Abstract
Introduction
Brain metastasis (BM) is an inauspicious finding in patients with pancreatic cancer, which significantly increases morbidity and mortality. The presentation is rare, and thus, outcomes on brain metastases from pancreatic cancer are limited.
Methods
This retrospective monocentric analysis included the electronic medical records of patients ≥ 18 years presenting with BM from pancreatic cancer. Clinical, demographic, and overall survival (OS) data were analyzed. Brain imaging (computed tomography, and magnetic resonance imaging) was used for BM diagnosis. OS was calculated from the date of diagnosis to either the date of last follow-up or death. Cox regression on OS time was used with an indicator variable for patients who ultimately developed a BM.
Results
Brain metastases was diagnosed in 34 (3.7%) patients with pancreatic cancer treated in our institution. Of the BM patients: 7 (22%) were non-White; 16 (47%) were female; the median age was 69; 35% were diagnosed at Stage II and III; and 65% were diagnosed at Stage IV. BM was the first diagnosed metastasis in 14 (41%) of the patients; in 5 individuals (15%) BM were detected within the first 100 days of pancreatic cancer diagnosis. OS from BM diagnosis was 9 months (95% CI [6.92,17.86]). The adjusted hazard ratio for patients with BM was .91(95% CI [.88, .94]; P = 0.60], which was not significant.
Conclusions
Due to the low incidence of BM in pancreatic cancer, identification of disease remains a current challenge. In this study, we found that BM from pancreatic cancer may be the first sign that the cancer has spread. Using advanced neuroimaging early may help physicians diagnose these metastases sooner and possibly improve survival. With a larger cohort size, the findings may be potentially impactful for clinicians and patients. Prospective, multicentric studies are warranted to identify prognostic factors for treatment and survival.
Plain Language Summary
Diagnosis remains a current challenge due to the low incidence of brain metastases in pancreatic cancer. Our study presents real-world data regarding the characteristics and survival of patients with brain metastases (BM) from pancreatic cancer. This study found that BM from pancreatic cancer can be the first sign that the cancer has spread. Using advanced brain imaging on higher risk patients could help physicians diagnose these metastases sooner, which may improve survival. This study collected initial data, which may be helpful for practitioners in gaining insights to help guide diagnosis and future treatments.
Keywords
Introduction
Pancreatic cancer is highly metastatic, difficult to diagnose due to its location and the commonality of the symptoms, and found at late stages. 1 Pancreatic cancer has a poor 5-year survival rate (5%), which has remained consistent over the past 30 years. 2 Further, it shows resistance to traditional and newer therapies making treatment of distant metastatic lesions problematic. 3 Metastatic pancreatic cancer is often observed in the liver (27%), bone (3.0%), and lung (2.3%) and seen less frequently in the brain (0.6%).4-7 Low prevalence may be due to poor survival leading to death before BM develops.8,9 Others speculate that life expectancy and length of time living with pancreatic cancer may increase the chance of metastasis to the brain. This could arise due to the cancer cells’ ability to exist in latency for prolonged periods of time. 10
The incidence rate of brain metastases from all cancers ranges from 9 to 17% in population-based studies of cancer patients. 11 This is likely an underestimate of the actual incidence rate, due to a myriad of factors including incomplete data sets and misdiagnosis resulting in patients with brain metastases infrequently undergoing autopsies.4,6,11 Pancreatic cancer tends to extend into adjacent structures such as the duodenum, the portal vein, or superior mesenteric vessels. This could potentially aid in the transport and metastasis of cancer to distant areas of the body, including the brain.
Brain metastases in patients with pancreatic cancer significantly increase mortality and brain metastases are seen with increasing incidence,12,13 making early detection critical. Existing literature lacks an adequate representation of patient characteristics, disease progression, and survival. 14 Therefore, research is needed to include these factors to guide strategies for early diagnosis and treatment. Treatment of metastatic brain tumors often involves surgical resection, radiotherapy, or radiosurgery. However, the indications for when to use surgery are not clearly defined and treatments are continually evolving. 9 Based on patient-specific factors, optimal treatment could be tailored for each patient that targets the primary tumor and the metastases to the brain, signaling better prognosis. 15
Diagnosis remains a current challenge due to the low incidence of BM in pancreatic cancer. The purpose of this investigation was to examine characteristics and survival of patients with BM from pancreatic cancer treated in a single institution. We hypothesized that early neuroimaging in high-risk patients would help diagnose brain metastasis earlier, which may improve overall survival. This study may help to identify a profile of individuals at risk for BM from pancreatic cancer. These findings have the potential to inform future prospective clinical studies as increased awareness of brain involvement is critical when neurological disorders emerge in patients with pancreatic cancer at earlier stages.
Methods
A retrospective observational study using de-identified data was conducted to query electronic health records to identify all patients diagnosed with BM from pancreatic cancer in our cancer center. The study design was operationalized by a multidisciplinary research team from HonorHealth and the Translational Genomics Research Institute. The team queried data from the healthcare’s electronic medical record (EMR) system. The EMR system included a patient’s medical history (e.g., radiology reports and visits), demographic information (age, sex, and race), and mortality data. Neuroimaging studies, of both the pancreas and brain (computed tomography CT, and magnetic resonance imaging, MRI), were obtained from the EMR system at the oncology center. International Classification of Diseases (ICD) codes were used to identify pancreatic cancer patients (C25.9 for pancreatic cancer) and BM patients (C79.31) for the study.
Data, such as age at pancreatic cancer and BM diagnosis, location of primary pancreas tumor, and stage at diagnosis were collected. All patients ≥ 18 years presenting with brain metastasis with the diagnosis of pancreatic cancer were used as an exploratory cohort. The reporting of this study conforms to STROBE guidelines. 16
Ethical Considerations
The study protocol was submitted to the Institutional Review Board (IRB), and it was determined that the project was exempt from IRB review according to federal regulations (IRB # 1453699-1). Informed consent was waived, as per IRB guidelines for retrospective studies of previously collected clinical data. The study is in accordance with the recommendations of the National Human Research Protections Advisory Committee. Confidentiality of information was maintained and each patient was assigned a discrete number in the study.
Statistical Analysis
Medians or means with standard deviations are reported for continuous variables, as appropriate. Descriptive statistics are presented, and frequencies and percentages are reported for categorical variables. All variables were assessed for normality. Statistical tests are 2-tailed, with a significance level of an α of .05. All statistical analyses were performed using SPSS Statistics for Windows (version 28.0; IBM Corp). Cox regression was used for time-to-event analyses. Cox regression on overall survival time was used with an indicator variable for patients who ultimately developed a brain metastasis. The model was also adjusted for age at diagnosis, race, and gender. Survival after brain metastasis was calculated from the date of diagnosis of BM by radiology or pathology to either date of last follow-up or death. This is a departure from the usual use of the regression, where one is trying to explain differences in survival time using factors known at the beginning of the study time period. If brain metastases were associated with extended pancreatic cancer survival time, the BM indicator variable would suggest a lower hazard ratio.
Results
Patient Demographics and Clinical Characteristics
Between 2009 and 2022, we identified 34 patients diagnosed with brain metastases from pancreatic cancer (3.7%). Non-BM diagnoses included 63% with liver metastases and 17% with lung metastases (Figure 1). Of the patients with pancreatic cancer,186 (21%) were non-White, 425 (48%) female, and the median age was 67 (IQR: 63.7,70.93). In addition, 27% indicated a family history of cancer and 14% indicated a personal history of cancer. The overall survival in the pancreatic cancer cohort was 14 months (95% CI [12.84, 28.92]). See Table 1. Time (in Months) Between Pancreatic Cancer Diagnosis and the First Non-BM Metastatic Diagnosis. Clinical Characteristics and Demographics of Patients With Brain Metastases (BM) and Non-brain Metastases (Non-BM) From Pancreatic Cancer.
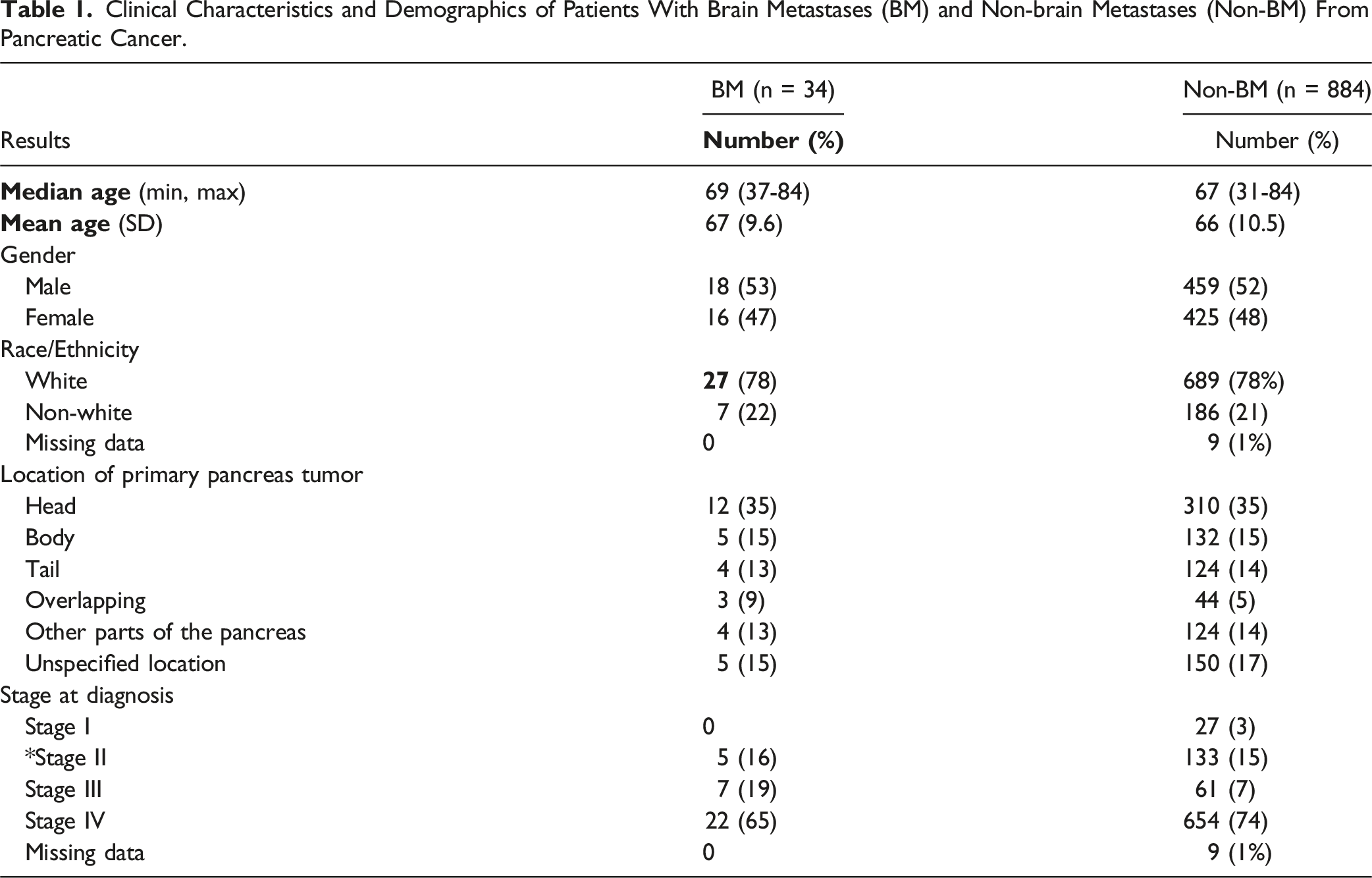
The site of the primary tumor within the pancreas was acquired from ICD10 codes and most were located in the head of the pancreas (12, 35% BM; 310, 35% pancreatic cancer).
Patients With Brain Metastases: Neurological Symptoms (n = 34).
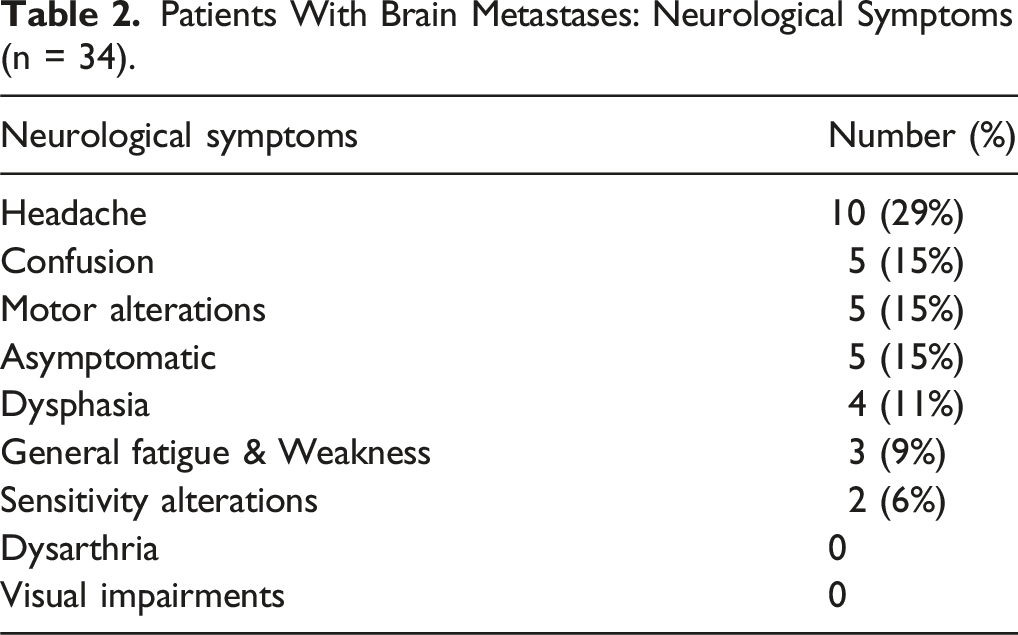
Overall survival (OS) in the BM group was nine months (95% CI [6.92,17.86]). A statistically significant difference in survival was observed between cohorts (P = .017). Regarding treatment, our study revealed surgery (30%) and radiotherapy (30%), radiosurgery (which included stereotactic radiosurgery, SRS 14%), and many chose best supportive care or no local treatment (26%). The median time to the diagnosis of brain metastases from initial diagnosis of pancreatic cancer was 233 days (IQR: 75 - 404) or 7.8 months. Figure 2 shows 34 patients and number of months between pancreatic cancer and BM diagnosis. Time (in Months) Between Pancreatic Cancer Diagnosis and the First BM Metastatic Diagnosis.
BM was the first diagnosed metastasis in 14 (41%) of the 34 patients; five individuals with BM were detected within the first 100 days of pancreatic cancer diagnosis. The model was adjusted for age at diagnosis, race, and gender. The adjusted hazard ratio for patients with brain metastasis was .91(95% CI [.88, .94]; P = .60], which was not significant. Cox regression analysis showed no significant relationship between extended pancreatic cancer survival time and brain metastasis development. There was no evidence of association between age at diagnosis of the BM and age at pancreatic cancer diagnosis, grouping patients by whether the brain metastasis was the first metastasis or a subsequent one. BM may develop from pancreatic cancer in initial stages of the primary disease and may not be dependent on increased survival time with pancreatic cancer (Figure 3). Overall Survival in Months in Total Study Population. Kaplan-Meier Curve Representing Overall Survival of Subjects.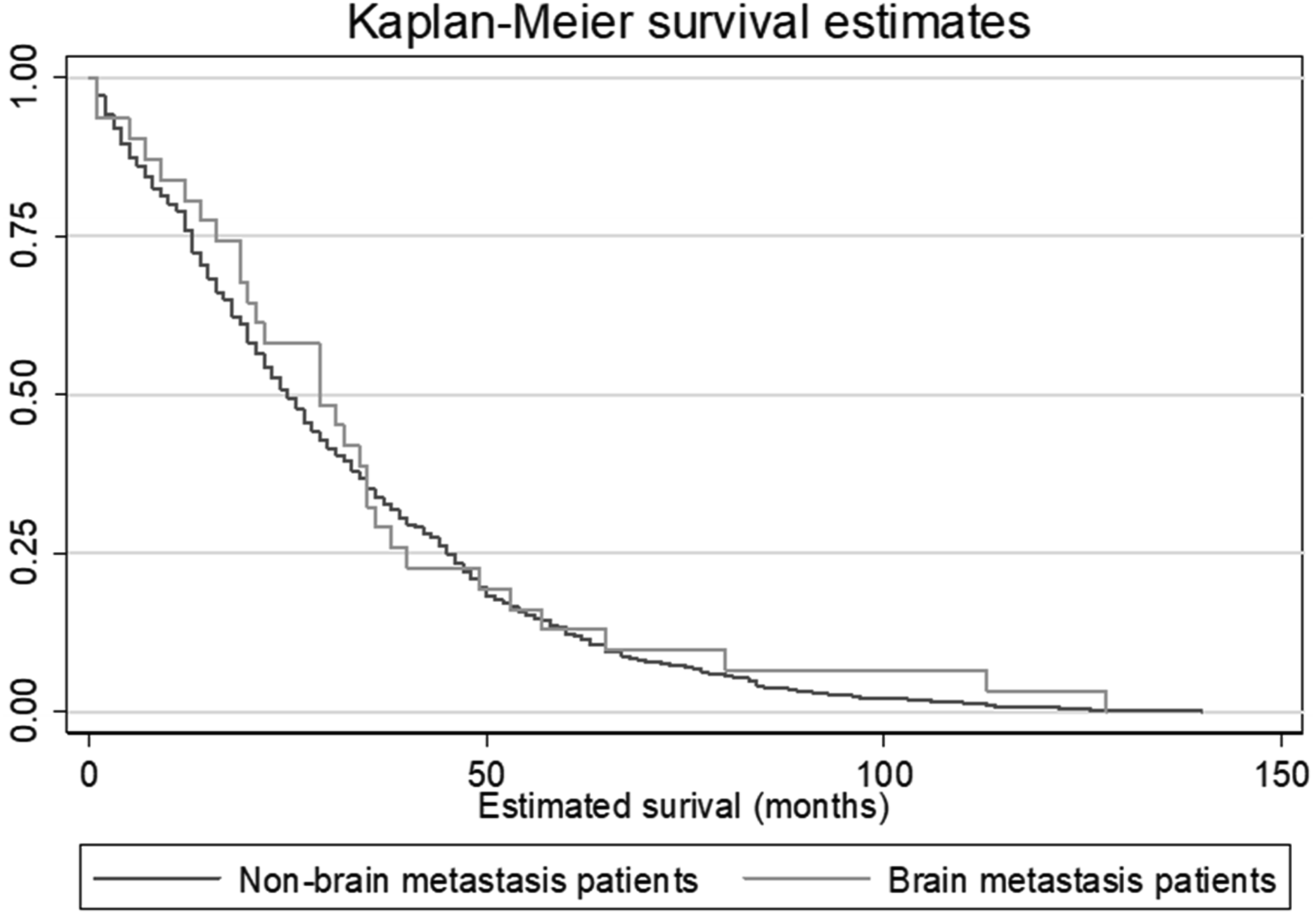
Discussion
Pancreatic cancer prognosis continues to be poor due to the fact most individuals have advanced-stage disease at diagnosis. Pancreatic selective surveillance and the emergence of biomarkers can enhance early detection and may provide a blueprint of innovative strategies to improve outcomes for those in high-risk groups.17-19 Despite challenges in disease identification, diagnosis of BM in pancreatic cancer has the potential to increase with early neuroimaging and screening, which may improve overall management and survival. 4
This retrospective observational study, conducted on pancreatic cancer patients, revealed earlier detection of BM from neuroimaging and improved survival in months. This larger cohort of 34 patients with BM from pancreatic cancer may be potentially beneficial to clinicians treating this rare subgroup. In this study, we found that BM from pancreatic cancer may be the first sign that the cancer has spread. Using advanced brain imaging early could help physicians find these metastases sooner and possibly improve survival. BM was the first diagnosed metastasis in 41% of the 34 patients and five individuals with BM were detected within the first 100 days of pancreatic cancer diagnosis. Some speculate there is potential for early brain metastases in specific subtypes. Despite not frequently discussed, brain metastases may originate from specific subtypes of pancreatic cancer, like those derived from intraductal papillary mucinous neoplasm (IPMN). While less common, brain metastases from the specific subtype, IPMN, may be more prone to early brain involvement. 20 Furthermore, this subtype of pancreatic cancer has a slower progression rate and is deemed a precursor to pancreatic ductal adenocarcinoma (PDAC).
Though average survival for patients with BM has been typically reported as six or less months, some studies show that subgroups of patients have longer survival.7,8 Our survival of nine months builds on recent findings showing survival of 8.25 months (95% CI [0.5-32.5]) and 12.5 months (95% CI [7.5,17.5]) respectively.21,22 Improvement in neuroimaging techniques have enabled earlier diagnosis, which may have impacted OS. 4 Our findings of increased BM survival support other current literature and the strategy of adding brain imaging early to initial screening for specific risk factors may help practitioners diagnose metastases sooner and potentially improve survival. All 34 patients with BM from pancreatic cancer had brain imaging. Although the majority presented with neurological symptoms, those who were asymptomatic (15%) were also selected for neuroimaging, which supports newer literature.7,21 Diagnosis delay can be a critical issue in patients with BM due to the lack of screening protocols. There is a potential for selection bias when only symptomatic patients receive imaging. Due to cost, participant burden, and lack of consensus regarding the need for routine brain imaging, neuroimaging studies are often performed in relatively small samples of participants, which may lead to selection bias. 23 For those who are asymptomatic, without imaging, brain metastases may be under-detected in early stages, leading to a potential delay in diagnosis and treatment. While integrating neuroimaging in all pancreatic cancer patients may not be feasible, selective screening for high risk patients is warranted; incorporating brain MRI in clinical practice in addition to CT scan and screening brain MRI in clinical trials may increase identification of BM patients from pancreatic cancer. 7
Alternative Clinical Factors
Important clinical risk factors have included diabetes and pancreatitis. Compared to patients without diabetes, individuals who received a recent diagnosis of diabetes had an almost 7-fold increase in risk of pancreatic cancer. 18 Both acute and chronic pancreatitis have been associated with risk for pancreatic cancer, being at its highest within one year of a pancreatitis episode. 17 Germline variants, such as BRCA1/2 mutations, have been identified as clinical risk factors, but as we experienced in our study, fewer patients had genomic data available, similar to other retrospective studies.7,21 Germline variants in BRCA1 and BRCA2 (potential pathogenic variants, namely BRCA1/2 PVs) have been reported to increase the risk of breast and ovarian cancers in female carriers and increase the risk for pancreatic and prostate cancer for males; knowledge of BRCA1/2 PVs informs recommended cancer screening strategies and options for risk reduction. 24 The clinical importance of genetic testing of BRCA1/2 in pancreatic cancers has been highlighted in recent literature as five to nine percent of patients with PDAC present with these mutations.25-27 In a current study, KRAS mutations were detected in BM patients who had molecular data available. 7 Further research is warranted to determine how germline mutations may lead to increased BM development in pancreatic cancer.
Clinical factors observed in this study included neurological symptoms, with headache and confusion as common presenting symptoms, consistent with recent studies.7,21 Additional risk factors have been associated with survival such as age and performance status. Researchers have administered the Karnofsky Performance Scale (KPS) to evaluate a patient’s functional status and the ability to complete activities of daily living. Recent literature revealed that age above 60 years (P = .10) and a KPS under 80% (P = .08) were significantly associated with overall survival. 28 Size and number of lesions have been associated with poorer prognosis. 29
The literature on BM from pancreatic cancer has been often limited to case reports and small series and optimal treatment approaches have not been established; however, surgery, radiotherapy, or radiosurgery have often been used. 9 A recent study indicated BM treatments included surgery (36%), radiotherapy (36%), radiosurgery (3%), and 25% had no local treatment. 21 Our study revealed similar BM treatment approaches. About one third of the patients had surgical resection, particularly when there was a single, accessible lesion. 30% had radiotherapy, also known as radiation therapy, which used high-energy radiation, such as X-rays or gamma rays to kill cancer cells and shrink tumors; 15% had radiosurgery, involving a precise radiation technique that delivered a concentrated dose of radiation specifically to the tumor, minimizing exposure to surrounding tissue. 30 Many patients had no local treatment and chose best supportive care (palliative care), which focused on managing symptoms and improving quality of life.
A recent meta-analysis 31 revealed no significant difference in 2-year survival among common treatment types, such as surgery, radiation involved stereotactic radiosurgery (SRS), and whole-brain radiotherapy (WBRT). While findings showed no difference in survival between surgery and SRS vs SRS alone 2 years after treatment or surgery and WBRT vs radiotherapy alone, patients with surgical resection showed greater risk for local tumor recurrence when compared to SRS. 31
Future Directions in Treatment
Technological advances that emerged over the last decade have prompted practitioners to think differently about cancer and its treatment. Improved neuroimaging offers additional opportunities for earlier diagnosis of BM32,33 Therapeutic tools have also expanded from whole brain therapy and surgical resection to laser-interstitial thermal therapy, systemic cytotoxic chemotherapy, targeted therapies, and immune-checkpoint inhibitors, which can be used alone or in combination.32-34 If detected earlier, synchronous surgery can be performed simultaneously on brain metastases and pancreatic cancer. 35 Recent literature also indicates stereotactic radiotherapy has been highly efficient in controlling brain metastases, with minimal side effects. 36 Newer medicines are designed to impede cancer cell reproduction or enable their death and cancer centers may employ immuno-oncology approaches, tumor antigen targeting, and specific tumor dependencies. Yet, BM treatment depends on the number of lesions, location, and the patient’s performance status.
As BM management has become more complex, with the onset of systemic therapies and advancements in radiation oncology and neurosurgery, a multidisciplinary approach is recommended to personalize treatment and improve clinical outcomes.34,37 The introduction of next-generation anticancer therapies, such as molecular targeted agents and immunotherapy, has improved the clinical decision-making process of this sub-population. 38 For patients with BM from pancreatic cancer, penetration of the blood-brain barrier has been a challenge for systemic treatments. More can be uncovered regarding molecular characteristics such as KRAS mutations and there may be a potential future role for KRAS-targeted therapy.7,39
Strengths and Limitations
Limitations include the retrospective nature and single-center study, which may result in unavoidable statistical bias, such as patient selection, and the results may not be widely generalizable. Availability and completeness of clinical records and medical documentation in the electronic medical records were limitations, inherent in retrospective studies. Variables which had many missing values (such as KRAS, BRCA1/2 germline mutations) were omitted as many patients did not have genetic testing or data available. We omitted individuals who did not have complete data in the larger pancreatic data set. Despite a larger cohort for BM, the relatively small sample size may affect the statistical power and the ability to detect significant associations. There is a chance the metastases are related to a primary cancer other than the pancreatic one, as the pathology information from the BM was not available. To mitigate limitations of the retrospective study, a multi-center prospective study is planned to detect survival associations and guide treatment and systemic therapies. Despite limitations, strengths of the current study include the valuable insights into the incidence and characteristics of brain metastasis in pancreatic cancer and the focus on early diagnosis and screening.
Conclusion
Due to the low incidence of BM in pancreatic cancer, diagnosis continues to be challenge. Our study presents real-world data regarding the characteristics and survival of patients with BM from pancreatic cancer. This study found that BM from pancreatic cancer can be the first sign that the cancer has spread. Using advanced brain imaging on higher risk patients could help physicians diagnose metastases sooner, which may improve survival. This study collected initial data, which may be helpful for practitioners in gaining insights to help guide diagnosis and future management. Prospective multicentric clinical trials will be valuable for advancing understanding of brain metastases, identifying prognostic factors, and improving patient outcomes.
Ethical Statement
Ethical Approval
The study was exempt from IRB review according to federal regulations (IRB # 1453699-1). The study is in accordance with the recommendations of the National Human Research Protections Advisory Committee. The requirement for obtaining informed consent was waived due to the retrospective nature of the study.
Footnotes
Acknowledgements
We acknowledge the Helios Scholar Internship Program, within the Translational Genomics Research Institute.
Author Contributions
SH: study design, provision of study materials, data integration, analysis, interpretation, manuscript writing, editing, and final manuscript. DC: study conception and design, provision of materials, data analysis and interpretation, manuscript writing and final manuscript.
Funding
The authors received no financial support for the research, author ship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article. Individual patient level data are not publicly available to maintain compliance with HIPPA regulations and IRB protocol.
