Abstract
Purpose
This investigation leveraged the SEER database to delve into the progression patterns of PTC when left untreated. Furthermore, it aimed to devise and authenticate a nomogram for prognosis prediction for such patients.
Methods
We extracted data from the SEER database, focusing on PTC-diagnosed individuals from 2004-2020. To discern disease progression intervals, median survival times across stages were gauged, and the disease progression time was estimated by subtracting the median survival time of a more severe stage from its preceding stage. Prognostic determinants in the training set were pinpointed using both univariate and multivariate Cox regression. Using these determinants, a prognostic nomogram was crafted.
Results
In untreated PTC patients, those in stages I and II had a favorable prognosis, with 10-year overall survival rates of 86.34% and 66.03%, respectively. Patients in stages III and IV had a relatively poorer prognosis. The median survival time of stage III, stage IVA, stage IVB and stage IVC patients was 108months, 43 months, 20 months and 8 months, respectively. The deduced progression intervals from stages III-IVC were 65, 23, and 12 months. In the training set, age, tumor stage, gender, and marital status were identified as independent risk factors influencing the prognosis of untreated PTC, and a nomogram was constructed using these variables.
Conclusion
In the absence of treatment intervention, early-stage PTC progressed slowly with an overall favorable prognosis. However, in mid to advanced-stage PTC, as tumor stage increased, disease progression accelerated, and prognosis gradually worsened. Age, tumor stage, marital status, and gender were independent risk factors influencing the prognosis of untreated PTC, and the nomogram based on these factors demonstrated good prognostic capability.
Plain Language Summary
Purpose
This investigation leveraged the SEER database to delve into the progression patterns of PTC when left untreated. Furthermore, it aimed to devise and authenticate a nomogram for prognosis prediction for such patients.
Methods
We extracted data from the SEER database, focusing on PTC-diagnosed individuals from 2004-2020. To discern disease progression intervals, median survival times across stages were gauged, and the disease progression time was estimated by subtracting the median survival time of a more severe stage from its preceding stage. Prognostic determinants in the training set were pinpointed using both univariate and multivariate Cox regression. Using these determinants, a prognostic nomogram was crafted.
Results
In untreated PTC patients, those in stages I and II had a favorable prognosis, with ten-year overall survival rates of 86.34% and 66.03%, respectively. Patients in stages III and IV had a relatively poorer prognosis. The median survival time of stage III, stage IVA, stage IVB and stage IVC patients was 108months, 43 months, 20 months and 8 months, respectively. The deduced progression intervals from stages III-IVC were 65, 23, and 12 months. In the training set, age, tumor stage, gender, and marital status were identified as independent risk factors influencing the prognosis of untreated PTC, and a nomogram was constructed using these variables.
Conclusion
In the absence of treatment intervention, early-stage PTC progressed slowly with an overall favorable prognosis. However, in mid to advanced-stage PTC, as tumor stage increased, disease progression accelerated, and prognosis gradually worsened. Age, tumor stage, marital status, and gender were independent risk factors influencing the prognosis of untreated PTC, and the nomogram based on these factors demonstrated good prognostic capability.
Keywords
Introduction
Thyroid cancer is the most common endocrine malignancy, accounting for approximately 3% of all cancers. 1 In the past decade, there has been a consistent rise in the rate of thyroid cancer diagnosis.2-4 Differentiated thyroid cancer (DTC) is the most common type of thyroid cancer, accounting for over 90% of all thyroid cancers. It primarily includes papillary thyroid cancer (PTC), follicular thyroid cancer, and Hürthle cell cancer of the thyroid, with PTC being the most common subtype of DTC.5-7 The prognosis of PTC is generally favorable. 8 As per the 2017’s 8th iteration of the TNM staging system, an overwhelming majority (beyond 90%) of PTC diagnoses were pinpointed at stages I or II. Following standardized care protocols, specific mortality associated with this thyroid malignancy remains under 2%. A minority of patients were diagnosed at stage III or IV, carrying a relatively higher risk of mortality. 9 Treatment options for PTC include surgical intervention, I131 therapy, TSH suppression therapy, and so on. Among these, surgical removal is the preferred treatment for the majority of PTC patients and plays a crucial role in prognosis.10,11 Even if patients with PTC have distant metastases, surgical removal is a prerequisite and foundation for radioiodine therapy and is an important factor affecting prognosis. 12 Furthermore, for a subset of low-risk patients with small PTC, given its relatively slow progression and low mortality, apart from surgical treatment, conservative approaches, such as active surveillance and close monitoring, may also be considered. 13
Understanding the inherent trajectory of a disease—its course without therapeutic interference—is pivotal for a comprehensive grasp of the ailment. 14 For PTC, such insights are invaluable for deepening disease comprehension, elucidating its evolutionary phases, and consequently deriving enhanced screening methodologies and more judicious diagnostic and therapeutic strategies. Initiating active surveillance during incipient stages of low-risk micro PTC sheds light on the untreated trajectory of this carcinoma variant. 15 Research led by Tuttle RM and colleagues indicated that under active surveillance, the majority of these cancers exhibited minimal growth or stability over an extended period, with some even demonstrating regression. Disease progression was negatively correlated with age at onset, with younger patients being more likely to progress. During active surveillance, no low-risk patients developed distant metastasis or died from thyroid cancer. 16 In another prospective study of active surveillance of micro PTC, it was found that within the first 10 years after the initial diagnosis, 8% of nodules showed disease progression, and 3.8% of nodules showed lymph node metastasis. Progression was more frequent in younger patients. 17 In patients with intermediate to advanced-stage PTC, there is a relatively higher risk of mortality. Under the current diagnostic and treatment framework, once diagnosed and in the absence of significant contraindications, it is advisable to promptly undergo standardized treatment. 13 Studying the natural progression of PTC requires a long-term follow-up period, including a substantial number of diagnosed but untreated patients. For intermediate to advanced-stage PTC, conducting large-scale observations of its natural course in clinical practice is nearly impossible.
The Surveillance, Epidemiology, and End Results (SEER) (https://seer.cancer.gov/) database is a large public database covering approximately 28% of the U.S. population, providing clinical information about patient demographics, tumor morphology, diagnosis, treatment, and prognosis. 18 Given its extensive records on PTC patients who haven’t undergone treatment, coupled with its exhaustive follow-up information spanning considerable durations, the SEER database presents a viable platform for our investigation. Therefore, in this study, we used the SEER database to statistically analyze the natural history of untreated PTC. Furthermore, we delve into the determinants impacting PTC prognosis and endeavor to formulate a prognostic model, thereby furnishing a robust framework to steer clinical diagnostic and therapeutic strategies.
Materials and Methods
Data Source and Study Population
Data for our research was sourced from the SEER program under the auspices of the National Cancer Institute. This study protocol was approved by the Ethics Committee of our research center. All information from the SEER program is available and free for public, and all patient data are de-identified, so informed consent is not required. Patient data from the SEER database (Version:8.4.1, https://seer.cancer.gov/data-software/) were screened. The research primarily centered around patients diagnosed with PTC between the years 2004 and 2020. The criteria for inclusion and exclusion for PTC patients are detailed subsequently. A schematic representation of the patient screening process can be observed in Figure 1. Flow chart detailing the selection of the patients in this study.
Criteria for inclusion comprised: (1) The tumor’s primary origin was identified within the thyroid gland. (2) Histological validation confirmed a PTC diagnosis, as reflected by the ICD-O-3 histology code references: 8050, 8260 and 8340-8344. (3) The time frame for diagnosis was established between the years 2004 and 2020. (4) Patients had surpassed the age of 18 at the time of diagnosis.
Exclusion criteria: (1) Not the only primary malignancy. (2) Incomplete clinical pathological data or unknown records. (3) Diagnosis of PTC from autopsy or death.
Assessing Variables
Since the SEER database currently lacks information on treatments other than surgery, radiation therapy, and chemotherapy, and considering that surgery and radiation therapy are the most crucial and commonly used treatment approaches for PTC,10,11 in this study, patients who have not undergone surgical treatment, radiation therapy, or chemotherapy are defined as the untreated group. Following the established inclusion and exclusion criteria, the study included 124,965 suitable patients, with 123,533 in the treated group and 1432 in the untreated group. Demographically, participants were categorized based on their age at the time of diagnosis into three age brackets: below 45, 45-59, and 60 and above. Racial profiles were grouped as white, black, and other ethnicities. Marital status differentiated between those married and those not. Gender considerations distinguished females from males. Clinically, we assessed tumors based on dimensions, grouping them into sizes of 0-20 mm, 21-40 mm, and exceeding 40 mm. The tumor stage, modified in line with the seventh edition of the AJCC staging criteria, spanned stages I through IV. T stage categories ranged from T1 to T4, while N stage classifications covered N1 to N3, and the M stage was split between M0 and M1. The patients in the untreated group were staged based on the clinical staging of PTC.
Survival Analysis
We employed the Kaplan-Meier methodology to predict survival outcomes, with the overall survival (OS) duration determined from the latest PTC diagnosis to the date of patient expiration. The point where half of the study population reached their survival endpoint was termed the Median Survival Time (MST). When fewer than half of the subjects achieved the survival endpoint, we turned to the survival rate for a more accurate representation. To determine “life loss,” the progression duration was discerned by subtracting the MST of a more severe stage from its preceding stage. 14 This study harnessed progression time to gauge the natural advancement in individuals diagnosed with intermediate-advanced PTC.
Construction and Validation of the Nomogram
To establish and verify the predictive nomogram, the untreated patients were randomly divided into the training set and the validation set in a 7:3 ratio. The initial analysis utilized univariate Cox regression to pinpoint prognostic risk factors in these untreated subjects. Factors that achieved statistical significance were then subjected to a more rigorous multivariate Cox regression examination. Subsequently, elements registering a P-value less than .05 in this multivariate analysis were recognized as standalone risk influencers. These factors formed the foundation of our nomogram aimed at forecasting the prognosis for PTC patients without treatment. The discriminative prowess of our nomogram was gauged through the concordance index (C-index) and the receiver operating characteristic curve (ROC). Additionally, calibration curves showcased the alignment between predicted likelihoods and actual patient outcomes. To ascertain the model’s clinical viability and advantages, we resorted to decision curve analysis (DCA).
Statistical Analysis
For categorical data analysis, we employed either the chi-square test or the Fisher’s exact test. The t test served to compare the continuous variables. We utilized the Kaplan-Meier technique for cumulative survival time computation, with the log-rank test analyzing survival curve discrepancies. Both single-variable and multi-variable Cox proportional hazards regression assessments were initiated to ascertain impactful variables on the prognosis for untreated PTC patients within the training dataset. All these statistical evaluations were executed with the aid of Stata15.0 Software. A threshold of P < .05 was set to indicate statistical significance.
Results
Clinical and Pathological Characteristics
Demographic and Clinicopathological Characteristics of the Papillary Thyroid Cancer Patients.
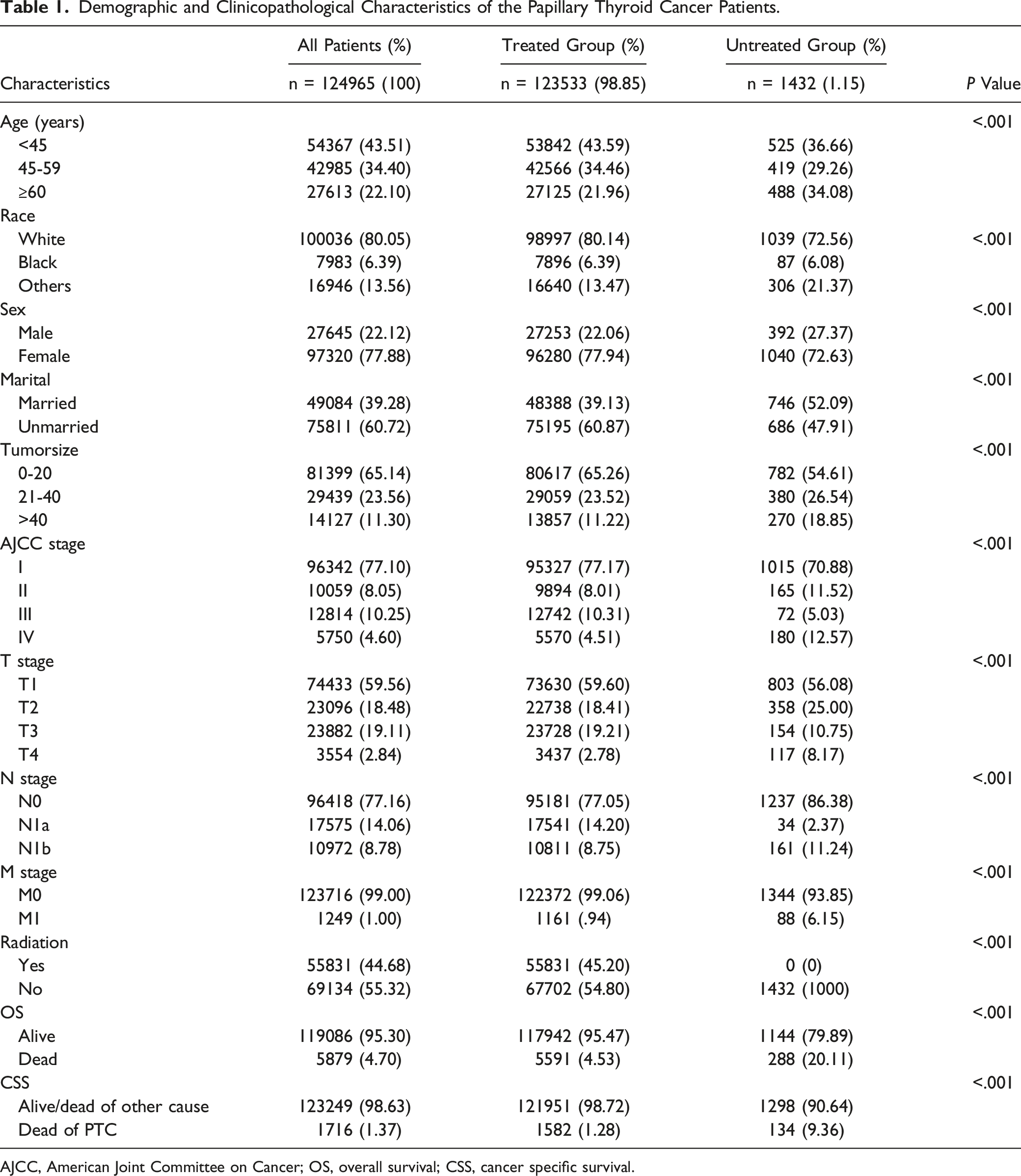
AJCC, American Joint Committee on Cancer; OS, overall survival; CSS, cancer specific survival.
Demographic and Clinicopathological Characteristics of the Papillary Thyroid Cancer Patients After PSM.
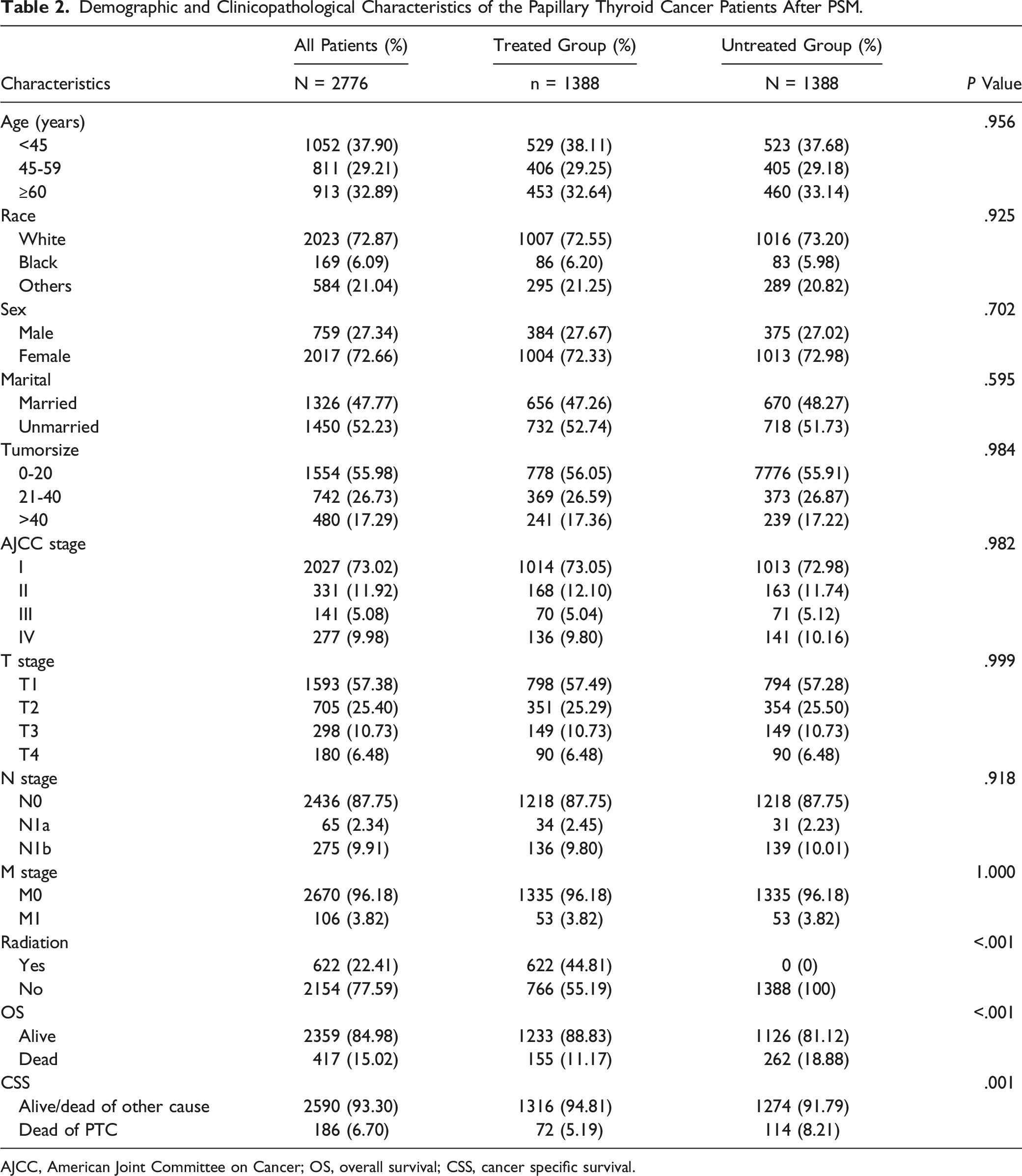
AJCC, American Joint Committee on Cancer; OS, overall survival; CSS, cancer specific survival.

Survival curves for PTC patients in treated and untreated groups. (A): overall survival (OS) before PSM, (B): overall survival (OS) after PSM, (C): cancer-specific survival (CSS) before PSM, (D): cancer-specific survival (CSS) after PSM.
Ten-Year Survival Rate After PSM in Treated and Untreated Groups.


Overall survival curve for PTC patients with tumor size less than 10 mm, age less than 45 years, T stage T1, N stage N0.
The Natural History of Untreated PTC
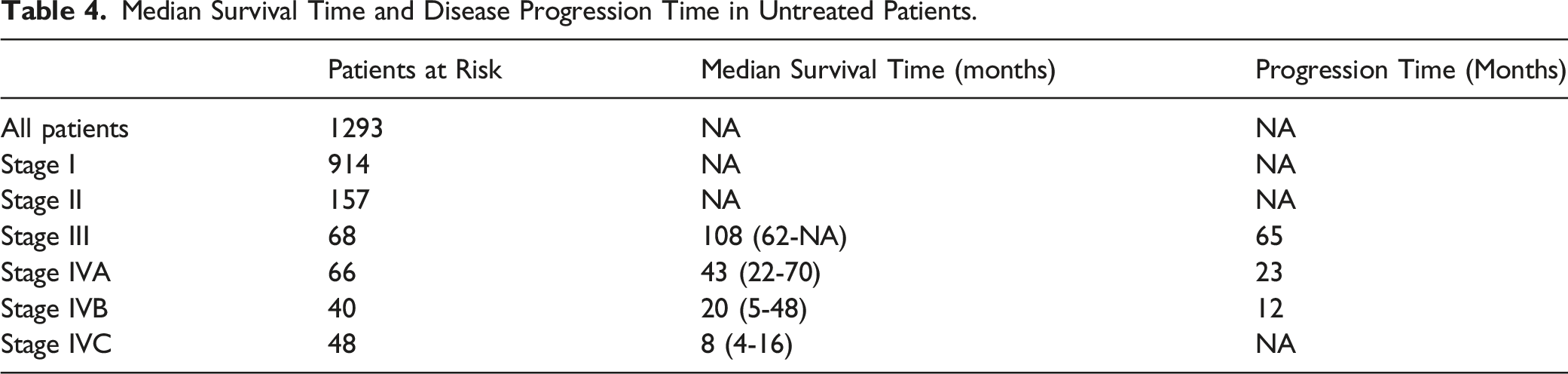
Kaplan-Meier survival curves for the untreated cohort are depicted in Figure 4, highlighting the decline in prognosis with escalating stage severity. The conclusion of our observation saw patients with stages I and II not attaining the median survival time, boasting 10-year survival percentages at 86.34% (95%CI: 82.89%-89.14%) and 66.03% (95%CI: 54.02%-75.59%) respectively. Furthermore, the 15-year survival rates stood at 83.87% (95%CI: 77.28%-88.69%) for stage I and 54.04% (95%CI: 37.87%-67.71%) for stage II. Stage III patients observed a median survival span of 108 months. In contrast, individuals in stages IVA, IVB, and IVC saw median survivals of 43, 20, and 8 months respectively. By analyzing the differential in median survival across stages, progression intervals between stages III to IVC were estimated at 65, 23, and 12 months (refer to Table 4). Yet, the early-stage PTC patient group, which demonstrated slow progression, failed to reach median survival, rendering its calculation via this method impractical. Overall survival curve for PTC patients in the untreated group. Median Survival Time and Disease Progression Time in Untreated Patients.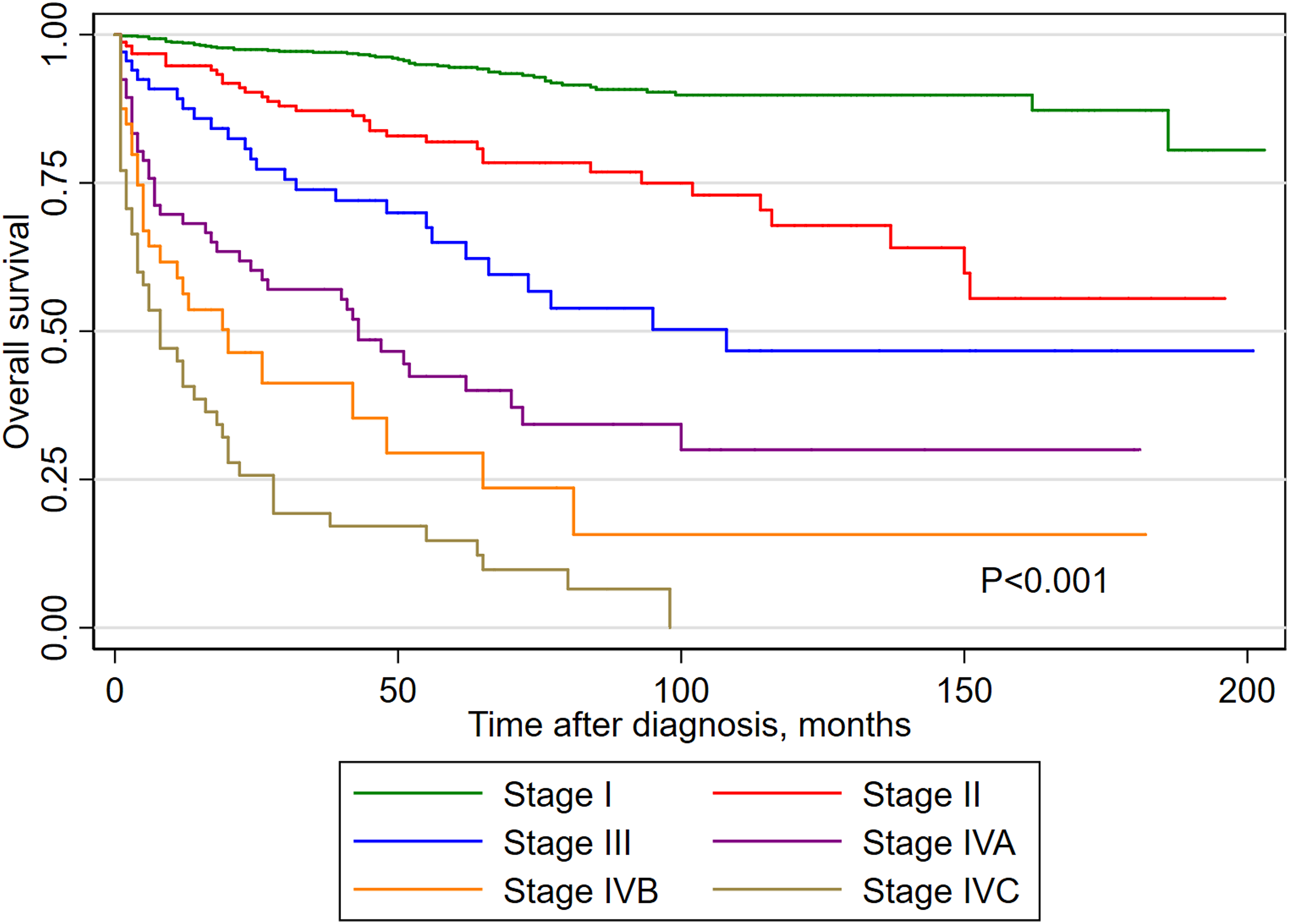
Variables Associated with the Prognosis of Untreated PTC
Univariate and Multivariate Cox Regression Analysis (Training Cohort).

Construction and Validation of a Nomogram
Utilizing the identified independent risk parameters (age, gender, marital status, tumor phase) from the cox regression analysis, a prognostic nomogram was constructed to forecast 5-year and 10-year survival outcomes in PTC patients (Refer to Figure 5). The analysis emphasized age as the most influential determinant in PTC prognosis, followed closely by tumor phase, whereas marital status and gender demonstrated lesser influence. By integrating the scores derived from each determinant, we derived a cumulative score to estimate 5-year and 10-year survival rates for individual patients. Nomogram predicting the 5- and 10-year overall survival (OS) of untreated patients with PTC using four factors (i.e. age, tumor stage, marital status and gender).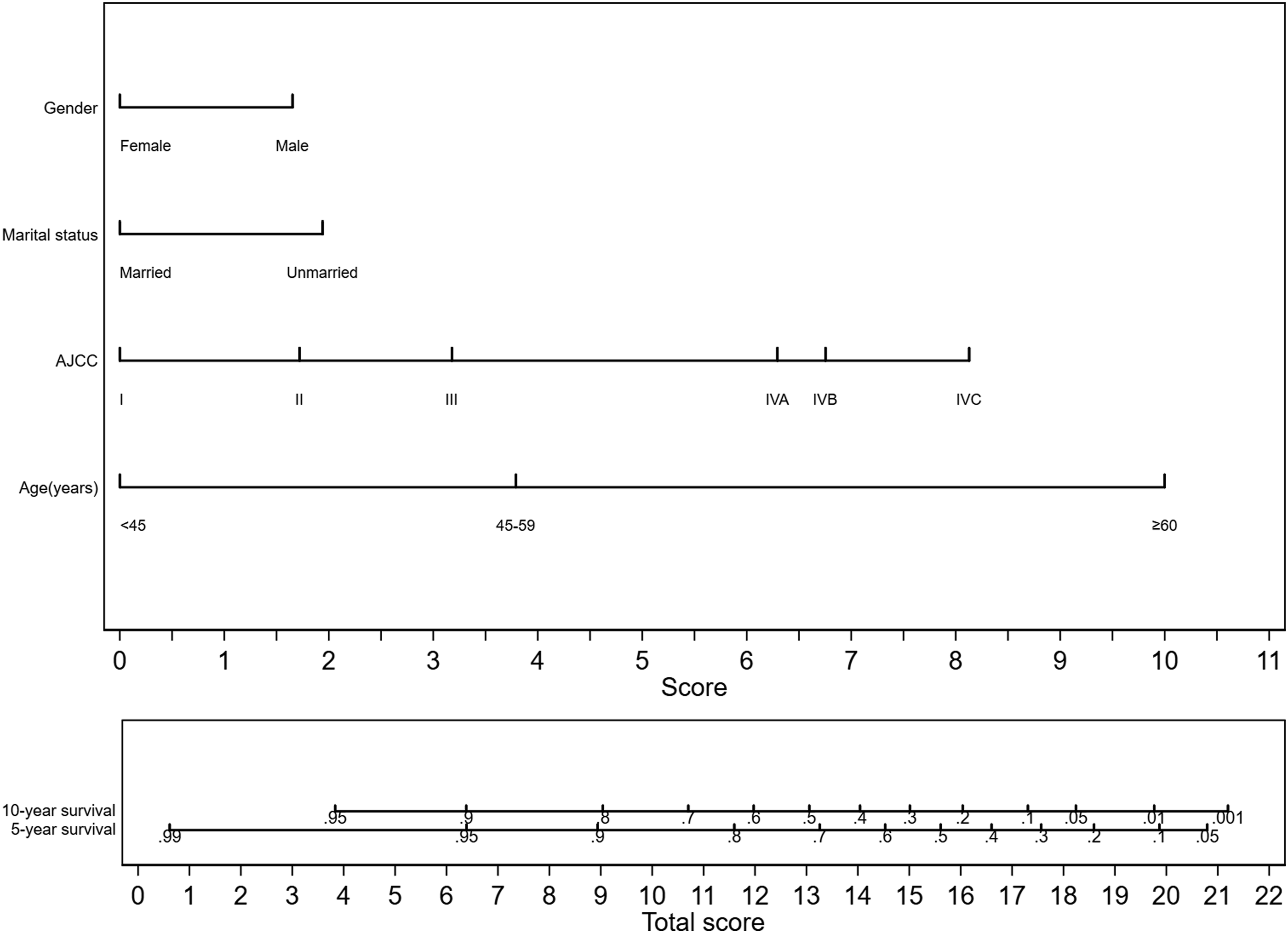
To validate the nomogram’s predictive prowess, we turned to the C-index and ROC curve methodologies. The C-index of the nomogram was calculated as .8887 (95% CI: .8681-.9093) in the training set and .9131 (95% CI: .8846-.9416) in the validation set. The accompanying ROC curves, showcased in Figure 6, were in alignment with the C-index outcomes, underscoring the model’s robust predictive capability. Figure 7 depicts a calibration plot, where the 95% CI of the calibration belt in both data sets closely adheres to the diagonal bisector, signifying an impressive alignment between forecasted probabilities and actual observations. Lastly, employing the DCA allowed for a comparative assessment of the utility and benefits of the constructed nomogram vs the conventional AJCC TNM staging method, as illustrated in Figure 8. The analysis unequivocally highlighted the nomogram’s enhanced practicality in gauging PTC patient prognosis compared to its TNM counterpart. ROC curves of the nomogram for predicting overall survival (OS) in training ((A), 5-year; (C), 10-year) and validation ((B), 5-year; (D), 10-year) sets. Calibration plots of the nomogram for predicting overall survival (OS) in training ((A), 5-year; (C), 10-year) and validation ((B), 5-year; (D), 10-year) sets. Decision curve analysis for the Nomogram and AJCC TNM stage system in prediction of overall survival (OS) in training ((A), 5-year; (C), 10-year) and validation ((B), 5-year; (D), 10-year) sets.


Discussion
Since the 1970s, the incidence of PTC has been steadily increasing, but the associated mortality rate has not shown a significant increase. 19 It is currently widely believed that the increase in the incidence of PTC is mainly due to the widespread use of advanced sensitive diagnostic equipment, leading to an increase in the detection rate of PTC, especially small cancer foci. Therefore, it is not considered a true increase in the incidence.20,21 The overdiagnosis and overtreatment of small PTC have not improved patient prognosis but have instead added additional burdens and may lead to surgery-related complications. 15 Therefore, according to the guidelines of the American Thyroid Association (ATA), surgical intervention was not considered the only available treatment option for micro papillary thyroid cancer; active surveillance was increasingly recognized as the best alternative strategy for low-risk groups. 22 Furthermore, Active surveillance in the research and application of PTC also provides real case data for understanding the natural history of early-stage PTC.
A recent review analyzed multiple global studies focusing on active surveillance of micro PTC. Within the seven studies assessed, the duration of monitoring varied between 6 to 312 months. The findings showed that tumor growth was observed in 1%–26% of the subjects, 1%–3% had new lymph node metastatic developments, and notably, over 70% did not show any signs of disease advancement. Only a minority exhibited tumor spread beyond the thyroid, with zero instances of distant metastasis or tumor-induced fatalities. 15 Intriguingly, in this study, in patients below the age of 45 with tumors under 10 mm, categorized as stage T1 and N0, there was a negligible difference in outcome between those who underwent treatment and those who did not. This was in line with earlier studies advocating active surveillance. These research findings demonstrate that micro PTC often maintained a favorable prognosis even without therapeutic intervention, with the disease typically not progressing or progressing slowly. A minority of patients who did exhibit disease progression could still derive substantial benefits from prompt surgical intervention, thereby highlighting that active surveillance was a safe and viable option for micro PTC. 23 The safe application of active surveillance in PTC patients with tumors smaller than 10 mm encourages further exploration of its use in PTC by researchers. Sakai T et al applied active surveillance to PTC staged as T1bN0M0 and found that after an average of 7.4 years of active surveillance, the size of 29 cases (8%) of T1aN0M0 tumors and 4 cases (7%) of T1bN0M0 tumors increased, with no significant difference in progression rates between the 2 groups. 24 Tuttle RM et al discovered that active surveillance is safe and feasible for low-risk PTC with a diameter smaller than 15 mm16. In this study, we found no significant difference in prognosis between the treated and untreated groups for PTC staged as T1bN0M0 in patients younger than 45 years. This indicates that active surveillance is safe and feasible in young patients with T1bN0M0 PTC. Gerwen M et al compared the prognosis of PTC patients who underwent surgery with those who refused surgery form the SEER database. The results showed that while the disease-specific survival rate of the surgery group was slightly higher than that of the refusal group when the tumor was confined locally, the difference was not statistically significant. 19 This suggests that active surveillance could have a broader application in PTC management and also reflects the predominantly slow progression of early-stage papillary thyroid cancer’s natural history.
Patients with mid to advanced-stage PTC face a higher risk of recurrence and metastasis, with the disease progressing more rapidly. The majority of thyroid cancer-related deaths occur among high-risk patients in the mid to advanced stages, who rightfully deserve more attention. 13 Therefore, in this study, we conducted an in-depth exploration of the natural history of mid to advanced-stage PTC. The results indicated that for PTC patients aged over 45 in mid to late stages, disease progression rates vary based on tumor stages: advancing from stage III to IVA took about 65 months, IVA to IVB spanned roughly 23 months, and transitioning from IVB to IVC was approximately 12 months. The data implies an acceleration in disease progression correlating with stage elevation. Therefore, advocating active surveillance for patients in these stages of PTC is inappropriate. Instead, once a diagnosis is confirmed for mid to late-stage PTC, immediate and intensive therapeutic measures are crucial to curb disease progression and optimize the prognosis.
To further understand the natural history of PTC and the factors affecting the prognosis of PTC patients without treatment intervention, we delved deeper into potential influencing factors. Our findings highlighted age, tumor staging, marital status, and gender as distinct determinants impacting PTC outcomes. Leveraging these identified risks, we crafted a nomogram to forecast the outcome of PTC when left untreated. Upon rigorous internal validation, this nomogram proved its efficacy and precision. Notably, when juxtaposed against the prevalent TNM tumor staging methodology, our nomogram exhibited superior utility and relevance. The determinants incorporated in this model stem from readily accessible clinical and pathological data, ensuring its broad clinical adaptability.
Age is a crucial component of our predictive model and a unique factor affecting the prognosis of PTC. 25 A consistent decline is seen in both general and malignancy-specific survival rates as patients age, implying an elevated mortality risk. 26 The currently recognized thyroid cancer prognosis systems, such as the Mayo Clinic’s MACIS score and the AJCC’s PTC staging model, both include patient age as part of their risk stratification algorithms, suggesting that PTC patients below a certain age have a lower risk than older individuals with the same tumor burden.27,28 Aligning with this, our research findings corroborate the idea that the prognosis of PTC diminishes as age advances. On one hand, older patients inherently have a relatively shorter life expectancy and are more likely to have other fatal diseases, such as cardiovascular and respiratory diseases.26,29 On the other hand, advancing age in PTC scenarios often manifests as heightened disease aggression, characterized by advanced tumor stages, escalated instances of distant metastases, and more expansive tumors, cumulatively exacerbating the prognosis. 27
Gender was another crucial component of our predictive model and a well-recognized factor that affected the incidence and prognosis of thyroid cancer.30,31 Gender played a significant role in the epidemiology of thyroid cancer, with women having a significantly higher risk of developing the disease than men. This disparity in incidence rates may be related to the effects of sex hormones or the fact that women are more likely to undergo thyroid examinations. 32 Interestingly, despite the higher incidence of thyroid cancer among women, female thyroid cancer patients generally had a better prognosis. 33 In our study participants, women constituted 77.88%, and men made up 22.12%, with a male-to-female ratio of 3.52:1. In the untreated cohort, women accounted for 72.63%, and men accounted for 27.37%, with a gender ratio of 2.65:1. We observed that women not only displayed a higher incidence but also were more inclined to receive treatment than men. Furthermore, female patients showed a better prognosis than male patients, regardless of whether they were in the treated or untreated group. Marital status was another component of our predictive model, with patients of different marital statuses potentially having varying mental health statuses. 34 Mental health, invariably shaped by both social backing and environmental dynamics, holds profound implications for treatment outcomes, particularly for those diagnosed with cancer. Evidence suggests that married individuals often experience heightened emotional resilience and consistently benefit from augmented social networks, spanning emotional and financial domains. This fortified support structure enables them to direct undivided attention to their treatment, fostering enhanced recuperative prospects from malignant conditions.34,35 Retrospective research has underscored the significance of marital status as a determinant of prognosis in PTC patients. The data indicates that, relative to their married counterparts, widowed patients confront an escalated risk of mortality from thyroid cancer, irrespective of gender, age stratification, or cancer progression stage. 36 Our investigation corroborates the predictive value of marital status in PTC prognosis, spotlighting superior outcomes for those married.
In a broader perspective, untreated PTC patients’ prognosis influencers largely mirror non-treatment determinants highlighted in earlier literature. Through our examination, age at diagnosis and tumor progression emerged as pronounced prognostic determinants for PTC, closely trailed by gender and marital status. Leveraging these identified risk facets, we’ve crafted a prognosis-centric model, surpassing the practicality of the AJCC system, aimed at guiding clinical application and practice.
Limitations
This study has certain limitations that should be taken into consideration. With the revision of the WHO classification for papillary thyroid carcinoma, the inclusion criteria for papillary thyroid carcinoma within the SEER database prior to 2017 might encompass a minority of noninvasive follicular thyroid neoplasms exhibiting papillary-like nuclear features (NIFTP) and thyroid neoplasms of indeterminate malignant potential (UMP). 37 This inclusion has the potential to skew the outcomes of prognostic evaluations. The lack of detailed patient comorbidity data within the SEER database precludes analysis of comorbidity influences on patient prognoses. Additionally, the database does not capture the motivations behind patient decisions to decline certain treatments, limiting our understanding of treatment refusal. The retrospective design of this study inherently carries biases, such as the exclusion of cases with incomplete documentation. Solely employing the SEER database for both the development and validation of the model might raise concerns regarding model overfitting. For a thorough validation of the model’s efficacy, it would be beneficial to test it against external datasets in future studies.
Conclusion
In our research, we delved into the natural trajectory of PTC left untreated to gain insights into its growth pattern. We observed that early-stage PTC patients had a good prognosis, with the disease progressing slowly even in the absence of treatment intervention; conversely, mid to advanced-stage PTC patients had a poorer prognosis, and the rate of disease progression increased significantly with the advancement of tumor staging. Among the variables studied, age, tumor stage, marital status, and gender emerged as standalone determinants influencing PTC prognosis. The model we crafted around these determinants exhibited strong predictive precision for the outcomes of PTC patients who went untreated.
Footnotes
Acknowledgments
The authors would like to thank SEER for the open access to the database.
Author Contributions
Dasong Wang contributed to the study conception and design. Dasong Wang, Yan Yang and Yu He participate in writing proposal, analyzed the data, wrote the result and discussion. Dasong Wang, Hongwei Yang and Lei Yang participate in analyzing the data, writing result and prepared manuscript. All authors commented on previous versions of the manuscript, and all authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Scientific research project of Suining Central Hospital (No. 2022ypj20).
Ethical Statement
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
