Abstract
Background
FOLFOX (leucovorin calcium [folinic acid], fluorouracil, and oxaliplatin) combined with or without anti-VEGF therapy represents one of the primary first-line treatment options for metastatic colorectal carcinoma (mCRC). However, there is limited comparative data on the impact of anti-VEGF therapy on treatment effectiveness, survival outcomes, and tumor location.
Methods
This retrospective, comparative study utilized data from the AIM Cancer Care Quality Program and commercially insured patients treated at medical oncology clinics in the US. We analyzed 1652 mCRC patients who received FOLFOX, of which 1015 (61.4%) were also treated with anti-VEGF therapy (VEGF cohort).
Results
Patients in the VEGF cohort exhibited a higher frequency of lung (33% vs 23%; P < .001) and liver metastases (74% vs 62%; P < .001), underwent fewer liver surgeries prior to treatment (1.2% vs 3.6%; P = .002), and had a higher proportion of right-sided tumors (27% vs 18%; P = .001). Adjusted analysis revealed no significant difference in overall survival (OS) between patients treated with and without anti-VEGF (median survival: 25.4 vs 26.0 months; P = .4). FOLFOX-only treated patients experienced higher rates of post-treatment hospitalizations (22% vs 15%; P < .001). Notably, left-sided tumors treated with anti-VEGF showed a trend toward decreased OS (median survival: 26.8 vs 33 months; P = .09).
Conclusion
Our real-world data analysis suggests that the addition of anti-VEGF to FOLFOX offers limited and short-lived benefits in the context of mCRC and may provide differential survival benefit based on tumor sidedness.
Introduction
Colorectal cancer, CRC, is the third most commonly diagnosed cancer in males and the second in females, worldwide. 1 Every year, approximately 150 000 new patients with CRC are diagnosed in the United States, and approximately 50 000 die of CRC. 2 Of new colorectal cancer diagnoses, 20% of patients have metastatic disease at presentation and another 25% who present with localized disease will later develop metastases. In early and locally advanced CRC, surgery and adjuvant chemotherapy are used with the aim to cure the disease, whereas treatment of metastatic disease is aimed at prolonging life and preserving quality of life.
The treatment for metastatic colorectal cancer (mCRC) has advanced significantly over the past two decades. Two decades ago, 5-fluorouracil (5-FU) was the only active treatment with a median overall survival of approximately one year, 3 while nowadays with newer agents and treatment combinations median survival can reach 2–3 years. Treatment agents for colorectal cancer today include the anti-topoisomerase irinotecan, the platinum agent oxaliplatin, the anti-EGFR monoclonal antibodies cetuximab and panitumumab, and the anti-VEGF antibody bevacizumab or its biosimilars. 2 The optimal combination of these agents in the first- and later treatment lines is a paradigm that is continually evolving, and different biomarkers are used to determine which is the best treatment for an individual patient. Another recently evolving predictive factor identified from post hoc analyses of various phase 3 trials suggested that the location of the primary tumor is an important predictive factor for first-line (1L) usage of EGFR inhibitors; thus, these drugs are usually recommended as 1L just for patients with left-sided primary tumors.4,5 The evidence for an overall survival (OS) benefit of VEGF inhibitors in the context of oxaliplatin- or irinotecan-based chemotherapies is limited and inconsistent and is based on clinical trials conducted almost two decades ago, with some studies showing a significant benefit in OS to anti-VEGF treatment6–8 and others do not.9–11 It should be noted that VEGF inhibitors are associated with increased toxicities, including proteinuria, hypertension, bleeding, bowel perforation, impaired wound healing, and arterial thromboembolic events, 12 along with added costs, emphasizing the need to monitor their efficacy.
Real-world studies are a very important tool to evaluate the actual usage and effectiveness of medical treatments which were already included into common practice by randomized control trials. Previous studies that compared real-world data to clinical trials and used proper adjustments have shown similar OS in NSCLC in the 2L setting. 13 Other studies have leveraged real-world data to provide evidence in the absence of a randomized phase 3 trial or to further characterize patient outcomes in the real world for metastatic breast cancer patients 14 or in metastatic pancreatic cancer,15,16 or to explore the prognostic and predictive role of tumor location in mCRC. 15
Although VEGF inhibitors together with FOLFOX are the most common treatment for mCRC in 1L setting, with strong evidence on positive impact on PFS, the evidence supporting the impact on survival is limited, as well as data supporting the role of tumor location for this treatment. Here, we utilized a large real-world clinical dataset from over 1600 mCRC patients across multiple regions and health care systems in the United States and explored survival outcomes and post-treatment hospitalizations associated with FOLFOX alone or in combination with VEGF inhibitors.
Materials and Methods
Data Source
This retrospective analysis utilizes nationally representative clinical data provided to the AIM Cancer Care Quality Program, along with corresponding claims and lab data. 17 Submissions via the AIM portal encompass various details, including the planned treatment regimen, treatment date, TNM staging, therapy sequence, ECOG PS, ICD-10 diagnosis code, and KRAS status (available for 30% of patients). The claim data covers medical and pharmacy claims for nearly 40 million members under 14 U.S. regional health plans with commercial insurance, excluding government, Medicare, and Medicaid claims. All data were anonymized for patient privacy. Mortality information was gathered from discharge status, national death registries, and obituary data. The patients originate from multiple outpatient centers and medical oncology clinics in numerous U.S. states. The index date is established as the start of first-line therapy as per the request, confirmed via administrative claims. Patients without mortality events were assessed until their final medical claim date or the study’s conclusion. The study adheres to STROBE guidelines. 18
Covariates and Outcomes
The primary measure of effectiveness centered on overall survival (OS), which was defined as the period from the treatment index date to the time of death or the most recent follow-up in cases of censorship. A secondary outcome involved post-treatment hospitalizations, identified within 30, 60, and 90 days after the index date. Initial characteristics considered encompassed prognostic stage, ECOG PS, sites of metastasis, instances of liver surgery before or after treatment, existing medical conditions, sociodemographic particulars, tumor orientation, and KRAS status. The dataset employed for analysis was rendered anonymous following the principles of Safe Harbor privacy. It excluded personal identifiers like names, precise dates (only relative intervals were retained), postal codes (only rounded SDI), and any other information that could lead to identification.
Covariate Calculation
The PS score is determined by the treating physician at the treatment’s outset. Comorbidities were evaluated using the Charlson Comorbidity Index (CCI) through patient claims within one year before and up to 10 days before the index date. The R comorbidity package 19 computed the CCI, consisting of 17 medical conditions; scores excluded metastasis and cancer diagnoses. Metastatic sites were identified using ICD-10 codes associated with metastasis in the year before and up to the index date. Sociodemographic data encompassed age, gender, and social deprivation index (SDI), determined from patient zip codes. ECOG PS, SDI score, and CCI, numerical variables, were normalized via min–max normalization. Prior liver surgeries were assessed using related CPT codes up to one year before the index date; post-treatment liver surgeries followed a similar evaluation with CPT codes within one year after the index date. KRAS status was available for only 30% of patients. Tumor sidedness was approximated using the ICD-10 diagnosis from the request and mapping provided in a previous study. 20 Rectal tumors were considered left-sided tumors after an analysis revealing comparable KRAS and MSI status in rectum patients and left-sided patients.
Adjustment
Matching
Employing the MatchIt R package (Version 4.3.1, R), 21 a 1:1 matching process was conducted, utilizing the Mahalanobis distance metric and incorporating all the mentioned covariates. Following matching, P-values were computed for the source variables to confirm the absence of significant disparities between the matched groups (see Table 1). Subsequently, the Kaplan–Meier estimator was applied to the matched cohort.
IPTW
Inverse probability of treatment weighting (IPTW) was employed through a propensity score analysis. The propensity score was derived using a logistic regression model with treatment type as the dependent variable, factored against potentially confounding variables. To compute and assess inverse propensity weighting, the Python causalib package (Version .1.3, IBM Python) was utilized. Evaluation of IPTW adjustment quality included ROC curve analysis with and without weights, assessing the propensity distribution, Absolute Standard mean difference before and after adjustment, and utilizing a calibration curve (refer to Supplementary Figure 2). For survival assessment, the Kaplan–Meier estimator was adjusted by the inverse propensity score.
Statistical Analysis
Patient Characteristics of the FOLFOX-Treated Cohorts.

aScores range from 1 to 100 with higher scores indicating greater deprivation (SDI).
bScores range from 0 to 2, with higher scores indicating worse status.
cCharlson index score was adjusted to exclude oncology diagnosis in the score.
Ethical Declaration
This study was designed as an analysis based on medical claims data, and there was no active enrollment or active follow-up of study subjects, and no data were collected directly from individuals. The study was not required to obtain additional IRB approval, as the HIPAA Privacy Rule permits protected health information (PHI) in a limited data set to be used or disclosed for research, without individual authorization, if certain criteria are met.
Results
Study Cohort
The study cohort included patients with a diagnosis of colorectal cancer stage IV between January 2016 and June 2021 at the start of their first-line treatment and ECOG performance score between 0 and 2. Based on this inclusion criteria, we identified 9003 patients. Only patients intended for treatment with FOLFOX-based regimens were included in analyses. Based on this inclusion criteria, we identified 6561 patients. Patients were followed until December 2021 or death (Figure 1). The treatment plan was defined using the clinical data available in the AIM Cancer Care Quality Program. Patients were excluded if the treatment plan could not be corroborated by the claims data, or if the line of treatment was higher than 1L, resulting in the “Treated cohort” with 2521 patients. Finally, only patients treated with FOLFOX only or FOLFOX and anti-VEGF were included in the final “FOLFOX cohort” resulting in 1652 patients. Flow chart of the FOLFOX mCRC 1L-treated patient cohorts.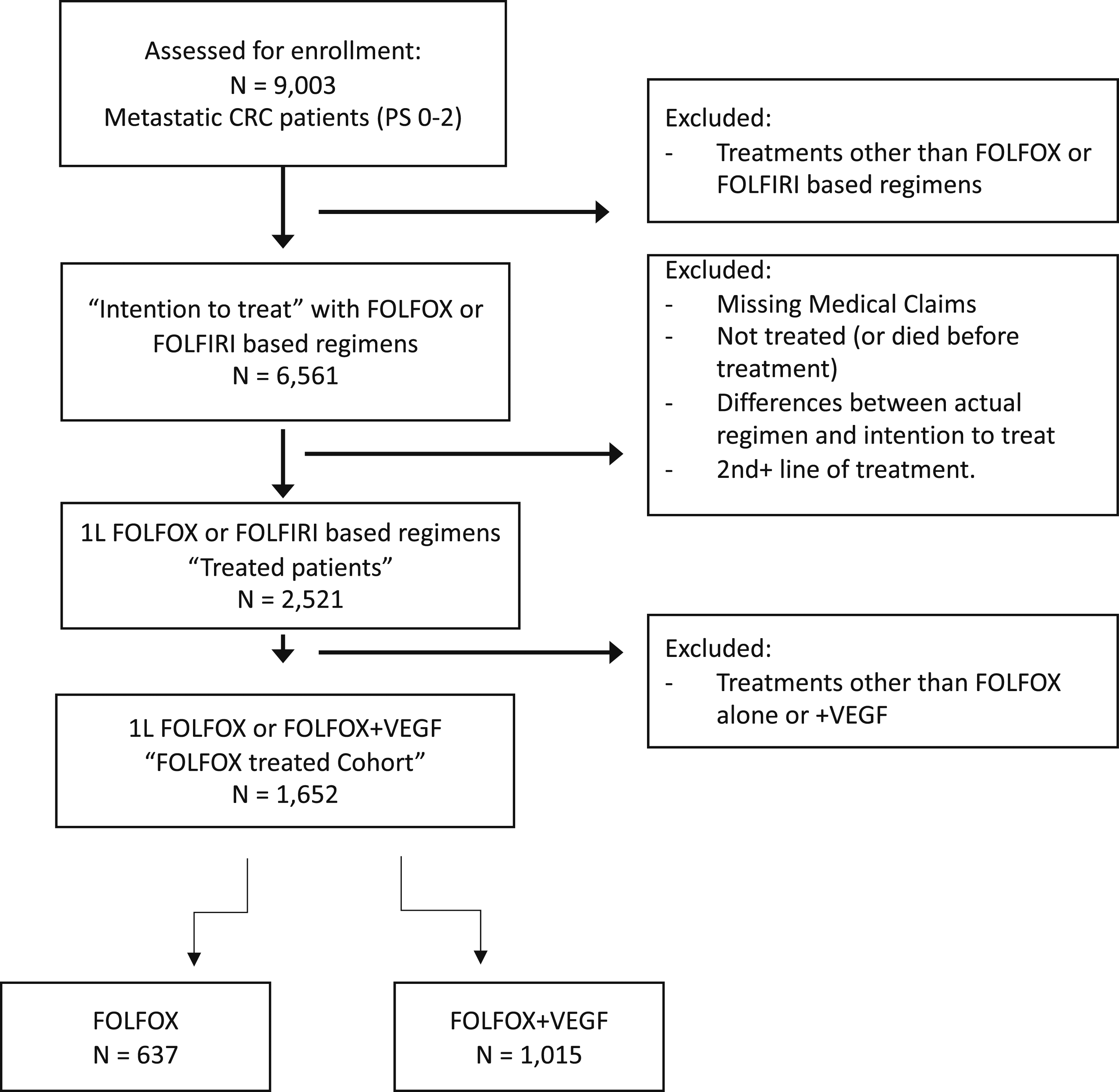
Trends in Treatments
Using clinical data and administrative claims, we identified 10 352 treatment requests (request for every treatment modification) for patients diagnosed with colorectal cancer stage IV and started between January 2016 and June 2021 their 1L treatment. The top regimens were FOLFOX- or FOLFIRI-based regimens (43% and 21%, respectively); however, many different combinations exist, including the FOLFIRINOX, Capecitabine, and CapeOX as other leading chemotherapies, and immune checkpoint blockade therapies as well (Figure 2A). We decided to focus our study on FOLFOX-based regimens, and for those requests we corroborated the clinical data with claims data to validate that the requests were executed, and patients were treated (Figure 1). In the validated “treated cohort.” FOLFOX combined with anti-VEGF (bevacizumab or MVASI) was the most common regimen accounting for 40.2% (1015/2521 patients), following FOLFOX-only (25.3%; 637/2521 patients). Interestingly, we observed a decline in the FOLFOX plus anti-VEGF combination in recent years with the FOLFOX-only regimen prevalence increasing (Figure 2B), suggesting clinicians started to reconsider the value of anti-VEGF treatment in the context of FOLFOX. Trends in treatments in 1L mCRC during 2016–2021. (A) Number of requests of different regimens for 1L mCRC in the intent to treat cohort. (B) Percentage of patients with first-line treatment of FOLFOX- and FOLFIRI-based regimens, normalized by year, between the years 2016 and 2021 in the treated cohort. (C) Length of cycles in days for patients treated with FOLFOX. (D–E) Stack density plot of the FOLFOX (in D) and FOLFOX plus VEGF patients (E) showing the percentage of patients who received any of these treatments: FOLFOX, FOLFOX + MVASI or VEGF, 5FU, 5FU + LV, 5FU + LV + Bev, or Bev only in each cycle (cycle length = 14 days) as well as patients that died, treatment was not reported, or don’t have a follow-up. (F–G) Similar plots, with only the treatment events in each cycle, in which case the 100% is different in each cycle and includes only patients who received any treatment from the list in this cycle.
We observed that even in the limited set of treatment decisions we focused on in this study, there are still many differences among patients in the treatment plan. First, cycle length differs among patients, as most patients receive treatment every 14 days, but cycle length can be 21 or 28 days (Figure 2C). Additionally, the treatment protocol is dynamic as treatment progresses, with the addition and removal of chemotherapies. In the FOLFOX-only cohort, we observed that after 10 cycles (with a cycle length of 14 days) of FOLFOX, there was an increase in the percentage of 5FU + LV treatment, suggesting oxaliplatin was removed due to toxicity or low tolerance (Figure 2D and 2F). After 20 cycles, there are less than 10% of patients that are still treated with FOLFOX, while the 5FU + LV is most dominant. We also observed that anti-VEGF may be added to the FOLFOX treatment after a few cycles for about 10% of the patients and there too oxaliplatin will be removed after around 10 cycles for most patients (Figure 2D and 2F). In the FOLFOX plus anti-VEGF cohort, we observed similar patterns, where oxaliplatin was removed even earlier, at around 7 cycles, as we observe an increase in 5FU + LV + Bev, and after around 15 cycles, this becomes the dominant treatment (Figure 2E and 2G).
Comparative Effectiveness of Addition of Anti-VEGF to FOLFOX
To study treatment effectiveness, we created a limited study cohort of 1652 patients treated with FOLFOX, with or without anti-VEGF treatment. Due to the observational nature of the study design, patients receiving FOLFOX differed in few demographic and clinical characteristics from those treated with FOLFOX plus anti-VEGF (Table 1). Some patients undergo liver resection (10.5% in our cohort), pre- or post-treatment, and treatment decision may be influenced by the planned surgery. Patients undergoing post-treatment liver resection may represent a better prognostic group and thus will be treated with FOLFOX only. Indeed, in our cohort there was a slight, but non-significant, enrichment of FOLFOX only compared to FOLFOX + anti-VEGF in patients undergoing post-treatment liver surgery (9.4% vs 7.8%; P-value = .28). Nevertheless, liver surgery was used as a confounder and was adjusted throughout our analyses. Of note, significant differences between the two groups are lung (33% vs 23% for FOLFOX plus anti-VEGF and FOLFOX alone, respectively; P-value <.001) and liver metastases (74% vs 62%; P-value <.001), liver surgeries before the treatment (1.2% vs 3.6%; P-value = .002), and right-sided tumors (27% vs 18% P-value = .001). There were no apparent socioeconomic differences in the decision to treat with anti-VEGF or not. Furthermore, we did not observe any significant difference between the treatment groups in terms of ECOG performance score, or biomarker status (KRAS), suggesting that both ECOG and KRAS status is not impacting the decision to treat with respect to anti-VEGF.
We compared the real-world effectiveness of FOLFOX with and without anti-VEGF with an end point of all-cause mortality. To adjust for the covariates, we performed IPTW (see method validation in Supplementary Figure 2a and in methods). Interestingly, the adjusted Kaplan–Meier fit showed no survival difference between the treatments (median survival: 25.4 vs 26 months for FOLFOX and FOLFOX and anti-VEGF, respectively, P-value = .7; Figure 3A). Similarly, an adjusted Cox proportional model using the IPTW as weights resulted in an HR of 1.08 (P-value = .36), again, suggesting no survival benefit for anti-VEGF treatment in the context of FOLFOX. We also used 1:1 matching to account for the confounders which resulted in sub-populations of 613 patients for each treatment, with similar results (Supplementary Figure 1b). Overall survival and hospitalization analysis of VEGF benefit. (A) Adjusted Kaplan–Meier fit for FOLFOX- and FOLFOX and anti-VEGF-treated patients; dashed lines and numbers show the median survival. (B) Percentage of hospitalization post-treatment, 30, 60, or 90 days after treatment start for patients treated with FOLFOX only or FOLFOX and anti-VEGF.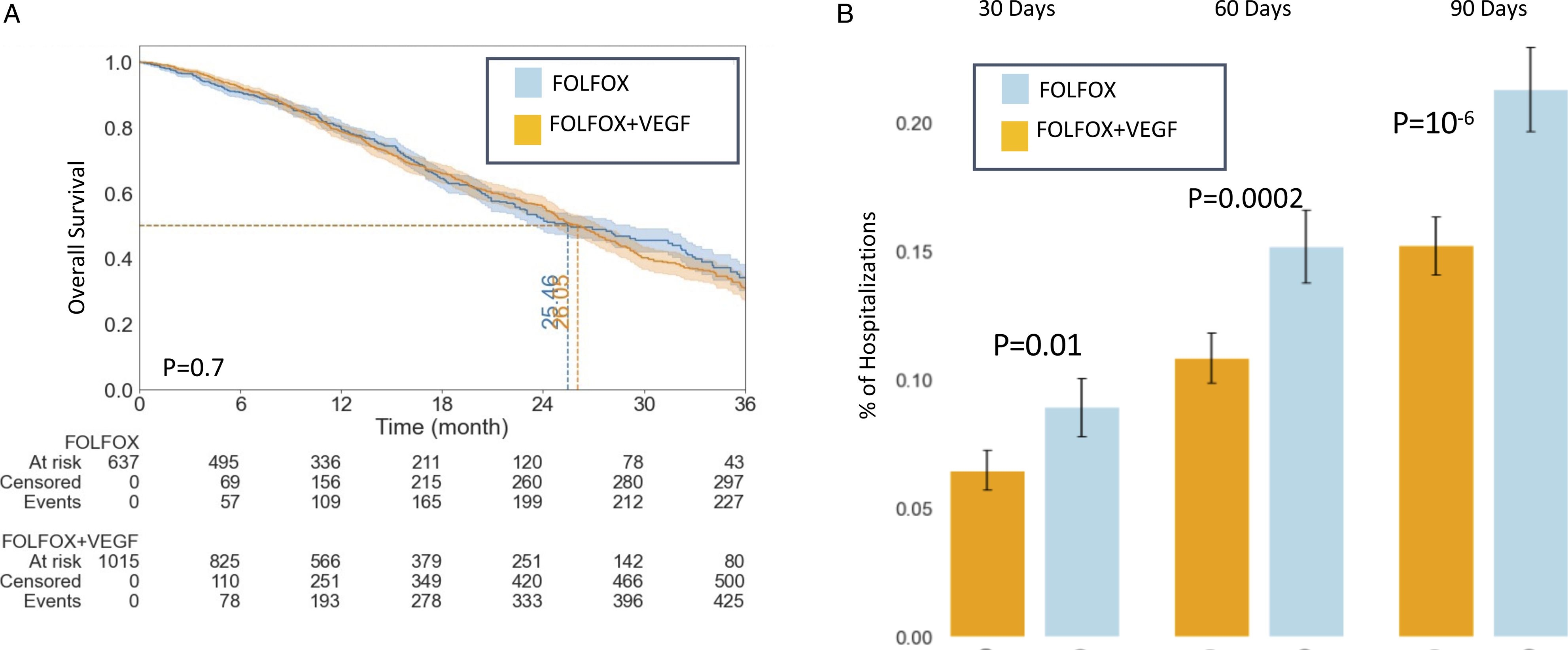
To study post-treatment effects, we compared hospitalization rates after the initiation of the treatment. Using both the matched cohort and IPTW adjustment, we found that anti-VEGF treatment leads to ∼29% less hospitalization events 90 days following the first treatment (P-value <.001; Figure 3B). A similar trend was observed for 30- and 60-day window. This analysis suggests some short-term benefits for anti-VEGF, supposedly reducing severe adverse reactions to the chemotherapies.
Tumor Sidedness
We next wanted to explore the impact of tumor sidedness on OS. About 65% of the patients could be annotated to right or left tumors based on their ICD-10 diagnosis code (Table 1). Right-sided CRC tumors are considered to have worse prognosis and different molecular landscapes. Similar to previous reports,
2
we found right-sided tumors were about twice as likely to be MSI-H (18% vs 9%, P-value = .37) and more commonly have KRAS sequence variations (59% vs 31%; P-value <.001). Tumor sidedness was also a strong prognostic feature in our data, where right-sided tumors are associated with shorter median OS (22.5 vs 28.3 months, P-value <.001; Figure 4A). KRAS mutational status was also shown to be associated with OS,
23
and indeed we observed worse survival for patients with KRAS mutation, although not significantly (24 vs 27 months, P-value = .19; Figure 4B). In terms of the benefit of anti-VEGF, in the left-sided tumors, after adjustments, anti-VEGF showed a trend toward decreased survival (26.8 vs 33.1 months, P-value = .09; Figure 4C), while in right-sided tumors, there was no difference in survival with or without anti-VEGF (22.3 vs 23.7 months, P-value = .9; Figure 4D). Interestingly, while 70% of the right-sided patients received anti-VEGF, only 58% of the left-sided patients received anti-VEGF (Figure 4E), suggesting clinicians might take tumor sidedness into their treatment decisions already. We also see within the tumor side a difference in anti-VEGF prevalence between KRAS status although less significant (Figure 4E). Overall survival analysis with sidedness of the tumor and KRAS status. (A–D) Adjusted Kaplan–Meier fits for left- and right-sided tumor patients treated with FOLFOX or FOLFOX + VEGF. Dashed lines and numbers show the median survival. (E) Pie charts representing how many patients were treated with FOLFOX (light blue) or FOLFOX + VEGF (orange) in different segments that include tumor sidedness (pink and gray chart) and KRAS status (green and red chart).
Discussion
In this study, we investigated the real-world effectiveness of the FOLFOX plus anti-VEGF combination in the first-line treatment of mCRC. Given the low probability of further randomized studies, real-world studies like ours play an important role in accumulating evidence on the efficacy of this treatment combination. Our results showed no statistically significant survival differences with the addition of anti-VEGF to the FOLFOX backbone. This finding is consistent with the phase 3 NO16966 trial, which also found no survival benefit when adding anti-VEGF to FOLFOX/XELOX regimens, 11 although there is conflicting evidence from other randomized controlled trials. 8
Although limited data support the OS benefit of anti-VEGF, it is still recommended for first-line treatment of mCRC by the NCCN guidelines. In our cohort, the FOLFOX plus anti-VEGF combination was the most prevalent treatment regimen. However, we observed a decline in the usage of this combination and a reciprocal rise in using FOLFOX alone. This might be due to the uncertainties regarding the efficacy of anti-VEGF, as well as concerns about toxicity and cost.
Tumor sidedness is recognized as a prognostic factor in colorectal cancer. 5 In our study, we confirmed the well-known worse prognosis of right-sided tumors and their tendency to harbor RAS mutation and MSI high tumors. We also found that anti-VEGF has no survival benefit for right-sided tumor patients and shows a trend toward decreased OS for patients with left-sided tumors (P-value = .09). This finding is in line with recent evidence suggesting that tumor sidedness is an important predictive factor for treatment in first-line therapies involving anti-EGFR agents, 4 although the evidence for anti-VEGF agents is less consistent.24-26
Treatment toxicity is an important consideration when evaluating the effectiveness of a treatment regimen. In our study, we measured treatment toxicity in terms of hospitalization during the treatment period. We found no evidence of increased toxicity when anti-VEGF was used in combination with FOLFOX, and in fact, we observed a significant reduction in the number of hospitalizations in the first months after treatment, suggesting a clinical and quality-of-life benefit from adding anti-VEGF to chemotherapy, consistent with previous reports of better PFS with the anti-VEGF. 11
Lastly, this study has several limitations. First, the use of claims data might result in coding differences by different practices and physicians. 16 However, our analysis is based on a combination of claims and clinical data from the AIM Cancer Care Quality Program. The clinical data was used to augment the administrative claims data, providing information such as the stage, line of treatment, ECOG performance status, and biomarkers. However, since the clinical data is only collected for pre-authorization decisions, information not relevant for the treatment decision is not collected, and therefore our data includes biomarker status data only for a subset of patients (in this case, KRAS status was only available for 30% of patients).
Second, our main outcomes were overall survival and post-treatment hospitalizations, and we may have ignored other clinically relevant outcomes such as progression-free survival (PFS) and quality of life metrics. Third, some patients could have left the insurance plan or used private services that are not well-documented. Finally, the observational nature of the study means that there may be inherent limitations from time-related biases and residual confounding from unmeasured factors.
Despite these limitations, our study provides valuable insights into the real-world effectiveness of the FOLFOX plus anti-VEGF combination in the first-line treatment of mCRC. Our findings support the use of this treatment combination in clinical practice, although the decision to use anti-VEGF should be made on a case-by-case basis, taking into account factors such as tumor sidedness and patient characteristics. Importantly, addition of anti-VEGF may not provide OS benefit but rather it seems to have more effect on toxic reactions. Further studies are needed to confirm our results and investigate the potential benefits and limitations of different treatment regimens in the first-line treatment of mCRC.
Conclusions
Although anti-VEGF therapy is very common in the treatment of mCRC 1L, there are inconsistent reports as to the efficacy and toxicity of it. Our study based on large cohort real-world data reveals that anti-VEGF is not associated with improved survival, although in the short term, it is associated with fewer post-treatment hospitalizations. Lastly, our study suggests that sidedness should be considered when treating with anti-VEGF in 1L of mCRC.
Supplemental Material
Supplemental Material - Comparative Analysis of First-Line FOLFOX Treatment With and Without Anti-VEGF Therapy in Metastatic Colorectal Carcinoma: A Real-World Data Study
Supplemental Material for Comparative Analysis of First-Line FOLFOX Treatment With and Without Anti-VEGF Therapy in Metastatic Colorectal Carcinoma: A Real-World Data Study by Ronen Brenner, Shlomit Amar-Farkash, Avital Klein-Brill, Keren Rosenberg-Katz, and Dvir Aran in Cancer Control.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Klein-Brill, Amar-Farkash, and Rosenberg-Katz are employees of Carelon Digital Platforms, Israel. Dr Aran reported receiving a salary from Carelon Digital Platforms Israel, Ltd.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
