Abstract
Background
Serum high-density lipoprotein cholesterol (HDL-c) may influence cancer development. However, its relationship with the histological grade of pancreatic ductal adenocarcinoma (PDAC) is not well understood. This study aims to explore the potential associations between serum HDL-c levels and different histological grades of PDAC.
Methods
This retrospective study included 181 patients with pathologically confirmed PDAC who underwent radical surgery. Clinical data, blood biochemical results, imaging features, and pathological details of the patients were collected, such as age, gender, diabetes, hypertension, tumor grade, tumor size and location, high-density lipoprotein (HDL-c), low-density lipoprotein (LDL), total cholesterol (TC), triglycerides (TG), carbohydrate antigen 19-9 (CA19-9), and carcinoembryonic antigen (CEA).
Results
Patients with high-grade PDAC had significantly lower HDL-c levels compared to those with low-grade PDAC across both training and validation cohorts (P < 0.05). Significant associations were found between HDL-c levels and high-grade PDAC in the training (P < 0.001) and validation (P = 0.044) groups. Moreover, HDL-c levels were inversely related to lymph node metastasis in the training (P = 0.001) and validation (P = 0.012) sets.
Conclusions
Lower HDL-c levels are associated with high-grade PDAC and lymph node metastasis, suggesting that HDL-c may play a protective role in the progression of PDAC.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is highly malignant with a 5-year survival rate below 5%. 1 PDAC is often diagnosed at advanced stages, resulting in high recurrence rates and low survival. 2 Histological grade is an independent prognostic factor for PDAC, with higher grades associated with shorter survival and increased mortality. 3 Poorly differentiated PDAC tends to be more invasive and prone to early metastasis. 4 Currently, histopathology remains the gold standard for diagnosing pancreatic cancer, requiring postoperative assessment to determine tumor grade.
Recent studies have identified multiple factors influencing PDAC prognosis, including clinical characteristics, imaging features, and novel biomarkers. Research indicates that tumors in the pancreatic head have significantly better prognoses compared to those located in the body and tail. This may be related to differences in clinical presentation and surgical approaches in patients.5,6 CA199 is the most commonly utilized biomarker for the diagnosis of pancreatic cancer patients, assisting in assessing treatment efficacy and predicting survival, however, its sensitivity and specificity still pose limitations.7,8 Promising new biomarkers like circulating tumor DNA (ctDNA), long non-coding RNAs (LncRNAs), and molecular metabolites are emerging as significant prognostic indicators.9,10
Additionally, there is evidence linking serum high-density lipoprotein cholesterol (HDL-c) levels and the risks of various types of cancer, including breast,2,11,12 prostate, 2 and gastric cancer. 13 Recent studies also suggest a relationship between HDL-c levels and aggressive features of pancreatic neuroendocrine tumors, as well as different breast cancer subtypes, indicating that lower HDL-c levels may correlate with aggressive tumor progression.14,15
However, the relationship between serum HDL-c concentrations and the histological grade of PDAC remain unclear. This study aims to investigate the potential association between histological grades of PDAC and serum HDL-c levels.
Materials and Methods
Patients
In this retrospective study, a total of 181 patients with pathologically confirmed pancreatic ductal adenocarcinoma (PDAC) were enrolled between 2016 and 2022. Of these, 121 patients were recruited from the Affiliated Hospital of Nanjing University of Chinese Medicine, serving as the training cohort. An additional 60 patients were independently recruited from the First Affiliated Hospital of Nanjing Medical University to serve as the external validation cohort. The inclusion criteria for participants from both centers included: (1) those who had undergone the radical surgery, and pathologically confirmed PDAC; (2) individuals with complete blood lipids data; (3) those who underwent an abdominal enhanced CT examination within 2 weeks before surgery. Exclusion criteria for participants from both centers were: (1) those who had incomplete clinical and laboratory data; (2) individuals with metastases of pancreatic tumor, other primary malignancies, or hematological disorders; (3) those who had been treated with hypolipidemic drugs within 1 month before surgery; (4) those who had obtained radiotherapy or chemotherapy before surgical resection; (5) those who had received palliative operation; (6) who died of complications related operation. The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Affiliated Hospital of Nanjing University of Chinese Medicine (2017NL-135-05). Given the retrospective nature of the study, the Institutional Review Board of Affiliated Hospital of Nanjing University of Chinese Medicine granted a waiver for obtaining informed consent. The reporting of this study conforms to STROBE guidelines. 16 All patient identifiable details were de-identified before further analysis.
Data Collection
Patient demographics, clinical data, blood biochemical results, and pathological details were gathered from medical records. Pathological characteristics included tumor grading, tumor size, tumor location, vascular invasion, lymph node metastasis, perineural invasion, surgical margin status, and TNM stage. Blood data including serum HDL-c, low density lipoprotein cholesterol (LDL-c), triglyceride (TG), total cholesterol (TC), serum carbohydrate antigen 19-9 (CA19-9), and serum carcinoembryonic antigen (CEA) were obtained within 7 days prior to operation. All blood samples were gathered into coagulant-coated tubes following a minimum of 8 hours of fasting. A reduced HDL-c level was characterized by an HDL-c level of ≤1.0 mmol/L (38.6 mg/dl), corresponding to the lowest quartile in the training cohort’s patient population.
Imaging Analysis
Two radiologists with 5 and 10 years of experience in abdominal diagnosis independently assessed the images without prior knowledge of the clinical and pathological results. Discrepancies in their assessments were resolved by consensus, following consultation with a senior abdominal radiologist. The following data were analyzed: pancreatic parenchymal atrophy, the dilatation of main pancreatic duct (MPD) and common bile duct (CBD). The criteria for MPD dilation were set at a diameter greater than 4 mm, while for the CBD, dilation was defined as a diameter exceeding 8 mm. All unenhanced and contrast-enhanced CT images were retrieved from the Pictures Archiving and Communication System (PACS).
Histological Grading
All surgically resected tumors were fixed in formalin, processed, and paraffin-embedded for subsequent histological analysis. The histological sections were evaluated blindly by 2 experienced pathologists and were classified in accordance with the World Health Organization (WHO) classification of the digestive system, 5th edition. In case of any discrepancies, a consensus was reached after discussion. Histological grades of tumors were categorized as well differentiated (Grade 1), moderately differentiated (Grade 2), and poorly differentiated (Grade 3). Considering the imbalance of differentiated types and the similarity in biological behavior, Grade 1 and Grade 2 were reclassified as a low-grade group, while Grade 3 was reclassified as a high-grade group in this study.17,18
Statistical Analysis
For continuous variables, the two-tailed t test or Mann-Whitney U test was utilized, whereas categorical variables were analyzed using the chi-square test or Fisher’s extract test. We employed logistic regression analyses to examine the potential connections between HDL-c and histological grade. The relationship between HDL-c concentrations and lymph node metastasis was evaluated using point-biserial correlation and logistic regression analysis. To evaluate the diagnostic efficiency of HDL-c level, receiver operating characteristic (ROC) curve was performed by SPSS 26.0 (SPSS lnc., Chicago, IL, USA) software and GraphPad Prism 9.0.0 (GraphPad Software, San Diego, CA, USA) were used for all statistical analyses. P-values less than 0.05 were regarded as statistically significant.
Results
Patient Characteristics
Characteristics of Patients With PDAC in the Training and Validation Cohorts.

PPA, pancreatic parenchymal atrophy; MPD, main pancreatic duct; CBD, common bile duct; LNM, lymph node metastasis; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; HDL-c, high-density lipoprotein-cholesterol; LDL, low density lipoprotein; TC, total cholesterol; TG, triglyceride.
aThe patients with new-onset diabetes (≦ 2 years duration).
High-grade PDAC patients exhibited significantly lower HDL-c levels compared to low-grade PDAC patients in both sets (P < 0.05). Furthermore, the high-grade PDAC group had a higher proportion of patients with low HDL-c levels compared to the low-grade group in both training (43.5% vs 3.4%) and validation sets (75.0% vs 50.0%). In both the training and validation sets, patients with low HDL-c levels exhibited a higher proportion of high-grade PDAC compared to those with low-grade PDAC (training set: 93.1% vs 6.9%; validation set: 63.2% vs 36.8%). As shown in Figure 1, the prevalence of high-grade PDAC showed a tendency to decrease as the HDL-c levels increased. The association between high-density lipoprotein-cholesterol (HDL-c) level and pancreatic ductal adenocarcinoma (PDAC) grades. The proportion of high-grade PDAC decreased with the increase of HDL-c level in training set (A) and validation set (B).
Univariate and Multivariate Logistic Regression Analysis
Associated Factors With High-Grade PDAC.

OR: odds ratio; CI: confidence interval; HDL-c: high-density lipoprotein cholesterol.
“/” means that the variables were not included in the regression analysis.
In addition, after adjusting for age, diabetes, and hypertension, HDL-c level was identified as an independent factor associated with PDAC grade in both training (OR = 22.86, 95%CI: 4.72-110.80) and validation sets (OR = 23.79, 95%CI: 1.12-503.74) (Table S2). In addition, gender and tumor location were also independent risk factors associated with high-grade PDAC in both training and validation sets (P < 0.05).
ROC Analysis
In Figure 2, the training set reveals that HDL-c exhibited an area under the curve (AUC) of 0.71 (95%CI: 0.62-0.81), with a cutoff value of 0.91 mmol/L, demonstrating a sensitivity of 96.6%, and a specificity of 41.9% in identifying high-grade PDAC. In addition, HDL-c plus gender and location had an AUC of 0.82 (95%CI: 0.75-0.90), a sensitivity of 94.9%, and a specificity of 64.5% in identifying high-grade PDAC (Figure 2A). The DeLong tests indicated that the inclusion of HDL-c significantly improve the diagnostic value compared to using either gender or tumor location alone, in the training set (P < 0.01) (Table S3). Receiver operating characteristics (ROC) analysis of variable in predicting high-grade pancreatic ductal adenocarcinoma (PDAC). Performance of high-density lipoprotein-cholesterol (HDL-c) levels, gender combined with location in predicting high-grade PDAC in training set (A) and validation set (B).
In the validation set, HDL-c displayed an AUC of 0.70 (95%CI: 0.57-0.83), with a sensitivity of 92.9%, and a specificity of 40.6% in identifying high-grade PDAC. In addition, HDL-c plus gender and location had an AUC of 0.81 (95%CI: 0.70-0.92), a sensitivity of 89.3%, and a specificity of 62.5% in identifying high-grade PDAC. (Figure 2B). The DeLong tests indicated that the inclusion of HDL-c significantly improve the diagnostic value compared to using either gender or tumor location alone in the validation set (P < 0.05) (Table S3).
The Association Between HDL-c Levels and PDAC Lymph Node Metastasis
Multivariate Logistic Regression to the Lymph Node Metastasis of PADC.
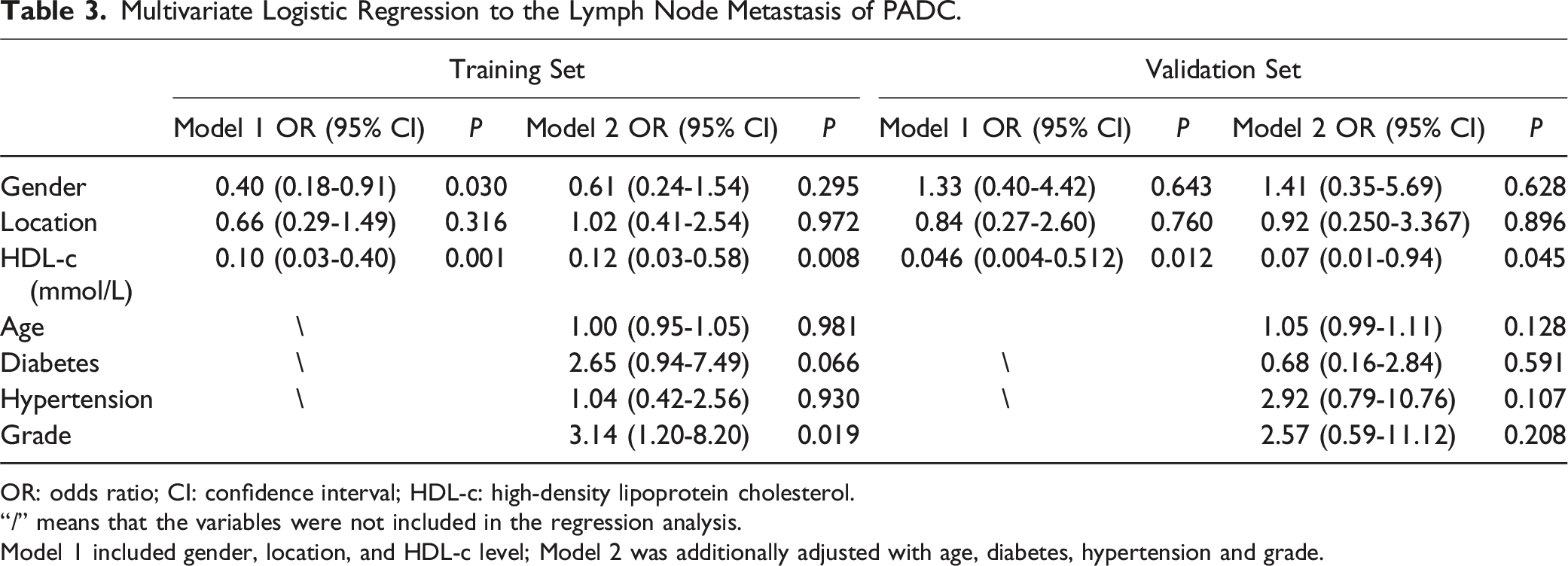
OR: odds ratio; CI: confidence interval; HDL-c: high-density lipoprotein cholesterol.
“/” means that the variables were not included in the regression analysis.
Model 1 included gender, location, and HDL-c level; Model 2 was additionally adjusted with age, diabetes, hypertension and grade.
Discussion
In this study, patients with high-grade PDAC exhibited significantly lower HDL-c levels than those with low-grade PDAC. Additionally, a significant correlation between reduced HDL-c levels and lymph node metastasis in PDAC patients was identified.
Numerous investigations have linked serum HDL-c levels to the risk of various cancers, including PDAC. A population-based cohort study showed that persistently low HDL-c and normal-to-low HDL-c change increase the risk of PDAC. 19 Moreover, multiple studies have identified a negative correlation between HDL-c concentrations and incidence of PDAC.20-22 In related pancreatic tumors, research has demonstrated notably lower HDL-c levels in patients with malignant IPMN, particularly those with branch duct IPMN. 23 Another study found a clear correlation between low HDL-c levels and aggressive features in pancreatic neuroendocrine tumors. 17 Furthermore, a significant randomized controlled trial highlighted an association between decreased HDL-c and higher cancer risk, accounting for variables like LDL-c, age, BMI, and smoking status. 24 Collectively, these findings suggest that serum HDL-c levels could be a crucial oncological biomarker for identifying patients with aggressive disease profiles, providing strong support for the conclusions of this study. Our findings align with a meta-analysis conducted in a Chinese population, which revealed a significant association between reduced levels of apolipoprotein A1, a key protein constituent of HDL, and both unfavorable histological grades and advanced tumor stages. 25 Furthermore, a retrospective study has revealed a positive correlation between low HDL-c levels and aggressive histological subtypes of breast cancer. 15 A clear association between HDL-c levels and lymph node metastasis has been established in various cancers, including gastric, lung, and colorectal cancer.26-28 In particular, it has been observed that patients with lymph node metastasis often present with notably lower HDL-c levels, a trend that aligns with our own findings.
The mechanisms by which specific HDL-c changes influence cancer risk are not fully understood. In tumor cells, scavenger receptor class B type 1(SR-B1) and ATP-binding cassette subfamily A member 1 (ABCA1) receptors are essential for maintaining the dynamic balance of cholesterol. 29 ABCA1 facilitates cholesterol efflux by transporting phospholipids and cholesterol out of the cell to form HDL particles. SR-B1 mediates cholesterol ester uptake and reverse cholesterol transport.14,30 There is a study showing that the anti-tumor capability of HDL is associated with the lower affinity of SR-BI-mediated lipid influx and the higher affinity of ABCA1-mediated cholesterol efflux. 30 It’s thus hypothesized that in low-grade PDAC, cholesterol efflux is relatively smooth, potentially leading to elevated HDL levels, whereas as the tumor progresses to a higher grade, rapid proliferation of tumor cells increases the demand for energy and lipids (including cholesterol), hindering cholesterol efflux and consequently reducing HDL synthesis in both the liver and the broader physiological context involved in HDL production. HDL-c also has anti-tumor properties due to its antioxidant, anti-inflammatory, anti-angiogenic, and immunomodulatory effects. 31 Previous research has suggested a potential correlation between reduced HDL-c levels and increased concentrations of pro-inflammatory cytokines, including tumor necrosis factor-alpha (TNFα) and interleukin-6 (ll6).32,33 Moreover, tumors characterized by high-grade or lymphatic invasion often exhibit higher proliferative activity. These aggressive tumors necessitate greater amounts of cholesterol for membrane synthesis, which may contribute to the decreased levels of HDL-c observed in high-grade PDAC. 34
While numerous studies have examined the link between HDL-c levels and cancer risk, research on its connection to tumor grade in PDAC remains limited. Compared to previous studies focusing on HDL levels and overall prognosis in PDAC, our research investigates the relationship between HDL levels and pancreatic cancer grading. By concentrating on pathological grading, we aim to reveal the potential role of HDL in early and mid-stages of tumor development, which differs from the prognostic research perspective that primarily focuses on the final outcomes of the disease. Our study suggests HDL could serve as a non-invasive biomarker for assessing tumor aggressiveness and adjusting treatment strategies, potentially leading to more personalized clinical treatments and guiding future research into deeper mechanistic links between HDL and pancreatic cancer development.
Our study has several limitations. Firstly,the absence of patient survival data limits our ability to explore the relationship between HDL-c levels and PDAC prognosis. Secondly, while previous research has linked elevated preoperative CA19-9 levels with higher histologic grades in PDAC patients,7,35,36 our study did not observe this correlation. The reliability of these findings requires confirmation with larger sample sizes. Third, although we included an independent external validation set, both sets were from the same region in China. Our findings would be strengthened by prospective research with larger, multi-center, and multi-regional cohorts. Fourth, apolipoprotein A-I and apolipoprotein B, as important components of high-density lipoprotein and key apolipoproteins in the metabolic process of lipoproteins, 30 will be included in future studies to further explore the biological mechanisms between pancreatic cancer progression and high-density lipoprotein. PDAC patients often have impaired liver function due to cancer cachexia, future research should investigate liver enzymes, plasma albumin levels, and inflammatory markers to understand their relationship with HDL production and liver function. Lastly, whether HDL is a biomarker or a causative factor for cancer is still debated. 37 This study only identified an association between HDL-c levels and the histological grade of PDAC, but it remains unclear whether this association indicates causation, selection bias, or a combination of both. Therefore, further research is needed to explore how HDL-c may influence PDAC tumorigenesis.
Conclusion
Our results demonstrate a significant inverse relationship between HDL-c concentrations and high-grade PDAC incidence, along with a notable link between lower HDL-c levels and lymph node metastasis. Further research is needed to understand the mechanisms behind this association.
Supplemental Material
Supplemental Material - Serum High-Density Lipoprotein Cholesterol Concentrations in Pancreatic Ductal Adenocarcinoma and Its Association With Histological Grade in a Chinese Population
Supplemental Material for Serum High-Density Lipoprotein Cholesterol Concentrations in Pancreatic Ductal Adenocarcinoma and Its Association With Histological Grade in a Chinese Population by Ying-Ying Cao, Xiao-Jing Lv, Hui Li, Li-Chao Qian, Hai-Peng Si, Yuan Li, Kai Guo, Shuai Ren, and Zhong-Qiu Wang in Cancer Control.
Footnotes
Acknowledgments
We thank all of the participants involved in this study.
Author Contributions
Zhongqiu Wang, Shuai Ren, Yingying Cao, Xiaojing Lv contributed to study design and data collection. Yingying Cao, Xiaojing Lv draft the manuscript. Zhongqiu Wang, Shuai Ren modified the final manuscript. Yingying Cao, Xiaojing Lv, Hui Li, Lichao Qian, Haipeng Si performed data analysis. Yuan Li, Kai Guo prepared all figures. And all authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (82371919, 82171925, 82202135, 82372017, and 81971681), Jiangsu Provincial Key research and development program (BE2023789, BK20241994), China Postdoctoral Science Foundation (2023M741808), Young Elite Scientists Sponsorship Program by China Association of Chinese Medicine (2024-QNRC2-B16), Shandong Provincial Postdoctoral Innovation Project (SDCX-ZG-202400053), Young Elite Scientists Sponsorship Program by Jiangsu Association for Science and Technology (JSTJ-2023-WJ027), Nanjing Postdoctoral Science Foundation, Foundation of Excellent Young Doctor of Jiangsu Province Hospital of Chinese Medicine (2023QB0112), The Open Project of Jiangsu Provincial Science and Technology Resources (Clinical Resources) Coordination Service Platform (TC2023B003), Natural Science Foundation of Nanjing University of Chinese Medicine (XZR2023036), Project of National Clinical Research Base of Traditional Chinese Medicine in Jiangsu Province, China (JD2023SZ16), Developing Program for High-level Academic Talent in Jiangsu Hospital of Chinese Medicine (y2021rc03 and y2021rc44), and Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX23_2175).
Ethical Statement
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
