuvant therapy, adjuvant CT or adjuvant CRT. 25 With a median follow-up of 18 months, patients who underwent adjuvant CT demonstrated a significant OS benefit on multivariable analysis; however, the patterns of recurrence were different amongst the groups. 25 Receipt of adjuvant CT or adjuvant CRT resulted in less local recurrence in patients with node-positive and margin-negative disease. 25 Further, some meta-analyses have supported the use of CRT in patients with positive resection margins.4, 26 These potential benefits to adjuvant CRT, as well as mixed results of previous trials, make it a potentially desirable addition to adjuvant systemic CT that warrants futher study.
Multiple prior studies have evaluated the role of CRT as a component of adjuvant therapy for resected PDAC, but due to lack of granular detail regarding ammount of CT received between treatment regimens, it remains unclear if there truly is a survival benefit associated with the combined use of CRT and adjuvant CT.27,28,29,30,31,32, 33 Kooby et al used the National Cancer Database to compare outcomes in patients who received no adjuvant therapy, adjuvant CT alone, and adjuvant CRT. They reported that CRT was associated with improved survival, but adjuvant CT remained a notable confounder as there was no classification, quantification, or comparison of systemic CT use between the groups. 28 A review of the SEER database, including 2532 patients with resected PDAC found an improvement in OS to 20 months among those treated with radiation after surgical resection compared to those not treated with radiation; however CT use was not evaluated. 28 A prior review of the SEER-Medicare linked database evaluated prognostic factors after surgical resection. 32 In this study of 396 patients, the receipt of adjuvant treatment (adjuvant CT and/or adjuvant radiation) significantly improved median OS; however, the study did not specifically compare receipt of adjuvant CT alone to adjuvant CMT, as in the present study. 32 Similarly, a few other national database studies have investigated the role of adjuvant CRT, but none have compared the addition of CRT to adjuvant CT in resected PDAC. Thus, despite prior randomized studies and more recent reviews, the effectiveness of CMT compared to CT alone for pancreatic cancer is still largely unknown. In the present study, contrary to what was hypothesized, there was no difference in the number of systemic chemotherapy doses administered to the CT and CMT groups. Yet, the addition of adjuvant CRT to CT was associated with an improvement in median OS. When stratified by nodal status, the association with improved OS was confined to the node-positive patients, as hypothesized, while the addition of CRT was not associated with improved OS in node-negative patients.
It is important to note some of the limitations of this study. The retrospective design and the use of a large epidemiological and billing database introduce inherent selection and coding biases. The data derived from databases is not collected to answer specific research questions, and may be subject to unmeasured confounding and have incomplete data. The Medicare fee-for-service population is restricted to patients aged ≥65; however, the median age of patients diagnosed with PDAC is 70, and the median age of our population was 73 years. 2 Although augmentation of the SEER database with Medicare claims data allowed description of types and doses of chemotherapy, the finer details of chemotherapeutic dosing, regimen choice, and regimen adherence, as well as the radiotherapeutic modality and dose used are not discernable through the data. We are also not able to comment on the reason for the low numbers of completion of adjuvant therapy, though it is known that many patients do not complete adjuvant therapy following upfront resection of PDAC.34,35 Finally, due to the nature of the SEER-Medicare linked data, granular patient- and tumor-related information such as patient functional status, perineural or neurovascular invasion, and resection margin status are not available. Nonetheless, the SEER-Medicare database provided insights into and quantification of adjuvant CT, which has been absent from previous national database investigations into the role of adjuvant CRT in resected PDAC.
Conclusion
Our data suggests that although CMT may not benefit all patients, those with node-positive disease seem to have improved survival. Future rigorous randomized controlled trials using modern adjuvant CT and CRT regimens are needed to further evaluate the utility of adjuvant CRT in resected PDAC.
Footnotes
Author Contributions
AA – study conception, design, data interpretation, manuscript preparation. MW –, data interpretation, manuscript preparation, manuscript revision. SM – study conception, data collection, data analysis and interpretation, manuscript revision. DS – data interpretation, manuscript preparation. KC – data interpretation, manuscript revision. EL – data interpretation, manuscript revision. CL – study conception, design, data interpretation, manuscript revision. JYCH – data interpretation, manuscript revision. TMT – data interpretation, manuscript revision. EHJ – study conception, design, data interpretation, manuscript revision. JD – study conception, design, data interpretation, manuscript revision, final approval of manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
This study was approved by the Institutional Review Board at the University of Minnesota (IRB), reference number STUDY00004830. All procedures in this study were conducted in accordance with the University of Minnesota IRB’s (STUDY00004830) approved protocols. A waiver of the consent process was granted by the above-named IRB because the study involved no more than minimal risk.