Abstract
Purpose:
It remained unclear whether tyrosine kinase inhibitors (TKIs) related renal impairment had impact on the survival of patients with metastatic renal cell carcinoma (mRCC).
Methods:
Clinicopathological parameters of patients with mRCC treated with TKIs were retrospectively reviewed. Blood urea nitrogen (BUN), proteinuria and estimated glomerular filtration rate (eGFR) at baseline and during TKIs treatment were recorded. BUN > 7.1mol/L, eGFR <60 ml/min/1.73m2 and/or proteinuria level > 0.3 g/L were defined as renal impairment. eGFR and proteinuria were furtherly classified into different levels. Treatment outcomes were defined as progression-free survival (PFS) and overall survival (OS).
Results:
At baseline, the presence of abnormal BUN, eGFR and proteinuria level were observed in 25 (22.7%), 27 (25.5%) and 30 (27.3%) patients, which increased to 46 (41.8%), 55 (50.0%) and 64 (58.2%) respectively after TKIs treatment. In the whole cohort (N = 110), survival analysis suggested that only post-treatment renal impairment was related to survival outcomes. Interestingly, sub-analysis showed that post-treatment eGFR level (p = 0.004), proteinuria (p = 0.014) and eGFR decrease >10% (p = 0.012) and elevated proteinuria compared with baseline (p = 0.006) were statistically correlated with OS among patients without RI at baseline (N = 51). On the contrary, deterioration of renal impairment after TKIs treatment in patients with renal impairment at baseline (N = 59) had no relationship with either PFS or OS. Furthermore, eGFR (p = 0.020) and eGFR decrease >10% (p = 0.016) within 1 year after TKIs therapy were potential biomarkers for OS.
Conclusion:
Dynamic changes of TKI-induced RI during TKIs treatment, especially eGFR and proteinuria level, could be considered as potential biomarkers predicting survival outcomes of mRCC patients.
Keywords
Introduction
Renal cell carcinoma (RCC) is the most common type of malignant kidney disease. 1 Due to its high aggressiveness, 25%-30% of patients were at the metastatic phase when initially diagnosed, while 20% of patients underwent recurrent disease after radical nephrectomy within 3 years. 2,3 In the last decade, tyrosine kinase inhibitors (TKIs) were designed to inhibit the vasculogenesis of tumor-targeting vascular endothelial growth factors (VEGFRs). TKIs had significantly improved the survival outcomes of mRCC patients and became a standard treatment. 4,5 However, patients often suffered from on-target adverse events (AEs) during TKIs treatment, leading to dosage reduction, schedule modification, interruption, or even cessation. This scenario was more frequently observed in Asian population. 6,7
Though the profile of AEs varies among different TKI drugs, the most common types are fatigue, hypertension (HTN), diarrhea, and hand-and-foot syndrome (HFS). 5,8 Previous evidence showed that certain types of AEs had predictive value for the prognosis of mRCC patients. Moreover, the severity and incidence of AEs were also related to survival outcomes. 9,10 Recently, accumulating evidence stated that, after receiving TKIs treatment, 60-70% of patients underwent on-set or progression of renal function damage, mainly eGFR decrease, which usually happened within 6.6 months. 11,12 However, the role of renal function in TKI-treated mRCC patients remains unclear.
Studies 12 demonstrated that mRCC patients with renal impairment (RI) at initial TKI treatment were tended to have a poor prognosis, while others 10,13 stated that mRCC patients with RI could equally benefit from TKIs treatment. The development of proteinuria, as well as deterioration of eGFR damage after targeted therapy, was reported to be a predictor for better survival outcomes in patients with TKIs treatment. 11 Yet, there are still limited data to clarify the exact role of TKI-induced RI in mRCC patients, and its predictive value for patients’ survival outcome. Hence, we conducted this study to verify the occurrence of RI during the TKIs treatment course and its predictive value in the prognosis of mRCC patients.
Materials and Methods
Characteristics of Patients
Patients older than 18 years, who were pathologically diagnosed with mRCC at West China Hospital between 2014 and 2018 were included. The clinicopathological data of patients were recorded in detail. All patients underwent nephrectomy and received further TKIs treatment. No patient received neoadjuvant therapy. Blood urea nitrogen (BUN), proteinuria, and estimated glomerular filtration rate (eGFR) were measured or calculated before and after TKIs treatment. In general conception, the normal BUN level was defined as ≤7.1 mmol/L. eGFR was calculated using the abbreviated MDRD-GFR equation. GFR in ml/min/1.73 m 2 = 186.3×creatinine −1.154 × age −0.203 (×0.742 if female). Proteinuria was measured with a rapid urine protein test. The positive result of the rapid urine protein test was defied as ≥0.3 g/L According to the KDIGO guideline, normal eGFR level was defined as eGFR >60 ml/min/1.73m 2 . eGFR was then classified into three degrees: >60, 30-60, and <30 ml/min/1.73m 2 , and proteinuria level was classified into 3 degrees: negative grade 1 (<0.3 g/L), grade 2 (0.3-3.0 g/L) and grade 3 (>3.0 g/L). Post-treatment levels of BUN, eGFR, and proteinuria were routinely evaluated every 4 weeks.
This study was approved by the Biomedical Research Ethics Committee of West China Hospital of Sichuan University. All procedures performed in studies involving human participants were following the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Treatment and Outcomes
Sunitinib and axitinib were given to patients as the 1st-line TKIs treatment. Sunitinib was administrated with 50 mg daily on a 4-weeks on and 2-weeks off schedule (4/2 schedule), and axitinib was administrated with 5 mg/bid. Primary and TKI-induced hypertension was documented. Treatment duration was identified as the time from initial TKIs treatment to cessation, death, or end of follow-up. All patients routinely underwent a contrast CT scan every 3 months to evaluate their treatment response, which was assessed using the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1. PFS was defined as the time from the first administration of TKIs to the progression of disease or death, and OS was defined as the time from the first administration of TKIs to all-cause mortality.
Statistical Analysis
Data were presented as categorical variables and continuous variables. Differences between quantitative variables were analyzed using a t-test, while differences in categorical variables were determined using the chi-square (χ2) test. Correlation between the two parameters was tested by Spearman’s rank correlation coefficient and logistic regression analysis. Kaplan-Meier survival curves were drawn and a log-rank test was used to analyze the statistical significance. Multivariate analysis was conducted using the Cox regression model with a forward stepwise selection procedure. SPSS version 24.0 (SPSS Inc., Chicago, IL, USA) software was used only for all statistical analyses. GraphPad Prism 7.0a was used to plot all the figures. P < 0.05 was considered statistically significant.
Results
Clinicopathological Characteristics
A total of 110 mRCC patients treated with TKIs at West China Hospital from Sep. 2014 to Dec. 2018 were included in this study, and their clinicopathological parameters were summarized in Table 1. Most of the patients were <65 yr of age, male, had good performance status and pathologically diagnosed with clear cell RCC (ccRCC). In the whole cohort, 65 (59.1%) patients received sunitinib treatment, while axitinib was administrated in 45 (40.9%) patients, separately. The mean duration of TKIs treatment was 26.2(±24.0) months.
Clinicopathological Parameters of Included Patients (N = 110).

BMI: body mass index; RCC: renal cell carcinoma; non-RCC: including papillary, chromophobe and other types of RCC; ECOG: Eastern Cooperative Oncology Group; IMDC: International Metastatic Renal-Cell Carcinoma Database Consortium; MSKCC: Memorial Sloan Kettering Cancer Center; AEs: adverse event; TKI, tyrosine kinase inhibitors; BUN, blood, urea nitrogen; eGFR estimated glomerular filtrating rate.
Deterioration of RI After TKIs Treatment
The incidence of RI pre- and post-TKIs treatment was summarized in Table 1. In the whole cohort, abnormal BUN, eGFR, and proteinuria levels were observed in 25 (22.7%), 27 (25.5%), and 30 (27.3%) patients at initial treatment. However, after receiving TKIs treatment, the incidence of abnormal BUN, eGFR, and proteinuria increased to 46 (41.8%), 55 (50%), and 64 (58.2%), respectively.
The trend of RI in mRCC patients after TKIs treatment was shown in Figure 1. In correlation analysis, in the whole cohort, BUN (r = 0.718, p < 0.001) and proteinuria level (r = 0.556, p = 0.002) were generally increased compared with baseline level after receiving TKIs (Figure 1A and C). On the contrary, the eGFR level steadily decreased during the follow-up (r = −0.574, p = 0.001) (Figure 1B). This trend was more obvious in patients without baseline RI (Figure 1D, E, and F). However, a similar trend was not observed in patients with baseline RI (Figure 1G, H, and I).
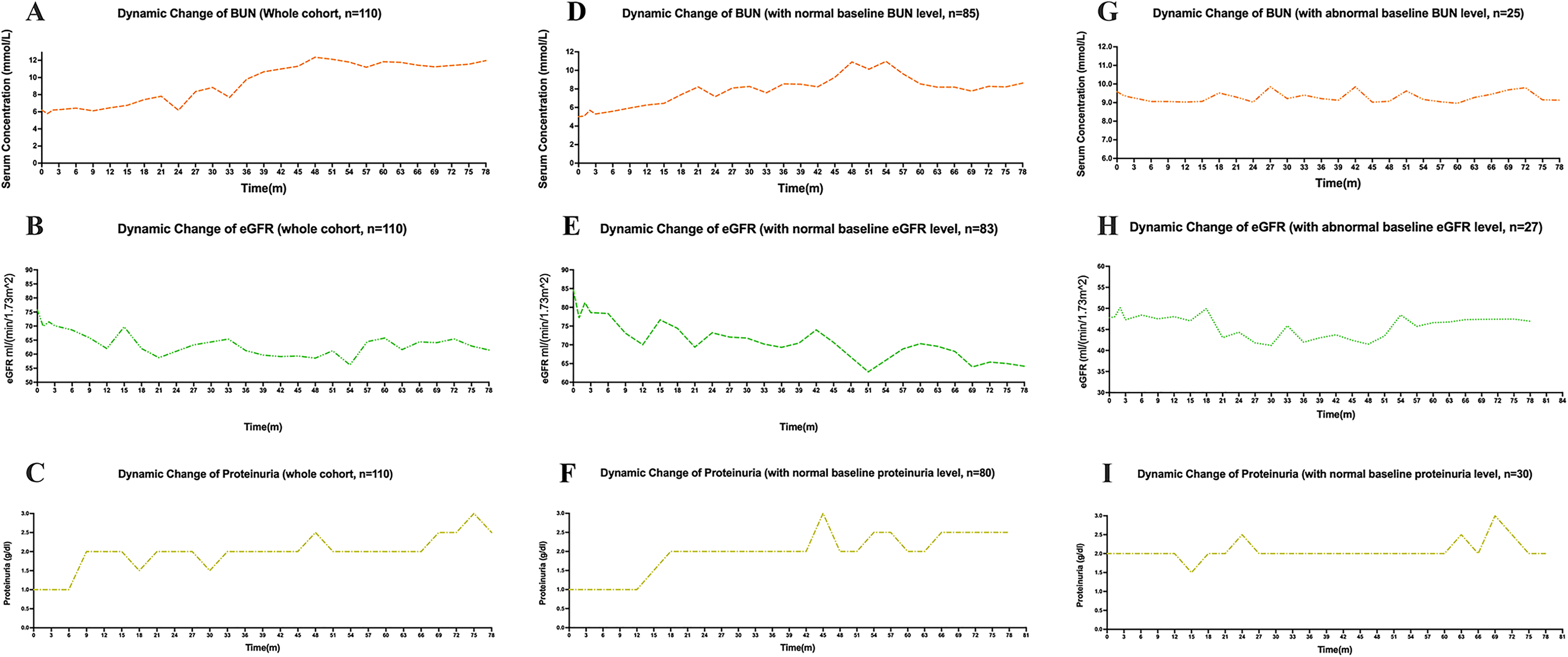
Dynamic change of renal function related parameters after TKIs treatment (n = 110). Tendency of renal function change after TKIs treatment: A-C stands for the dynamic change of BUN, eGFR, and proteinuria in the whole cohort; D-F stands for the dynamic change of BUN, eGFR, and proteinuria in patients without RI at baseline; G-I stands for the dynamic change of BUN, eGFR and proteinuria in patients with RI at baseline.
Association Between Clinicopathological Parameters and the Occurrence of Post-Treatment RI
In searching for baseline predictors for post-treatment RI, baseline clinicopathological parameters were included for analysis. In correlation analysis, age >65 years old was significantly related to abnormal eGFR level (OR: 5.972, 95%CI: 2.316-15.399, p < 0.001) after TKIs treatment. Moreover, the male gender (OR: 3.268, 95%CI: 1.178-9.091, p = 0.023) was statistically indicated for the occurrence of TKI-related proteinuria. Interestingly, proteinuria was significantly associated with the use of axitinib (42.5% vs 20.0%. p = 0.007). Other baseline variables, including IMDC risk group and histology, had no statistical relationship with the occurrence of post-treatment RI (Supplementary Table 1).
To clarify the role of hypertension in the occurrence of RI, TKI-induced HTN was also included and analyzed. Primary and TKI-induced HTN were reported in 37 (33.6%) and 32 (29.1%) patients, separately (Table 1). After the Spearman Correlation test, primary and TKI-induced HTN were only correlated with proteinuria (p = 0.044 and 0.013, separately). eGFR and BUN had no relationship with the occurrence of hypertension.
The Impact of Baseline and Post-Treatment Renal Function Related Parameters on PFS
Among all 110 patients, 83 of them (75.7%) experienced the progressed disease. Median PFS time in the whole cohort was 13.0(± 2.1) months. In univariate analysis, baseline renal function related parameters had no impact on patients’ PFS. However, post-treatment proteinuria (p = 0.024), elevated proteinuria (p = 0.014) and eGFR (p = 0.006) were significantly related to PFS (Supplementary Table 2 and Figure 2). In multivariate analysis, post-treatment eGFR (p = 0.048), proteinuria (p = 0.039), along with elevated proteinuria (p = 0.022) indicated longer PFS (Table 2).

Correlations between post-treatment renal function related parameters and PFS of mRCC treated with TKIs (n = 110). Kaplan-Meier curves of different renal function related parameters in patients with normal renal function related parameters at baseline: (A) Relationship of post-treatment eGFR level and PFS; (B) Relationship of post-treatment proteinuria level and PFS; (C) relationship of elevated proteinuria level after TKIs treatment and PFS.
Multivariate Analysis of PFS and OS in mRCC Patients (n = 110).

ECOG: Eastern Cooperative Oncology Group; IMDC: International Metastatic Renal-Cell Carcinoma Database Consortium; BUN, blood, urea nitrogen; eGFR estimated glomerular filtrating rate.
* p < 0.05.
We furtherly evaluated the impact of post-treatment renal function related parameters on PFS in patients without RI at baseline (n = 51). In these patients, elevated proteinuria level after TKIs treatment was independently related to longer PFS (p = 0.023). However, other RI-related parameters had no relationship with PFS in multivariate analysis (Table 3). In the subgroup of patients with RI at baseline, the occurrence of RI had no relationship with PFS (Supplementary Table 3).
Multivariate Analysis of PFS and OS in mRCC Patients Without Renal Impairment at Initial Diagnosis.

ECOG: Eastern Cooperative Oncology Group; IMDC: International Metastatic Renal-Cell Carcinoma Database Consortium; TKI, tyrosine kinase inhibitors; BUN, blood, urea nitrogen; eGFR estimated glomerular filtrating rate.
* p < 0.05.
The Impact of Pre- and Post-Treatment Renal Function Related Parameters on OS
Among all 110 patients, 56 of them (50.9%) died of renal cell carcinoma. Median OS time in the whole cohort was 30.0(±3.6) months. In univariate analysis, compared with baseline renal function related parameters, post-TKIs treatment renal function related parameters were correlated with OS (Supplementary Table 2 and Figure 3). In multivariate analysis, eGFR grade (p = 0.009), eGFR decrease >10% (p = 0.034), proteinuria level (p < 0.001), as well as elevated proteinuria level (p < 0.001) were identified as independent predictors for patients’ OS (Table 2).
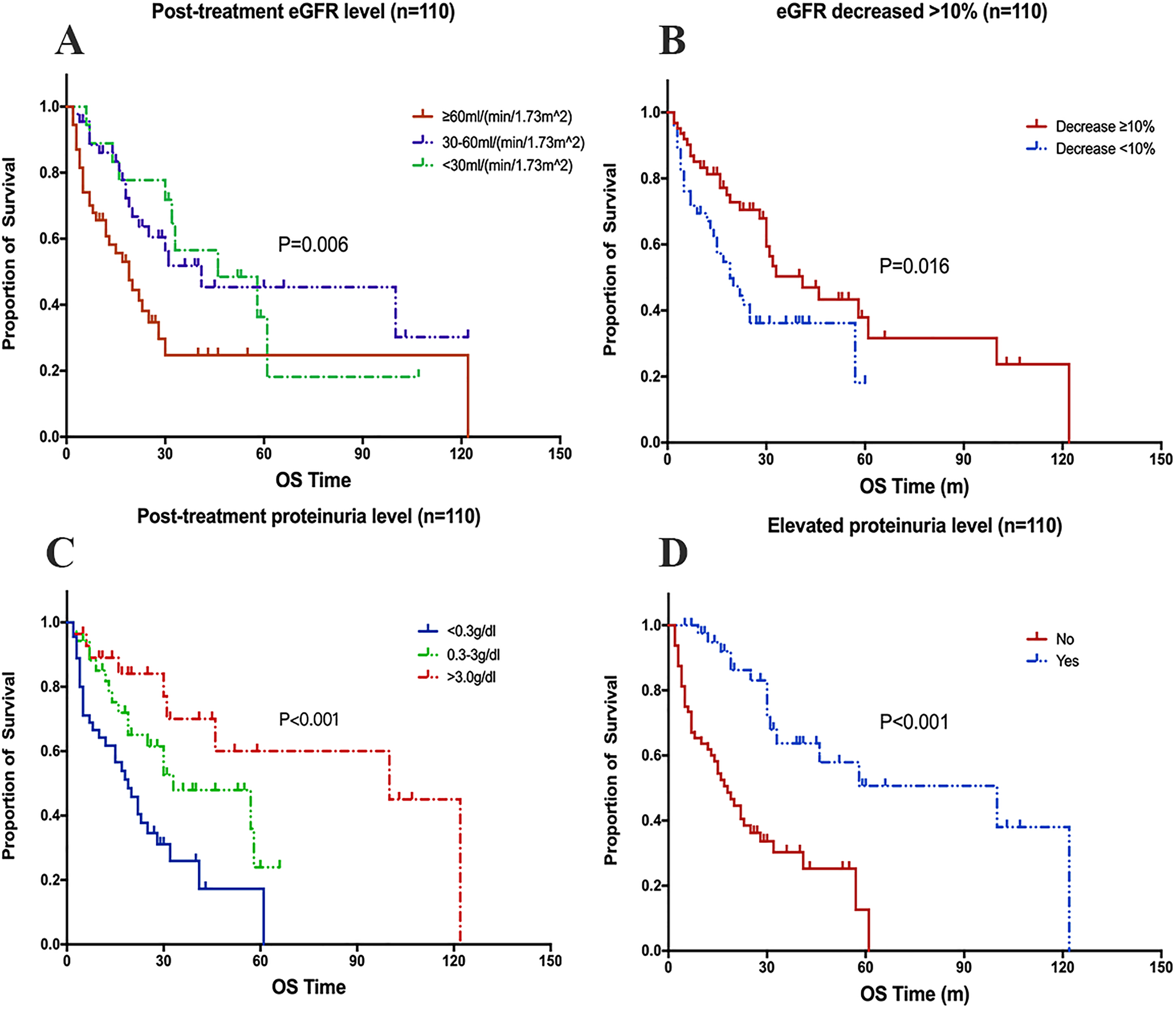
Correlations between post-treatment renal function related parameters and OS of mRCC treated with TKIs (n = 110). Kaplan-Meier curves of different renal function related parameters in patients with normal renal function related parameters at baseline: (A) Relationship of post-treatment eGFR level and OS; (B) Relationship of eGFR decreased >10% after TKIs treatment and OS; (C) Relationship of post-treatment proteinuria level and OS; (D) Relationship of elevated roteinuria level after TKIs treatment and OS.
We furtherly evaluated the impact of post-treatment renal function related parameters on OS in the subgroup of patients without RI at baseline (n = 51). In these patients, post-treatment eGFR (p = 0.004), eGFR decreased >10% (p = 0.012), proteinuria (p = 0.014) and elevated proteinuria level (p = 0.006) were independently associated with OS (Table 3). In the subgroup of patients with RI at baseline, only proteinuria level elevated after TKIs treatment was independently associated with longer OS (p = 0.002) (Supplementary Table 3).
The Relationship of Renal Impairment in Different IMDC Risk Groups with Patients’ Prognosis
The median PFS of favorable-, intermediate- and poor-risk groups were 20.0, 12.0, and 8.0 months, separately (p = 0.464). No relationship was found between post-treatment renal impairment and PFS time in neither favorable-, intermediate- or poor-risk group (Supplementary Table 4). The median OS time of favorable-, intermediate- and poor-risk groups were 41.0, 30.0, and 14.0 months, separately (p = 0.007). No relationship was found between post-treatment renal impairment and PFS time in the favorable-risk group. In intermediate-risk group, patients with eGFR level > 60 ml/min/1.73m2 had significantly shorter OS time than those with eGFR level 30-60 and <30 ml/min/1.73m 2 (19.0, 30.0 and 31.0 months, separately, p = 0.002) (Figure 4A). In addition, patients with eGFR decreased > 10% than baseline had longer OS time than those with eGFR decreased < 10% (30.0 vs 19.0, p = 0.047) (Figure 4B). As for patients in the poor-risk group, no relationship was found between post-treatment renal impairment and PFS time. Patients with elevated proteinuria level than baseline were indicated for longer OS time (25.0 vs 8.0, p = 0.008) (Figure 4C). Other renal function parameters had no relationship with OS.
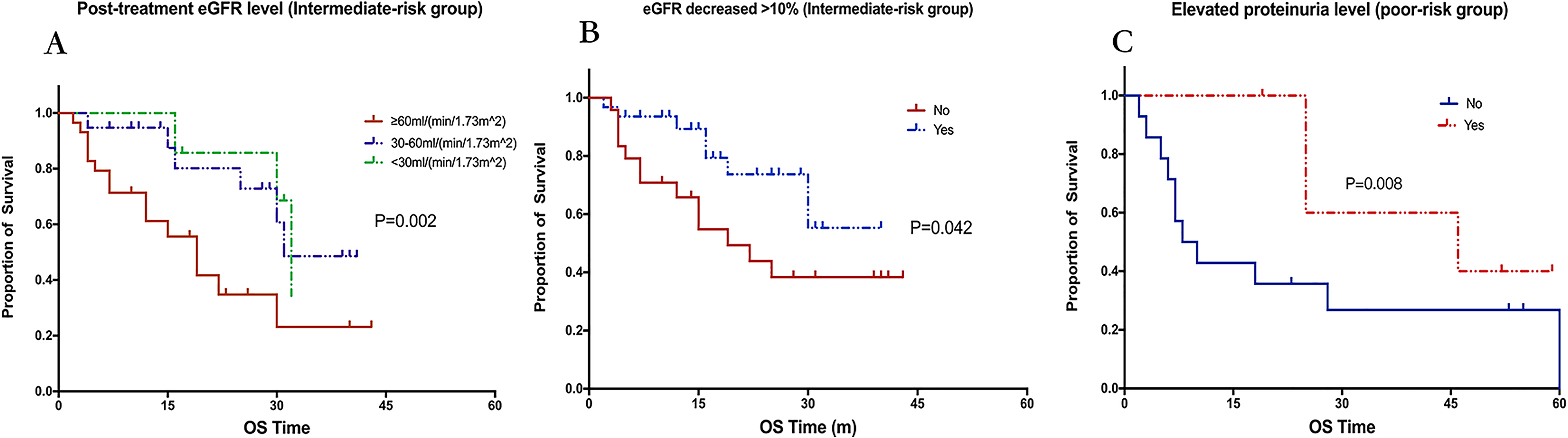
Correlations between post-treatment renal impairment and survival outcomes of mRCC patients in different IMDC risk groups (n = 110). Kaplan-Meier curves of renal impairment in patients with different IMDC risk groups: (A) Relationship of post-treatment eGFR level and OS in IMDC intermediate-risk group; (B) Relationship of eGFR decreased >10% after TKIs treatment and OS in IMDC intermediate-risk group; (C) Relationship of elevated proteinuria level after TKIs treatment and OS in IMDC poor-risk group.
The Occurrence of Early Renal Impairment and Patients’ Prognosis
To evaluate the predictive value of early change (<1 year) of renal function for patients’ prognosis, we furtherly analyzed the relationship of renal function related parameters within one year after TKIs treatment with PFS and OS. In univariate analysis, the eGFR decreased >10% within one year was statistically related to PFS (p = 0.039) and OS (p = 0.042) (Supplementary Table 5 and Figure 5). In multivariate analysis, eGFR decreased >10% within one year was independently correlated with longer PFS time (p = 0.026) and OS (p = 0.016) (Table 4). That is to say, the dynamic change of eGFR after TKIs using could offer early prognostic information of patients.
Multivariate Analysis of the Dynamic Change of Renal Function Within 1 Year and PFS and OS in mRCC Patients (n = 110).
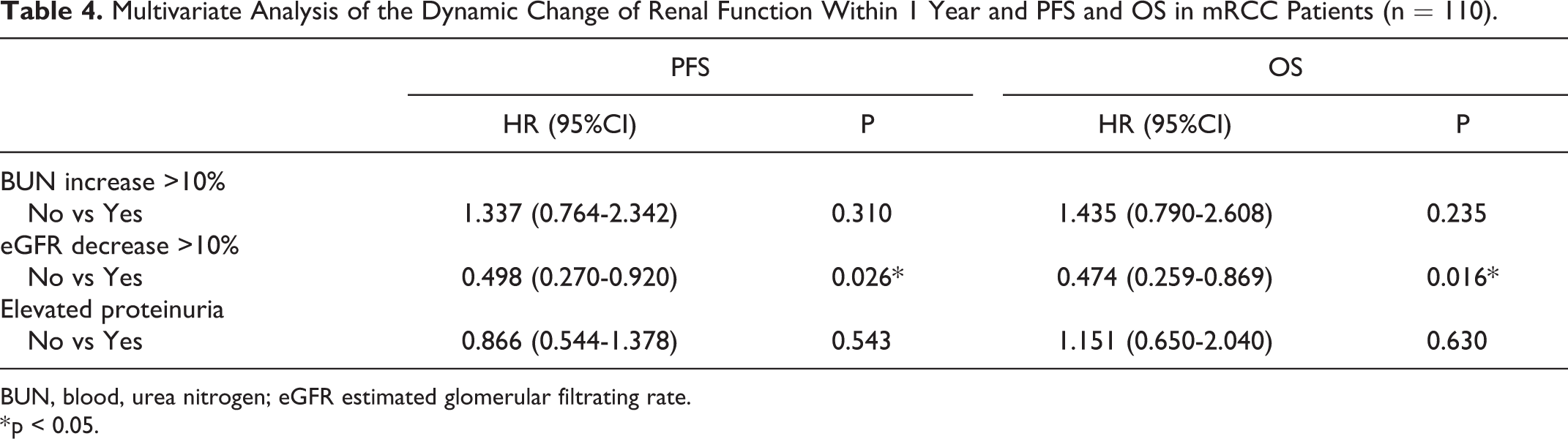
BUN, blood, urea nitrogen; eGFR estimated glomerular filtrating rate.
* p < 0.05.

Correlations between post-treatment eGFR level within 1 year and PFS and OS of mRCC treated with TKIs (n = 110). Kaplan-Meier curves of eGFR level within 1 year: (A) Relationship of post-treatment eGFR level within 1 year and PFS; (B) Relationship of post-treatment eGFR level within 1 year and OS.
Relationship Between Pre- and Post-Treatment Renal Function and Tumor Response
In the whole cohort, CR was observed in only 2 (1.8%), PR in 17 (15.5%), and SD in 57 (51.8%) patients, while 34 (30.9%) patients underwent disease progression (PD). Patients with BUN >7.1 mmol/L after TKIs treatment was tended to have favorable ORR (p = 0.020), while lower post-treatment eGFR was associated with higher DCR (p = 0.044). Other renal function related parameters, including both baseline and post-treatment index, had no impact on tumor response (Supplementary Table 6).
Discussion
Along with adequate anti-cancer efficacy by TKIs drugs, some patients with mRCC would suffer from one or more types of adverse events, including hypertension, fatigue, hand-and-foot syndrome. Certain types of AEs, like hypertension, during TKIs treatment, were found to be associated with patients’ survival outcomes. 8 -10 In this study, we observed that the renal function of mRCC patients was tended to deteriorate after receiving TKIs treatment. In the meantime, both primary and TKIs-induced hypertension was testified to be correlated with the occurrence of post-treatment RI. Furthermore, we identified the predictive value for eGFR change within one year after receiving targeted therapy for survival outcomes of patients.
Studies reported controversial results about how renal function changes after VEGFR-targeted therapy. Boursiquot et al 14 stated that the use of VEGF TKIs did not adversely affect long-term renal function. On the contrary, Khan et al10 demonstrated eGFR tended to decrease after sunitinib and sorafenib treatment, and the median time from TKIs treatment to maximum renal function change was 6.6 months. Moreover, the role of RI in mRCC patients with TKIs treatment remained controversial. Accumulating evidence 10,15 demonstrated that mRCC patients with RI at baseline could equally benefit from sunitinib and sorafenib treatment, with adequate monitoring of renal function. However, Nouhaud et al 12,16 found that renal function impairment before treatment was significantly associated with a longer median time to treatment failure and OS in mRCC patients treated with TKIs. Moreover, Fukuda et al 17 reported that eGFR decreasing >10% after TKIs treatment was indicated for longer PFS and OS time. A similar result was reported by Baek et al, 11 who also stressed the impact of post-treatment proteinuria on patients’ PFS. Our study supported that TKI-induced RI was strongly related to better prognosis in mRCC patients with TKIs treatment. Moreover, the dynamic change of eGFR within 1 year was a predictive biomarker for both PFS and OS time. In mRCC patients with TKIs treatment, the monitoring of renal function was recommended during treatment.
TKI-induced severe renal function damage was also associated with treatment modification. Baek et al 11 stated that most patients with proteinuria were asymptomatic, and it would be resolved by drug discontinuation or dose reduction in 62% of patients. Khan et al10 also reported a dose reduction in 52% and treatment discontinued in 10% of patients secondary to toxicity in patients with RI at the start of treatment. Fukuda et al 17 reported that, in 62 included patients, 77% of patients underwent dose reduction or interruption, but only 5 patients were caused by renal dysfunction. Most patients underwent treatment modification because of other TKI-induced AEs, including fatigue, hypertension, and hand-and-foot syndrome. 9,10 However, none of these studies clarified in indication for treatment modification due to renal toxicity, and no study was performed to determine the standard management for TKI-related renal toxicity.
Recently, in the treatment of mRCC patients, the immune checkpoint inhibitors (ICIs) based therapy demonstrated favorable survival benefit and safety over TKIs, and gradually became a superior option for the management of mRCC patients. 18 -21 Axitinib was often combined with ICIs in the treatment of mRCC. 20,21 In this study, we found that axitinib was more likely to cause proteinuria than sunitinib during treatment (42.5% was 20.0%). In a previous study, the incidence of proteinuria after axitinib treatment was about 35.0% to 66.0%. 22,23 In the era of immune therapy, when combined with ICIs, the dosage of axitinib was lower than it was used alone, which could decrease the incidence of post-treatment proteinuria. However, there was still 17.5% of mRCC patients would experience proteinuria during axitinib plus pembrozulimab treatment. 20 Thus, the monitoring of renal function was needed in the era of ICIs therapy.
Several hypotheses have been proposed about the mechanism of VEGF-TKI-induced renal dysfunction, and its relationship with the anti-tumor process. Eremina et al 24 demonstrated that VEGF-α decrease in podocytes and tubular cells resulting in TKIs use would lead to dysfunction of the filtration barrier, and caused proteinuria and endothelin. Izzedine et al 25 also reported the appearance of renal thrombotic microangiopathy, mainly minimal change disease and/or collapsing-like focal segmental glomerulosclerosis in TKIs-used patients. 26 These pathological changes happened at various times (from 1 day to more than 2 years) and might be related to drug dosage. 27,28 Hypertension, proteinuria, and renal insufficiency were common manifestations of these changes. 28 Renal function damage alleviates after controlling hypertension. Studies in patients with anti-VEGF agents suggested the ACEI and ARA use in anti-VEGF-treated patients with proteinuria. 27 ACEI drugs were highly recommended in TKI-treated patients with hypertension and proteinuria. Gatenby et al 29 found that serum urea nitrogen would accumulate in RCC patients after cytoreductive nephrectomy, which led to decreased system pH levels. Alterations in systemic pH could decrease the pH level in the tumor extracellular space and inhibit the outflow of the acid within tumor cells, thus resulting in the accumulation of acid in the tumor. This process contributes to tumor cell necrosis altogether.
Limitations of this study included the small size of sample and retrospective data collection, which might contribute to information and selection bias, as well as insufficient data for comprehensive analysis. Proteinuria level was tested by a rapid urine protein test, which was less accurate compared to the 24-hour urinary protein quantity test. What’s more, due to the diversity of post-treatment follow-up, data were not collected in the same period. Thus, the conclusion needs further validation with a large-sample scale and prospective studies.
Conclusion
TKI-induced RI could indicate for longer survival and better tumor response, and provide valuable information for the individual treatment of mRCC patients. Thus, routinely monitoring of renal function, especially eGFR and proteinuria, was highly recommended in mRCC patients with TKIs treatment.
Supplemental Material
Supplemental Material, sj-docx-1-ccx-10.1177_1073274820977143 - The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors
Supplemental Material, sj-docx-1-ccx-10.1177_1073274820977143 for The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors by Haoran Zhang, Xingming Zhang, Xudong Zhu, Yuchao Ni, Jindong Dai, Sha Zhu, Guangxi Sun, Zhipeng Wang, Junru Chen, Jinge Zhao, Hao Zeng, Zi Li and Pengfei Shen in Cancer Control
Supplemental Material
Supplemental Material, sj-docx-2-ccx-10.1177_1073274820977143 - The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors
Supplemental Material, sj-docx-2-ccx-10.1177_1073274820977143 for The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors by Haoran Zhang, Xingming Zhang, Xudong Zhu, Yuchao Ni, Jindong Dai, Sha Zhu, Guangxi Sun, Zhipeng Wang, Junru Chen, Jinge Zhao, Hao Zeng, Zi Li and Pengfei Shen in Cancer Control
Supplemental Material
Supplemental Material, sj-docx-3-ccx-10.1177_1073274820977143 - The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors
Supplemental Material, sj-docx-3-ccx-10.1177_1073274820977143 for The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors by Haoran Zhang, Xingming Zhang, Xudong Zhu, Yuchao Ni, Jindong Dai, Sha Zhu, Guangxi Sun, Zhipeng Wang, Junru Chen, Jinge Zhao, Hao Zeng, Zi Li and Pengfei Shen in Cancer Control
Supplemental Material
Supplemental Material, sj-docx-4-ccx-10.1177_1073274820977143 - The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors
Supplemental Material, sj-docx-4-ccx-10.1177_1073274820977143 for The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors by Haoran Zhang, Xingming Zhang, Xudong Zhu, Yuchao Ni, Jindong Dai, Sha Zhu, Guangxi Sun, Zhipeng Wang, Junru Chen, Jinge Zhao, Hao Zeng, Zi Li and Pengfei Shen in Cancer Control
Supplemental Material
Supplemental Material, sj-docx-5-ccx-10.1177_1073274820977143 - The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors
Supplemental Material, sj-docx-5-ccx-10.1177_1073274820977143 for The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors by Haoran Zhang, Xingming Zhang, Xudong Zhu, Yuchao Ni, Jindong Dai, Sha Zhu, Guangxi Sun, Zhipeng Wang, Junru Chen, Jinge Zhao, Hao Zeng, Zi Li and Pengfei Shen in Cancer Control
Supplemental Material
Supplemental Material, sj-docx-6-ccx-10.1177_1073274820977143 - The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors
Supplemental Material, sj-docx-6-ccx-10.1177_1073274820977143 for The Impact of Renal Impairment on Survival Outcomes in Patients With Metastatic Renal Cell Carcinoma Treated With Tyrosine Kinase Inhibitors by Haoran Zhang, Xingming Zhang, Xudong Zhu, Yuchao Ni, Jindong Dai, Sha Zhu, Guangxi Sun, Zhipeng Wang, Junru Chen, Jinge Zhao, Hao Zeng, Zi Li and Pengfei Shen in Cancer Control
Footnotes
Authors’ Note
Haoran Zhang, Xingming Zhang, and Xudong Zhu are co-first authors and contributed equally. Our study was approved by The Biomedical Ethics Committee of West China Hospital, Sichuan University (approval no. 2012 (15)). All patients provided written informed consent prior to enrollment in the study. Informed consent was obtained from all individual participants included in the study.
Acknowledgments
The authors sincerely wish to thank all of the patients agreed to be involved in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of China (NSFC 81672547, 81974398 and 81902577), and 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
